3.1. Salt Rejection
The salt rejection rates for the respective membranes in this study are presented in
Figure 2. At an external pressure of 30 MPa, all the defective membranes exhibited complete salt rejection (100%), but the pristine ZIF-8 membrane did not (98.2%). This observation can be explained by the pristine ZIF-8 (11.5 Å) membrane possessing one of the two largest cavity diameters among all the membranes in this study, which may allow the ions to pass through it and enter the permeate region easily. While the ion rejection rate of the pristine ZIF-8 membrane in our study is lower than the value obtained by Hu et al. [
5], it should be noted that in their research, 0.5 M NaCl was employed with a fully rigid membrane, which can restrict ion movement. Additionally, only Cl
− ions were present in the permeate solution, which suggests that atomic interactions between the ions and the membranes may restrict the movement of Na
+ ions. Such a proposition will be elaborated later.
To study the presence of salts within the ZIF-8 membranes, the density distribution graphs of the individual ions were plotted for the respective ZIF-8 membranes (
Figure 3). With respect to the distribution of Na
+ and Cl
− ions within the membranes, the Na
+ ions were highly concentrated just below the surface of the membrane on the feed side, while the Cl
− ions were relatively uniformly distributed within the membrane. Based on
Figure 3d, Cl
− ions were embedded deep in the linker1 and linker3 membranes, and they were present in the permeate regions of the pristine ZIF-8 membrane, as mentioned above. While a few Cl
− ions were able to enter the membranes, most ions were unable to do so, and they remained outside the membranes in high concentrations due to the size exclusion effect. By comparing the hydrated radii of the ions (Na
+ = 2.76 Å [
39] and Cl
− = 3.3 Å [
40]) to the aperture diameters (
Table S1), one could deduce that the ions can enter the apertures by losing their hydration shells, since the aperture diameters are larger than the ionic radii (Na
+ = 0.95 Å [
39] and Cl
− = 1.81 Å [
41]).
Generally, the lack of ions within the Zn substitutional defective membranes can be attributed to their small pore (aperture and cavity) diameters, which render it difficult for the ions to enter the membranes and occupy their interiors. Therefore, only some of the ions managed to occupy the region directly below the surfaces of the membranes. Despite the linker-defective membranes possessing relatively larger pores, most of the Na+ ions remained on their surfaces. As a corollary, only a few Na+ ions were located directly below the membrane surfaces. Based on the average velocities of the different ions in the ZIF-8 membranes, the Na+ ions were mostly faster than the Cl− ions as a result of their smaller mass and size. Thus, the Na+ ions can move more rapidly than the Cl− ions within the membranes. However, Cl− ions were present deep within the linker1 and linker3 membranes, as mentioned above, which indicates that atomic interactions between the ions and the ZIF membranes may influence ion movement in the membranes.
To further investigate the movement of ions in the different ZIF-8 membranes, the radial distribution functions (RDFs) of the ions–O (oxygen of water), ZIF–O, and ZIF–ion interactions were plotted. The RDF is defined as the probability of finding two atoms separated at a specific distance from each other. Therefore, if the
r values of the ZIF–ion interactions are smaller than those of the ion–O interactions, there will be a high possibility of membrane adsorption due to a greater affinity between the ZIFs and the ions [
20]. In addition, the average interaction energies of the ZIF–ion and ZIF–O interactions throughout the entire simulation were also calculated and plotted [
42]. The more negative an interaction energy value is, the stronger the interaction will be, which corresponds to stronger adsorption of ions and water molecules by the atoms constituting ZIF-8.
The appearance of the first peaks in the RDF of the ion–O interactions (
Figure 4) indicates the formation of the first hydration shell around the ions, and the values of
r for the first peak for the Na
+ and Cl
− ions were reported as 2.05 Å and 2.75 Å, respectively. The first peak height of the Na
+ ions decreases in the following order: Zn2 > Zn3 > pristine > linker1 > linker3 > linker2 >Zn1, while the first peak height of the Cl
− ions decreases in the following order: pristine > Zn2 > linker1 > linker3 > Zn3 > Zn1 > linker2. The hydration level of the Cl
− ions was found to be higher than that of the Na
+ ions, which is in agreement with the results of Dahanayaka et al. [
34] These observations imply that the hydration levels of the Na
+ and Cl
− ions are affected by the type and concentration of defects in the ZIF-8 membrane.
