Journal Description
Catalysts
Catalysts
is a peer-reviewed open access journal of catalysts and catalyzed reactions published monthly online by MDPI. The Romanian Catalysis Society (RCS) are partners of Catalysts journal and its members receive a discount on the article processing charge.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), Inspec, CAPlus / SciFinder, CAB Abstracts, and other databases.
- Journal Rank: JCR - Q2 (Chemistry, Physical) / CiteScore - Q1 (General Environmental Science)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 14.3 days after submission; acceptance to publication is undertaken in 2.7 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Impact Factor:
3.9 (2022);
5-Year Impact Factor:
4.2 (2022)
Latest Articles
Valorization of Chlorella Microalgae Residual Biomass via Catalytic Acid Hydrolysis/Dehydration and Hydrogenolysis/Hydrogenation
Catalysts 2024, 14(5), 286; https://0-doi-org.brum.beds.ac.uk/10.3390/catal14050286 - 23 Apr 2024
Abstract
Microalgal biomass can be utilized for the production of value-added chemicals and fuels. Within this research, Chlorella vulgaris biomass left behind after the extraction of lipids and proteins was converted to valuable sugars, organic acids and furanic compounds via hydrolysis/dehydration using dilute aqueous
[...] Read more.
Microalgal biomass can be utilized for the production of value-added chemicals and fuels. Within this research, Chlorella vulgaris biomass left behind after the extraction of lipids and proteins was converted to valuable sugars, organic acids and furanic compounds via hydrolysis/dehydration using dilute aqueous sulfuric acid as a homogeneous catalyst. Under mild conditions, i.e., low temperature and low sulfuric acid concentration, the main products of hydrolysis/dehydration were monomeric sugars (glucose and xylose) and furanic compounds (HMF, furfural) while under more intense conditions (i.e., higher temperature and higher acid concentration), organic acids (propionic, formic, acetic, succinic, lactic, levulinic) were also produced either directly from sugar conversion or via intermediate furans. As a second valorization approach, the residual microalgal biomass was converted to value-added sugar alcohols (sorbitol, glycerol) via hydrogenation/hydrogenolysis reactions over metallic ruthenium catalysts supported on activated carbons (5%Ru/C). It was also shown that a low concentration of sulfuric acid facilitated the conversion of biomass to sugar alcohols by initiating the hydrolysis of carbohydrates to monomeric sugars. Overall, this work aims to propose valorization pathways for a rarely utilized residual biomass towards useful compounds utilized as platform chemicals and precursors for the production of a wide variety of solvents, polymers, fuels, food ingredients, pharmaceuticals and others.
Full article
(This article belongs to the Special Issue State-of-the-Art in Molecular Catalysis in Europe)
Open AccessEditorial
Catalytic Epoxidation Reaction
by
Sébastien Leveneur, Pasi Tolvanen and Vincenzo Russo
Catalysts 2024, 14(5), 285; https://0-doi-org.brum.beds.ac.uk/10.3390/catal14050285 - 23 Apr 2024
Abstract
The epoxidation of unsaturated groups is a well-known process [...]
Full article
(This article belongs to the Special Issue Catalytic Epoxidation Reaction)
Open AccessArticle
Enhancing Oxygen Evolution Reaction with Two-Dimensional Nickel Oxide on Au (111)
by
Handing Zhang, Haoyu Zhang, Ruijing Wang, Jiayu Lv, Wugen Huang, Chenyan Guo and Fan Yang
Catalysts 2024, 14(5), 284; https://0-doi-org.brum.beds.ac.uk/10.3390/catal14050284 - 23 Apr 2024
Abstract
The nature of the active sites of transition metal oxides during the oxygen evolution reaction (OER) has attracted much attention. Herein, we constructed well-defined nickel oxide/Au (111) model catalysts to study the relationship between the structures and their OER activity using scanning tunneling
[...] Read more.
The nature of the active sites of transition metal oxides during the oxygen evolution reaction (OER) has attracted much attention. Herein, we constructed well-defined nickel oxide/Au (111) model catalysts to study the relationship between the structures and their OER activity using scanning tunneling microscopy (STM), X-ray photoelectron spectroscopy (XPS), electrochemical measurements, and density functional theory (DFT) calculations. The deposited nickel oxides on Au (111) were found to exhibit a two-dimensional (2D)/three-dimensional (3D) structure by regulating the annealing temperature. Combining STM, XPS and electrochemical measurements, our results demonstrated an optimal OER reactivity could be achieved for NiOx with a 2D structure on Au and provided a morphological description of the active phase during electrocatalysis.
Full article
(This article belongs to the Topic Surface Chemistry of Catalysis)
►▼
Show Figures
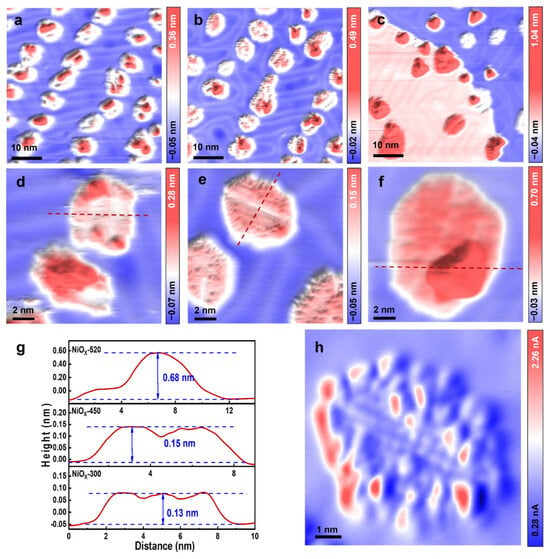
Figure 1
Open AccessArticle
Platinum-Modified Rod-like Titania Mesocrystals with Enhanced Photocatalytic Activity
by
Zhishun Wei, Yuanyuan Ji, Zuzanna Bielan, Xin Yue, Yuqi Xu, Jiajie Sun, Sha Chen, Guoqiang Yi, Ying Chang and Ewa Kowalska
Catalysts 2024, 14(4), 283; https://0-doi-org.brum.beds.ac.uk/10.3390/catal14040283 - 22 Apr 2024
Abstract
Photocatalysis is considered as an environmentally friendly method for both solar energy conversion and environmental purification of water, wastewater, air, and surfaces. Among various photocatalytic materials, titania is still the most widely investigated and applied, but more efforts must be carried out considering
[...] Read more.
