Molecular Signaling Pathways and Networks on Cancer
Share This Topical Collection
Editor
 Dr. Shihori Tanabe
Dr. Shihori Tanabe
 Dr. Shihori Tanabe
Dr. Shihori Tanabe
E-Mail
Website
Collection Editor
Division of Risk Assessment, Center for Biological Safety and Research, National Institute of Health Sciences, Kawasaki 210-9501, Japan
Interests: molecular network in diseases; signaling pathway in cancer; molecular mechanism in cancer therapy; therapeutic response in cancer
Special Issues, Collections and Topics in MDPI journals
Topical Collection Information
Dear Colleagues,
Dynamic regulation in the activity of molecular signaling pathways and networks is critical in cancer. Cancer signaling in various types and conditions of cancers triggers, progresses, and promotes cancer development. Molecular signaling pathway networks involved in progression, metastasis, drug resistance, and development in cancer are the main focuses in this collection.
In the meantime, specific Adverse Outcome Pathways (AOPs) in cancer are also in the main scope of this collection. Any authors who have been developing AOPs in cancer are greatly welcome to submit their papers.
Dr. Shihori Tanabe
Collection Editor
Manuscript Submission Information
Manuscripts should be submitted online at www.mdpi.com by registering and logging in to this website. Once you are registered, click here to go to the submission form. Manuscripts can be submitted until the deadline. All submissions that pass pre-check are peer-reviewed. Accepted papers will be published continuously in the journal (as soon as accepted) and will be listed together on the collection website. Research articles, review articles as well as short communications are invited. For planned papers, a title and short abstract (about 100 words) can be sent to the Editorial Office for announcement on this website.
Submitted manuscripts should not have been published previously, nor be under consideration for publication elsewhere (except conference proceedings papers). All manuscripts are thoroughly refereed through a single-blind peer-review process. A guide for authors and other relevant information for submission of manuscripts is available on the Instructions for Authors page. Cancers is an international peer-reviewed open access semimonthly journal published by MDPI.
Please visit the Instructions for Authors page before submitting a manuscript.
The Article Processing Charge (APC) for publication in this open access journal is 2900 CHF (Swiss Francs).
Submitted papers should be well formatted and use good English. Authors may use MDPI's
English editing service prior to publication or during author revisions.
Keywords
- molecular signaling pathway
- adverse outcome pathway (AOP)
- signaling pathway network
Published Papers (5 papers)
Open AccessArticle
Advanced Cell Culture Models Illuminate the Interplay between Mammary Tumor Cells and Activated Fibroblasts
by
Martina Del Nero, Alessandro Colombo, Stefania Garbujo, Chiara Baioni, Linda Barbieri, Metello Innocenti, Davide Prosperi, Miriam Colombo and Luisa Fiandra
Cited by 3 | Viewed by 1827
Abstract
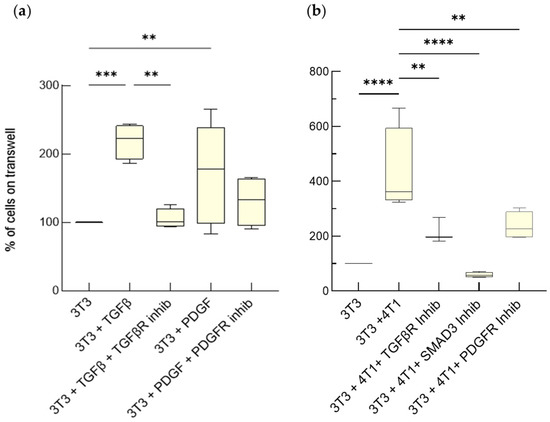
The interaction between tumor cells and activated fibroblasts determines malignant features of desmoplastic carcinomas such as rapid growth, progression towards a metastatic phenotype, and resistance to chemotherapy. On one hand, tumor cells can activate normal fibroblasts and even reprogram them into CAFs through
[...] Read more.