The interactions between the atoms from the ZIF-8 membranes and the ions were explored by calculating their RDFs and the interaction energies between the ions and water molecules with the ZIF atoms. The RDF results of the pristine ZIF-8 membrane indicate that the positively charged Na
+ ions and the negatively charged Cl
− ions interact strongly with the N (
Figure 5a) and Zn (
Figure 5b) atoms, respectively. The interaction energy of the N–Na
+ pairs (
Figure S4a) decreases in magnitude in the following order: Zn2 > pristine > linker3 > Zn1 > linker2 > linker1 > Zn3. This implies that the adsorption of Na
+ ions is the strongest and weakest in the Zn2 and Zn3 membranes, respectively. According to the graph in
Figure S4b, the magnitude of the Zn–Cl
− interaction energy decreases in the following order: linker3 > pristine > Zn2 > Zn1 > linker2 > linker1 > Zn3, with linker3 and Zn3 membranes exhibiting the strongest and weakest affinity toward Cl
− ions, respectively. However, based on the RDF graph in
Figure 5b, aside from the Zn1 and pristine ZIF-8 membranes, the other defective ZIF-8 membranes did not display distinct curves. This could be attributed to the ZIF-8 membranes exhibiting a stronger affinity toward Na
+ than Cl
− ions, which is evident by the Na
+ ions possessing higher RDF peak values and greater interaction energies than the Cl
− ions. Hence, it is possible to dampen ion adsorption on a ZIF-8 membrane by introducing Zn substitutional defects, as evidenced by the Zn3 membrane exhibiting the weakest ion adsorption at its Zn and N sites. However, it is also possible to improve ion adsorption on ZIF-8 membranes by increasing the number of linker defect sites within them.
To explore the possibility of ion adsorption occurring at the defect sites, the RDF curves of the atoms in the defect sites with respect to the solute ions were plotted (
Figure 6 and
Figure 7). Furthermore, the interaction energies between the ions and the atoms were measured (
Figures S4a,b, S5a, S6a, S7a, and S8a). By analyzing Na
+ adsorption at the Zn substitutional defect sites, the first peak height of the H3–Na
+ curve was observed to decrease in the following order: Zn3 > Zn1 > Zn2, with the first RDF peaks of the Zn3 and Zn2 membranes corresponding to a common
r value of 2.65 Å, and the first RDF peak of Zn1 being located at 3.55 Å. Unlike the H3–Na
+ interactions, the N–Na
+ interactions are sufficiently strong enough to overcome the Na
+–O interactions, which indicates that the Zn defects are not involved in Na
+ adsorption. This is also evident from the positive interaction energy values for H3–Na
+ pairs, indicating that no Na
+ adsorption had taken place. Therefore, the H3 atoms’ peak value in the Zn defects can be attributed to the proximity of the H3 atoms to the N atoms. For linker defects, only the O2 atoms in the linker3 membrane showed distinct interactions with Na
+. This is also supported by
Figure S8a, which shows the Na
+ adsorption taking place at the O2 atoms of the three ZIF-8 membranes with linker defects. The lack of well-defined curves in the RDF diagrams of the linker1 and 2 membranes could be due to the huge differences between their interaction energies and that of the linker3 membrane. Meanwhile, both the
r values of O2–Na
+ (
Figure 6b) and N–Na
+ (
Figure 5a) interactions are equal at 1.95 Å. However, their respective interaction energy graphs (
Figures S4a and S8a) reveal that the N–Na
+ pairs exhibit a higher interaction energy than the O2–Na
+ pairs. The stronger N–Na
+ interactions may have also dampened the O2–Na
+ interactions, which may explain the lack of distinct curves in the RDF diagrams of the linker1 and linker2 membranes.
