Common Regulators of Lipid Metabolism and Bone Marrow Adiposity in Postmenopausal Women
Abstract
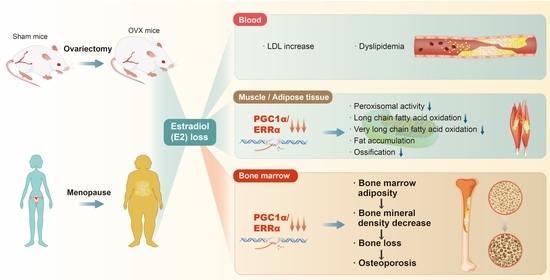
:1. Introduction
2. Abnormal Blood Lipids and Fat Accumulation in Postmenopausal Women
2.1. Blood Lipid Abnormalities in Postmenopausal Women
2.2. Fat Accumulation in Skeletal Muscle and Adipose Tissue in Postmenopausal Women
3. Postmenopausal Bone Marrow Adiposity and Fatty Acid Metabolism Abnormalities
3.1. Basic Roles of Bone Marrow Adipose Tissue and the Loss of E2
3.2. Menopause-Associated Fat Accumulation in Bone Marrow
3.3. Roles of PGC-1α and ERRα in Abnormal Lipid Metabolism in Bones of Postmenopausal Women
3.3.1. PGC-1α
3.3.2. ERRα
3.4. Bone Marrow Adiposity and Bone Loss in Postmenopausal Women
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ko, S.H.; Kim, H.S. Menopause-Associated Lipid Metabolic Disorders and Foods Beneficial for Postmenopausal Women. Nutrients 2020, 12, 102. [Google Scholar] [CrossRef] [Green Version]
- Cervellati, C.; Bergamini, C.M. Oxidative damage and the pathogenesis of menopause related disturbances and diseases. Clin. Chem. Lab. Med. 2016, 54, 739–753. [Google Scholar] [CrossRef] [Green Version]
- Ozbey, N.; Sencer, E.; Molvalilar, S.; Orhan, Y. Body fat distribution and cardiovascular disease risk factors in pre- and postmenopausal obese women with similar. BMI Endocr. J. 2002, 49, 503–509. [Google Scholar] [CrossRef] [Green Version]
- Boldarine, V.T.; Pedroso, A.P.; Brandão-Teles, C.; LoTurco, E.G.; Nascimento, C.M.O.; Oyama, L.M.; Bueno, A.A.; Martins-de-Souza, D.; Ribeiro, E.B. Ovariectomy modifies lipid metabolism of retroperitoneal white fat in rats: A proteomic approach. Am. J. Physiol. Endocrinol. Metab. 2020, 319, e427–e437. [Google Scholar] [CrossRef]
- Yoshida, T.; Takahashi, K.; Yamatani, H.; Takata, K.; Kurachi, H. Impact of surgical menopause on lipid and bone metabolism. Climacteric 2011, 14, 445–452. [Google Scholar] [CrossRef]
- Mogarekar, M.R.; Kulkarni, S.K. Small Dense Low Density Lipoprotein Cholesterol, Paraoxonase 1 and Lipid Profile in Postmenopausal Women: Quality or Quantity? Arch. Med. Res. 2015, 46, 534–538. [Google Scholar] [CrossRef]
- Ko, S.H.; Jung, Y. Energy Metabolism Changes and Dysregulated Lipid Metabolism in Postmenopausal Women. Nutrients 2021, 13, 4556. [Google Scholar] [CrossRef]
- Kamei, Y.; Suzuki, M.; Miyazaki, H.; Tsuboyama-Kasaoka, N.; Wu, J.; Ishimi, Y.; Ezaki, O. Ovariectomy in mice decreases lipid metabolism-related gene expression in adipose tissue and skeletal muscle with increased body fat. J. Nutr. Sci. Vitaminol. 2005, 51, 110–117. [Google Scholar] [CrossRef]
- Bonnelye, E.; Kung, V.; Laplace, C.; Galson, D.L.; Aubin, J.E. Estrogen receptor-related receptor alpha impinges on the estrogen axis in bone: Potential function in osteoporosis. Endocrinology 2002, 143, 3658–3670. [Google Scholar] [CrossRef] [Green Version]
