A Method for Simultaneous Determination of 20 Fusarium Toxins in Cereals by High-Resolution Liquid Chromatography-Orbitrap Mass Spectrometry with a Pentafluorophenyl Column
Abstract
:1. Introduction
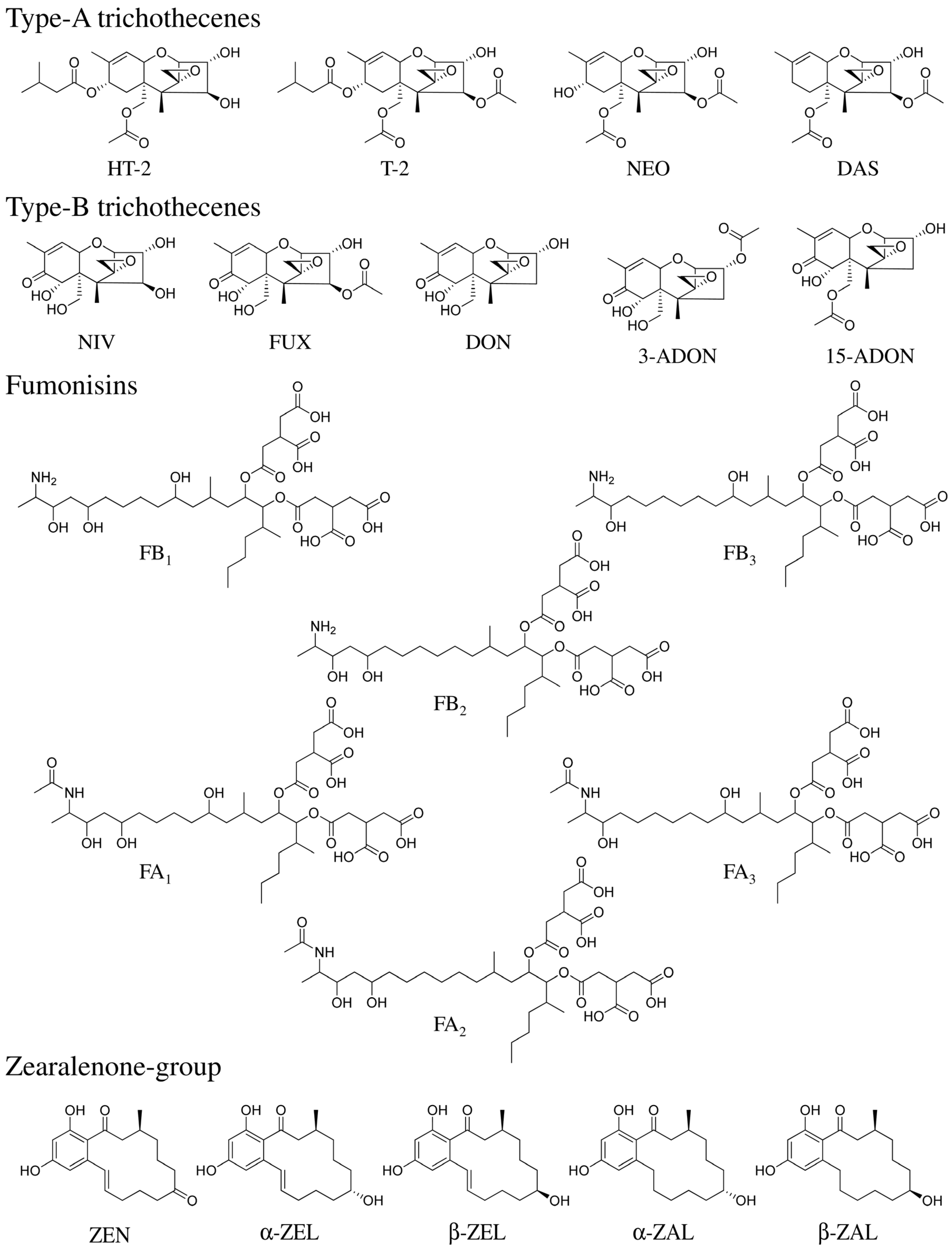
2. Results and Discussion
2.1. Separation of 20 Fusarium Toxins Using a Pentafluorophenyl Column
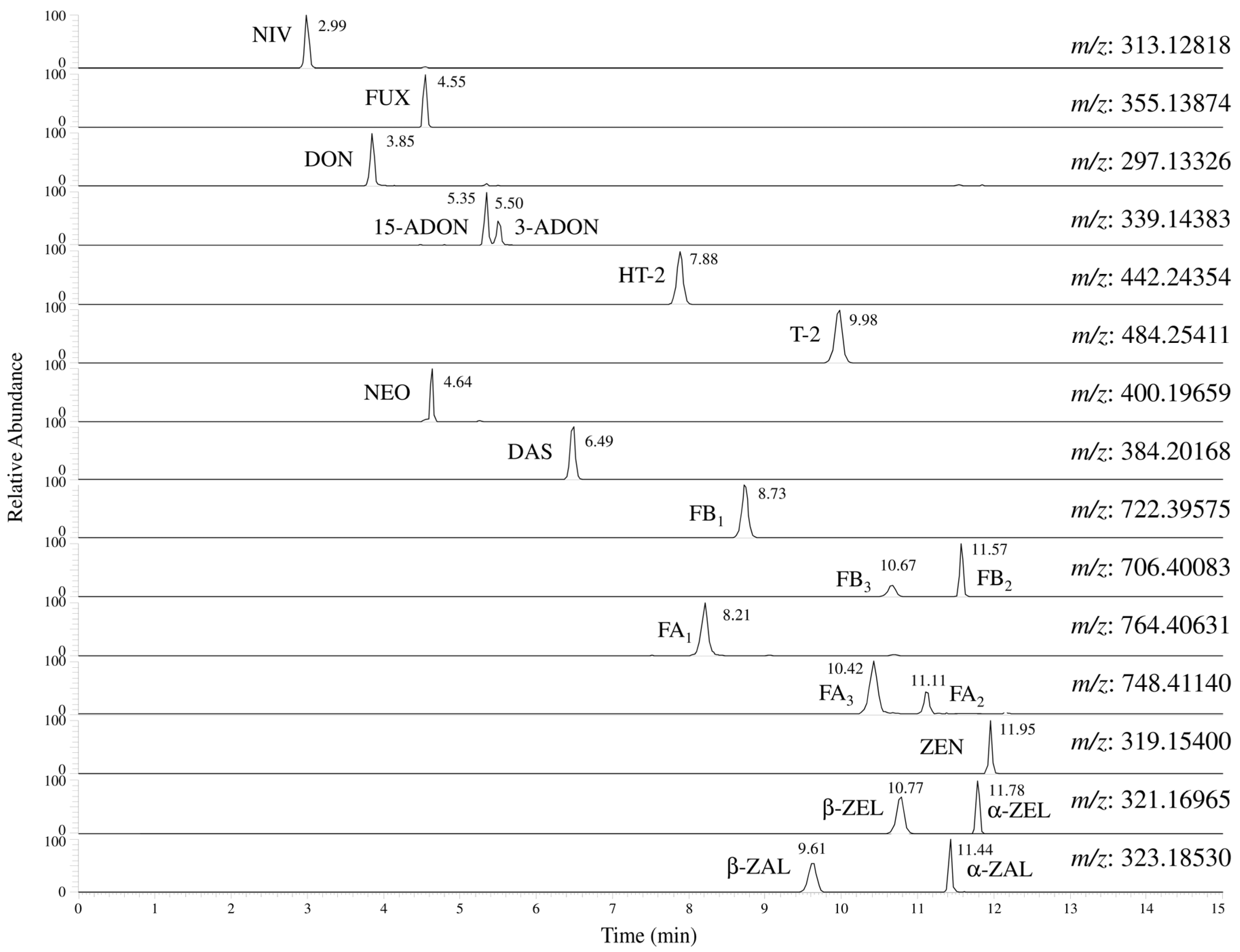

2.2. Detection of 20 Fusarium Toxins Using an LC-Orbitrap MS
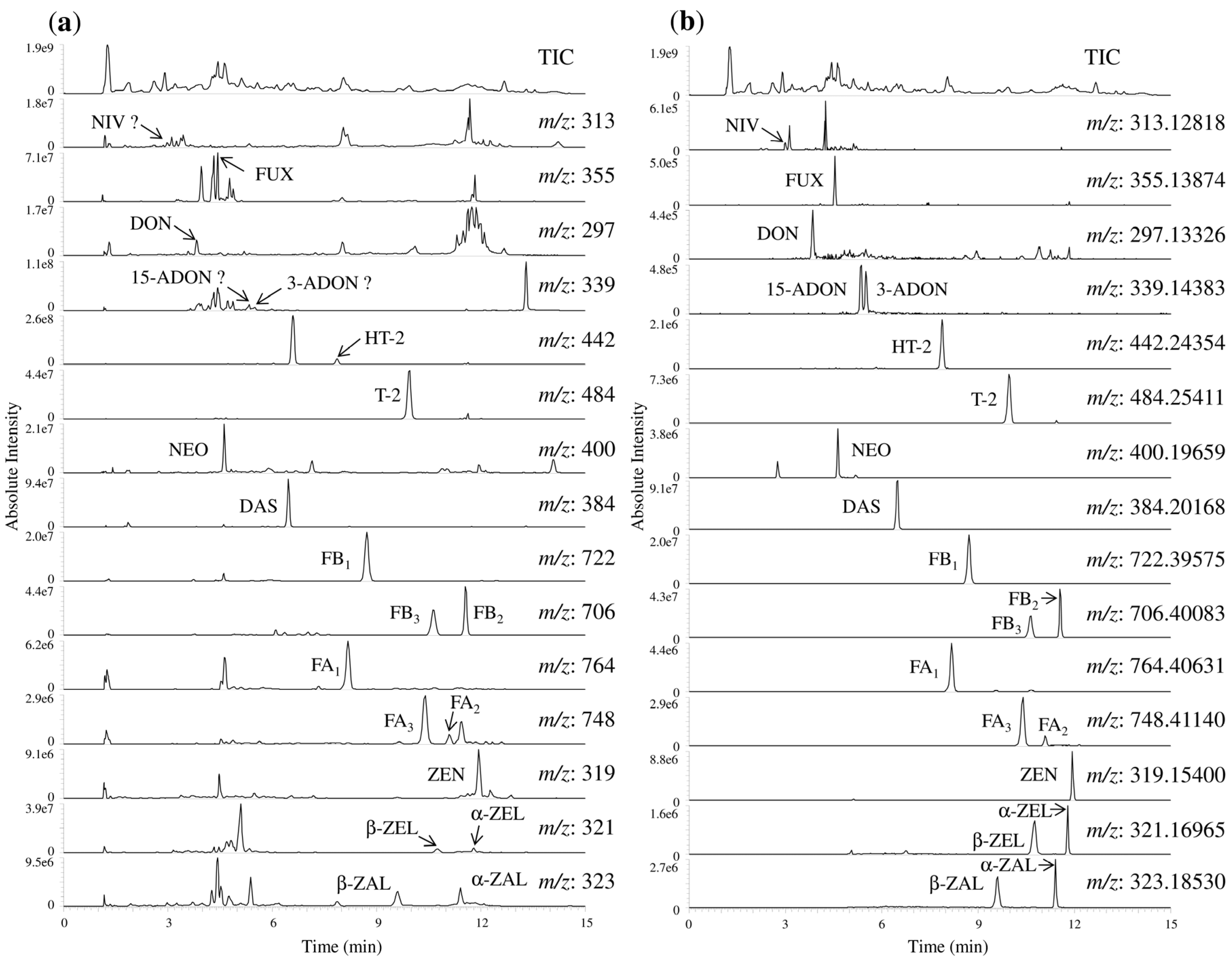
2.3. Method Validation for Determination of 20 Fusarium Toxins
2.4. Quantification of the 20 Fusarium Toxins in Cereal Samples
| Fusarium Toxins | Corn | Wheat | Barley | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Linearity (r) (a) | Repeatability (%) (b) | Recovery (%) (c) | Linearity (r) | Repeatability (%) | Recovery (%) | Linearity (r) | Repeatability (%) | Recovery (%) | ||
| Trichothecenes | NIV | 0.9997 | 2.1 | 76 | 0.9999 | 5.4 | 71 | 0.9996 | 5.1 | 78 |
| FUX | 0.9995 | 4.4 | 87 | 0.9998 | 6.2 | 93 | 0.9995 | 7.8 | 102 | |
| DON | 0.9993 | 4.2 | 80 | 0.9996 | 6.0 | 89 | 0.9968 | 4.6 | 82 | |
| 3-ADON | 0.9995 | 5.7 | 92 | 0.9992 | 5.6 | 89 | 0.9978 | 5.8 | 88 | |
| 15-ADON | 0.9989 | 2.4 | 95 | 0.9990 | 5.0 | 99 | 0.9982 | 6.8 | 96 | |
| HT-2 | 0.9999 | 1.4 | 98 | 0.9998 | 1.0 | 101 | 0.9991 | 3.4 | 98 | |
| T-2 | 0.9998 | 0.9 | 93 | 0.9994 | 1.7 | 95 | 0.9989 | 1.0 | 94 | |
| NEO | 0.9986 | 4.7 | 98 | 0.9987 | 2.9 | 99 | 0.9964 | 7.0 | 100 | |
| DAS | 0.9989 | 1.0 | 97 | 0.9979 | 1.5 | 97 | 0.9967 | 2.5 | 97 | |
| Fumonisins | FB1 | 0.9999 | 1.6 | 96 | 0.9992 | 2.7 | 85 | 0.9998 | 2.6 | 93 |
| FB2 | 0.9994 | 2.4 | 102 | 0.9985 | 3.8 | 91 | 0.9991 | 3.6 | 94 | |