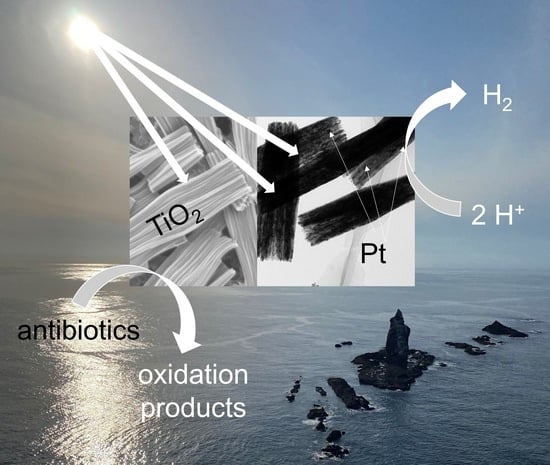
Photocatalysis is considered as an environmentally friendly method for both solar energy conversion and environmental purification of water, wastewater, air, and surfaces. Among various photocatalytic materials, titania is still the most widely investigated and applied, but more efforts must be carried out considering the synthesis of highly efficient photocatalysts for multifarious applications. It is thought that nanoengineering design of titania morphology might be the best solution. Accordingly, here, titania mesocrystals, assembled from crystallographically oriented nanocrystals, have been synthesized by an easy, cheap, and “green” solvothermal method (without the use of surfactants and templates), followed by simple annealing. The obtained materials have been characterized by various methods, including transmission electron microscopy (TEM), scanning electron microscopy (SEM), X-ray photoelectron spectroscopy (XPS), X-ray powder diffraction (XRD) and diffuse reflectance spectroscopy (DRS). It has been found that the as-obtained photocatalysts exhibit a unique nanorod-like subunit structure with excellent crystalline and surface properties. However, pristine titania is hardly active for a hydrogen evolution reaction, and thus additional modification has been performed by platinum photodeposition (and silver as a reference). Indeed, the modification with only 2 wt% of noble metals results in a significant enhancement in activity, i.e., ca. 75 and 550 times by silver- and platinum-modified samples, respectively, reaching the corresponding reaction rates of 37 μmol h−1 and 276 μmol h−1. Additionally, titania mesocrystals exhibit high oxidation power under simulated solar light irradiation for the degradation of antibiotics within the tetracycline group (tetracycline (TC), ciprofloxacin (CIP), norfloxacin (NOR) and oxytetracycline hydrochloride (OTC)). It has been found that both experimental results and the density functional theory (DFT) calculations confirm the high ability of titania mesocrystals for oxidative decomposition of tetracycline antibiotics.
Full article
(This article belongs to the Special Issue Novel Nanocatalysts for Sustainable and Green Chemistry)
►▼
Show Figures
Graphical abstract
Open AccessArticle
Catalytic Decomposition of CH4 to Hydrogen and Carbon Nanotubes Using the Pt(1)-Fe(30)/MCM-41 Catalyst
by
Ho Joon Seo
Catalysts 2024, 14(4), 282; https://0-doi-org.brum.beds.ac.uk/10.3390/catal14040282 - 20 Apr 2024
Abstract
The catalytic decomposition of CH4 to H2 and carbon nanotubes (CNTs) was investigated regarding Pt(1)-Fe(30)/MCM-41 and Fe(30)/MCM-41 using a fixed-bed flow reactor under an atmosphere. X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), scanning electron microscopy (SEM), energy dispersive spectroscopy (EDS), transmission
[...] Read more.
The catalytic decomposition of CH4 to H2 and carbon nanotubes (CNTs) was investigated regarding Pt(1)-Fe(30)/MCM-41 and Fe(30)/MCM-41 using a fixed-bed flow reactor under an atmosphere. X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), scanning electron microscopy (SEM), energy dispersive spectroscopy (EDS), transmission electron microscope (TEM), and Raman spectroscopy were used to characterize the behavior of Pt(1)-Fe(30)/MCM-41 and Fe(30)/MCM-41. The hydrogen yield of Pt(1)-Fe(30)/MCM-41 was 3.2 times higher than that of Fe(30)/MCM-41. When 1 wt% of Pt was added to Fe(30)/MCM-41(Mobil Composition of Matter No. 41), the atomic percentage of Fe2p increased from 13.39% to 16.14% and the core Fe2p1/2 electron levels of Fe0 and Fe2+ chemically shifted to lower energies (0.2 eV and 0.1 eV, respectively) than those of Fe(30)/MCM-41. The Fe, Pt, Si, and O nanoparticles were uniformly distributed on the catalyst surface, and the average iron particle sizes of the Pt(1)-Fe(30)/MCM-41 and Fe(30)/MCM-41 were about 33.4 nm and 58.5 nm, respectively. This is attributed to the uniform distribution of the nano-sized iron particles on the MCM-41 surface, which was due to the suitable metal-carrier interaction (SMCI) between Fe, Pt, and MCM-41 and the high reduction degree of Fe due to the spillover effect of H2 from Pt to Fe. Pt(1)-Fe(30)/MCM-41 produced multiwalled CNTs and bamboo-shaped CNTs with high crystallinity and graphitization degree using the tip-growth mechanism, with an ID/IG ratio of 0.93 and a C(101)/C(002) ratio of 0.64.
Full article
(This article belongs to the Special Issue Study of Novel Catalysts for Methane Conversion)
►▼
Show Figures
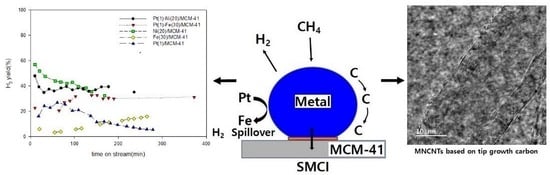
Graphical abstract
Open AccessArticle
Taurine-Functionalized Carbon Nanotubes as Electrode Catalysts for Improvement in the Performance of Vanadium Redox Flow Battery
by
Lian Wei, Tao Liu, Yimin Zhang, Hong Liu and Ling Ge
Catalysts 2024, 14(4), 281; https://0-doi-org.brum.beds.ac.uk/10.3390/catal14040281 - 20 Apr 2024
Abstract
The vanadium redox flow battery (VRFB) is a highly favorable tool for storing renewable energy, and the catalytic activity of electrode materials is crucial for its development. Taurine-functionalized carbon nanotubes (CNTs) were prepared with the aim of augmenting the redox process of vanadium
[...] Read more.