- Yu, B.; Huo, L.; Liu, Y.; Deng, P.; Szymanski, J.; Li, J.; Luo, X.; Hong, C.; Lin, J.; Wang, C.Y. PGC-1α Controls Skeletal Stem Cell Fate and Bone-Fat Balance in Osteoporosis and Skeletal Aging by Inducing TAZ. Cell Stem Cell 2018, 23, 193–209.e195. [Google Scholar] [CrossRef] [Green Version]
- Beekman, K.M.; Veldhuis-Vlug, A.G.; den Heijer, M.; Maas, M.; Oleksik, A.M.; Tanck, M.W.; Ott, S.M.; van′t Hof, R.J.; Lips, P.; Bisschop, P.H.; et al. The effect of raloxifene on bone marrow adipose tissue and bone turnover in postmenopausal women with osteoporosis. Bone 2019, 118, 62–68. [Google Scholar] [CrossRef]
- Attané, C.; Estève, D.; Chaoui, K.; Iacovoni, J.S.; Corre, J.; Moutahir, M.; Valet, P.; Schiltz, O.; Reina, N.; Muller, C. Human Bone Marrow Is Comprised of Adipocytes with Specific Lipid Metabolism. Cell Rep. 2020, 30, 949–958.e946. [Google Scholar] [CrossRef] [Green Version]
- Li, S.; Jiang, H.; Wang, B.; Gu, M.; Zhang, N.; Liang, W.; Wang, Y. Effect of Leptin on Marrow Adiposity in Ovariectomized Rabbits Assessed by Proton Magnetic Resonance Spectroscopy. J. Comput. Assist. Tomogr. 2018, 42, 588–593. [Google Scholar] [CrossRef]
- Elbaz, A.; Rivas, D.; Duque, G. Effect of estrogens on bone marrow adipogenesis and Sirt1 in aging C57BL/6J mice. Biogerontology 2009, 10, 747–755. [Google Scholar] [CrossRef]
- Limonard, E.J.; Veldhuis-Vlug, A.G.; van Dussen, L.; Runge, J.H.; Tanck, M.W.; Endert, E.; Heijboer, A.C.; Fliers, E.; Hollak, C.E.; Akkerman, E.M.; et al. Short-Term Effect of Estrogen on Human Bone Marrow Fat. J. Bone Miner. Res. 2015, 30, 2058–2066. [Google Scholar] [CrossRef]
- Wong, A.K.; Chandrakumar, A.; Whyte, R.; Reitsma, S.; Gillick, H.; Pokhoy, A.; Papaioannou, A.; Adachi, J.D. Bone Marrow and Muscle Fat Infiltration Are Correlated among Postmenopausal Women With Osteoporosis: The AMBERS Cohort Study. J. Bone Miner. Res. 2020, 35, 516–527. [Google Scholar] [CrossRef]
- Milišić, L.; Vegar-Zubović, S.; Valjevac, A. Bone marrow adiposity is inversely associated with bone mineral density in postmenopausal females. Med. Glas. 2020, 17, 15–21. [Google Scholar] [CrossRef]
- Justesen, J.; Stenderup, K.; Ebbesen, E.N.; Mosekilde, L.; Steiniche, T.; Kassem, M. Adipocyte tissue volume in bone marrow is increased with aging and in patients with osteoporosis. Biogerontology 2001, 2, 165–171. [Google Scholar] [CrossRef]
- Landgren, B.-M.; Collins, A.; Csemiczky, G.; Burger, H.G.; Baksheev, L.; Robertson, D.M. Menopause Transition: Annual Changes in Serum Hormonal Patterns over the Menstrual Cycle in Women during a Nine-Year Period Prior to Menopause. J. Clin. Endocrinol. Metab. 2004, 89, 2763–2769. [Google Scholar] [CrossRef]
- Maglione, A.; Rolla, S.; Mercanti, S.F.D.; Cutrupi, S.; Clerico, M. The Adaptive Immune System in Multiple Sclerosis: An Estrogen-Mediated Point of View. Cells 2019, 8, 1280. [Google Scholar]
- Wei, X.; Zhang, Y.; Sun, C.; Qi, B.; Huang, X.; Chen, M.; Liu, N.; Sun, K.; Chen, X.; Qin, X.; et al. Elucidating the relationship between dyslipidemia and osteoporosis: A multicenter, prospective cohort study protocol. Front. Cardiovasc. Med. 2022, 9, 901786. [Google Scholar] [CrossRef]