| FB3 | 0.9998 | 0.8 | 104 | 0.9998 | 3.4 | 93 | 0.9997 | 3.1 | 94 | |
| FA1 | 0.9991 | 1.5 | 100 | 0.9997 | 2.3 | 97 | 0.9997 | 1.6 | 96 | |
| FA2 | 0.9999 | 11.9 | 93 | 0.9999 | 6.7 | 106 | 0.9986 | 14.7 | 98 | |
| FA3 | 0.9999 | 2.6 | 97 | 0.9996 | 2.6 | 97 | 0.9992 | 2.0 | 97 | |
| Zearalenone-group | ZEN | 0.9998 | 3.4 | 82 | 0.9995 | 4.0 | 90 | 0.9997 | 7.3 | 84 |
| α-ZEL | 0.9998 | 6.1 | 86 | 0.9994 | 2.4 | 91 | 0.9979 | 4.3 | 79 | |
| β-ZEL | 0.9993 | 3.1 | 95 | 0.9997 | 5.6 | 84 | 0.9988 | 7.6 | 92 | |
| α-ZAL | 0.9998 | 2.7 | 78 | 0.9998 | 6.4 | 97 | 0.9984 | 12.4 | 82 | |
| β-ZAL | 0.9990 | 6.8 | 86 | 0.9999 | 4.1 | 99 | 0.9984 | 6.2 | 78 | |
| Sample | Concentration of Fusarium toxins [μg/kg] | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Trichothecenes | Fumonisins | Zearalenone-group | ||||||||||||||
| NIV | FUX | DON | 3-ADON | 15-ADON | HT-2 | T-2 | NEO | DAS | FB1 | FB2 | FB3 | FA1 | FA2 | FA3 | ZEN | |
| C-1 | (a) | 63.7 | 10.6 | <5 | 373 | 70.9 | 54.8 | 34.7 | 53.7 | 5.44 | <5 | |||||
| C-2 | <5 | 15.3 | <5 | <5 | 229 | 41.4 | 19.0 | 17.6 | 23.9 | <5 | ||||||
| C-3 | 16.0 | <5 | 32.3 | 8.66 | <5 | <5 | <5 | |||||||||
| C-4 | <5 | <5 | 154 | 30.6 | 13.8 | 11.0 | 13.3 | <5 | ||||||||
| C-5 | 10.5 | <5 | 67.3 | 12.5 | 11.5 | 8.29 | 11.0 | <5 | <5 | |||||||
| C-6 | 53.3 | 14.8 | <5 | <5 | 924 | 171 | 122 | 87.8 | 168 | 11.6 | 13.7 | |||||
| C-7 | 96.1 | 18.0 | <5 | 526 | 82.9 | 60.2 | 38.5 | 66.6 | <5 | 30.2 | ||||||
| C-8 | <5 | 401 | 145 | <5 | 38.3 | <5 | <5 | 81.7 | ||||||||
| C-9 | 154 | <5 | 34.9 | 40.8 | 10.9 | <5 | <5 | <5 | 19.9 | |||||||