The interaction between tumor cells and activated fibroblasts determines malignant features of desmoplastic carcinomas such as rapid growth, progression towards a metastatic phenotype, and resistance to chemotherapy. On one hand, tumor cells can activate normal fibroblasts and even reprogram them into CAFs through complex mechanisms that also involve soluble factors. Among them, transforming growth factor beta (TGF-β) and Platelet-Derived Growth Factor (PDGF) have an established role in the acquisition of pro-tumorigenic phenotypes by fibroblasts. On the other hand, activated fibroblasts release Interleukin-6 (IL-6), which increases tumor-cell invasiveness and chemoresistance. However, the interplay between breast cancer cells and fibroblasts, as well as the modes of action of TGF-β, PDGF, and IL-6, are difficult to investigate in vivo. Here, we validated the usage of advanced cell culture models as tools to study the interplay between mammary tumor cells and fibroblasts, taking mouse and human triple-negative tumor cells and fibroblasts as a case study. We employed two different settings, one permitting only paracrine signaling, the other both paracrine and cell-contact-based signaling. These co-culture systems allowed us to unmask how TGF-β, PDGF and IL-6 mediate the interplay between mammary tumor cells and fibroblasts. We found that the fibroblasts underwent activation induced by the TGF-β and the PDGF produced by the tumor cells, which increased their proliferation and IL-6 secretion. The IL-6 secreted by activated fibroblasts enhanced tumor-cell proliferation and chemoresistance. These results show that these breast cancer avatars possess an unexpected high level of complexity, which resembles that observed in vivo. As such, advanced co-cultures provide a pathologically relevant tractable system to study the role of the TME in breast cancer progression with a reductionist approach.
Full article
►▼
Show Figures
Open AccessArticle
Procaine Abrogates the Epithelial-Mesenchymal Transition Process through Modulating c-Met Phosphorylation in Hepatocellular Carcinoma
by
Min Hee Yang, Chakrabhavi Dhananjaya Mohan, Amudha Deivasigamani, Arunachalam Chinnathambi, Sulaiman Ali Alharbi, Kanchugarakoppal S. Rangappa, Sang Hoon Jung, Hyejin Ko, Kam Man Hui, Gautam Sethi and Kwang Seok Ahn
Cited by 5 | Viewed by 1787
Abstract
EMT is a critical cellular phenomenon that promotes tumor invasion and metastasis. Procaine is a local anesthetic agent used in oral surgeries and as an inhibitor of DNA methylation in some types of cancers. In this study, we have investigated whether procaine can
[...] Read more.
EMT is a critical cellular phenomenon that promotes tumor invasion and metastasis. Procaine is a local anesthetic agent used in oral surgeries and as an inhibitor of DNA methylation in some types of cancers. In this study, we have investigated whether procaine can inhibit the EMT process in HCC cells and the preclinical model. Procaine suppressed the expression of diverse mesenchymal markers but induced the levels of epithelial markers such as E-cadherin and occludin in HGF-stimulated cells. Procaine also significantly reduced the invasion and migration of HCC cells. Moreover, procaine inhibited HGF-induced c-Met and its downstream oncogenic pathways, such as PI3K/Akt/mTOR and MEK/ERK. Additionally, procaine decreased the tumor burden in the HCC mouse model and abrogated lung metastasis. Overall, our study suggests that procaine may inhibit the EMT process through the modulation of a c-Met signaling pathway.