- Mumusoglu, S.; Yildiz, B.O. Metabolic Syndrome During Menopause. Curr. Vasc. Pharmacol. 2019, 17, 595–603. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, Y.; Liu, L.; Cui, H. Ovariectomy induces abdominal fat accumulation by improving gonadotropin-releasing hormone secretion in mouse. Biochem. Biophys. Res. Commun. 2022, 588, 111–117. [Google Scholar] [CrossRef]
- Yeh, J.H.; Tung, Y.T.; Yeh, Y.S.; Chien, Y.W. Effects of Dietary Fatty Acid Composition on Lipid Metabolism and Body Fat Accumulation in Ovariectomized Rats. Nutrients 2021, 13, 2022. [Google Scholar] [CrossRef]
- Toda, K.; Takeda, K.; Akira, S.; Saibara, T.; Okada, T.; Onishi, S.; Shizuta, Y. Alternations in hepatic expression of fatty-acid metabolizing enzymes in ArKO mice and their reversal by the treatment with 17beta-estradiol or a peroxisome proliferator. J. Steroid Biochem. Mol. Biol. 2001, 79, 11–17. [Google Scholar] [CrossRef]
- Panghiyangani, R.; Soeharso, P.; Andrijono; Suryandari, D.A.; Wiweko, B.; Kurniati, M.; Pujianto, D.A. CYP19A1 Gene Expression in Patients with Polycystic Ovarian Syndrome. J. Hum. Reprod. Sci. 2020, 13, 100–103. [Google Scholar] [CrossRef]
- Jones, M.E.; Thorburn, A.W.; Britt, K.L.; Hewitt, K.N.; Wreford, N.G.; Proietto, J.; Oz, O.K.; Leury, B.J.; Robertson, K.M.; Yao, S.; et al. Aromatase-deficient (ArKO) mice have a phenotype of increased adiposity. Proc. Natl. Acad. Sci. USA 2000, 97, 12735–12740. [Google Scholar] [CrossRef] [Green Version]
- Moise, A.R.; Kuksa, V.; Imanishi, Y.; Palczewski, K. Identification of all-trans-retinol:all-trans-13,14-dihydroretinol saturase. J. Biol. Chem. 2004, 279, 50230–50242. [Google Scholar] [CrossRef] [Green Version]
- Lindberg, M.R.; Lamps, L.W. Bone Marrow. Diagnostic Pathology: Normal Histology, 2nd ed.; Lindberg, M.R., Lamps, L.W., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 130–137. [Google Scholar]
- Monga, I.; Kaur, K.; Dhanda, S.K. Revisiting hematopoiesis: Applications of the bulk and single-cell transcriptomics dissecting transcriptional heterogeneity in hematopoietic stem cells. Brief. Funct. Genom. 2022, 21, 159–176. [Google Scholar] [CrossRef]
- Comazzetto, S.; Shen, B.; Morrison, S.J. Niches that regulate stem cells and hematopoiesis in adult bone marrow. Dev. Cell 2021, 56, 1848–1860. [Google Scholar] [CrossRef]
- Hindorf, C.; Glatting, G.; Chiesa, C.; Lindén, O.; Flux, G. EANM Dosimetry Committee guidelines for bone marrow and whole-body dosimetry. Eur. J. Nucl. Med. Mol. Imaging 2010, 37, 1238–1250. [Google Scholar] [CrossRef]
- Scheller, E.L.; Cawthorn, W.P.; Burr, A.A.; Horowitz, M.C.; MacDougald, O.A. Marrow Adipose Tissue: Trimming the Fat. Trends Endocrinol. Metab. 2016, 27, 392–403. [Google Scholar] [CrossRef] [Green Version]
- Rharass, T.; Lucas, S. Mechanisms in endocrinology: Bone marrow adiposity and bone, a bad romance? Eur. J. Endocrinol. 2018, 179, R165–R182. [Google Scholar] [CrossRef] [Green Version]
