Tremorgenic Mycotoxins: Structure Diversity and Biological Activity
Abstract
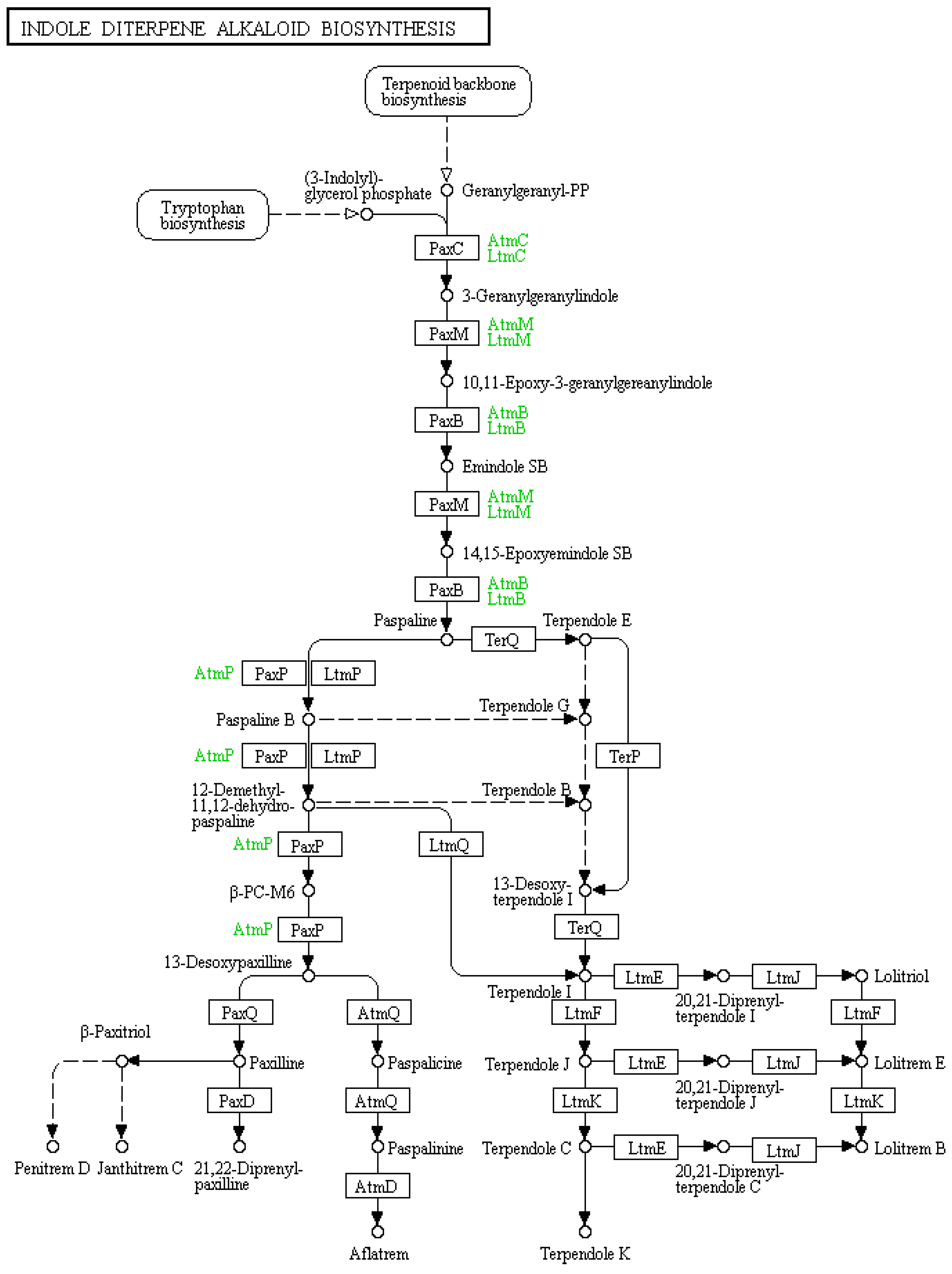
:1. Introduction
2. Reported Animal Toxicity for Indole-Diterpenes
2.1. Classes of Indole-Diterpenes and Their Reported Activities
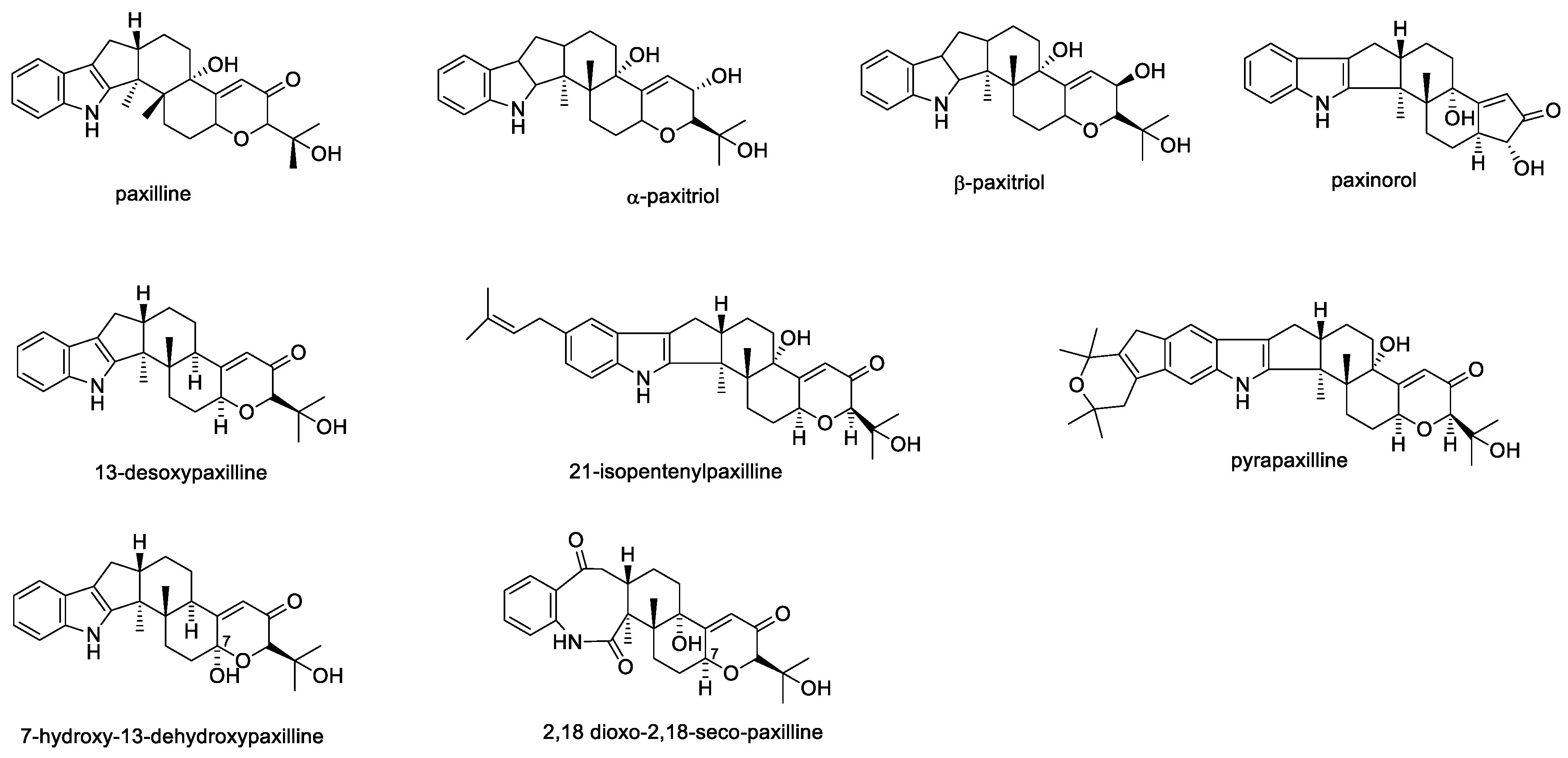
2.1.1. Paxilline
2.1.2. Lolitrems
2.1.3. Penitrems
2.1.4. Paspaline
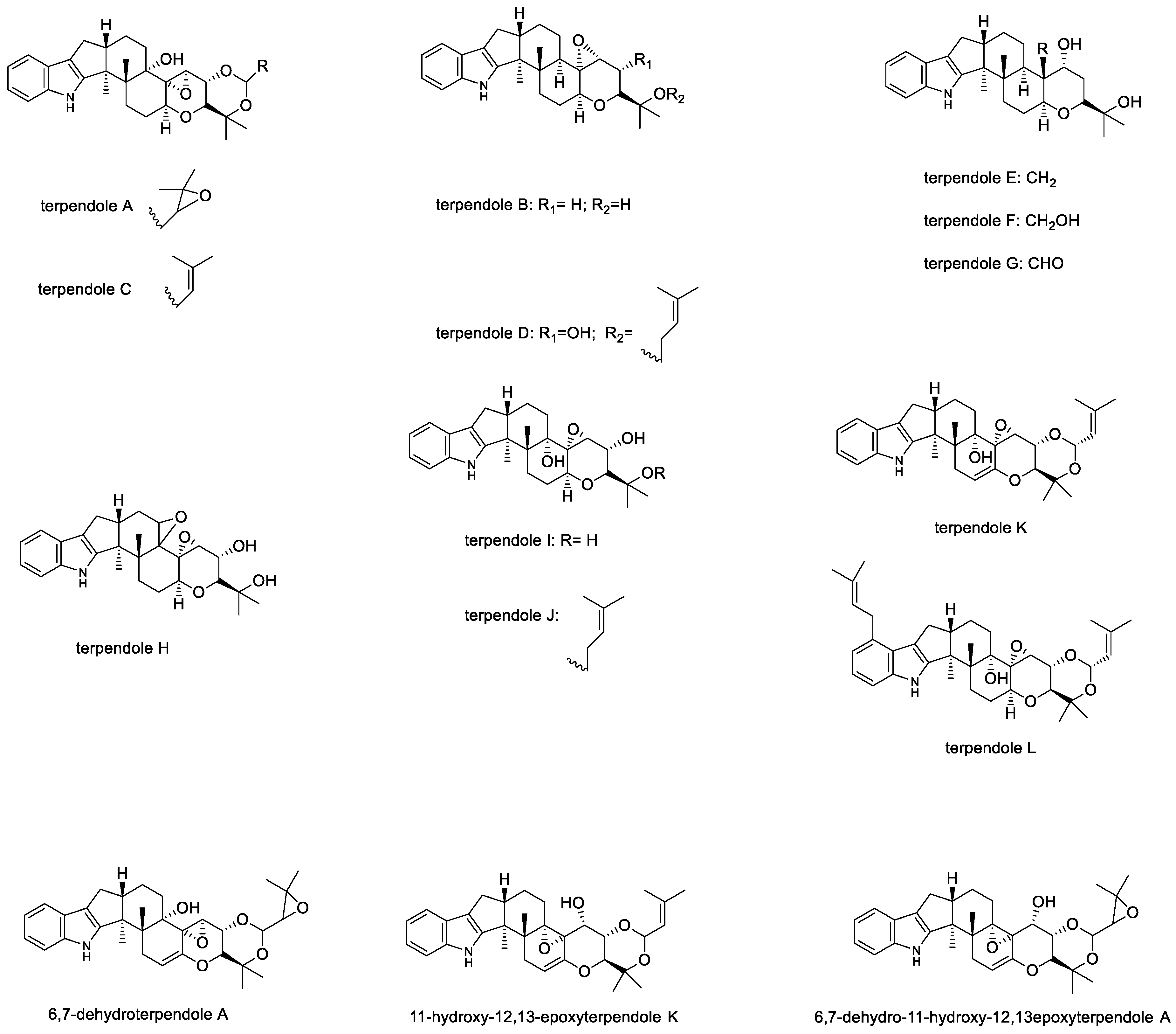
2.1.5. Terpendoles
2.1.6. Sulpinines
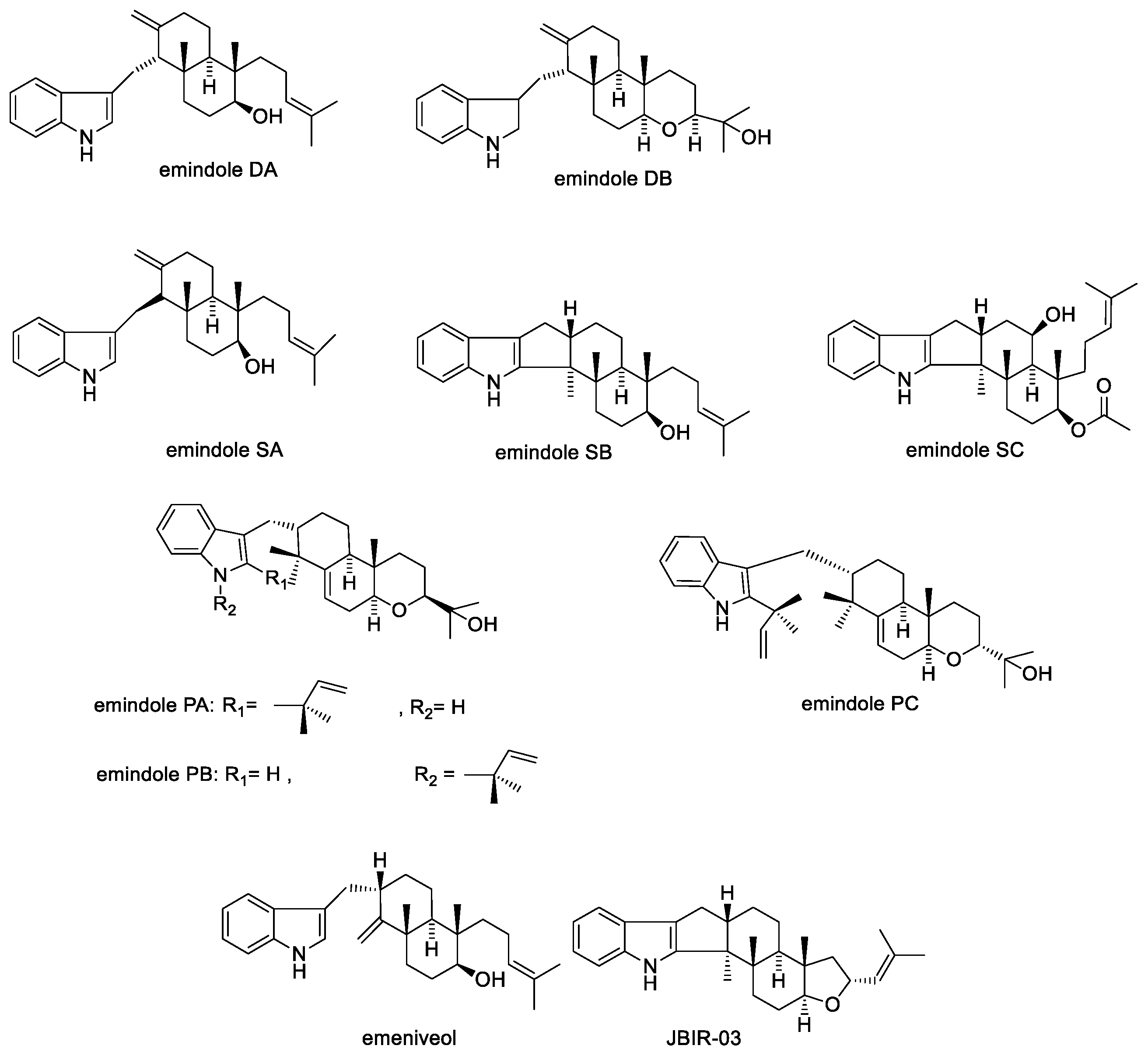
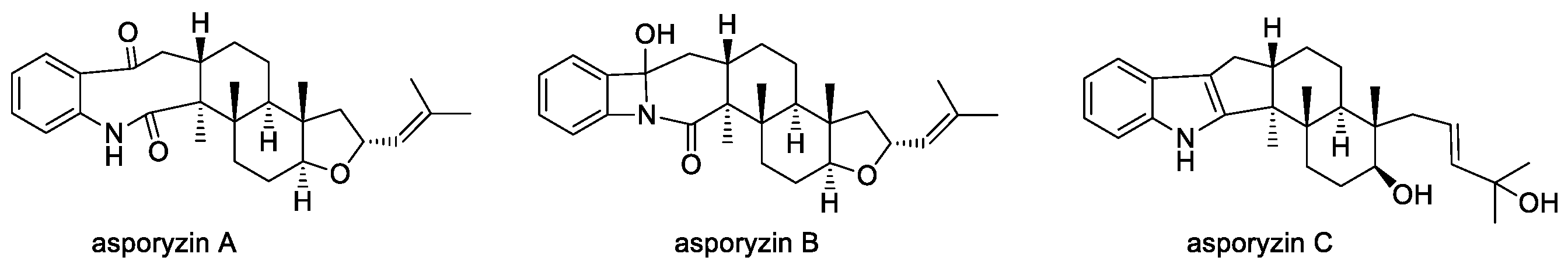
2.1.7. Emindoles and Asporyzins
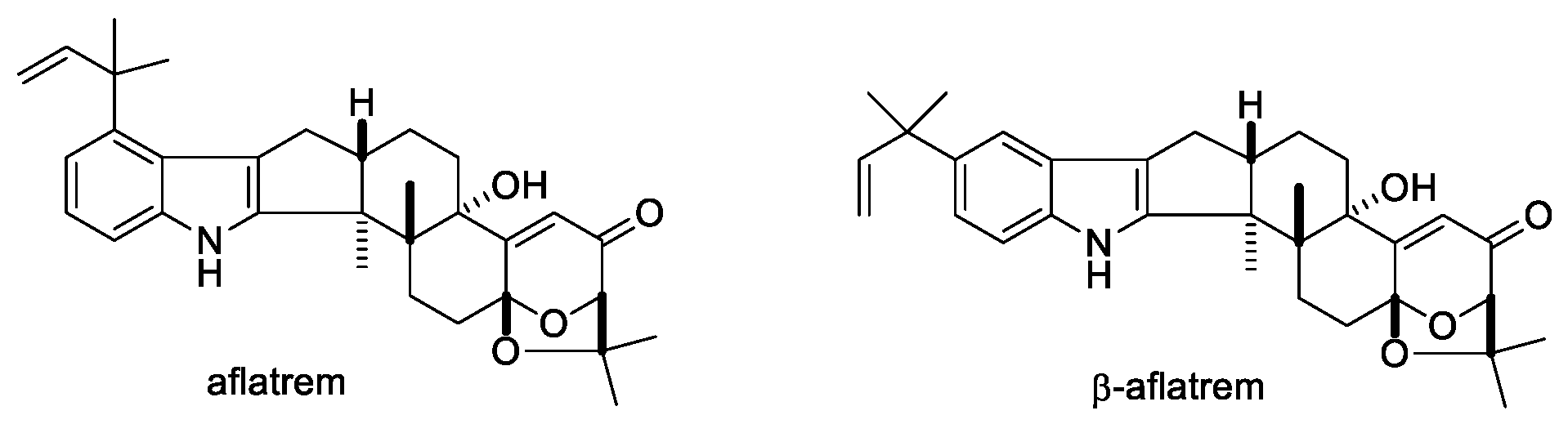
2.1.8. Aflatrems
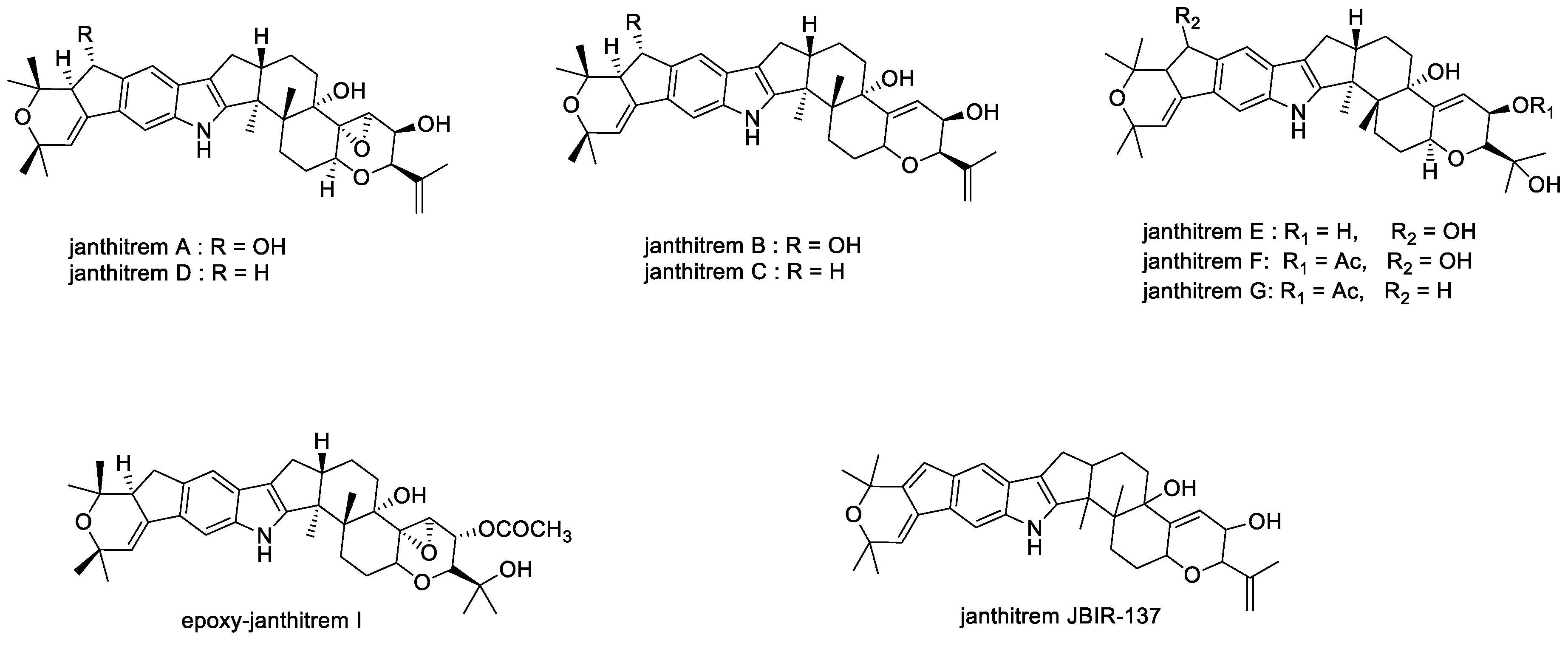
2.1.9. Janthitrems
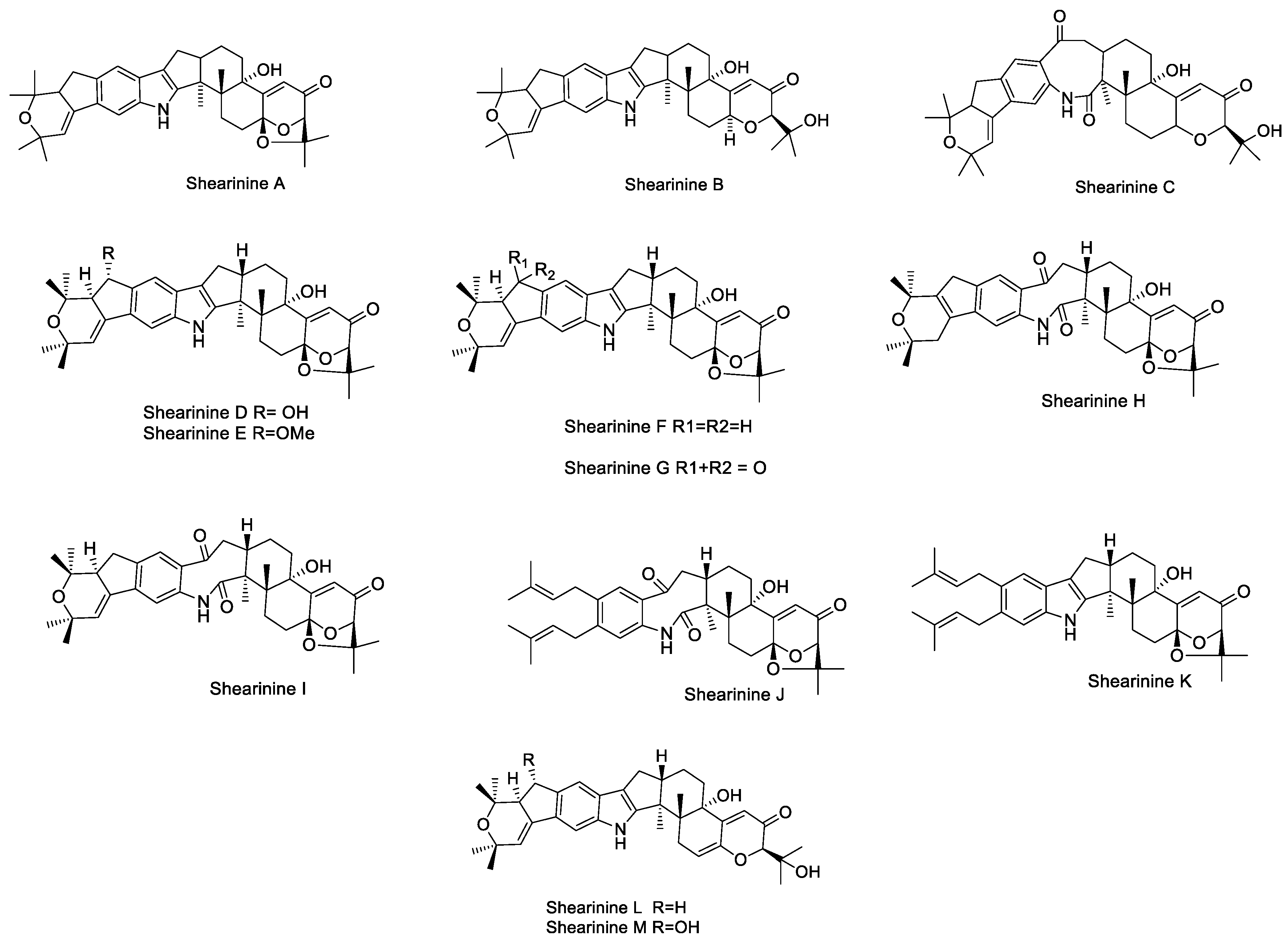
2.1.10. Shearinines
2.1.11. Other Pharmaceutical Applications of Indole-Diterpenes
3. Toxicity of Ergovaline and Lolitrem B in the Field
4. Indole-Diterpenes Reported in the Environment
5. High Throughput Methods for Determining Endophyte Toxicity
Funding
Acknowledgments
Conflicts of Interest
References
- Forster, J.W.; Cogan, N.O.I.; Dobrowolski, M.P.; Francki, M.G.; Spangenberg, G.C.; Smith, K.F. Functionally associated molecular genetic markers for temperate pasture plant improvement. In Plant Genotyping II: SNP Technology; Henry, R.J., Ed.; CABI: Wallingford, UK, 2008; pp. 154–186. [Google Scholar]
- Young, C.A.; Hume, D.E.; McCulley, R.L. Forages and pastures symposium: Fungal endophytes of tall fescue and perennial ryegrass: Pasture friend or foe? J. Anim. Sci. 2013, 91, 2379–2394. [Google Scholar] [CrossRef]
- Cunningham, P.J.; Foot, J.Z.; Reed, K.F.M. Perennial ryegrass (Lolium perenne) endophyte (Acremonium lolii) relationships: the Australian experience. Agric. Ecosyst. Environ. 1993, 44, 157–168. [Google Scholar] [CrossRef]
- Tian, P.; Le, T.N.; Ludlow, E.J.; Smith, K.F.; Forster, J.W.; Guthridge, K.M.; Spangenberg, G.C. Characterisation of novel perennial ryegrass host-Neotyphodium endophyte associations. Crop Pasture Sci. 2013, 64, 716–725. [Google Scholar] [CrossRef]
- Van Zijll De Jong, E.; Dobrowolski, M.P.; Bannan, N.R.; Stewart, A.V.; Smith, K.F.; Spangenberg, G.C.; Forster, J.W. Global genetic diversity of the perennial ryegrass fungal endophyte Neotyphodium lolii. Crop Sci. 2008, 48, 1487–1501. [Google Scholar] [CrossRef]