| C-10 | 8.39 | 135 | 5.57 | 38.1 | 413 | 45.6 | 60.5 | 34.7 | 47.3 | 7.52 | 6.79 | |||||
| C-11 | 214 | <5 | 26.1 | <5 | 1.30c) | 291 | 193 | 106 | 93.9 | 6.76 | 65.8 | |||||
| C-12 | 5.62 | 1.11 (c) | 12.6 | 47.4 | 54.8 | 14.9 | <5 | <5 | 148 | |||||||
| C-13 | 466 | 85.0 | 77.3 | 51.4 | 42.6 | 5.09 | ||||||||||
| DC-617 | <5 (b) | 4.82 (c) [4.2–6.4] (c) (d) | 372 | 29.5 | 13.9 | <5 | 2.48 (c) | 486 | 263 | 563 | 677 | 71.0 | 592 | |||
| FC-443 | 99.8 | 16.3 | <5 | <5 | <5 | 3.69 (c) [2.3–4.9] (c) | 786 [0.5–1.1] (c) | 244 [0.2–0.4] (c) | 522 | 705 | 74.9 | <5 | ||||
| ZC-327 | 2.57 (c) | 27.5 | 241 | 1.56 (c) | 291 | 150 | 262 | 378 | 30.2 | 1.72 [1.08–1.85] | ||||||
| MTC-9990 | <5 | 1.78 (c) [1.6–2.2] (c) | 10.5 | 153 | 16.2 | 6.73 | <5 | <5 | 1.14 (c) [1.0–1.6] (c) | 181 [0.15–0.25] (c) | 125 | 257 | 370 | 40.8 | 284 | |
| MTC-9999E | 23.0 | <5 | 2.16 (c) [2.1–3.1] (c) | 7.01 | 168 | 368 [350–697] | 153 [142–386] | 19.4 | <5 | 28.3 (c) [20.7–35.9] (c) | 5.39 (c) [5.2–9.0] (c) | 1.36 (c) [1.2–2.2] (c) | 2.37 | 2.61 | 182 | 323 [239–465] |
| Sample | Concentration of Fusarium Toxins [μg/kg] | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Trichothecenes | Fumonisins | Zearalenone-group | |||||||||||
| NIV | FUX | DON | 3-ADON | 15-ADON | HT-2 | T-2 | NEO | DAS | FB1 | FB2 | ZEN | ||
| Wheat | W-1 | <5 (a) | 102 | <5 | <5 | <5 | <5 | <5 | <5 | ||||
| W-2 | (b) | 35.1 | <5 | ||||||||||
| W-3 | <5 | <5 | 19.0 | ||||||||||
| W-4 | <5 | 71.6 | <5 | <5 | |||||||||
| W-5 | <5 | 405 | 21.3 | 16.9 | 10.1 | <5 | <5 | ||||||
| W-6 | 110 | 7.08 | <5 | <5 | |||||||||
| W-7 | <5 | 352 | <5 | <5 | |||||||||
| W-8 | <5 | 6.76 | 451 | 9.88 | <5 | <5 | 33.1 | ||||||