The vanadium redox flow battery (VRFB) is a highly favorable tool for storing renewable energy, and the catalytic activity of electrode materials is crucial for its development. Taurine-functionalized carbon nanotubes (CNTs) were prepared with the aim of augmenting the redox process of vanadium ions and enhancing the efficiency of the VRFB. Sulfonated CNTs were synthesized through a simple modification process in a taurine solution and used as electrocatalysts for redox reactions involving VO2+/VO2+ and V2+/V3+. The SO3H-CNTs modified at 60 °C for 2 h exhibit the best electrocatalytic activity, showing higher redox peak current values compared to pristine carboxylated CNTs (COOH-CNTs). Sulfonic acid groups added to the surface of CNTs increase active sites for redox reactions and act as carriers for mass transfer and bridges for charge transfer, accelerating the rate of the electrode reactions. A battery consisting of SO3H-CNTs as catalysts demonstrates the outstanding charge–discharge performance at a current density of 300 mA∙cm−2. This configuration displays voltage and energy efficiencies of 81.46% and 78.83%, respectively, representing enhancements of 6.15% and 6.12% compared to that equipped with conventional graphite felts (75.31%, 72.71%). This study illustrates that taurine-functionalized carbon nanotubes serve as an efficient and promising catalyst for both the anode and cathode, leading to the improved performance of the VRFB.
Full article
(This article belongs to the Section Electrocatalysis)
►▼
Show Figures
Figure 1
Open AccessArticle
Simple Fabrication of Hydrophobicity-Controlled Fe-ZSM-5 for Aqueous-Phase Partial Oxidation of Methane with Hydrogen Peroxide
by
Seok Young Hwang, Minjae Kwon, Jongkook Hwang and Eun Duck Park
Catalysts 2024, 14(4), 280; https://0-doi-org.brum.beds.ac.uk/10.3390/catal14040280 - 20 Apr 2024
Abstract
Surface hydrophobicity is an important factor in controlling the catalytic activity of heterogeneous catalysts in various reactions, particularly liquid-phase reactions using water as the (co)solvent. In this study, the surface hydrophobicity of Fe-ZSM-5 was successfully controlled using a simple coating method in which
[...] Read more.
Surface hydrophobicity is an important factor in controlling the catalytic activity of heterogeneous catalysts in various reactions, particularly liquid-phase reactions using water as the (co)solvent. In this study, the surface hydrophobicity of Fe-ZSM-5 was successfully controlled using a simple coating method in which furfuryl alcohol was used as the carbon precursor. Various techniques, such as N2 physisorption, temperature-programmed desorption of ammonia, and contact angle measurements of water droplets, were used to characterize the catalysts. Fe-ZSM-5 catalysts with different degrees of hydrophobicity were used for the aqueous-phase selective oxidation of methane with H2O2. The positive effect of the surface carbon coating on the catalytic performance was confirmed when the carbon content was not sufficiently high to block the pores.
Full article
(This article belongs to the Special Issue Catalytic Conversion of Low Carbon Alkane)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Preparation of High-Efficiency Fe/N-Doped Carbon Catalysts Derived from Graphite Phase Carbon Nitride for Reduction of Oxygen
by
Yan Wang, Wuxin Liu, Rongzhe Wang, Qing Wang, Shaohua Luo, Pengqing Hou, Yahui Zhang, Shengxue Yan, Xin Liu and Jing Guo
Catalysts 2024, 14(4), 279; https://0-doi-org.brum.beds.ac.uk/10.3390/catal14040279 - 19 Apr 2024
Abstract
Fe/N-doped carbon (Fe-NC) is an excellent base-metal catalyst for use in an electrocatalytic oxygen reduction reaction (ORR) with high activity. In this paper, graphite phase carbon nitride (g-C3N4) was first obtained from the pyrolyzing of melamine, and then different
[...] Read more.
Fe/N-doped carbon (Fe-NC) is an excellent base-metal catalyst for use in an electrocatalytic oxygen reduction reaction (ORR) with high activity. In this paper, graphite phase carbon nitride (g-C3N4) was first obtained from the pyrolyzing of melamine, and then different proportions of FeCl3 were separately doped into g-C3N4 to further prepare the Fe-NC catalyst. The Fe-NC catalyst was applied in an ORR reaction, and the results show that the Fe-NC catalyst doped with 0.5 mmol FeCl3 possesses exceptional electrocatalytic performance, with an onset potential of 0.96 V and a half-wave potential of 0.81 V, which approaches that of a Pt/C catalyst. Meanwhile, the Fe-NC catalyst displays high stability and methanol resistance. The results supply a new way to prepare efficient ORR electrocatalysts.
Full article
(This article belongs to the Section Electrocatalysis)
►▼
Show Figures

Figure 1
Open AccessArticle
Stably Improving the Catalytic Activity of Oxygen Evolution Reactions via Two-Dimensional Graphene Oxide-Incorporated NiFe-Layered Double Hydroxides
by
Ling Chen, Yue Lu, Manman Duanmu, Xin Zhao, Shenglu Song, Liyue Duan, Zhipeng Ma, Ailing Song and Guangjie Shao
Catalysts 2024, 14(4), 278; https://0-doi-org.brum.beds.ac.uk/10.3390/catal14040278 - 19 Apr 2024
Abstract
NiFe-layered double hydroxides (NiFe-LDH) have been reported to possess exceptional oxygen evolution reaction (OER) activity. However, maintaining the stability of high activity over a long time remains a critical challenge that needs to be addressed for their practical application. Here, we report a
[...] Read more.
NiFe-layered double hydroxides (NiFe-LDH) have been reported to possess exceptional oxygen evolution reaction (OER) activity. However, maintaining the stability of high activity over a long time remains a critical challenge that needs to be addressed for their practical application. Here, we report a custom-sized deep recombination of 2D graphene oxide with NiFe-LDH (NiFe-LDH/GO/NF) through a simple electrodeposition method that improves OER activity and achieves excellent stability. The excellent performance of the catalyst mainly comes from the three-phase interface and electron transport channel dredged by the three-dimensional structure constructed by the deep composite, which can not only significantly reduce its charge and electron transfer resistance, improving the material conductivity, but it also effectively increases the specific surface area, inhibits aggregation, and exposes rich active sites. In addition, GO with good conductivity not only supports NiFe-LDH well but also increases the heterogeneous interface, putting the NiFe-LDH/GO composites in close contact with Ni foam and increasing the electrocatalytic stability of the NiFe-LDH/GO/NF. The experimental results show that the overpotential of NiFe-LDH/20,000GO/NF is only 295 mV at a current density of 100 mA cm−2; the Tafel slope is 52 mV dec−1, and the charge transfer resistance (Rct) is only 0.601 Ω in 1 M KOH. This indicates that GO has excellent potential to assist in constructing geometric and electronic structures of NiFe-LDH in long-term applications.