- Pierce, J.L.; Begun, D.L.; Westendorf, J.J.; McGee-Lawrence, M.E. Defining osteoblast and adipocyte lineages in the bone marrow. Bone 2019, 118, 2–7. [Google Scholar] [CrossRef]
- Li, J.; Liu, X.; Zuo, B.; Zhang, L. The Role of Bone Marrow Microenvironment in Governing the Balance between Osteoblastogenesis and Adipogenesis. Aging Dis. 2016, 7, 514–525. [Google Scholar] [CrossRef] [Green Version]
- Onji, M.; Werschler, N.; Penninger, J. A critical relationship between bone and fat: The role of bone marrow adipose-derived RANKL in bone metabolism. EMBO Rep. 2021, 22, e52986. [Google Scholar] [CrossRef]
- Woods, G.N.; Ewing, S.K.; Sigurdsson, S.; Kado, D.M.; Eiriksdottir, G.; Gudnason, V.; Hue, T.F.; Lang, T.F.; Vittinghoff, E.; Harris, T.B.; et al. Greater Bone Marrow Adiposity Predicts Bone Loss in Older Women. J. Bone Miner. Res. 2020, 35, 326–332. [Google Scholar] [CrossRef]
- Griffith, J.F.; Yeung, D.K.; Ma, H.T.; Leung, J.C.; Kwok, T.C.; Leung, P.C. Bone marrow fat content in the elderly: A reversal of sex difference seen in younger subjects. J. Magn. Reson. Imaging 2012, 36, 225–230. [Google Scholar] [CrossRef]
- Beekman, K.M.; Zwaagstra, M.; Veldhuis-Vlug, A.G.; van Essen, H.W.; den Heijer, M.; Maas, M.; Kerckhofs, G.; Parac-Vogt, T.N.; Bisschop, P.H.; Bravenboer, N. Ovariectomy increases RANKL protein expression in bone marrow adipocytes of C3H/HeJ mice. Am. J. Physiol. Endocrinol. Metab. 2019, 317, e1050–e1054. [Google Scholar] [CrossRef]
- Martin, R.B.; Zissimos, S.L. Relationships between marrow fat and bone turnover in ovariectomized and intact rats. Bone 1991, 12, 123–131. [Google Scholar] [CrossRef]
- Marie, J.C.; Bonnelye, E. Effects of Estrogens on Osteoimmunology: A Role in Bone Metastasis. Front. Immunol. 2022, 13, 899104. [Google Scholar] [CrossRef]
- Bukowska, J.; Frazier, T.; Smith, S.; Brown, T.; Bender, R.; McCarthy, M.; Wu, X.; Bunnell, B.A.; Gimble, J.M. Bone Marrow Adipocyte Developmental Origin and Biology. Curr. Osteoporos. Rep. 2018, 16, 312–319. [Google Scholar] [CrossRef]
- Khalid, A.B.; Krum, S.A. Estrogen receptors alpha and beta in bone. Bone 2016, 87, 130–135. [Google Scholar] [CrossRef] [Green Version]
- Okazaki, R.; Inoue, D.; Shibata, M.; Saika, M.; Kido, S.; Ooka, H.; Tomiyama, H.; Sakamoto, Y.; Matsumoto, T. Estrogen Promotes Early Osteoblast Differentiation and Inhibits Adipocyte Differentiation in Mouse Bone Marrow Stromal Cell Lines that Express Estrogen Receptor (ER) α or β. Endocrinology 2002, 143, 2349–2356. [Google Scholar] [CrossRef]
- Kumar, A.; Ruan, M.; Clifton, K.; Syed, F.; Khosla, S.; Oursler, M.J. TGF-β Mediates Suppression of Adipogenesis by Estradiol through Connective Tissue Growth Factor Induction. Endocrinology 2012, 153, 254–263. [Google Scholar] [CrossRef] [Green Version]
- Zhao, J.W.; Gao, Z.L.; Mei, H.; Li, Y.L.; Wang, Y. Differentiation of human mesenchymal stem cells: The potential mechanism for estrogen-induced preferential osteoblast versus adipocyte differentiation. Am. J. Med. Sci. 2011, 341, 460–468. [Google Scholar] [CrossRef]