- Moate, P.J.; Williams, S.R.O.; Grainger, C.; Hannah, M.C.; Mapleson, D.; Auldist, M.J.; Greenwood, J.S.; Popay, A.J.; Hume, D.E.; Mace, W.J.; et al. Effects of wild-type, AR1 and AR37 endophyte-infected perennial ryegrass on dairy production in Victoria, Australia. Anim. Prod. Sci. 2012, 52, 1117–1130. [Google Scholar] [CrossRef]
- Leuchtmann, A.; Bacon, C.W.; Schardl, C.L.; White Jr, J.F.; Tadych, M. Nomenclatural realignment of Neotyphodium species with genus Epichloë. Mycologia 2014, 106, 202–215. [Google Scholar] [CrossRef] [PubMed]
- Popay, A.J.; Hume, D.E.; Mainland, R.A.; Saunders, C.J. Field resistance to Argentine stem weevil (Listronotus bonariensis) in different ryegrass cultivars infected with an endophyte deficient in lolitrem B. New Zeal. J. Agr. Res. 1995, 38, 519–528. [Google Scholar] [CrossRef]
- Prestidge, R.A.; Ball, O.J.P. The role of endophytes in alleviating plant biotic stress in New Zealand. In Proceedings of the Second International Symposium on Acremonium/Grass Interactions; Hume, D.E., Latch, G.C.M., Easton, H.S., Eds.; AgResearch: Palmerston North, New Zealand, 1993; pp. 141–151. [Google Scholar]
- Porter, J.K. Analysis of endophyte toxins: Fescue and other grasses toxic to livestock. J. Animal. Sci. 1995, 73, 871–880. [Google Scholar] [CrossRef] [PubMed]
- Hovermale, J.T.; Craig, A.M. Correlation of ergovaline and lolitrem B levels in endophyte-infected perennial ryegrass (Lolium perenne). J. Vet. Diagn. Invest. 2001, 13, 323–327. [Google Scholar] [CrossRef]
- Weedon, C.M.; Mantle, P.G. Paxilline biosynthesis by Acremonium loliae; a step towards defining the origin of lolitrem neurotoxins. Phytochemistry 1987, 26, 969–971. [Google Scholar] [CrossRef]
- CHAPTER 14—Neurological Diseases. In Diagnostic Techniques in Equine Medicine, 2nd ed.; Taylor, F.G.R.; Brazil, T.J.; Hillyer, M.H. (Eds.) W.B. Saunders: Edinburgh, UK, 2009; pp. 287–304. [Google Scholar]
- CHAPTER 14—Diseases of the Nervous System. In Veterinary Medicine, 11th ed.; Constable, P.D.; Hinchcliff, K.W.; Done, S.H.; Grünberg, W. (Eds.) W.B. Saunders: Edinburgh, UK, 2017; pp. 1155–1370. [Google Scholar]
