Rapid Detection of Escherichia coli O157 and Shiga Toxins by Lateral Flow Immunoassays
Abstract
:1. Introduction
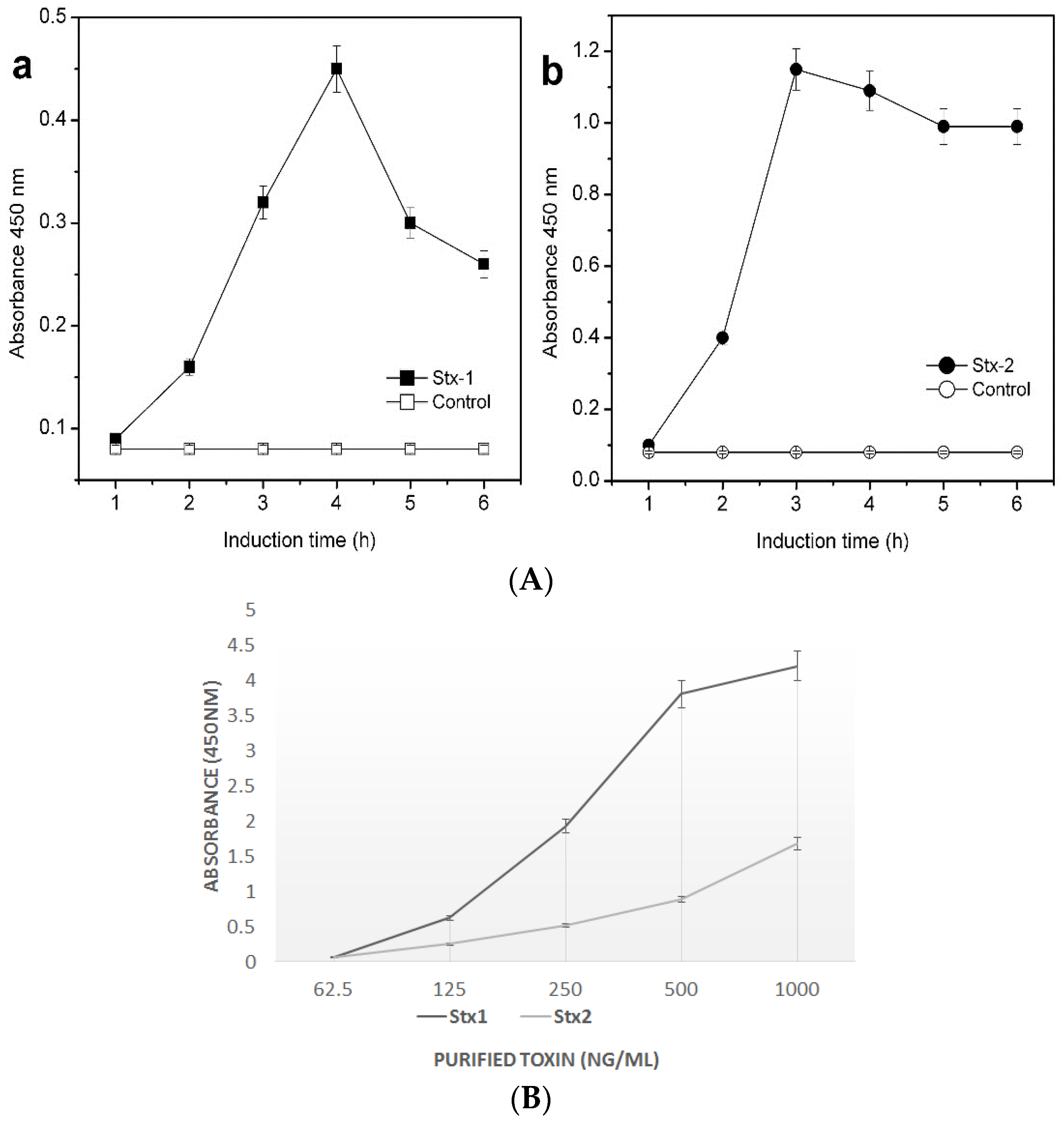
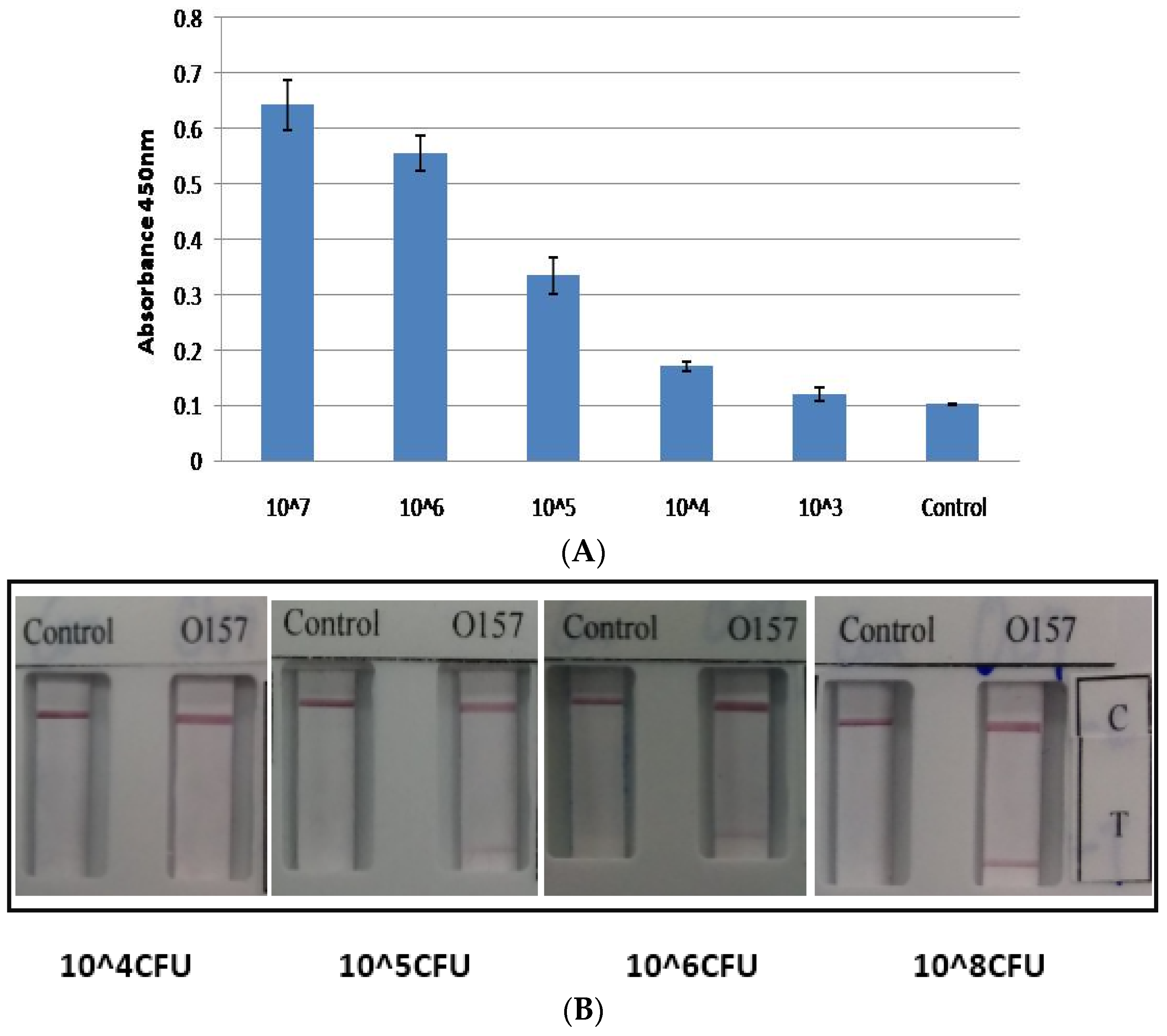
2. Results and Discussion
3. Materials and Methods
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mead, P.S.; Slutsker, L.; Dietz, V.; McCaig, L.F.; Bresee, J.S.; Shapiro, C.; Griffin, P.M.; Tauxe, R.V. Food-related illness and death in the United States. Emerg. Infect. Dis. 1999, 5, 607–625. [Google Scholar] [CrossRef] [PubMed]
- Scallan, E.; Hoekstra, R.M.; Angulo, F.J.; Tauxe, R.V.; Widdowson, M.A.; Roy, S.L.; Jones, J.L.; Griffin, P.M. Foodborne illness acquired in the United States-major pathogens. Emerg. Infect. Dis. 2011, 17, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Brooks, J.T.; Sowers, E.G.; Wells, J.G.; Greene, K.D.; Griffin, P.M.; Hoekstra, R.M.; Strockbine, N.A. Non-0157 Shiga toxin producing Escherichia coli infections in the United States, 1983–2002. J. Infect. Dis. 2005, 192, 1422–1429. [Google Scholar] [CrossRef] [PubMed]
- USDA Food Safety and Inspection Service. Risk Profile for Pathogenic Non-O157 Shiga Toxin-Producing Escherichia coli (Non-O157 STEC); USDA Food Safety and Inspection Service: Washington, DC, USA, 2012.