- Rooney, A.M.; van der Meulen, M.C.H. Mouse models to evaluate the role of estrogen receptor α in skeletal maintenance and adaptation. Ann. N. Y. Acad. Sci. 2017, 1410, 85–92. [Google Scholar] [CrossRef]
- Gavin, K.M.; Sullivan, T.M.; Kohrt, W.M.; Majka, S.M.; Klemm, D.J. Ovarian Hormones Regulate the Production of Adipocytes From Bone Marrow-Derived Cells. Front. Endocrinol. 2018, 9, 276. [Google Scholar] [CrossRef] [Green Version]
- Liu, P.; Ji, Y.; Yuen, T.; Rendina-Ruedy, E.; DeMambro, V.E.; Dhawan, S.; Abu-Amer, W.; Izadmehr, S.; Zhou, B.; Shin, A.C.; et al. Blocking FSH induces thermogenic adipose tissue and reduces body fat. Nature 2017, 546, 107–112. [Google Scholar] [CrossRef]
- Veldhuis-Vlug, A.G.; Woods, G.N.; Sigurdsson, S.; Ewing, S.K.; Le, P.T.; Hue, T.F.; Vittinghoff, E.; Xu, K.; Gudnason, V.; Sigurdsson, G.; et al. Serum FSH Is Associated With BMD, Bone Marrow Adiposity, and Body Composition in the AGES-Reykjavik Study of Older Adults. J. Clin. Endocrinol. Metab. 2021, 106, e1156–e1169. [Google Scholar] [CrossRef]
- Mirza, F.S.; Padhi, I.D.; Raisz, L.G.; Lorenzo, J.A. Serum sclerostin levels negatively correlate with parathyroid hormone levels and free estrogen index in postmenopausal women. J. Clin. Endocrinol. Metab. 2010, 95, 1991–1997. [Google Scholar] [CrossRef] [Green Version]
- Mödder, U.I.; Clowes, J.A.; Hoey, K.; Peterson, J.M.; McCready, L.; Oursler, M.J.; Riggs, B.L.; Khosla, S. Regulation of circulating sclerostin levels by sex steroids in women and in men. J. Bone Miner. Res. 2011, 26, 27–34. [Google Scholar] [CrossRef]
- Drake, M.T.; Khosla, S. Hormonal and systemic regulation of sclerostin. Bone 2017, 96, 8–17. [Google Scholar] [CrossRef] [Green Version]
- Maeda, K.; Kobayashi, Y.; Koide, M.; Uehara, S.; Okamoto, M.; Ishihara, A.; Kayama, T.; Saito, M.; Marumo, K. The Regulation of Bone Metabolism and Disorders by Wnt Signaling. Int. J. Mol. Sci. 2019, 20, 5525. [Google Scholar] [CrossRef] [Green Version]
- Ueland, T.; Stilgren, L.; Bollerslev, J. Bone Matrix Levels of Dickkopf and Sclerostin are Positively Correlated with Bone Mass and Strength in Postmenopausal Osteoporosis. Int. J. Mol. Sci. 2019, 20, 2896. [Google Scholar] [CrossRef] [Green Version]
- Fairfield, H.; Falank, C.; Harris, E.; Demambro, V.; McDonald, M.; Pettitt, J.A.; Mohanty, S.T.; Croucher, P.; Kramer, I.; Kneissel, M.; et al. The skeletal cell-derived molecule sclerostin drives bone marrow adipogenesis. J. Cell. Physiol. 2018, 233, 1156–1167. [Google Scholar] [CrossRef]
- Liang, H.; Ward, W.F. PGC-1alpha: A key regulator of energy metabolism. Adv. Physiol. Educ. 2006, 30, 145–151. [Google Scholar] [CrossRef]
- Villena, J.A. New insights into PGC-1 coactivators: Redefining their role in the regulation of mitochondrial function and beyond. FEBS J. 2015, 282, 647–672. [Google Scholar] [CrossRef]
- Rius-Pérez, S.; Torres-Cuevas, I.; Millán, I.; Ortega, Á.L.; Pérez, S. PGC-1α, Inflammation, and Oxidative Stress: An Integrative View in Metabolism. Oxidative Med. Cell. Longev. 2020, 2020, 1452696. [Google Scholar] [CrossRef] [Green Version]