| W-9 | <5 | 12.2 | |||||||||||
| W-10 | 11.6 | 15.3 | 198 | 13.1 | 5.22 | <5 | <5 | <5 | <5 | 5.95 | |||
| W-11 | 6.99 | 25.5 | 271 | <5 | |||||||||
| W-12 | 10.6 | 16.0 | 272 | 12.4 | <5 | <5 | 9.85 | ||||||
| Barley | B-1 | 7.41 | |||||||||||
| B-2 | <5 | 6.56 | 34.1 | ||||||||||
| B-3 | <5 | ||||||||||||
| B-4 | <5 | ||||||||||||
| B-5 | <5 | ||||||||||||
| B-6 | <5 | <5 | 116 | 8.32 | <5 | <5 | 60.9 | ||||||
| B-7 | <5 | ||||||||||||
| B-8 | 5.93 | <5 | <5 | <5 | <5 | <5 | |||||||
| B-9 | 77.7 | <5 | <5 | <5 | <5 | 0.43 | |||||||
3. Experimental Section
3.1. Samples, Chemicals, and Reagents
3.2. Sample Preparation
3.3. LC-Orbitrap MS Analysis
3.4. Method Validation
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Antonissen, G.; Martel, A.; Pasmans, F.; Ducatelle, R.; Verbrugghe, E.; Vandenbroucke, V.; Li, S.; Haesebrouck, F.; Van Immerseel, F.; Croubels, S. The impact of Fusarium mycotoxins on human and animal host susceptibility to infectious diseases. Toxins 2014, 6, 430–452. [Google Scholar] [CrossRef] [Green Version]
- Marroquín-Cardona, A.G.; Johnson, N.M.; Phillips, T.D.; Hayes, A.W. Mycotoxins in a changing global environment—A review. Food Chem. Toxicol. 2014, 69, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Hussein, H.S.; Brasel, J.M. Toxicity, metabolism, and impact of mycotoxins on humans and animals. Toxicol 2001, 167, 101–134. [Google Scholar] [CrossRef]
- D’Mello, J.P.F.; Placinta, C.M.; Macdonald, A.M.C. Fusarium mycotoxins: A review of global implications for animal health, welfare and productivity. Anim. Feed Sci. Technol. 1999, 80, 183–205. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). Scientific Opinion on the risks to human and animal health related to the presence of beauvericin and enniatins in food and feed: EFSA Panel on Contaminants in the Food Chain (CONTAM). EFSA J. 2014, 12, 3802. [Google Scholar]