Full article
(This article belongs to the Section Catalytic Materials)
►▼
Show Figures
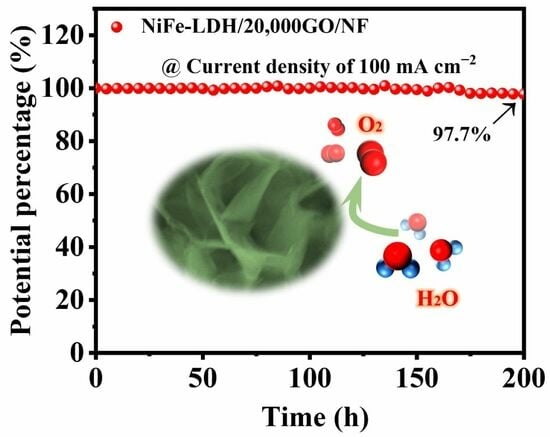
Graphical abstract
Open AccessArticle
Preparation of Poly(Butadiene–Styrene–Vinyl Pyridine)/Poly(Acrylonitrile–Butadiene) Core–Shell Nanoparticles by Intermittent Seeded Emulsion Polymerization and Their Catalytic Latex Hydrogenation
by
Fei Yuan, Xudong Li, Jianying Dou, Baojia Zhang, Xueling Song, Lin Li, Junjie Liu, Yanyan Li, Yigao Jiang and Hui Wang
Catalysts 2024, 14(4), 277; https://0-doi-org.brum.beds.ac.uk/10.3390/catal14040277 - 19 Apr 2024
Abstract
Seed emulsion polymerization was an effective modification method to improve not only the properties of polymers but also the compatibility between different polymers by designing special core-shell structures. In this study, poly (butadiene-styrene-vinyl pyridine) (VPR)/poly (acrylonitrile-butadiene) (NBR) core–shell nanoparticles (VPR/NBR) were prepared by
[...] Read more.
Seed emulsion polymerization was an effective modification method to improve not only the properties of polymers but also the compatibility between different polymers by designing special core-shell structures. In this study, poly (butadiene-styrene-vinyl pyridine) (VPR)/poly (acrylonitrile-butadiene) (NBR) core–shell nanoparticles (VPR/NBR) were prepared by seed emulsion polymerization using VPR as seed emulsion and butadiene and acrylonitrile as monomers. Subsequently, HVPR/HNBR was obtained by direct hydrogenation of the core–shell nanoparticles in latex using Wilkinson’s catalyst under high temperature and H2 pressure. It is noteworthy that the unsaturated C=C double bonds in the core (VPR) and shell (NBR) of HVPR/HNBR nanoparticles were reduced simultaneously during the hydrogenation process without obvious sequence. The particle size and size distribution of the particles remained consistent before and after hydrogenation, indicating that the synthesized core-shell nanoparticles have excellent stability. This study provides a new perspective on the chemical modification of NBR and promises an environmentally friendly “green” process for the industrial hydrogenation of unsaturated elastomers.
Full article
(This article belongs to the Section Catalysis in Organic and Polymer Chemistry)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Direct Conversion of Ethanol to Propylene over Zn-Modified HBeta Zeolite: Influence of Zinc Precursors
by
Ting Bai, Xiaohui Li, Liang Ding, Jin Wang, Yong-Shan Xiao and Bin Cao
Catalysts 2024, 14(4), 276; https://0-doi-org.brum.beds.ac.uk/10.3390/catal14040276 - 19 Apr 2024
Abstract
A series of Zn-modified HBeta (Zn/HBeta) catalysts were prepared via the wetness impregnation method with different zinc precursors such as ZnSO4·7H2O, ZnCl2, C4H6O4Zn·2H2O and Zn(NO3)2·6H
[...] Read more.
A series of Zn-modified HBeta (Zn/HBeta) catalysts were prepared via the wetness impregnation method with different zinc precursors such as ZnSO4·7H2O, ZnCl2, C4H6O4Zn·2H2O and Zn(NO3)2·6H2O, and their catalytic performance in the conversion of ethanol to propylene reaction was evaluated. Results indicate that the amount and strength distribution of the acid sites of the Zn/HBeta catalysts were easily tuned by employing different types of zinc precursors. More importantly, when the zinc species were introduced to the HBeta, the propylene yield was significantly enhanced, whereas the yields of ethylene and C2–C4 alkanes were remarkably suppressed. For the catalyst prepared by using the ZnCl2 precursor, a higher propylene yield of up to 43.4% for Zn/HBeta-C was achieved as a result of the moderate amount and strength distribution of acid sites. The average coking rate of the used Zn/HBeta catalysts strongly depended on the amount of total acid sites, especially the strong acid sites, i.e., the higher the amount of total acid sites of the catalyst, the greater the average coking rate. For the catalyst prepared by using the ZnSO4·7H2O precursor, Zn/HBeta-S exhibited a better stability even after depositing more coke, which was due to the higher amount of strong acid sites.