- Fahed, G.; Aoun, L.; Bou Zerdan, M.; Allam, S.; Bou Zerdan, M.; Bouferraa, Y.; Assi, H.I. Metabolic Syndrome: Updates on Pathophysiology and Management in 2021. Int. J. Mol. Sci. 2022, 23, 786. [Google Scholar] [CrossRef]
- Huang, T.Y.; Zheng, D.; Houmard, J.A.; Brault, J.J.; Hickner, R.C.; Cortright, R.N. Overexpression of PGC-1α increases peroxisomal activity and mitochondrial fatty acid oxidation in human primary myotubes. Am. J. Physiol. Endocrinol. Metab. 2017, 312, e253–e263. [Google Scholar] [CrossRef] [Green Version]
- Uldry, M.; Yang, W.; St-Pierre, J.; Lin, J.; Seale, P.; Spiegelman, B.M. Complementary action of the PGC-1 coactivators in mitochondrial biogenesis and brown fat differentiation. Cell Metab. 2006, 3, 333–341. [Google Scholar] [CrossRef] [Green Version]
- Kong, S.; Cai, B.; Nie, Q. PGC-1α affects skeletal muscle and adipose tissue development by regulating mitochondrial biogenesis. Mol. Genet. Genom. Med. 2022, 297, 621–633. [Google Scholar] [CrossRef]
- Schreiber, S.N.; Emter, R.; Hock, M.B.; Knutti, D.; Cardenas, J.; Podvinec, M.; Oakeley, E.J.; Kralli, A. The estrogen-related receptor alpha (ERRalpha) functions in PPARgamma coactivator 1alpha (PGC-1alpha)-induced mitochondrial biogenesis. Proc. Natl. Acad. Sci. USA 2004, 101, 6472–6477. [Google Scholar] [CrossRef] [Green Version]
- Calvo, J.A.; Daniels, T.G.; Wang, X.; Paul, A.; Lin, J.; Spiegelman, B.M.; Stevenson, S.C.; Rangwala, S.M. Muscle-specific expression of PPARgamma coactivator-1alpha improves exercise performance and increases peak oxygen uptake. J. Appl. Physiol. 2008, 104, 1304–1312. [Google Scholar] [CrossRef]
- Kushwaha, P.; Wolfgang, M.J.; Riddle, R.C. Fatty acid metabolism by the osteoblast. Bone 2018, 115, 8–14. [Google Scholar] [CrossRef]
- Bagattin, A.; Hugendubler, L.; Mueller, E. Transcriptional coactivator PGC-1alpha promotes peroxisomal remodeling and biogenesis. Proc. Natl. Acad. Sci. USA 2010, 107, 20376–20381. [Google Scholar] [CrossRef] [Green Version]
- Kushwaha, P.; Alekos, N.S.; Kim, S.P.; Li, Z.; Wolfgang, M.J.; Riddle, R.C. Mitochondrial fatty acid β-oxidation is important for normal osteoclast formation in growing female mice. Front. Physiol. 2022, 13, 997358. [Google Scholar] [CrossRef]
- Kim, S.P.; Li, Z.; Zoch, M.L.; Frey, J.L.; Bowman, C.E.; Kushwaha, P.; Ryan, K.A.; Goh, B.C.; Scafidi, S.; Pickett, J.E.; et al. Fatty acid oxidation by the osteoblast is required for normal bone acquisition in a sex- and diet-dependent manner. JCI Insight 2017, 2, e92704. [Google Scholar] [CrossRef] [Green Version]
- Feng, C.; Xu, Z.; Tang, X.; Cao, H.; Zhang, G.; Tan, J. Estrogen-Related Receptor α: A Significant Regulator and Promising Target in Bone Homeostasis and Bone Metastasis. Molecules 2022, 27, 3976. [Google Scholar] [CrossRef]