- Kamala, A.; Ortiz, J.; Kimanya, M.; Haesaert, G.; Donoso, S.; Tiisekwa, B.; de Meulenaer, B. Multiple mycotoxin co-occurrence in maize grown in three agro-ecological zones of Tanzania. Food Control. 2015, 54, 208–215. [Google Scholar] [CrossRef]
- Streit, E.; Naehrer, K.; Rodrigues, I.; Schatzmayr, G. Mycotoxin occurrence in feed and feed raw materials worldwide: Long-term analysis with special focus on Europe and Asia. J. Sci. Food Agric. 2013, 93, 2892–2899. [Google Scholar] [CrossRef] [PubMed]
- Perkowski, J.; Stuper, K.; Buśko, M.; Góral, T.; Jeleń, H.; Wiwart, M.; Suchowilska, E. A comparison of contents of group A and B trichothecenes and microbial counts in different cereal species. Food Addit. Contam. B 2012, 5, 151–159. [Google Scholar] [CrossRef]
- Alkadri, D.; Rubert, J.; Prodi, A.; Pisi, A.; Mañes, J.; Soler, C. Natural co-occurrence of mycotoxins in wheat grains from Italy and Syria. Food Chem. 2014, 157, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Tamura, M.; Matsumoto, K.; Watanabe, J.; Iida, J.; Nagatomi, Y.; Mochizuki, N. Minimization of carryover for high-throughput liquid chromatography with tandem mass spectrometry analysis of 14 mycotoxins in corn grits. J. Sep. Sci. 2014, 37, 1552–1560. [Google Scholar] [CrossRef] [PubMed]
- JECFA. Evaluation of certain mycotoxins in food. Fifty-sixth report of the Joint FAO/WHO Export Committee on Food Additives, WHO Technical Report Committee on Food Additives. 2002. Available online: http://whqlibdoc.who.int/trs/WHO_TRS_906.pdf?ua=1 (accessed on 7 May 2015).