As for Cl
− adsorption at the defect sites, the first peak height of the H3–Cl
− (
Figure 7a) curves in the Zn defective membranes decreases in the following order: Zn2 > Zn3 > Zn1 at 2.35 Å. Such a trend is similar to the pattern exhibited by the interaction energy graphs (
Figure S5a) in which they decreases in the same order. Therefore, there exists an ideal number of Zn defect sites per unit cell for optimal Cl
− adsorption. For the linker defective membranes, only the linker3 membrane exhibits a distinct curve for H3–Cl
− interactions, which suggests that only the H3 atoms within it can adsorb Cl
− ions. However, based on the interaction energy graphs (
Figures S6a and S7a), involving the H3 and H4 atoms, Cl
− adsorption occurred in the cases of the three ZIF-8 membranes with linker defects. The discrepancy between the RDF results and the interaction energy graphs can be attributed to the difference in the interaction energy values between the linker3 membrane and the other two linker defective membranes (linker1 and linker2). Since they correspond to relatively weak Cl
− adsorption, the RDF curves of the linker1 and linker2 membranes do not exhibit distinct peaks (
Figure 7). Therefore, even if Cl
− ions could be potentially adsorbed onto the H3 and H4 atoms of the linker1 and linker2 membranes, such an effect may not be sufficiently strong to overcome the Cl
−–O interactions. Since the
r values of the H3–Cl
− interactions in the defect sites of the Zn2, Zn3, and linker3 membranes are smaller than those of the Cl
−–O interactions, the defect sites of these defective membranes can facilitate Cl
− adsorption.
Based on the interaction energy graphs (
Figures S5a, S6a, S7a, and S8a), all the atoms except the O2 atoms from the linker defect sites can facilitate Cl
− adsorption, while the O2 atoms are capable of can adsorbing Na
+ ions. Although ion adsorption is weaker at the defect sites than at the Zn and N atoms, it is possible to improve ion adsorption in ZIF-8 membranes by introducing linker substitutional defects. In RO applications, since ZIF-8 membranes are constantly in contact with water, more linker defects can be expected to be formed, which in turn enhances ion adsorption on the membranes without involving any additional modifications.
According to the different RDF curves, the ZIF-8 membranes exhibit a higher affinity toward Na+ ions than Cl− ions. As a result of such a relatively high affinity, the Na+ ions could only occupy a few angstroms below the surfaces of the membranes despite being smaller than the Cl− ions. Meanwhile, based on the RDF results, the pristine ZIF-8 membrane has the highest affinity toward Cl− ions among all the membranes. Despite their relatively low Cl− affinity, the defective ZIF-8 membranes may also facilitation Cl− adsorption. However, such weak Cl− ion adsorption implies that while Cl− ions can still be adsorbed onto the defective ZIF-8 membranes, they cannot be trapped within the membranes. Instead, the Cl− ions will pass through these membranes despite their relatively large ionic radius.
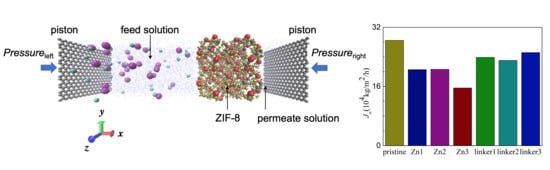
3.2. Water Flux
In RO, water flux across the different ZIF-8 membranes (
Figure 8) is affected by the size of the pore diameters, the activation energy for water permeation, ZIF hydrophilicity, and the ordering of water molecules in the entire system. The water flux values at different pressures in this study are higher than those obtained in the ZIF-8 study by Hu et al. [
5], which can be attributed to Hu et al. employing a rigid ZIF-8 membrane in their study [
5], while this study utilized more flexible membranes.
As shown in
Figure 8, the pristine ZIF-8 membrane corresponds to the highest water flux, followed by the linker substitutional defective membranes (linker3, linker1, and linker2) and the Zn substitutional defective membranes (Zn1, Zn2, and Zn3). As evident from the relationship between the pore diameter and the water flux, large cavities and apertures increase the water flux, as evident in the cases of the pristine ZIF-8 membrane that possesses the largest cavity diameter and exhibit the highest water flux and the linker3 membrane that possesses the largest aperture diameter and exhibit the second-highest water flux. With respect to the type of defects, the ZIF-8 membranes with linker defects generally exhibit the second-highest water flux due to them possessing larger cavities than the Zn defects and the largest aperture diameters collectively. However, even if the cavities or the apertures are large, the water flux will still be low if the other kind of pores are too small. This is evident in the case of the Zn3 membrane (3.52 Å), which despite having a larger aperture diameter than pristine ZIF-8 membrane (3.40 Å), possesses the second smallest cavity diameter (11.0 Å) among the studied ZIF-8 membranes. Based on
Figure 9, the pristine ZIF-8 membrane displays the highest water flux among all the membranes at higher pressure values. Furthermore, under an applied pressure of 60 MPa, the ZIF-8 membranes with linker substitutional defects (linker1, linker2, and linker3) generally exhibit the second-highest water flux after the pristine ZIF-8 membrane.