- Kozák, L.; Szilágyi, Z.; Tóth, L.; Pócsi, I.; Molnár, I. Tremorgenic and neurotoxic paspaline-derived indole-diterpenes: Biosynthetic diversity, threats and applications. Appl. Microbiol. Biotechnol. 2019, 103, 1599–1616. [Google Scholar] [CrossRef]
- Gallagher, R.T.; Hawkes, A.D. The potent tremorgenic neurotoxins lolitrem B and aflatrem: A comparison of the tremor response in mice. Experientia 1986, 42, 823–825. [Google Scholar] [CrossRef] [PubMed]
- Latch, G.C.M. Trichothecenes and other mycotoxins. In Proceedings of the International Mycotoxin Symposium, Sydney, Australia, August 1984. [Google Scholar]
- Plumlee, K.H.; Galey, F.D. Neurotoxic mycotoxins: A review of fungal toxins that cause neurological disease in large animals. J. Vet. Intern. Med. 1994, 8, 49–54. [Google Scholar] [CrossRef]
- Thom, E.R.; Clark, D.A.; Waugh, C.D. Growth, persistence, and alkaloid levels of endophyte-infected and endophyte-free ryegrass pastures grazed by dairy cows in northern New Zealand. New Zeal. J. Agr. Res. 1999, 42, 241–253. [Google Scholar] [CrossRef]
- Miles, C.O.; Munday, S.C.; Wilkins, A.L.; Ede, R.M.; Towers, N.R. Large-scale isolation of lolitrem B and structure determination of lolitrem E. J. Agric. Food Chem. 1994, 42, 1488–1492. [Google Scholar] [CrossRef]
- Russell, C.A. Letter: “Rye grass staggers”. Vet. Rec. 1975, 97, 295. [Google Scholar] [CrossRef]
- Young, C.A.; Tapper, B.A.; May, K.; Moon, C.D.; Schardl, C.L.; Scott, B. Indole-diterpene biosynthetic capability of Epichloë endophytes as predicted by ltm gene analysis. Appl. Environ. Microbiol. 2009, 75, 2200–2211. [Google Scholar] [CrossRef]
- Lewis, P.R.; Donoghue, M.B.; Cook, L.; Granger, L.V.; Hocking, A.D. Tremor syndrome associated with a fungal toxin: sequelae of food contamination. Med. J. Aust. 2005, 182, 582–584. [Google Scholar] [CrossRef] [PubMed]
- Evans, T.J.; Gupta, R.C. Chapter 74—Tremorgenic Mycotoxins. In Veterinary Toxicology, 3rd ed.; Gupta, R.C., Ed.; Academic Press: Amsterdam, Netherlands, 2018; pp. 1033–1041. [Google Scholar]
- Bluett, S.J.; Thom, E.R.; Clark, D.A.; Macdonald, K.A.; Minneé, E.M.K. Effects of perennial ryegrass infected with either AR1 or wild endophyte on dairy production in the Waikato. New Zeal. J. Agr. Res. 2005, 48, 197–212. [Google Scholar] [CrossRef]