Full article
►▼
Show Figures
Open AccessArticle
Identification of Src Family Kinases as Potential Therapeutic Targets for Chemotherapy-Resistant Triple Negative Breast Cancer
by
Ishwar N. Kohale, Jia Yu, Yongxian Zhuang, Xiaoyang Fan, Raven J. Reddy, Jason Sinnwell, Krishna R. Kalari, Judy C. Boughey, Jodi M. Carter, Matthew P. Goetz, Liewei Wang and Forest M. White
Cited by 6 | Viewed by 2106
Abstract
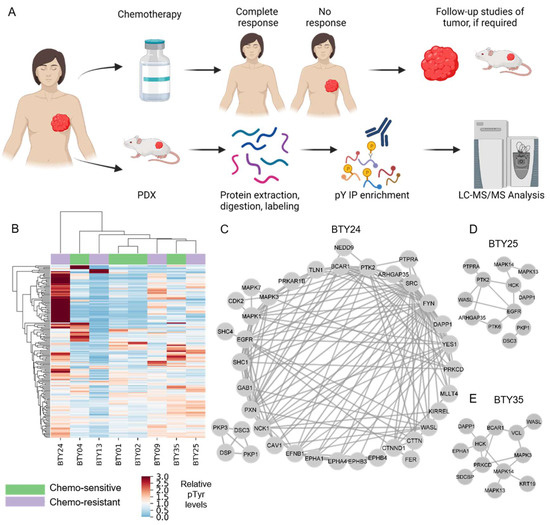
Neoadjuvant chemotherapy (NAC) remains the cornerstone of the treatment for triple negative breast cancer (TNBC), with the goal of complete eradication of disease. However, for patients with residual disease after NAC, recurrence and mortality rates are high and the identification of novel therapeutic
[...] Read more.
Neoadjuvant chemotherapy (NAC) remains the cornerstone of the treatment for triple negative breast cancer (TNBC), with the goal of complete eradication of disease. However, for patients with residual disease after NAC, recurrence and mortality rates are high and the identification of novel therapeutic targets is urgently needed. We quantified tyrosine phosphorylation (pTyr)-mediated signaling networks in chemotherapy sensitive (CS) and resistant (CR) TNBC patient-derived xenografts (PDX), to gain novel therapeutic insights. The antitumor activity of SFK inhibition was examined in vivo. Treated tumors were further subjected to phosphoproteomic and RNAseq analysis, to identify the mechanism of actions of the drug. We identified Src Family Kinases (SFKs) as potential therapeutic targets in CR TNBC PDXs. Treatment with dasatinib, an FDA approved SFK inhibitor, led to inhibition of tumor growth in vivo. Further analysis of post-treatment PDXs revealed multiple mechanisms of actions of the drug, confirming the multi-target inhibition of dasatinib. Analysis of pTyr in tumor specimens suggested a low prevalence of SFK-driven tumors, which may provide insight into prior clinical trial results demonstrating a lack of dasatinib antitumor activity in unselected breast cancer patients. Taken together, these results underscore the importance of pTyr characterization of tumors, in identifying new targets, as well as stratifying patients based on their activated signaling networks for therapeutic options. Our data provide a strong rationale for studying SFK inhibitors in biomarker-selected SFK-driven TNBC.
Full article
►▼
Show Figures
Open AccessArticle
Expression of SARS-CoV-2-Related Surface Proteins in Non-Small-Cell Lung Cancer Patients and the Influence of Standard of Care Therapy
by
Christophe Deben, Maxim Le Compte, Vasiliki Siozopoulou, Hilde Lambrechts, Christophe Hermans, Ho Wa Lau, Manon Huizing, Kevin Lamote, Jeroen M. H. Hendriks, Peter Van Dam, Patrick Pauwels, Evelien L. J. Smits, Marc Peeters and Filip Lardon
Cited by 2 | Viewed by 1814
Abstract
In this study, we aimed to study the expression of SARS-CoV-2-related surface proteins in non-small-cell lung cancer (NSCLC) cells and identify clinicopathological characteristics that are related to increased membranous (m)ACE2 protein expression and soluble (s)ACE2 levels, with a particular focus on standard of
[...] Read more.