Full article
(This article belongs to the Special Issue Heterogeneous Catalysis and Advanced Oxidation Processes (AOP) for Environmental Protection (VOCs Oxidation, Air and Water Purification), 2nd Edition)
►▼
Show Figures
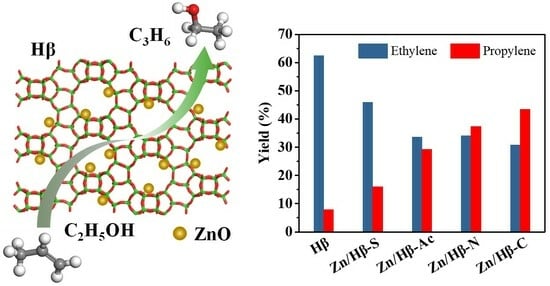
Graphical abstract
Open AccessArticle
Plasmonic Bi-Modified Bi2Sn2O7 Nanosheets for Efficient Photocatalytic NO Removal
by
Ning Li, Wenwen Zhao, Jiatong Zhang, Xuhui Liu, Yangqin Gao and Lei Ge
Catalysts 2024, 14(4), 275; https://0-doi-org.brum.beds.ac.uk/10.3390/catal14040275 - 18 Apr 2024
Abstract
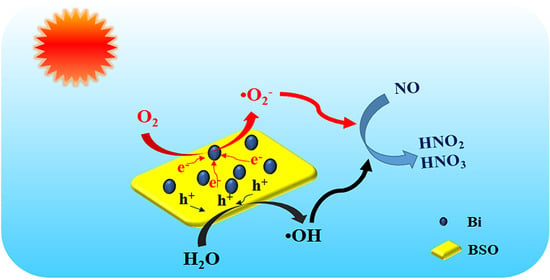
The photocatalytic removal of nitric oxide (NO) is a promising technology used to reduce the level of harmful gaseous pollutants in parts per billion (ppb). As a potential photocatalyst, Bi2Sn2O7 has a low quantum efficiency due to its
[...] Read more.
The photocatalytic removal of nitric oxide (NO) is a promising technology used to reduce the level of harmful gaseous pollutants in parts per billion (ppb). As a potential photocatalyst, Bi2Sn2O7 has a low quantum efficiency due to its fast recombination rate of photo-generated carriers. In this paper, Bi/Bi2Sn2O7 was prepared by the in situ deposition of Bi. The structural, electrical, and optical properties of the attained sample were investigated through a series of analyses. The results demonstrate that Bi nanoparticles not only enhance the photoabsorption ability of Bi2Sn2O7 due to their surface plasmon resonance (SPR) effect, but also improve its photocatalytic activity. Photocatalytic performance was evaluated by the oxidation of NO at ppb level under xenon lamp (λ > 400 nm) irradiation. It was found that the photocatalytic NO removal rate increased from 7.2% (Bi2Sn2O7) to 38.6% (Bi/Bi2Sn2O7). The loading of Bi promotes the separation and migration of photo-generated carriers and enhances the generation of •O2− and •OH radicals responsible for the oxidation of NO. The Bi/Bi2Sn2O7 composite photocatalyst also exhibits excellent photocatalytic stability, which makes it a potential candidate for use in air purification systems.
Full article
(This article belongs to the Special Issue Two-Dimensional Materials in Photo(electro)catalysis)
►▼
Show Figures
Graphical abstract
Open AccessArticle
Synthesis and Catalytic Activity of 1,2-Benzenediamine-Derived Organocatalysts Based on (1R,2R)-Cyclohexane-1,2-Diamine
by
Luka Ciber, Klara Klemenčič, Ana Golob, Helena Brodnik, Franc Požgan, Jurij Svete, Bogdan Štefane and Uroš Grošelj
Catalysts 2024, 14(4), 274; https://0-doi-org.brum.beds.ac.uk/10.3390/catal14040274 - 18 Apr 2024
Abstract
A four-step synthesis process of bifunctional, noncovalent organocatalysts based on the chiral (1R,2R)-cyclohexane-1,2-diamine scaffold containing a 1,2-benzenediamine H-bond donor was developed. Nucleophilic aromatic substitution of the 2-fluoronitrobenzene derivative with the commercial (1R,2R)-cyclohexane-1,2-diamine was followed by
[...] Read more.
A four-step synthesis process of bifunctional, noncovalent organocatalysts based on the chiral (1R,2R)-cyclohexane-1,2-diamine scaffold containing a 1,2-benzenediamine H-bond donor was developed. Nucleophilic aromatic substitution of the 2-fluoronitrobenzene derivative with the commercial (1R,2R)-cyclohexane-1,2-diamine was followed by selective alkylation of the primary amino group, reduction of the aromatic nitro group and final derivatization of the primary aromatic amino group, i.e., acylation, sulfonation, reductive alkylation and arylation, leading to the four subtypes of organocatalysts. All new compounds were fully characterized. The prepared organocatalysts (32 examples) were tested in the Michael addition of acetylacetone to trans-β-nitrostyrene, yielding the addition product with incomplete conversions (up to 93%) and enantioselectivities of up to 41% ee.
Full article
(This article belongs to the Special Issue Organocatalysis in the Chemical Transformations, 2nd Edition)
►▼
Show Figures
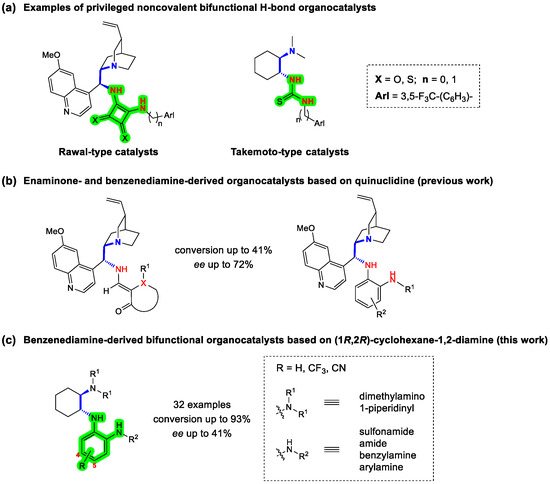
Figure 1
Open AccessArticle
Determination of Activation Energy from Decolorization Reactions of Synthetic Dyes by Fenton Processes Using the Behnajady–Modirshahla–Ghanbary Kinetic Model
by
Márcio Daniel Nicodemos Ramos, Juan Pablo Pereira Lima and André Aguiar
Catalysts 2024, 14(4), 273; https://0-doi-org.brum.beds.ac.uk/10.3390/catal14040273 - 18 Apr 2024
Abstract
The present work used the Behnajady–Modirshahla–Ghanbary (BMG) kinetic model to determine the initial reaction rates (1/m), which were used to calculate the activation energy (Ea) from the decolorization of synthetic dyes by Fenton processes (Fe2+/H2O
[...] Read more.