- Reddy, P.; Deseo, M.A.; Ezernieks, V.; Guthridge, K.; Spangenberg, G.; Rochfort, S. Toxic indole diterpenes from endophyte-infected perennial ryegrass Lolium perenne L.: Isolation and stability. Toxins 2019, 11, 16. [Google Scholar] [CrossRef]
- Munday-Finch, S.C.; Wilkins, A.L.; Miles, C.O. Isolation of lolicine A, lolicine B, lolitriol, and lolitrem N from Lolium perenne infected with Neotyphodium lolii and evidence for the natural occurrence of 31-epilolitrem N and 31-epilolitrem F. J. Agric. Food Chem. 1998, 46, 590–598. [Google Scholar] [CrossRef]
- Munday-Finch, S. Aspects of the chemistry and toxicology of indole-diterpenoid mycotoxins involved in tremorganic disorder of livestock. Mycotoxin Res. 1997, 13, 88. [Google Scholar] [CrossRef]
- Kanehisa, M.; Furumichi, M.; Tanabe, M.; Sato, Y.; Morishima, K. KEGG: New perspectives on genomes, pathways, diseases and drugs. Nucleic. Acids. Res. 2017, 45, D353–D361. [Google Scholar] [CrossRef] [PubMed]
- Kanehisa, M.; Goto, S. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic. Acids. Res. 2000, 28, 27–30. [Google Scholar] [CrossRef] [PubMed]
- Kanehisa, M.; Sato, Y.; Furumichi, M.; Morishima, K.; Tanabe, M. New approach for understanding genome variations in KEGG. Nucleic. Acids. Res. 2019, 47, D590–D595. [Google Scholar] [CrossRef]
- Gallagher, R.T.; Campbell, A.G.; Hawkes, A.D.; Holland, P.T.; McGaveston, D.A.; Pansier, E.A. Ryegrass staggers: The presence of lolitrem neurotoxins in perennial ryegrass seed. N. Z. Vet. J. 1982, 30, 183–184. [Google Scholar] [CrossRef] [PubMed]
- McLeay, L.M.; Smith, B.L.; Munday-Finch, S.C. Tremorgenic mycotoxins paxilline, penitrem and lolitrem B, the nontremorgenic 31-epilolitrem B and electromyographic activity of the reticulum and rumen of sheep. Res. Vet. Sci. 1999, 66, 119–127. [Google Scholar] [CrossRef]
- Dalziel, J.E.; Dunlop, J.; Finch, S.C.; Wong, S.S. Immune Response Inhibition Using Indole Diterpene Compound. WO2006115423A1, 2006. Available online: https://patents.google.com/patent/WO2006115423A1/ko (accessed on 2 November 2018).