- Tripathi, M.; Yen, P.M.; Singh, B.K. Estrogen-Related Receptor Alpha: An Under-Appreciated Potential Target for the Treatment of Metabolic Diseases. Int. J. Mol. Sci. 2020, 21, 1645. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Liu, Z.; Gou, Y.; Yu, H.; Siminelakis, S.; Wang, S.; Kong, D.; Zhou, Y.; Liu, Z.; Ding, Y.; et al. Estradiol mediates vasculoprotection via ERRα-dependent regulation of lipid and ROS metabolism in the endothelium. J. Mol. Cell. Cardiol. 2015, 87, 92–101. [Google Scholar] [CrossRef]
- Yamatani, H.; Takahashi, K.; Yoshida, T.; Soga, T.; Kurachi, H. Differences in the fatty acid metabolism of visceral adipose tissue in postmenopausal women. Menopause 2014, 21, 170–176. [Google Scholar] [CrossRef]
- Bonnelye, E.; Vanacker, J.M.; Dittmar, T.; Begue, A.; Desbiens, X.; Denhardt, D.T.; Aubin, J.E.; Laudet, V.; Fournier, B. The ERR-1 orphan receptor is a transcriptional activator expressed during bone development. Mol. Endocrinol. 1997, 11, 905–916. [Google Scholar] [CrossRef]
- Yoshitake, H.; Rittling, S.R.; Denhardt, D.T.; Noda, M. Osteopontin-deficient mice are resistant to ovariectomy-induced bone resorption. Proc. Natl. Acad. Sci. USA 1999, 96, 8156–8160. [Google Scholar] [CrossRef] [Green Version]
- Laflamme, N.; Giroux, S.; Loredo-Osti, J.C.; Elfassihi, L.; Dodin, S.; Blanchet, C.; Morgan, K.; Giguère, V.; Rousseau, F. A frequent regulatory variant of the estrogen-related receptor alpha gene associated with BMD in French-Canadian premenopausal women. J. Bone Miner. Res. 2005, 20, 938–944. [Google Scholar] [CrossRef]
- Liu, D.; Zhang, Z.; Gladwell, W.; Teng, C.T. Estrogen stimulates estrogen-related receptor alpha gene expression through conserved hormone response elements. Endocrinology 2003, 144, 4894–4904. [Google Scholar] [CrossRef]
- Bonnelye, E.; Merdad, L.; Kung, V.; Aubin, J.E. The orphan nuclear estrogen receptor-related receptor alpha (ERRalpha) is expressed throughout osteoblast differentiation and regulates bone formation in vitro. J. Cell Biol. 2001, 153, 971–984. [Google Scholar] [CrossRef]
- Cai, C.; Yuan, G.J.; Huang, Y.; Yang, N.; Chen, X.; Wen, L.; Wang, X.; Zhang, L.; Ding, Y. Estrogen-related receptor α is involved in the osteogenic differentiation of mesenchymal stem cells isolated from human periodontal ligaments. Int. J. Mol. Med. 2013, 31, 1195–1201. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.; Wang, J. Estrogen-related receptor alpha interacts cooperatively with peroxisome proliferator-activated receptor-gamma coactivator-1alpha to regulate osteocalcin gene expression. Cell Biol. Int. 2013, 37, 1259–1265. [Google Scholar] [CrossRef]
- Wang, J.S.; Mazur, C.M.; Wein, M.N. Sclerostin and Osteocalcin: Candidate Bone-Produced Hormones. Front. Endocrinol. 2021, 12, 584147. [Google Scholar] [CrossRef]
- Raehtz, S.; Bierhalter, H.; Schoenherr, D.; Parameswaran, N.; McCabe, L.R. Estrogen Deficiency Exacerbates Type 1 Diabetes-Induced Bone TNF-α Expression and Osteoporosis in Female Mice. Endocrinology 2017, 158, 2086–2101. [Google Scholar] [CrossRef] [Green Version]