To explore the temperature dependency of water flux through the membranes, the Arrhenius equation [
43] was invoked. It is defined as:
where
Ea, R, and
T are the apparent activation energy, gas constant, and temperature, respectively. The water flux
JW against the reciprocal of
T is plotted in
Figure 10a, and the linear fitting of the curves provides the apparent activation energy values for water permeation through the membranes. From the gradients of the Arrhenius plots for the respective ZIF-8 membranes, the activation energy value for water permeation decreases in the following order: Zn3 (20.5 kJ/mol) > Zn2 (18.9 kJ/mol) > Zn1 (17.2 kJ/mol) > linker2 (16.0 kJ/mol) > pristine (15.5 kJ/mol) > linker3 (13.6 kJ/mol) > linker1 (12.3 kJ/mol). Positive values of the activation energy indicate that water flux can be improved with a higher feed temperature [
23]. Based on the values of
Ea, the presence of Zn substitutional defects in a ZIF-8 membrane can generally increase the value of
Ea, while it is possible for linker substitutional defects to lower the value of
Ea, with the latter depending on the concentration of linker substitutional defects in the membrane. Therefore, water flux may not improve significantly for ZIF-8 membranes with Zn substitutional defects even by increasing the feed temperature. According to the value of
Ea of the pristine ZIF-8 membrane, its corresponding activation energy is lower than that recorded in the study of Hu et al. (24.4 kJ/mol) [
5], in which used a fully rigid ZIF-8 membrane that hinders water flow was utilized, as mentioned above.
To study the diffusion of water through the membranes, the mean square displacements (MSDs) of the water molecules were evaluated (
Figure 10b). The measured displacements of the water molecules were measured with respect to their equilibrium positions. The linear fitting of an MSD
curve provides the diffusion coefficient
D according to the relation
, where
t is the simulation time. This approximation method is commonly used in MD simulations to obtain the membrane diffusion coefficients [
23]. Based on our results, the value of
D decreases in the following order: linker3 > Zn2 > Zn3 > Zn1 > linker1 > linker2 > pristine, which indicates that water molecules moved more quickly in substitutional defective ZIF-8 membranes than pristine ZIF-8 membranes. However, such an observation appears to contradict the water flux records, which show that the presence of substitutional defects reduces the water flux through a ZIF-8 membrane. Therefore, the obtained diffusion coefficient values suggest that the rate of diffusion does not depend on the size of the cavities or apertures. Instead, it depends on the interactions between the water molecules and the constituent atoms of the ZIF-8 membranes, as described in the subsequent paragraphs.
Another factor that contributed to the reduced water flux in the linker substitutional defect membranes is their enhanced hydrophilicity, which was caused by the loss of hydrophobic organic linkers and the oxygenation of the ZIF-8 membranes. The hydrophilicity of ZIF-8 membranes can be assessed according to their RDFs, which is necessary to analyze the interactions between the membrane atoms and the water molecules. As the Zn atoms in the pristine ZIF-8 membranes exhibit the strongest interactions with the O atoms in the water molecules, the RDFs and the interaction energies of Zn–O atom pairs (
Figure 11a and
Figure S4c) from the different ZIF-8 membranes were compared. The heights of the first peaks of the Zn–O atom pairs at an
r value of 1.95 Å decrease in the following order: pristine > linker1 > Zn1 > linker2 > Zn2 > linker3 > Zn3, while the magnitudes of the interaction energies decrease in the following order: linker3 > linker2 > pristine > linker1 > Zn1 > Zn2 > Zn3. Both of these trends indicate that the presence of Zn defects will weaken the Zn–O interactions, since the weakened Zn–O interactions in the Zn substitutional defects may be attributed to the decreasing number of Zn atoms in the membranes.