- JECFA. Safety evaluation of certain contaminants in food. Seventy-second meeting of the Joint FAO/WHO Export Committee on Food Additives. 2011. Available online: http://whqlibdoc.who.int/publications/2011/9789241660631_eng.pdf (accessed on 7 May 2015).
- European Commission. Commission recommendation of 27 March 2013 on the presence of T-2 and HT-2 toxins in cereals and cereal products (2013/165/EU). Off. J. Eur. Union. 2013, L91, pp. 12–15. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32013H0165&qid=1424778563603&from=EN (accessed on 24 March 2015).
- European Commission. Commission regulation (EC) No 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs. Consolidated version 01.09.2014. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:02006R1881-20140901&qid=1424778289701&from=EN (accessed on 24 March 2015).
- Food and Drug Administration (FDA). FDA regulatory guidance for mycotoxins. Available online: http://www.ngfa.org/wp-content/uploads/NGFAComplianceGuide-FDARegulatoryGuidanceforMycotoxins8-2011.pdf (accessed on 20 February 2015).
- IARC. Monographs on the Evaluation of Carcinogenic Risks to Humans; IARC: Lyon, France, 2002; Volume 82, pp. 301–366. Available online: http://monographs.iarc.fr/ENG/Monographs/vol82/mono82.pdf (accessed on 7 May 2015).
- CODEX. Joint FAO/WHO food standards programme CODEX alimentarius commission 37th Session, Geneva, Switzerland, 14–18 July 2014. Available online: ftp://ftp.fao.org/Codex/Reports/Reports_2014/REP14_CFe.pdf (accessed on 7 May 2015).