The present work used the Behnajady–Modirshahla–Ghanbary (BMG) kinetic model to determine the initial reaction rates (1/m), which were used to calculate the activation energy (Ea) from the decolorization of synthetic dyes by Fenton processes (Fe2+/H2O2, Fe2+/H2O2/reducer and Fe3+/H2O2/reducer). When increasing the temperature and adding Fe3+-reducing compounds (3-Hydroxyanthranilic Acid, Hydroquinone, Gallic Acid, Cysteine or Ascorbic Acid), increases in the 1/m values were observed. When studying the classical Fenton reaction (Fe2+/H2O2), almost all added reducers had decreased Ea. For example, 3-Hydroxyanthranilic Acid decreased the Ea related to the decolorization of the Phenol Red dye by 39%, while Ascorbic Acid decreased the Ea of Safranin T decolorization by 23%. These results demonstrate that the reducers increased the initial reaction rate and decreased the energy barrier to improve Fenton-based decolorization of dyes. When comparing the reaction systems in presence of reducers (Fen+/H2O2/reducer), the reactions initially containing Fe2+ presented lower Ea than reactions catalyzed by Fe3+. That way, the activation energy obtained through the 1/m values of the BMG model highlighted the pro-oxidant effect of reducers in Fenton processes to degrade dyes.
Full article
(This article belongs to the Special Issue Emerging Research in Integrated Real Wastewater Treatments via Advanced Catalytic Oxidation)
►▼
Show Figures
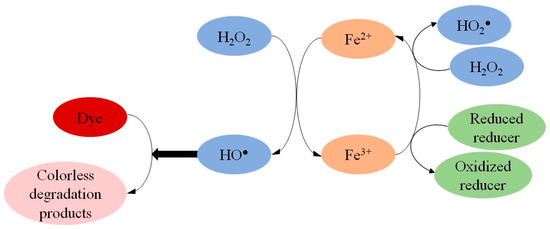
Figure 1
Open AccessArticle
Cobalt Nanoparticles Supported on TiO2 for Highly Selective Formation of N-Benzylideneanilines from Nitroarenes and Benzaldehyde via Reductive Imination Reaction
by
Daniela González-Vera, Tatiana M. Bustamante, J. Noé Díaz de León, Cecilia C. Torres and Cristian H. Campos
Catalysts 2024, 14(4), 272; https://0-doi-org.brum.beds.ac.uk/10.3390/catal14040272 - 17 Apr 2024
Abstract
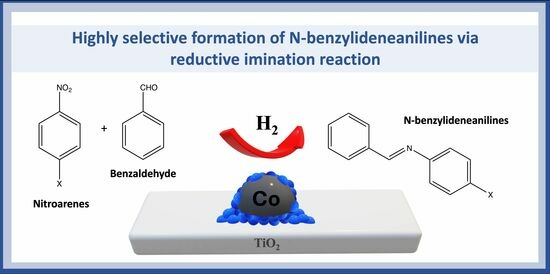
The search for active, inexpensive, and stable heterogeneous catalysts to produce desired imines in fine chemistry presents an ongoing challenge for both academia and industry. This work reports the utilization of Co nanoparticles supported on TiO2 derived from the H2-assisted
[...] Read more.
The search for active, inexpensive, and stable heterogeneous catalysts to produce desired imines in fine chemistry presents an ongoing challenge for both academia and industry. This work reports the utilization of Co nanoparticles supported on TiO2 derived from the H2-assisted reduction of the perovskite-type mixed oxide CoTiO3. The entire preparation process is operationally simple and straightforward, enabling scalability for practical applications. The resulting catalyst comprises metallic cobalt nanoparticles responsible for the hydrogenation process, whereas the TiOx thin layer surrounding the cobalt promotes the adsorption of C=O, thereby enhancing the formation of desired products. Notably, at lower temperatures, the reaction yields the target imine product. Our study demonstrates a synergistic effect between nitrobenzene and benzaldehyde in the presence of a Co-TiOx interface, which reduces the apparent activation energy for the hydrogenation of the-NO2 group. Furthermore, under moderate reaction conditions, the catalytic system offers applicability to various nitrobenzene compounds substituted at the 4-position and benzaldehyde, resulting in high yields of the corresponding imines with electron-density-donating substituent groups. Finally, the catalyst exhibits facile separation for subsequent reuse, displaying moderate stability with minimal selectivity for the desired product.
Full article
(This article belongs to the Special Issue Heterogeneous Catalysis for Environmentally Compatible Reactions and Processes)
►▼
Show Figures
Graphical abstract
Open AccessArticle
Investigating the Long-Term Kinetics of Pd Nanoparticles Prepared from Microemulsions and the Lindlar Catalyst for Selective Hydrogenation of 3-Hexyn-1-ol
by
Faeze Tari, Sebastian Hertle, Hongguang Wang, Julian Fischer, Peter A. van Aken, Thomas Sottmann, Elias Klemm and Yvonne Traa
Catalysts 2024, 14(4), 271; https://0-doi-org.brum.beds.ac.uk/10.3390/catal14040271 - 17 Apr 2024
Abstract
The effect of non-saturated corner and edge sites of Pd particles on the long-term selectivity of cis-3-hexen-1-ol in the hydrogenation of 3-hexyn-1-ol was studied in this work. Non-supported Pd agglomerates were synthesized through the microemulsion synthesis route and used at
[...] Read more.
The effect of non-saturated corner and edge sites of Pd particles on the long-term selectivity of cis-3-hexen-1-ol in the hydrogenation of 3-hexyn-1-ol was studied in this work. Non-supported Pd agglomerates were synthesized through the microemulsion synthesis route and used at
(This article belongs to the Section Nanostructured Catalysts)
►▼
Show Figures
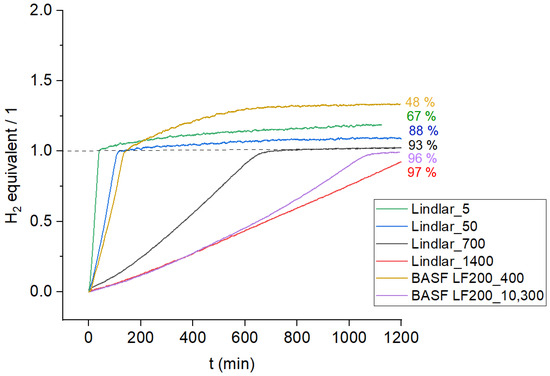
Figure 1
Open AccessArticle
CO2 Reduction Performance with Double-Layered Cu/TiO2 and P4O10/TiO2 as Photocatalysts under Different Light Illumination Conditions
by
Akira Nishimura, Hiroki Senoue, Homare Mae, Ryo Hanyu and Eric Hu
Catalysts 2024, 14(4), 270; https://0-doi-org.brum.beds.ac.uk/10.3390/catal14040270 - 17 Apr 2024
Abstract
This paper presents an experimental study of using a double-layered Cu/TiO2 and P4O10/TiO2 as photocatalysts for CO2 reduction with an extended wavelength of range light from ultraviolet light (UV) to infrared light (IR). The lights studied
[...] Read more.