- Gao, B.; Huang, Q.; Lin, Y.S.; Wei, B.Y.; Guo, Y.S.; Sun, Z.; Wang, L.; Fan, J.; Zhang, H.Y.; Han, Y.H.; et al. Dose-dependent effect of estrogen suppresses the osteo-adipogenic transdifferentiation of osteoblasts via canonical Wnt signaling pathway. PLoS ONE 2014, 9, e99137. [Google Scholar] [CrossRef] [Green Version]
- Walker, V.R.; Korach, K.S. Estrogen receptor knockout mice as a model for endocrine research. ILAR J. 2004, 45, 455–461. [Google Scholar] [CrossRef] [Green Version]
- Wend, K.; Wend, P.; Drew, B.G.; Hevener, A.L.; Miranda-Carboni, G.A.; Krum, S.A. ERα regulates lipid metabolism in bone through ATGL and perilipin. J. Cell Biochem. 2013, 114, 1306–1314. [Google Scholar] [CrossRef] [Green Version]
- Kenkre, J.S.; Bassett, J. The bone remodelling cycle. Ann. Clin. Biochem. 2018, 55, 308–327. [Google Scholar] [CrossRef]
- Krishnan, V.; Bryant, H.U.; Macdougald, O.A. Regulation of bone mass by Wnt signaling. J. Clin. Investig. 2006, 116, 1202–1209. [Google Scholar] [CrossRef]
- Moorer, M.C.; Riddle, R.C. Regulation of Osteoblast Metabolism by Wnt Signaling. Endocrinol. Metab. 2018, 33, 318–330. [Google Scholar] [CrossRef]
- Suthon, S.; Lin, J.; Perkins, R.S.; Crockarell, J.R.; Miranda-Carboni, G.A.; Krum, S.A. Estrogen receptor alpha and NFATc1 bind to a bone mineral density-associated SNP to repress WNT5B in osteoblasts. Am. J. Hum. Genet. 2022, 109, 97–115. [Google Scholar] [CrossRef]
- Schwartz, A.V.; Sigurdsson, S.; Hue, T.F.; Lang, T.F.; Harris, T.B.; Rosen, C.J.; Vittinghoff, E.; Siggeirsdottir, K.; Sigurdsson, G.; Oskarsdottir, D.; et al. Vertebral bone marrow fat associated with lower trabecular BMD and prevalent vertebral fracture in older adults. J. Clin. Endocrinol. Metab. 2013, 98, 2294–2300. [Google Scholar] [CrossRef]
- Shen, W.; Chen, J.; Gantz, M.; Punyanitya, M.; Heymsfield, S.B.; Gallagher, D.; Albu, J.; Engelson, E.; Kotler, D.; Pi-Sunyer, X.; et al. MRI-measured pelvic bone marrow adipose tissue is inversely related to DXA-measured bone mineral in younger and older adults. Eur. J. Clin. Nutr. 2012, 66, 983–988. [Google Scholar] [CrossRef] [Green Version]
- Ali, D.; Tencerova, M.; Figeac, F.; Kassem, M.; Jafari, A. The pathophysiology of osteoporosis in obesity and type 2 diabetes in aging women and men: The mechanisms and roles of increased bone marrow adiposity. Front. Endocrinol. 2022, 13, 981487. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, D.-Y.; Ko, S.-H. Common Regulators of Lipid Metabolism and Bone Marrow Adiposity in Postmenopausal Women. Pharmaceuticals 2023, 16, 322. https://0-doi-org.brum.beds.ac.uk/10.3390/ph16020322
Kim D-Y, Ko S-H. Common Regulators of Lipid Metabolism and Bone Marrow Adiposity in Postmenopausal Women. Pharmaceuticals. 2023; 16(2):322. https://0-doi-org.brum.beds.ac.uk/10.3390/ph16020322
Chicago/Turabian StyleKim, Dae-Yong, and Seong-Hee Ko. 2023. "Common Regulators of Lipid Metabolism and Bone Marrow Adiposity in Postmenopausal Women" Pharmaceuticals 16, no. 2: 322. https://0-doi-org.brum.beds.ac.uk/10.3390/ph16020322