In this study, we aimed to study the expression of SARS-CoV-2-related surface proteins in non-small-cell lung cancer (NSCLC) cells and identify clinicopathological characteristics that are related to increased membranous (m)ACE2 protein expression and soluble (s)ACE2 levels, with a particular focus on standard of care (SOC) therapies. ACE2 (
n = 107), TMPRSS2, and FURIN (
n = 38) protein expression was determined by immunohistochemical (IHC) analysis in NSCLC patients. sACE2 levels (
n = 64) were determined in the serum of lung cancer patients collected before, during, or after treatment with SOC therapies. Finally, the TCGA lung adenocarcinoma (LUAD) database was consulted to study the expression of ACE2 in EGFR- and KRAS-mutant samples and ACE2 expression was correlated with EGFR/HER, RAS, BRAF, ROS1, ALK, and MET mRNA expression. Membranous (m)ACE2 was found to be co-expressed with mFURIN and/or mTMPRSS2 in 16% of the NSCLC samples and limited to the adenocarcinoma subtype. TMPRSS2 showed predominantly atypical cytoplasmic expression. mACE2 and sACE2 were more frequently expressed in mutant EGFR patients, but not mutant-KRAS patients. A significant difference was observed in sACE2 for patients treated with targeted therapies, but not for chemo- and immunotherapy. In the TCGA LUAD cohort, ACE2 expression was significantly higher in EGFR-mutant patients and significantly lower in KRAS-mutant patients. Finally, ACE2 expression was positively correlated with ERBB2-4 and ROS1 expression and inversely correlated with KRAS, NRAS, HRAS, and MET mRNA expression. We identified a role for EGFR pathway activation in the expression of mACE2 in NSCLC cells, associated with increased sACE2 levels in patients. Therefore, it is of great interest to study SARS-CoV-2-infected EGFR-mutated NSCLC patients in greater depth in order to obtain a better understanding of how mACE2, sACE2, and SOC TKIs can affect the course of COVID-19.
Full article
►▼
Show Figures
Open AccessArticle
The AKT1E17K Allele Promotes Breast Cancer in Mice
by
Donatella Malanga, Carmelo Laudanna, Teresa Mirante, Fabiana Colelli, Simona Migliozzi, Pietro Zoppoli, Gianluca Santamaria, Luca Roberto, Carmela De Marco, Marzia Scarfò, Donatella Montanaro, Orlando Paciello, Serenella Papparella, Chiara Mignogna, Alfonso Baldi and Giuseppe Viglietto
Cited by 1 | Viewed by 1830
Abstract
The gain-of-function mutation in the pleckstrin homology domain of AKT1 (AKT1E17K) occurs in lung and breast cancer. Through the use of human cellular models and of a AKT1E17K transgenic Cre-inducible murine strain (R26-AKT1E17K mice), we have demonstrated that AKT1E17K is a bona fide
[...] Read more.
The gain-of-function mutation in the pleckstrin homology domain of AKT1 (AKT1E17K) occurs in lung and breast cancer. Through the use of human cellular models and of a AKT1E17K transgenic Cre-inducible murine strain (R26-AKT1E17K mice), we have demonstrated that AKT1E17K is a bona fide oncogene for lung epithelial cells. However, the role of AKT1E17K in breast cancer remains to be determined. Here, we report the generation and the characterization of a MMTV-CRE; R26-AKT1E17K mouse strain that expresses the mutant AKT1E17K allele in the mammary epithelium. We observed that AKT1E17K stimulates the development of mammary tumors classified as ductal adenocarcinoma of medium–high grade and presented a variety of proliferative alterations classified as adenosis with low-to-high grade dysplasia in the mammary epithelium. A subsequent immunohistochemical characterization suggested they were PR
−/HER2
−/ER
+, basal-like and CK8
−/CK10
−/CK5
+/CK14
+. We also observed that, in parallel with an increased proliferation rate, tumors expressing mutant AKT1E17K presented an activation of the GSK3/cyclin D1 pathway in the mammary epithelium and cluster significantly with the human basal-like tumors. In conclusion, we demonstrate AKT1E17K is a bona fide oncogene that can initiate tumors at high efficiency in murine mammary epithelium in vivo.
Full article
►▼
Show Figures