This paper presents an experimental study of using a double-layered Cu/TiO2 and P4O10/TiO2 as photocatalysts for CO2 reduction with an extended wavelength of range light from ultraviolet light (UV) to infrared light (IR). The lights studied were UV + visible light (VIS) + IR, VIS + IR and IR only. This study also investigated the impact of the molar ratio of CO2:H2O on the CO2 reduction performance. This study revealed that the optimum molar ratio of CO2:H2O to produce CO was 1:1, irrespective of light illumination condition, which matched the theoretical molar ratio to produce CO according to the reaction scheme of CO2 reduction with H2O. Comparing the results of double-layered Cu/TiO2 and P4O10/TiO2 with those of double-layered TiO2 obtained under the UV + VIS + IR light illumination condition, the highest concentration of formed CO and the molar quantity of formed CO per unit weight of the photocatalyst increased by 281 ppmV and 0.8 μmol/g, in the case of the molar ratio of CO2:H2O = 1:1. With IR-only illumination, the highest concentration of formed CO and the molar quantity of CO formed per unit weight of the photocatalyst was 251 ppmV and 4.7 μmol/g, respectively.
Full article
(This article belongs to the Special Issue Enhancement of the Performance of Photocatalytic CO2 Reduction)
►▼
Show Figures
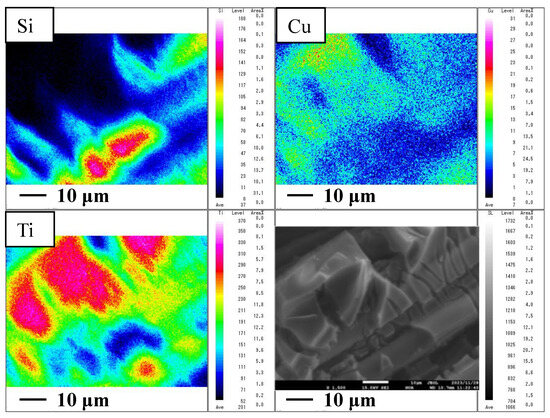
Figure 1
Open AccessReview
Performance of Traditional and Emerging Water-Treatment Technologies in the Removal of Tetracycline Antibiotics
by
Fanjin Wang, Ziyi Wang, Yue Zhao and Jian Zhang
Catalysts 2024, 14(4), 269; https://0-doi-org.brum.beds.ac.uk/10.3390/catal14040269 - 17 Apr 2024
Abstract
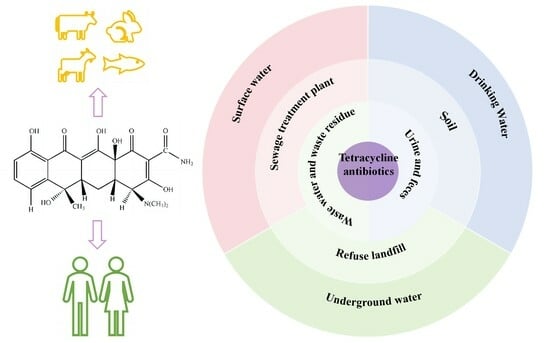
Tetracycline antibiotics are widely used in human medical treatment, control of animal disease, and agricultural feed because of their broad spectrum of action, high efficiency, and low cost. The excessive use of antibiotics and arbitrary discharge of antibiotic wastewater have become increasingly serious
[...] Read more.
Tetracycline antibiotics are widely used in human medical treatment, control of animal disease, and agricultural feed because of their broad spectrum of action, high efficiency, and low cost. The excessive use of antibiotics and arbitrary discharge of antibiotic wastewater have become increasingly serious problems, and the current sewage-treatment process is not ideal for treating water contaminated with tetracycline antibiotics, leading to increasingly prominent antibiotic pollution in water and the imminent need for its removal. In order to understand the necessity of removing tetracycline antibiotics from the water environment, this paper first expounds on their source, harms, and pollution status in oceans and in surface water, groundwater, wastewater, and drinking water. It next introduces the research status of conventional treatment methods such as adsorption methods, biological methods, and physical and chemical methods, then introduces new treatment methods such as advanced oxidation methods and comprehensive treatment technology in sewage plants. The degradation effects, mechanisms of action, and challenges of these methods were summarized. The advantages and disadvantages of each treatment technology are compared. Finally, potential future processing technologies are discussed.
Full article
(This article belongs to the Topic Advanced Oxidation Processes for Wastewater Purification)
►▼
Show Figures
Graphical abstract
Open AccessReview
Ethylene Oligomerization Catalyzed by Different Homogeneous or Heterogeneous Catalysts
by
Anfeng Peng, Zheng Huang and Gang Li
Catalysts 2024, 14(4), 268; https://0-doi-org.brum.beds.ac.uk/10.3390/catal14040268 - 17 Apr 2024
Abstract
Linear α-olefins (LAOs) are linear alkenes with double bonds at the ends of the molecular chains. LAOs with different chain lengths can be widely applied in various fields. Ethylene oligomerization has become the main process for producing LAOs. In this review, different homogeneous
[...] Read more.
Linear α-olefins (LAOs) are linear alkenes with double bonds at the ends of the molecular chains. LAOs with different chain lengths can be widely applied in various fields. Ethylene oligomerization has become the main process for producing LAOs. In this review, different homogeneous or heterogeneous catalysts recently reported in ethylene oligomerization with Ni, Fe, Co, Cr, etc., as active centers will be discussed. In the homogeneous catalytic system, we mainly discuss the effects of the molecular structure and the electronic and coordination states of complexes on their catalytic activity and selectivity. The Ni, Fe, and Co homogeneous catalysts are discussed separately based on different ligand types, while the Cr-based homogeneous catalysts are discussed separately for ethylene trimerization, tetramerization, and non-selective oligomerization. In heterogeneous catalytic systems, we mainly concentrate on the influence of various supports (metal–organic frameworks, covalent organic frameworks, molecular sieves, etc.) and different ways to introduce active centers to affect the performance in ethylene oligomerization. Finally, a summary and outlook on ethylene oligomerization catalysts are provided based on the current research. The development of highly selective α-olefin formation processes remains a major challenge for academia and industry.