- Tamura, M.; Mochizuki, N.; Nagatomi, Y.; Toriba, A.; Hayakawa, K. Characterization of fumonisin A-series by high-resolution liquid chromatography-orbitrap mass spectrometry. Toxins 2014, 6, 2580–2593. [Google Scholar] [CrossRef] [PubMed]
- Tamura, M.; Mochizuki, N.; Nagatomi, Y.; Harayama, K.; Toriba, A.; Hayakawa, K. Identification and quantification of fumonisin A1, A2, and A3 in corn by high-resolution liquid chromatography-orbitrap mass spectrometry. Toxins 2015, 7, 582–592. [Google Scholar] [CrossRef] [PubMed]
- Van der Westhuizen, L.; Shephard, G.S.; Snyman, S.D.; Abel, S.; Swanevelder, S.; Gelderblom, W.C.A. Inhibition of sphingolipid biosynthesis in rat primary hepatocyte cultures by fumonisin B1 and other structurally related compounds. Food Chem. Toxicol. 1998, 36, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Zinedine, A.; Soriano, J.M.; Moltó, J.C.; Mañes, J. Review on the toxicity, occurrence, metabolism, detoxification, regulations and intake of zearalenone: An oestrogenic mycotoxin. Food Chem. Toxicol. 2007, 45, 1–18. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority (EFSA). Scientific Opinion on the risks for public health related to the presence of zearalenone in food: EFSA Panel on Contaminants in the Food Chain. EFSA J. 2011, 9, 2197. [Google Scholar]
- Adejumo, T.O.; Hettwer, U.; Karlovsky, P. Survey of maize from south-western Nigeria for zearalenone, α - and β –zearalenols, fumonisn B1 and eniatines produced by Fusarium species. Food Addit. Comtam. 2007, 24, 993–1000. [Google Scholar] [CrossRef]
- Shier, W.T.; Shier, A.C.; Xie, W.; Mirocha, C.J. Structure-activity relationships for human estrogenic activity in zearalenone mycotoxins. Toxicon 2001, 39, 1435–1438. [Google Scholar] [CrossRef] [PubMed]
- Omemu, A.M.; Bankole, M.O.; Oyewole, O.B.; Akintokun, A.K. Yeasts and molds associated with ogi-A cereal based weaning food during storage. Res. J. Microbiol. 2007, 2, 141–148. [Google Scholar] [CrossRef]
- Brodehl, A.; Möller, A.; Kunte, H.J.; Koch, M.; Maul, R. Biotransformation of the mycotoxin zearalenone by fungi of the genera Rhizopus and Aspergillus. FEMS Microbiol. Lett. 2014, 359, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Rahmani, A.; Jinap, S.; Soleimany, F. Qualitative and quantitative analysis of mycotoxins. Compr. Rev. Food Sci. Food Saf. 2009, 8, 202–251. [Google Scholar] [CrossRef]
- Pereira, V.L.; Fernandes, J.O.; Cunha, S.C. Mycotoxins in cereals and related foodstuffs: A review in occurrence and recent methods of analysis. Trends Food Sci. Technol. 2014, 36, 96–136. [Google Scholar] [CrossRef]
- Berthiller, F.; Schuhmacher, R.; Buttinger, G.; Krska, R. Rapid simultaneous determination of major type A- and B-trichothecenes as well as zearalenone in maize by high performance liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2005, 1062, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Mizutani, K.; Nagatomi, Y.; Mochiduki, N. Metabolism of zearalenone in the course of beer fermentation. Toxins 2011, 3, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, H.; Naito, S.; Kitani, Y.; Ito, Y.; Aoyama, Y.; Koyama, M.; Hiejima, Y.; Nakamura, K.; Miyazaki, H.; Morita, H.I.; et al. Harmonized collaborative validation of a simultaneous and multiple determination method for nivalenol, deoxynivalenol, T-2 toxin, HT-2 toxin, and zearalenone in wheat barley by liquid chromatography coupled to tandem mass spectrometry (LC-MS/MS). J. Anal. Bioanal. Tech. 2014, S6:002, 1–9. [Google Scholar]
- Senyuva, H.Z.; Gilbert, J.; Türköz, G.; Leeman, D.; Donnelly, C. Analysis of deoxynivalenol, zearalenone, T-2, and HT-2 toxins in animal feed by LC/MS/MS—A critical comparison of immunoaffinity column cleanup with no cleanup. J. AOAC Int. 2012, 95, 1701–1708. [Google Scholar] [CrossRef]