Based on the interactions between the O atoms in the water molecules and the atoms within the defects (Zn and linker), RDF curves and interaction energy graphs of these O atoms were plotted (
Figure 12). By analyzing the H3–O interactions in the Zn1, Zn2, and Zn3 membranes (
Figure 12a), the heights of the peaks at an
r value of 2.35 Å decrease in the following order: Zn2 > Zn1 > Zn3. The negative values from the interaction energy graphs (
Figure S5b) indicate that by increasing the number of Zn defect sites, the strength of the H3–O interactions can be improved. However, these interactions are not as strong as the Zn–O interactions, which that Zn substitutional defects could not improve ZIF-8 hydrophilicity.
In a linker substitutional defect, all three atoms (H3, H4, and O2) that replace the missing linker exhibit distinct peaks in their RDFs with respect to the O and H atoms from the water molecules. For the H3–O, H4–O, and O2–H interactions, the first peaks correspond to
r values of 2.35, 2.35, and 1.55 Å, respectively, and their heights all decrease in the following order: linker3 > linker2 > linker1. A similar trend can be observed for the corresponding interaction energy graphs (
Figures S6b, S7b, and S8b), which could be attributed to the defect sites introducing additional interactions. By comparing the
r values of the H3–O, H4–O, and O2–H interactions with those of the O–O (2.75 Å) and Zn–O interactions (1.95 Å), the H3–O and H4–O interactions were determined to be weaker than the Zn–O interactions despite the fact that their
r values are smaller than that of the O–O interactions, which implies that the defect site is hydrophilic in nature. Overall, since the Zn–O interactions correspond to a higher interaction energy than the H3–O, H4–O, and O2–H interactions, the Zn atoms exhibit the strongest interactions with water.
Furthermore, the heights of the first peaks of the O–O curves (
Figure 11b) decrease in the following order: Zn3 > linker2 > Zn1 > Zn2 > linker1 > pristine > linker3, which indicate that the hydrophilicity of a ZIF-8 membrane can be potentially improved by introducing a sufficient amount of linker defects to it. However, such an enhanced hydrophilicity will reduce the water flux through the membrane, as evident from the relatively low water flux through the linker substitutional defective ZIF-8 membranes compared with that of the pristine ZIF-8 membrane. According to the heights of the first peaks of the RDFs pertaining to the interactions between the salt ions and water molecules and the atoms at the defect sites, the H3 atoms at the Zn defect sites (Zn1, Zn2, and Zn3) generally exhibit a higher affinity towards Cl
− ions than water molecules. Meanwhile, the H3, H4, and O2 atoms at the linker1, linker2, and linker3 defect sites generally display a stronger affinity toward water molecules than the salt ions.
According to the interaction energy graphs corresponding to the interactions between the salt ions and water molecules and the membrane atoms at the defect sites, all the energy values pertaining to the membrane atoms and the water molecules are several times higher than those regarding the membrane atoms and the ions. As mentioned previously, this phenomenon is due to the simulation systems containing more water molecules than membrane atoms at the defect sites, and thus interactions between the ZIF-8 atoms and the water molecules occurred much more frequently than the interactions between the ZIF-8 atoms and the salt ions.
The results of this study suggest that the RO seawater desalination performance of the pristine ZIF-8 membrane is superior to that of the defective membranes, which is in good agreement with the study of Zhang et al. [
10], in which even a small number of defects in a ZIF-8 unit cell significantly affected the performance of the corresponding RO desalination membrane. As mentioned previously, linker substitutional defects are more likely to be formed than Zn substitutional defects. Experimental studies regarding the presence of water-based defects have indicated that the presence of defects within a ZIF-8 membrane allows more water molecules to pass through it, which can be attributed to the structural collapse of ZIF-8 unit cells due to the formation of a large number of defect sites in each unit cell. Such structural collapse results in the loss of large ZIF-8 clusters, which in turn creates large voids within the ZIF-8 membranes. In the scanning electron microscopy [
44] images of defective ZIF-8 membranes obtained by Zhang et al. [
9], holes are visible within the membranes on a scale of 2 µm and a magnification of ×10,000. As the duration of the ZIF-8 submersion in water was prolonged, the holes were enlarged. Such an observation indicates that within defective ZIF-8 membranes, whole ZIF-8 unit cells are missing instead of individual Zn atoms and/or linkers. Therefore, the formation of water-based substitutional defects within a ZIF-8 unit cell does not contribute significantly to the increase in the water flux of the corresponding ZIF-8 membrane, but it may improve ZIF-8 hydrophilicity of the membrane if sufficient linker substitutional defect sites exist.