- Dalziel, J.E.; Finch, S.C.; Dunlop, J. The fungal neurotoxin lolitrem B inhibits the function of human large conductance calcium-activated potassium channels. Toxicol. Lett. 2005, 155, 421–426. [Google Scholar] [CrossRef]
- Knaus, H.-G.; McManus, O.B.; Lee, S.H.; Schmalhofer, W.A.; Garcia-Calvo, M.; Helms, L.M.H.; Sanchez, M.; Giangiacomo, K.; Reuben, J.P.; Smith, A.B.; et al. Tremorgenic indole alkaloids potently inhibit smooth muscle high-conductance calcium-activated potassium channels. Biochemistry 1994, 33, 5819–5828. [Google Scholar] [CrossRef]
- Imlach, W.L.; Finch, S.C.; Dunlop, J.; Meredith, A.L.; Aldrich, R.W.; Dalziel, J.E. The molecular mechanism of “ryegrass staggers,” a neurological disorder of K+ channels. J. Pharmacol. Exp. Ther. 2008, 327, 657–664. [Google Scholar] [CrossRef]
- McMillan, L.K.; Carr, R.L.; Young, C.A.; Astin, J.W.; Lowe, R.G.; Parker, E.J.; Jameson, G.B.; Finch, S.C.; Miles, C.O.; McManus, O.B.; et al. Molecular analysis of two cytochrome P450 monooxygenase genes required for paxilline biosynthesis in Penicillium paxilli, and effects of paxilline intermediates on mammalian maxi-K ion channels. Mol. Genet. Genomics. 2003, 270, 9–23. [Google Scholar] [CrossRef] [PubMed]
- Imlach, W.L.; Finch, S.C.; Zhang, Y.; Dunlop, J.; Dalziel, J.E. Mechanism of action of lolitrem B, a fungal endophyte derived toxin that inhibits BK large conductance Ca2+-activated K+ channels. Toxicon 2011, 57, 686–694. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, M.; McManus, O.B. Paxilline inhibition of the alpha-subunit of the high-conductance calcium-activated potassium channel. Neuropharmacology 1996, 35, 963–968. [Google Scholar] [CrossRef]
- Imlach, W.L.; Finch, S.C.; Dunlop, J.; Dalziel, J.E. Structural determinants of lolitrems for inhibition of BK large conductance Ca2+-activated K+ channels. Eur. J. Pharmacol. 2009, 605, 36–45. [Google Scholar] [CrossRef]
- Imlach, W.L.; Finch, S.C.; Miller, J.H.; Meredith, A.L.; Dalziel, J.E. A role for BK channels in heart rate regulation in rodents. PLoS One 2010, 5, e8698. [Google Scholar] [CrossRef] [PubMed]
- Munday-Finch, S.C.; Wilkins, A.L.; Miles, C.O.; Tomoda, H.; Omura, S. Isolation and structure elucidation of lolilline, a possible biosynthetic precursor of the lolitrem family of tremorgenic mycotoxins. J. Agric. Food Chem. 1997, 45, 199–204. [Google Scholar] [CrossRef]
- Munday-Finch, S.C.; Miles, C.O.; Wilkins, A.L.; Hawkes, A.D. Isolation and structure elucidation of lolitrem A, a tremorgenic mycotoxin from perennial ryegrass infected with Acremonium lolii. J. Agric. Food Chem. 1995, 43, 1283–1288. [Google Scholar] [CrossRef]
- Munday-Finch, S.C.; Wilkins, A.L.; Miles, C.O. Isolation of paspaline B, an indole-diterpenoid from Penicilium paxilli. Phytochemistry 1996, 41, 327–332. [Google Scholar] [CrossRef]
- Munday-Finch, S.C.; Wilkins, A.L.; Miles, C.O.; Ede, R.M.; Thomson, R.A. Structure elucidation of lolitrem F, a naturally occurring stereoisomer of the tremorgenic mycotoxin lolitrem B, isolated from Lolium perenne infected with Acremonium lolii. J. Agric. Food Chem. 1996, 44, 2782–2788. [Google Scholar] [CrossRef]
- Miles, C.O.; Wilkins, A.L.; Gallagher, R.T.; Hawkes, A.D.; Munday, S.C.; Towers, N.R. Synthesis and tremorgenicity of paxitriols and lolitriol: Possible biosynthetic precursors of lolitrem B. J. Agric. Food Chem. 1992, 40, 234–238. [Google Scholar] [CrossRef]
- Gardner, D.R.; Welch, K.D.; Lee, S.T.; Cook, D.; Riet-Correa, F. Tremorgenic indole diterpenes from Ipomoea asarifolia and Ipomoea muelleri and the identification of 6,7-dehydro-11-hydroxy-12,13-epoxyterpendole A. J. Nat. Prod. 2018, 81, 1682–1686. [Google Scholar] [CrossRef]
- Lee, S.T.; Gardner, D.R.; Cook, D. Identification of indole diterpenes in Ipomoea asarifolia and Ipomoea muelleri, plants tremorgenic to livestock. J. Agric. Food Chem. 2017, 65, 5266–5277. [Google Scholar] [CrossRef]
- Gatenby, W.A.; Munday-Finch, S.C.; Wilkins, A.L.; Miles, C.O. Terpendole M, a novel indole-diterpenoid isolated from Lolium perenne infected with the endophytic fungus Neotyphodium lolii. J. Agric. Food Chem. 1999, 47, 1092–1097. [Google Scholar] [CrossRef]
- De Jesus, A.E.; Gorst-Allman, C.P.; Steyn, P.S.; Van Heerden, F.R.; Vleggaar, R.; Wessels, P.L.; Hull, W.E. Tremorgenic mycotoxins from Penicillium crustosum. Biosynthesis of penitrem A. J. Chem. Soc. Perkin. Trans. 1 1983, 1863–1868. [Google Scholar] [CrossRef]
- Ciegler, A. Mycotoxins: Occurrence, chemistry, biological activity. Lloydia 1975, 38, 21–35. [Google Scholar] [PubMed]
- Penny, R.H.; O’Sullivan, B.M.; Mantle, P.G.; Shaw, B.I. Clinical studies on tremorgenic mycotoxicoses in sheep. Vet. Rec. 1979, 105, 392–393. [Google Scholar] [CrossRef]
- Kyriakidis, N.; Waight, E.S.; Day, J.B.; Mantle, P.G. Novel metabolites from Penicillium crustosum, including penitrem E, a tremorgenic mycotoxin. Appl. Environ. Microbiol. 1981, 42, 61–62. [Google Scholar] [PubMed]
- Cole, R.J.; Dorner, J.W.; Lansden, J.A.; Cox, R.H.; Pape, C.; Cunfer, B.; Nicholson, S.S.; Bedell, D.M. Paspalum staggers: Isolation and identification of tremorgenic metabolites from sclerotia of Claviceps paspali. J. Agric. Food Chem. 1977, 25, 1197–1201. [Google Scholar] [CrossRef] [PubMed]
- Cole, R.J.; Dorner, J.W.; Springer, J.P.; Cox, R.H. Indole metabolites from a strain of Aspergillus flavus. J. Agric. Food Chem. 1981, 29, 293–295. [Google Scholar] [CrossRef]
- Gallagher, R.T.; Finer, J.; Clardy, J.; Leutwiler, A.; Weibel, F.; Acklin, W.; Arigoni, D. Paspalinine, a tremorgenic metabolite from Claviceps paspali Stevens et Hall. Tetrahedron. Lett. 1980, 21, 235–238. [Google Scholar] [CrossRef]
- Babu, J.V.; Popay, A.J.; Miles, C.O.; Wilkins, A.L.; Di Menna, M.E.; Finch, S.C. Identification and structure elucidation of janthitrems A and D from Penicillium janthinellum and determination of the tremorgenic and anti-insect activity of janthitrems A and B. J. Agric. Food Chem. 2018, 66, 13116–13125. [Google Scholar] [CrossRef] [PubMed]
- Lauren, D.R.; Gallagher, R.T. High-performance liquid chromatography of the janthitrems: Fluorescent tremorgenic mycotoxins produced by Penicillium janthinellum. J. Chromatogr. A 1982, 248, 150–154. [Google Scholar] [CrossRef]
- Gallagher, F.T.; Latch, G.C.M.; Keogh, R.G. The janthitrems: Fluorescent tremorgenic toxins produced by Penicillium janthinellum isolates from ryegrass pastures. Appl. Environ. Microbiol. 1980, 39, 272–273. [Google Scholar]
- Cole, R.J.; Kirksey, J.W.; Wells, J.M. A new tremorgenic metabolite from Penicillium paxilli. Can. J. Microbiol. 1974, 20, 1159–1162. [Google Scholar] [CrossRef] [PubMed]
- Springer, J.P.; Clardy, J.; Wells, J.M.; Cole, R.J.; Kirksey, J.W. The structure of paxilline, a tremorgenic metabolite of Penicillium paxilli Bainier. Tetrahedron. Lett. 1975, 16, 2531–2534. [Google Scholar] [CrossRef]
- CHAPTER 8—Tremorgen Group. Handbook of Toxic Fungal Metabolites; Cole, R.J., Cox, R.H., Eds.; Academic Press: New York, NY, USA, 1981; pp. 355–509. [Google Scholar]
- Zhou, Y.; Lingle, C.J. Paxilline inhibits BK channels by an almost exclusively closed-channel block mechanism. J. Gen. Physiol. 2014, 144, 415–440. [Google Scholar] [CrossRef] [Green Version]
- DeFarias, F.P.; Carvalho, M.F.; Lee, S.H.; Kaczorowski, G.J.; Suarez-Kurtz, G. Effects of the K+ channel blockers paspalitrem-C and paxilline on mammalian smooth muscle. Eur. J. Pharmacol. 1996, 314, 123–128. [Google Scholar] [CrossRef]
- Saikia, S.; Parker, E.J.; Koulman, A.; Scott, B. Defining paxilline biosynthesis in Penicillium paxilli: Functional characterization of two cytochrome P450 monooxygenases. J. Biol. Chem. 2007, 282, 16829–16837. [Google Scholar] [CrossRef]
- Matsui, C.; Ikeda, Y.; Iinuma, H.; Kushida, N.; Kunisada, T.; Simizu, S.; Umezawa, K. Isolation of a novel paxilline analog pyrapaxilline from fungus that inhibits LPS-induced NO production. J. Antibiot. 2014, 67, 787–790. [Google Scholar] [CrossRef] [Green Version]
- Nozawa, K.; Nakajima, S.; Kawai, K.; Udagawa, S.; Horie, Y.; Yamazaki, M. Novel indoloditerpenes, emindoles, and their related compounds from Emericella spp. Proceedings of Tennen Yuki Kagobutsu Toronkai Koen Yoshishu (Symposium on the Chemistry of Natural Products), Sapporo, Japan, 23–26 May 1987; pp. 637–643. Available online: https://www.worldcat.org/title/29-tennen-yuki-kagobutsu-toronkai-koen-yoshishu-sapporo-august-26-28-1987-2/oclc/833622249 (accessed on 5 February 2018).