- Liao, C.D.; Wong, J.W.; Zhang, K.; Hayward, D.G.; Lee, N.S.; Trucksess, M.W. Multi-mycotoxin analysis of finished grain and nut products using high-performance liquid chromatography-triple-quadrupole mass spectrometry. J. Agric. Food Chem. 2013, 61, 4771–4782. [Google Scholar] [CrossRef]
- Sulyok, M.; Krska, R.; Schuhmacher, R. A liquid chromatography/tandem mass spectrometric multi-mycotoxin method for the quantification of 87 analytes and its application to semi-quantitative screening of moldy food samples. Anal. Bioanal. Chem. 2007, 389, 1505–1523. [Google Scholar] [CrossRef]
- Zachariasova, M.; Lacina, O.; Malachova, A.; Kostelanska, M.; Poustka, J.; Godula, M.; Hajslova, J. Novel approaches in analysis of Fusarium mycotoxins in cereals employing ultra performance liquid chromatography coupled with high resolution mass spectrometry. Anal. Chem. Acta 2010, 662, 51–61. [Google Scholar] [CrossRef]
- Zachariasova, M.; Cajka, T.; Godula, M.; Malachova, A.; Veprikova, Z.; Hajslova, J. Analysis of multiple mycotoxins in beer employing (ultra)-high-resolution mass spectrometry. Rapid Contam. Mass Spectrom. 2010, 24, 3357–3367. [Google Scholar] [CrossRef]
- Shao, L.K.; Locke, D.C. Determination of paclitaxel and related taxanes in bulk drug and injectable dosage forms by reversed phase liquid chromatography. Anal. Chem. 1997, 69, 2008–2016. [Google Scholar] [CrossRef] [PubMed]
- Grebenstein, N.; Frank, J. Rapid baseline-separation of all eight tocopherols and tocotrienols by reverse-phase liquid-chromatography with a solid-core pentafluorophenyl column and their sensitive quantification in plasma and liver. J. Chromatogr. A 2012, 1243, 39–46. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Method validation and quality control procedures for pesticide residues analysis in food and feed. Available online: http://ec.europa.eu/food/plant/protection/resources/qualcontrol_en.pdf (accessed on 18 February 2015).
- Anastassiades, M.; Lehotay, S.J.; Stajnbaher, D.; Schenck, F.J. Fast and easy multiresidue method employing acetonitrile extraction/partitioning and “dispersive solid-phase extraction” for the determination of pesticide residues in produce. J. AOAC Int. 2003, 86, 412–431. [Google Scholar] [PubMed]
- Tamura, M.; Takahashi, A.; Uyama, A.; Mochizuki, N. A method for multiple mycotoxin analysis in wines by solid phase extraction and multifunctional cartridge purification, and ultra-high-performance liquid chromatography coupled to tandem mass spectrometry. Toxins 2012, 4, 476–486. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tamura, M.; Mochizuki, N.; Nagatomi, Y.; Harayama, K.; Toriba, A.; Hayakawa, K. A Method for Simultaneous Determination of 20 Fusarium Toxins in Cereals by High-Resolution Liquid Chromatography-Orbitrap Mass Spectrometry with a Pentafluorophenyl Column. Toxins 2015, 7, 1664-1682. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins7051664
Tamura M, Mochizuki N, Nagatomi Y, Harayama K, Toriba A, Hayakawa K. A Method for Simultaneous Determination of 20 Fusarium Toxins in Cereals by High-Resolution Liquid Chromatography-Orbitrap Mass Spectrometry with a Pentafluorophenyl Column. Toxins. 2015; 7(5):1664-1682. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins7051664
Chicago/Turabian StyleTamura, Masayoshi, Naoki Mochizuki, Yasushi Nagatomi, Koichi Harayama, Akira Toriba, and Kazuichi Hayakawa. 2015. "A Method for Simultaneous Determination of 20 Fusarium Toxins in Cereals by High-Resolution Liquid Chromatography-Orbitrap Mass Spectrometry with a Pentafluorophenyl Column" Toxins 7, no. 5: 1664-1682. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins7051664