Full article
(This article belongs to the Special Issue Exclusive Papers of the Editorial Board Members and Topical Advisory Panel Members of Catalysts in Section "Catalytic Materials")
►▼
Show Figures
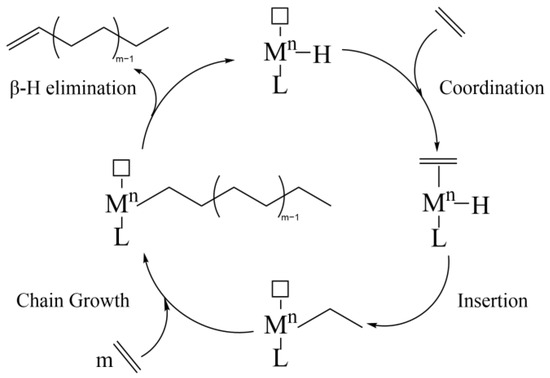
Figure 1
Open AccessReview
An Overview of Environmental Catalysis Mediated by Hydrogen Peroxide
by
Monica Rigoletto, Enzo Laurenti and Maria Laura Tummino
Catalysts 2024, 14(4), 267; https://0-doi-org.brum.beds.ac.uk/10.3390/catal14040267 - 17 Apr 2024
Abstract
The use of hydrogen peroxide (produced in situ or ex situ) as the main agent in oxidative processes of environmental pollutant removal is widely studied. The degradation of water pollutants, such as dyes, pharmaceuticals, cosmetics, petroleum derivatives, and even pathogens, has been successfully
[...] Read more.
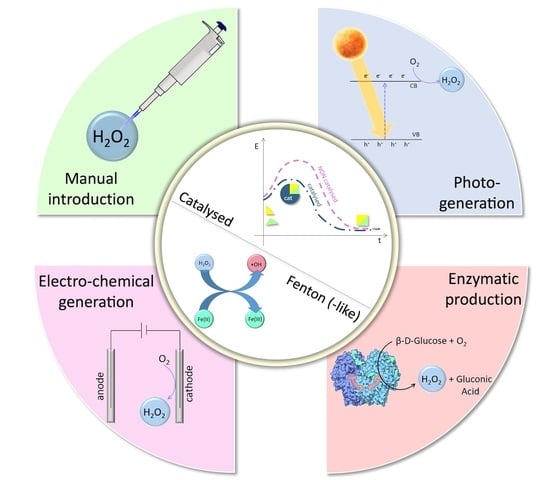
The use of hydrogen peroxide (produced in situ or ex situ) as the main agent in oxidative processes of environmental pollutant removal is widely studied. The degradation of water pollutants, such as dyes, pharmaceuticals, cosmetics, petroleum derivatives, and even pathogens, has been successfully obtained by different techniques. This review gives an overview of the more recent methods developed to apply oxidative processes mediated by H2O2 and other reactive oxygen species (ROS) in environmental catalysis, with particular attention to the strategies (Fenton-like and Bio-Fenton, photo- and electro-catalysis) and the materials employed. A wide discussion about the characteristics of the materials specifically studied for hydrogen peroxide activation, as well as about their chemical composition and morphology, was carried out. Moreover, recent interesting methods for the generation and use of hydrogen peroxide by enzymes were also presented and their efficiency and applicability compared with the Fenton and electro-Fenton methods discussed above. The use of Bio-Fenton and bi-enzymatic methods for the in situ generation of ROS seems to be attractive and scalable, although not yet applied in full-scale plants. A critical discussion about the feasibility, criticalities, and perspectives of all the methods considered completes this review.
Full article
(This article belongs to the Topic Advanced Oxidation Processes for Wastewater Purification)
►▼
Show Figures
Graphical abstract

Journal Menu
► ▼ Journal Menu-
- Catalysts Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Batteries, Catalysts, Energies, Hydrogen, Sustainability
Preparation, Storage, and Transportation of Green Hydrogen and Multi-Scenario Application Technologies
Topic Editors: Weihua Cai, Chao Xu, Zhonghao Rao, Fuqiang Wang, Ming GaoDeadline: 30 June 2024
Topic in
Catalysts, Coatings, Crystals, Energies, Materials, Nanomaterials
Interfacial Bonding Design and Applications in Structural and Functional Materials
Topic Editors: Junlei Qi, Pengcheng WangDeadline: 20 July 2024
Topic in
Energies, Catalysts, Fermentation, Processes, Waste
Valorizing Waste through Thermal and Biological Processes for Sustainable Energy Production
Topic Editors: Margarida Gonçalves, Cândida VilarinhoDeadline: 31 August 2024
Topic in
Cancers, Catalysts, Current Oncology, Plasma, Sci
Advances in Low-Temperature Plasma Cancer Therapy
Topic Editors: Michael Keidar, Li Lin, Dayun YanDeadline: 20 September 2024

Conferences
Special Issues
Special Issue in
Catalysts
Applications and New Trends in Catalysts and Photocatalytic Nanomaterials for Environmental Remediation
Guest Editors: Mehrab Mehrvar, Lesly Tejeda-Benitez, Fiderman Machuca-Martínez, Ciro Bustillo-LecompteDeadline: 30 April 2024
Special Issue in
Catalysts
Heterogeneous Catalysis for Selective Hydrogenation
Guest Editors: Shihui Zou, Juanjuan LiuDeadline: 15 May 2024
Special Issue in
Catalysts
Advances in Photocatalytic and Photothermal Catalytic CO2 Reduction
Guest Editor: Fernando FresnoDeadline: 6 June 2024
Special Issue in
Catalysts
Modern Catalytic Reactor: From Active Center to Application Tests, 2nd Edition
Guest Editors: Anna Gancarczyk, Agnieszka CiemięgaDeadline: 15 June 2024
Topical Collections
Topical Collection in
Catalysts
Photocatalytic Water Splitting
Collection Editors: Weilong Shi, Guigao Liu
Topical Collection in
Catalysts
Catalytic Conversion of Biomass to Bioenergy
Collection Editors: Sergio Nogales Delgado, Juan Félix González, Simona M. Coman