- Belofsky, G.N.; Gloer, J.B.; Wicklow, D.T.; Dowd, P.F. Antiinsectan alkaloids: Shearinines A-C and a new paxilline derivative from the ascostromata of Eupenicillium shearii. Tetrahedron 1995, 51, 3959–3968. [Google Scholar] [CrossRef]
- Miles, C.O.; Wilkins, A.L.; Garthwaite, I.; Ede, R.M.; Munday-Finch, S.C. Immunochemical techniques in natural products chemistry: Isolation and structure determination of a novel indole-diterpenoid aided by TLC-ELISAgram. J. Org. Chem. 1995, 60, 6067–6069. [Google Scholar] [CrossRef]
- Gallagher, R.T.; White, E.P.; Mortimer, P.H. Ryegrass Staggers: Isolation of Potent Neurotoxins Lolitrem A and Lolitrem B From Staggers-Producing Pastures. N. Z. Vet. J. 1981, 29, 189–190. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, R.T.; Hawkes, A.D.; Steyn, P.S.; Vleggaar, R. Tremorgenic neurotoxins from perennial ryegrass causing ryegrass staggers disorder of livestock: structure elucidation of lolitrem B. J. Chem. Soc. Chem. Commun. 1984, 9, 614–616. [Google Scholar] [CrossRef]
- De Jesus, A.E.; Steyn, P.S.; Van Heerden, F.R.; Vleggaar, R.; Wessels, P.L.; Hull, W.E. Structure and biosynthesis of the penitrems A-F, six novel tremorgenic mycotoxins from Penicillium crustosum. J. Chem. Soc. Chem. Commun. 1981, 6, 289–291. [Google Scholar] [CrossRef]
- De Jesus, A.E.; Steyn, P.S.; Van Heerden, F.R.; Vleggaar, R.; Wessels, P.L.; Hull, W.E. Tremorgenic mycotoxins from Penicillium crustosum. Structure elucidation and absolute configuration of penitrems B—F. J. Chem. Soc. Perkin. 1 1983, 0, 1857–1861. [Google Scholar] [CrossRef]
- Cavanagh, J.B.; Holton, J.L.; Nolan, C.C.; Ray, D.E.; Naik, J.T.; Mantle, P.G. The effects of the tremorgenic mycotoxin penitrem A on the rat cerebellum. Vet. Pathol. 1998, 35, 53–63. [Google Scholar] [CrossRef]
- Wilson, B.J.; Hoekman, T.; Dettbarn, W.D. Effects of a fungus tremorgenic toxin (penitrem A) on transmission in rat phrenic nerve-diaphragm preparations. Brain Res. 1972, 40, 540–544. [Google Scholar] [CrossRef]
- Norris, P.J.; Smith, C.C.T.; de Belleroche, J.; Bradford, H.F.; Mantle, P.G.; Thomas, A.J.; Penny, R.H. Actions of tremorgenic fungal toxins on neurotransmitter release. J. Neurochem. 1980, 34, 33–42. [Google Scholar] [CrossRef]
- Bradford, H.F.; Norris, P.J.; Smith, C.C.T. Changes in transmitter release patterns in vitro induced by tremorgenic mycotoxins. J. Environ. Pathol. Toxicol. Oncol. 1990, 10, 17–30. [Google Scholar]
- Sobotka, T.J.; Brodie, R.E.; Spaid, S.L. Neurobehavioral studies of tremorgenic mycotoxins verruculogen and penitrem A. Pharmacology 1978, 16, 287–294. [Google Scholar] [CrossRef]
- Cho, Y.; Cha, S.H.; Sok, D.E. Presynaptic effects of verruculogen on gamma-aminobutyric acid(GABA) uptake and release in rat brain. Korean Biochemical. Journal 1994, 27, 353–356. [Google Scholar]
- Hotujac, L.; Stern, P. Pharmacological examination of verruculogen induced tremor. Acta. Medica. Iugoslavica. 1974, 28, 223–229. [Google Scholar]
- Hotujac, L.J.; Muftić, R.H.; Filipović, N. Verruculogen: A new substance for decreasing of gaba levels in CNS. Pharmacology 1976, 14, 297–300. [Google Scholar] [CrossRef]
- Ouadid-Ahidouch, H.; Roudbaraki, M.; Delcourt, P.; Ahidouch, A.; Joury, N.; Prevarskaya, N. Functional and molecular identification of intermediate-conductance Ca 2+-activated K+ channels in breast cancer cells: Association with cell cycle progression. Am. J. Physiol. Cell. Physiol. 2004, 287, C125–C134. [Google Scholar] [CrossRef]
- Goda, A.A.; Siddique, A.B.; Mohyeldin, M.; Ayoub, N.M.; El Sayed, K.A. The Maxi-K (BK) channel antagonist penitrem a as a novel breast cancer-targeted therapeutic. Marine Drugs 2018, 16, 157. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.; Lin, C.; Roberts, M.J.; Waud, W.R.; Piazza, G.A.; Li, Y. Niclosamide suppresses cancer cell growth by inducing Wnt co-receptor LRP6 degradation and inhibiting the Wnt/β-catenin pathway. PLoS ONE 2011, 6, e29290. [Google Scholar] [CrossRef] [PubMed]
- Sallam, A.A.; Houssen, W.E.; Gissendanner, C.R.; Orabi, K.Y.; Foudah, A.I.; El Sayed, K.A. Bioguided discovery and pharmacophore modeling of the mycotoxic indole diterpene alkaloids penitrems as breast cancer proliferation, migration, and invasion inhibitors. MedChemComm 2013, 4, 1360–1369. [Google Scholar] [CrossRef]
- Sallam, A.A.; Ayoub, N.M.; Foudah, A.I.; Gissendanner, C.R.; Meyer, S.A.; El Sayed, K.A. Indole diterpene alkaloids as novel inhibitors of the Wnt/β-catenin pathway in breast cancer cells. Eur. J. Med. Chem. 2013, 70, 594–606. [Google Scholar] [CrossRef]
- Goda, A.A.; Naguib, K.M.; Mohamed, M.M.; Amra, H.A.; Nada, S.A.; Abdel-Ghaffar, A.R.B.; Gissendanner, C.R.; El Sayed, K.A. Astaxanthin and docosahexaenoic acid reverse the toxicity of the maxi-K (BK) channel antagonist mycotoxin penitrem A. Marine Drugs 2016, 14, 208. [Google Scholar] [CrossRef]
- Selala, M.I.; Musuku, A.; Schepens, P.J.C. Isolation and determination of paspalitrem-type tremorgenic mycotoxins using liquid chromatography with diode-array detection. Anal. Chim. Acta. 1991, 244, 1–8. [Google Scholar] [CrossRef]
- Springer, J.P.; Clardy, J. Paspaline and paspalicine, two indole-mevalonate metabolites from Claviceps paspali. Tetrahedron Lett. 1980, 21, 231–234. [Google Scholar] [CrossRef]
- Huang, X.H.; Nishida, H.; Tomoda, H.; Tabata, N.; Shiomi, K.; Yang, D.J.; Takayanagi, H.; Omura, S. Correction: Terpendoles, novel AC AT inhibitors produced by Albophoma yamanashiensis. II. Structure elucidation of terpendoles A, B, C and D. J. Antibiot. (Tokyo) 1995, 48, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Tomoda, H.; Tabata, N.; Yang, D.-J.; Takayanagi, H.; Omura, S. Terpendoles, novel AC AT inhibitors produced by Albophoma yamanashiensis. III. Production, isolation and structure elucidation of new components. J. Antibiot. 1995, 48, 793–804. [Google Scholar] [CrossRef] [PubMed]
- Chiwata, T.; Aragane, K.; Fujinami, K.; Kojima, K.; Ishibashi, S.; Yamada, N.; Kusunoki, J. Direct effect of an acyl-CoA:cholesterol acyltransferase inhibitor, F-1394, on atherosclerosis in apolipoprotein E and low density lipoprotein receptor double knockout mice. Br. J. Pharmacol. 2001, 133, 1005–1012. [Google Scholar] [CrossRef] [Green Version]
- Ohshiro, T.; Rudel, L.L.; Omura, S.; Tomoda, H. Selectivity of microbial acyl-CoA: Cholesterol acyltransferase inhibitors toward isozymes. J. Antibiot. (Tokyo) 2007, 60, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.A.; Joyce, C.; Davis, M.; Reagan, J.W.; Clark, M.; Shelness, G.S.; Rudel, L.L. Identification of a form of acyl-CoA: Cholesterol acyltransferase specific to liver and intestine in nonhuman primates. J. Biol. Chem. 1998, 273, 26747–26754. [Google Scholar] [CrossRef] [PubMed]
- Nakazawa, J.; Yajima, J.; Usui, T.; Ueki, M.; Takatsuki, A.; Imoto, M.; Toyoshima, Y.Y.; Osada, H. A novel action of terpendole E on the motor activity of mitotic Kinesin Eg5. Chem. Biol. 2003, 10, 131–137. [Google Scholar] [CrossRef]
- Rosenfeld, S.S.; Rich, J.; Venere, M. Mitotic Kinesin Eg5 Inhibiting Anticancer Agents. WO2015153967A1, 2015. Available online: https://patents.google.com/patent/US20150352114A1/en (accessed on 3 January 2019).
- Laakso, J.A.; Gloer, J.B.; Wicklow, D.T.; Dowd, P.F. Sulpinines A-C and secopenitrem B: New antiinsectan metabolites from the sclerotia of Aspergillus sulphureus. J. Org. Chem. 1992, 57, 2066–2071. [Google Scholar] [CrossRef]
- Laakso, J.A.; Tepaske, M.R.; Dowd, P.F.; Gloer, J.B.; Wicklow, D.T.; Staub, G.M. Indole Antiinsectan Aspergillus Metabolites. US5227396A, 1993. Available online: https://patents.google.com/patent/US5227396A/en (accessed on 5 February 2019).
- Hosoe, T.; Itabashi, T.; Kobayashi, N.; Udagawa, S.-i.; Kawai, K.-i. Three new types of indoloditerpenes, emindole PA-PC, from Emericella purpurea. Revision of the structure of emindole PA. Chem. Pharm. Bull. 2006, 54, 185–187. [Google Scholar] [CrossRef] [PubMed]
- Petersen, L.M.; Frisvad, J.C.; Knudsen, P.B.; Rohlfs, M.; Gotfredsen, C.H.; Larsen, T.O. Induced sclerotium formation exposes new bioactive metabolites from Aspergillus sclerotiicarbonarius. J. Antibiot. 2015, 68, 603–608. [Google Scholar] [CrossRef] [PubMed]
- Kimura, Y.; Nishibe, M.; Nakajima, H.; Hamasaki, T.; Shigemitsu, N.; Sugawara, F.; Stout, T.J.; Clardy, J. Emeniveol; A new pollen growth inhibitor from the fungus, Emericella nivea. Tetrahedron Lett. 1992, 33, 6987–6990. [Google Scholar] [CrossRef]
- Ogata, M.; Ueda, J.Y.; Hoshi, M.; Hashimoto, J.; Nakashima, T.; Anzai, K.; Takagi, M.; Shin-Ya, K. A novel indole-diterpenoid, JBIR-03 with anti-MRSA activity from Dichotomomyces cejpii var. cejpii NBRC 103559. J. Antibiot. (Tokyo) 2007, 60, 645–648. [Google Scholar] [CrossRef]
- Qiao, M.F.; Ji, N.Y.; Liu, X.H.; Li, K.; Zhu, Q.M.; Xue, Q.Z. Indoloditerpenes from an algicolous isolate of Aspergillus oryzae. Bioorg. Med. Chem. Lett. 2010, 20, 5677–5680. [Google Scholar] [CrossRef]
- TePaske, M.R.; Gloer, J.B.; Wicklow, D.T.; Dowd, P.F. Aflavarin and β-aflatrem: New anti-insectan metabolites from the sclerotia of Aspergillus flavus. J. Nat. Prod. 1992, 55, 1080–1086. [Google Scholar] [CrossRef]
- Penn, J.; Swift, R.; Wigley, L.J.; Mantle, P.G.; Bilton, J.N.; Sheppard, R.N. Janthitrems B and C, two principal indole-diterpenoids produced by Penicillium janthinellum. Phytochemistry 1993, 32, 1431–1434. [Google Scholar] [CrossRef]
- Wilkins, A.L.; Miles, C.O.; Ede, R.M.; Gallagher, R.T.; Munday, S.C. Structure elucidation of janthitrem B, a tremorgenic metabolite of Penicillium janthinellum, and relative configuration of the A and B rings of janthitrems B, E, and F. J. Agric. Food Chem. 1992, 40, 1307–1309. [Google Scholar] [CrossRef]
- Tapper, B.; Lane, G.A. Janthitrems found in a Neotyphodium endophyte of perennial ryegrass. In Proceedings of the 5th International Symposium on Neotyphodium/Grass Interactions, Fayetteville, AR, USA, 23–26 May 2004; p. 301. [Google Scholar]
- De Jesus, A.E.; Steyn, P.S.; Van Heerden, F.R.; Vleggaar, R. Structure elucidation of the janthitrems, novel tremorgenic mycotoxins from Penicillium janthinellum. J. Chem. Soc. Perkin. 1 1984, 697–701. [Google Scholar] [CrossRef]
- Kawahara, T.; Nagai, A.; Takagi, M.; Shin-Ya, K. JBIR-137 and JBIR-138, new secondary metabolites from Aspergillus sp. fA75. J. Antibiot. 2012, 65, 535–538. [Google Scholar] [CrossRef]
- Xu, M.; Gessner, G.; Groth, I.; Lange, C.; Christner, A.; Bruhn, T.; Deng, Z.; Li, X.; Heinemann, S.H.; Grabley, S.; et al. Shearinines D-K, new indole triterpenoids from an endophytic Penicillium sp. (strain HKI0459) with blocking activity on large-conductance calcium-activated potassium channels. Tetrahedron 2007, 63, 435–444. [Google Scholar] [CrossRef]
- You, J.; Du, L.; King, J.B.; Hall, B.E.; Cichewicz, R.H. Small-molecule suppressors of Candida albicans biofilm formation synergistically enhance the antifungal activity of amphotericin B against clinical Candida isolates. ACS. Chem. Biol. 2013, 8, 840–848. [Google Scholar] [CrossRef] [PubMed]
- Dhodary, B.; Schilg, M.; Wirth, R.; Spiteller, D. Secondary metabolites from Escovopsis weberi and their role in attacking the garden fungus of Leaf-cutting ants. Chem. Eur. J. 2018, 24, 4445–4452. [Google Scholar] [CrossRef]
- Goetz, M.A.; Kaczorowski, G.J.; Monaghan, R.L.; Strohl, W.R.; Tkacz, J.S. Maxi-K Potassium Channel Blockers for Treatment of Glaucoma and as Ocular Neuroprotective Agents. WO2003105868A1, 2003. Available online: https://patents.google.com/patent/WO2003105868A1/ru (accessed on 22 March 2018).
- Garcia, M.L.; Goetz, M.A.; Kaczorowski, G.J.; McManus, O.B.; Monaghan, R.L.; Strohl, W.R.; Tkacz, J.S. Indole Diterpene Compound Maxi-K Potassium Channel Blockers, Methods Using Them for the Treatment of Glaucoma and Other Conditions, and Fermentation Process for Production. WO2003105724A2, 2003. Available online: https://patents.google.com/patent/WO2003105724A2 (accessed on 22 March 2018).
- Brnardic, E.; Doherty, J.B.; Dorsey, J.; Ellwood, C.; Fillmore, M.; Malaska, M.; Nelson, K.; Soukri, M. Preparation of Indole Diterpene Alkaloids as Maxi-K Channel Blockers for the Treatment of Glaucoma. WO2009048559A1, 2009. Available online: https://patents.google.com/patent/WO2009048559A1/en (accessed on 22 March 2018).
- Fan, Y.; Wang, Y.; Liu, P.; Fu, P.; Zhu, T.; Wang, W.; Zhu, W. Indole-diterpenoids with anti-H1N1 activity from the aciduric fungus Penicillium camemberti OUCMDZ-1492. J. Nat. Prod. 2013, 76, 1328–1336. [Google Scholar] [CrossRef] [PubMed]
- Easton, H.S.; Lane, G.A.; Tapper, B.A.; Keogh, R.G.; Cooper, B.M.; Blackwell, M.; Anderson, M.; Fletchers, L.R. Ryegrass endophyte-related heat stress in cattle. Proc. N. Z. Grassl. Assoc. 1996, 57, 37–41. [Google Scholar]
- Craig, A.M.; Blythe, L.L.; Duringer, J.M. The role of the oregon state university endophyte service laboratory in diagnosing clinical cases of endophyte toxicoses. J. Agric. Food Chem. 2014, 62, 7376–7381. [Google Scholar] [CrossRef] [PubMed]
- Repussard, C.; Zbib, N.; Tardieu, D.; Guerre, P. Ergovaline and lolitrem B concentrations in perennial ryegrass in field culture in Southern France: Distribution in the plant and impact of climatic factors. J. Agric. Food Chem. 2014, 62, 12707–12712. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, J.A.; Rottinghaus, G.E.; González, E. Ergovaline concentration in perennial ryegrass infected with a lolitrem B-free fungal endophyte in north-west Spain. New Zeal. J. Agr. Res. 2003, 46, 117–122. [Google Scholar] [CrossRef]
- Andersen, B.; Frisvad, J.C. Natural Occurrence of Fungi and Fungal Metabolites in Moldy Tomatoes. J. Agric. Food Chem. 2004, 52, 7507–7513. [Google Scholar] [CrossRef] [PubMed]
- Gordon, K.E.; Masotti, R.E.; Waddell, W.R. Tremorgenic encephalopathy: A role of mycotoxins in the production of CNS disease in humans? Can. J. Neurol. Sci. 1993, 20, 237–239. [Google Scholar] [CrossRef] [PubMed]
- Talcott, P.A. Chapter 63—Mycotoxins. In Small Animal Toxicology, 3rd ed.; Peterson, M.E., Talcott, P.A., Eds.; W.B. Saunders: Saint Louis, MO, USA, 2013; pp. 677–682. [Google Scholar]
- Walter, S.L. Acute penitrem A and roquefortine poisoning in a dog. Can. Vet. J. 2002, 43, 372–374. [Google Scholar]
- Gallagher, R.T.; Hawkes, A.D. High-performance liquid chromatography with stop-flow ultraviolet spectral chracterization of lolitrem neu-rotoxins from perennial ryegrass. J. Chromatogr. A 1985, 322, 159–167. [Google Scholar] [CrossRef]
- Gallagher, R.T.; Hawkes, A.D.; Stewart, J.M. Rapid determination of the neurotoxin lolitrem B in perennial ryegrass by high-performance liquid chromatography with fluorescence detection. J. Chromatogr. A 1985, 321, 217–226. [Google Scholar] [CrossRef]
- Repussard, C.; Tardieu, D.; Alberich, M.; Guerre, P. A new method for the determination of lolitrem B in plant materials. Anim. Feed. Sci. Tech. 2014, 193, 141–147. [Google Scholar] [CrossRef]
- Laganà, A. Introduction to the toxins special issue on LC-MS/MS methods for mycotoxin analysis. Toxins 2017, 9, 325. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, M.J.; Koulman, A.; Monahan, B.J.; Pritchard, B.L.; Payne, G.A.; Scott, B. Identification of two aflatrem biosynthesis gene loci in Aspergillus flavus and metabolic engineering of Penicillium paxilli to elucidate their function. Appl. Environ. Microbiol. 2009, 75, 7469–7481. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, S.; Lane, G.A.; Mace, W.; Parsons, A.J.; Fraser, K.; Xue, H. The use of genomics and metabolomics methods to quantify fungal endosymbionts and alkaloids in grasses. In Plant Metabolomics Methods in Molecular Biology (Methods and Protocols); Hardy, N., Hall, R., Eds.; Humana Press: New York City, NY, USA, 2012; Volume 860, pp. 213–226. [Google Scholar]





| Compound Name | Toxicity as Per Biological Activity on Mice (mg of Compound/kg of Body Weight) | Observation on Mice | Biological Activity on BK/Maxi or hSlo Channel | Biological Activity on Animal Model (EMG Activity/Observation in Sheep) | Reference |
|---|---|---|---|---|---|
| Lolitrem A | 2 mg/kg | Severe and prolonged tremor | ¯ | ¯ | [44] |
| Lolitrem B | 0.5 to 8.0 mg/kg | Severe and prolonged tremor | IC50 = 4 nM (No recovery after wash out) | At 70 µg/kg dose, tremors were observed after 15 min and slowly increased in severity lasting for duration of 12 h. The reticulum and rumen showed inhibition after 20–30 min, coinciding with tremors. | [16,35,41] |
| 31-epi-lolitrem B | 4 mg/kg | Nontremorgenic | IC50 = 50 nM (>50% recovery after 10 min) | The lolitrem B isomer was administered at a dose of 70 µg/kg and there was no effect either on the skeletal muscle EMG activity or EMG activity of the reticulum and rumen. | [33,41,45] |
| Lolitrem E | 2 mg/kg | Nontremorgenic | IC50 = 6 nM (No recovery after wash out) | ¯ | [20,41] |
| Lolitrem E acetate | 16 mg/kg | Weakly tremorgenic | IC50 = 2 nM (No recovery after wash out) | ¯ | [28,41] |
| 31-epi-lolitrem Fa | 4 mg/kg | Slightly less tremorgenic than lolitrem B | ¯ | ¯ | [46] |
| Lolitrem Fa | 4 mg/kg | Slightly less tremorgenic than lolitrem B | IC50 = 8 nM (No recovery after wash out) | ¯ | [41,46] |
| Lolitrem M | Nontremorgenic | IC50 = 78 nM (>50% recovery after 10 min) | ¯ | [41] | |
| Paxilline | 4 to 8 mg/kg and an 80 mg/kg dose | Severe but short term tremorgenicity compared to lolitrem B | Complete inhibition = 1 µM (recovery after wash out) Fraction current blocked by 10 nM = 70% (recovery after wash out) | At 1.0 mg/kg dose, moderate to strong tremors; the onset was immediate after 2 min administration; tremors gradually disappeared over the next hour. EMG activity showed both excitatory and inhibitory on the reticulum and rumen. Also, within a minute of infusion, elevations of the EMG activity coincided with induction of marked tremoring. | [33,36,38,47] |
| 13-Desoxypaxilline | 8 mg/kg | Nontremorgenic | < 50% inhibition = 30 µM | ¯ | [38] |
| α-Paxitriol | 100 mg/kg | Lethargy and rough coats, also normal activities such as walking, rearing and preening were greatly reduced for several hours. Animals recovered to normal by 24 h post-injection. | ¯ | ¯ | [47] |
| β-Paxitriol | 100 mg/kg | Lethargy and rough coats, also normal activities such as walking, rearing and preening were greatly reduced for several hours. Animals recovered to normal by 24 h post-injection. | ¯ | ¯ | [47] |
| Lolitriol | 20 mg/kg | Nontremorgenic | IC50 = 196 nM (>50% recovery after 10 min) | ¯ | [47] |
| Lolitriol acetate | - | Nontremorgenic | IC50 = 43 nM (>50% recovery after 10 min) | ¯ | [41] |
| Lolitriol and β-Paxitrol | As a mixture: 16 mg/kg and 100 mg/kg respectively (200 µL dosage) | The single administration of both β-paxitriol and the nontremorgenic lolitriol proved lethal after an initial period of lethargy | - | - | [47] |
| Lolilline | 8 mg/kg | Nontremorgenic | - | - | [43] |
| 6,7-dehydroterpendole A | 8 mg/kg | Produced more intense tremors than terpendole C and K at the same dose level | - | - | [48,49] |
| Terpendole C | 4 mg/kg and 8 mg/kg | A fast-acting tremorgen that produced more intense tremors than paxilline, 11-hydroxy12,13-epoxyterpendole K and 6,7-dehydro-11-hydroxy-12, 13 epoxyterpendole A at the same dose level, but the activity ceased after 2 h, as compared to paxilline which ceased after 6 h. | ¯ | ¯ | [43,48] |
| Terpendole D, E, F, G, H and I | 8 mg/kg | Nontremorgenic | ¯ | ¯ | [28] |
| Terpendole K | 8 mg/kg | Produced more intense tremors than paxilline, 11-hydroxy12,13-epoxyterpendole K and 6,7-dehydro-11-hydroxy-12,13 epoxyterpendole A at the same dose level | ¯ | ¯ | [48] |
| Terpendole M | 8 mg/kg | Weakly tremorgenic | ¯ | ¯ | [50] |
| 6,7-dehydro-11-hydroxy-12,13 epoxyterpendole A and 11-hydroxy12,13-epoxyterpendole K | 8 mg/kg | Mild tremors | ¯ | ¯ | [48,49] |
| Mixture of 88.3% Penitrem A, 6.4% Penitrem B, 5.3% Penitrem E | ¯ | ¯ | A dose of 5.5 mg/kg showed no significant skeletal EMG activity, although exhibited strong inhibition on the reticulum and rumen. This was apparent at 15 to 30 min and lasted 2 h. The maximum period of inhibition coincided with the period of greatest tremoring. | [33,51] | |
| Penitrem A | 0.75 mg/kg (dose range 0.5 mg/kg to 1.5 mg/kg) | Elicited moderate tremors. Tremor duration reported as several hours. | Fraction current blocked by 10 nM = 100% (no recovery after wash out) | Tremorgenic observation in sheep when given at a dose of 20 µg/kg intravenously. | [36,52,53,54] |
| Penitrem E | 2.25 mg/kg (dose range 1.0 mg/kg to 3.6 mg/kg) | Elicited moderate tremors. Tremor duration reported as several hours. No difference to penitrem A in the rates of onset of tremors observed, and the symptomatology were like-wise similar. | - | - | [54] |
| Paspaline | ¯ | ¯ | Slight inhibition at concentrations up to 1 µM | ¯ | [38] |
| Paspalinine | 8 mg/kg | Short duration tremors | Fraction current blocked by 10 nM = 100% (no recovery after wash out) | ¯ | [28,36,55,56] |
| Paspalicine | 250 mg/kg | Nontremorgenic | Fraction current blocked by 10 nM = 83% (recovery after wash out) | ¯ | [36,55,57] |
| Paspalitrems | 14 mg/kg | Short duration tremors | Fraction current blocked by 10 nM of paspalitrem A and paspalitrem C = 98% and 100% respectively (no recovery after wash out) | ¯ | [36,55] |
| Aflatrem | 1 mg/kg (dose range 0.5 mg/kg to 4.0 mg/kg) | Short duration tremors | Fraction current blocked by 10 nM = 100% (no recovery after wash out) | ¯ | [16,36] |
| Janthitrem A | 4 mg/kg | Tremor duration was reported as 8 h and peaked at 15 min. Tremors produced were more intense than janthitrem B, from 2 h post exposure. | ¯ | ¯ | [58] |
| Janthitrem B | 6 mg/kg | Tremor duration reported as 6 h and peaked at 30 min. Un-coordination and hypersensitivity to sound and touch is also reported. | ¯ | ¯ | [58,59,60] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reddy, P.; Guthridge, K.; Vassiliadis, S.; Hemsworth, J.; Hettiarachchige, I.; Spangenberg, G.; Rochfort, S. Tremorgenic Mycotoxins: Structure Diversity and Biological Activity. Toxins 2019, 11, 302. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins11050302
Reddy P, Guthridge K, Vassiliadis S, Hemsworth J, Hettiarachchige I, Spangenberg G, Rochfort S. Tremorgenic Mycotoxins: Structure Diversity and Biological Activity. Toxins. 2019; 11(5):302. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins11050302
Chicago/Turabian StyleReddy, Priyanka, Kathryn Guthridge, Simone Vassiliadis, Joanne Hemsworth, Inoka Hettiarachchige, German Spangenberg, and Simone Rochfort. 2019. "Tremorgenic Mycotoxins: Structure Diversity and Biological Activity" Toxins 11, no. 5: 302. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins11050302