- Fremaux, B.; Prigent-Combaret, C.; Vernozy-Rozand, C. Long-term survival of Shiga toxin-producing Escherichia coli in cattle effluents and environment: An updated review. Vet. Microbiol. 2008, 132, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Gyles, C.L. Shiga toxin-producing Escherichia coli: An overview. J. Anim. Sci. 2007, 85, E45–E62. [Google Scholar] [CrossRef] [PubMed]
- Orth, D.; Grif, K.; Khan, A.B.; Naim, A.; Dierich, M.P.; Wurzner, R. Shiga toxin genotype rather than the amount of Shiga toxin or the cytotoxicity of Shiga toxin in vitro correlates with the appearance of hemolytic uremic syndrome. Diagn. Microbiol. Infect. Dis. 2007, 59, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; McDaniel, A.D.; Wolf, L.E.; Keusch, G.T.; Waldor, M.K.; Acheson, D.W. Quinolone antibiotics induce Shiga toxin-encoding bacteriophages, toxin production, and death in mice. J. Infect. Dis. 2000, 181, 664–670. [Google Scholar] [CrossRef] [PubMed]
- Thorpe, C.M.; Hurley, B.P.; Lincicome, L.L.; Jacewicz, M.S.; Keusch, G.T.; Acheson, D.W.K. Shiga toxins stimulate secretion of interleukin-8 from intestinal epithelial cells. Infect. Immun. 1999, 67, 5985–5993. [Google Scholar] [PubMed]
- DebRoy, C.; Roberts, E.; Valadez, A.M.; Dudley, E.G.; Cutter, C.N. Detection of Shiga toxin-producing Escherichia coli O26, O45, O103, O111, O113, O121, O145, and O157 serogroups by multiplex PCR of the wzx gene of the O-antigen gene cluster. Foodborne Pathog. Dis. 2011, 8, 651–652. [Google Scholar] [CrossRef] [PubMed]
- Shridhar, P.B.; Noll, L.W.; Shi, X.; An, N.; Cernicchiaro, N.; Renter, D.G.; Nagaraja, T.G.; Bai, J. Multiplex quantitative PCR assays for the detection and quantification of the six major non-O157 Escherichia coli serogroups in cattle feces. J. Food Prot. 2016, 79, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Margot, H.; Cernela, N.; Iversen, C.; Zweifel, C.; Stephan, R. Evaluation of seven different commercially available real-time PCR assays for detection of Shiga toxin 1 and 2 gene subtypes. J. Food Prot. 2013, 76, 871–873. [Google Scholar] [CrossRef] [PubMed]
- Kehl, K.S.; Havens, P.; Behnke, C.E.; Acheson, D.W. Evaluation of the premier EHEC assay for detection of Shiga toxin-producing Escherichia coli. J. Clin. Microbiol. 1997, 35, 2051–2054. [Google Scholar] [PubMed]
- Hegde, N.V.; Hulet, R.; Jayarao, B.M.; Muldoon, M.; Lindpaintner, K.; Kapur, V.; DebRoy, C. Detection of the top six non-O157 Shiga toxin-producing Escherichia coli O groups by ELISA. Foodborne Pathog. Dis. 2012, 9, 1044–1048. [Google Scholar] [CrossRef] [PubMed]
- Ashkenazi, S.; Cleary, T.G. Rapid method to detect Shiga toxin and Shiga-like toxin I based on binding to globotriosyl ceramide (Gb3), their natural receptor. J. Clin. Microbiol. 1989, 27, 1145–1150. [Google Scholar] [PubMed]
- Teel, L.D.; Daly, J.A.; Jerris, R.C.; Maul, D.; Svanas, G.; O’Brien, A.D.; Park, C.H. Rapid detection of Shiga toxin-producing Escherichia coli by optical immunoassay. J. Clin. Microbiol. 2007, 45, 3377–3380. [Google Scholar] [CrossRef] [PubMed]
- Skinner, C.; Patfield, S.; Stanker, L.H.; Fratamico, P.; He, X. New high-affinity 4 monoclonal antibodies against Shiga toxin 1 facilitate the detection of hybrid Stx1/Stx2 in vivo. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- He, X.; McMahon, S.; Skinner, C.; Merrill, P.; Scotcher, M.C.; Stanker, L.H. Development and characterization of monoclonal antibodies against Shiga toxin 2 and their application for toxin detection in milk. J. Immunol. Methods 2013, 389, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Ching, K.H. Lateral Flow Immunoassay. Methods Mol. Biol. 2015, 1318, 127–137. [Google Scholar] [PubMed]
- Chen, M.; Yu, Z.; Liu, D.; Peng, T.; Liu, K.; Wang, S.; Xiong, Y.; Wei, H.; Xu, H.; Lai, W. Dual gold nanoparticle late flow immunoassay for sensitive detection of Escherichia coli O157:H7. Anal. Chim. Acta 2015, 876, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Ching, K.H.; He, X.; Stanker, L.H.; Lin, A.V.; McGarvey, J.A.; Hnasko, R. Detection of shiga toxins by lateral flow assay. Toxins 2015, 7, 1163–1173. [Google Scholar] [CrossRef] [PubMed]
- Schramm, W.; Angulo, G.B.; Torres, P.C.; Burgess-Cassler, A. A simple saliva-based test for detecting antibodies to human immunodeficiency virus. Clin. Diagn. Lab. Immunol. 1999, 6, 577–580. [Google Scholar] [PubMed]
- Fernandez-Sanchez, C.; McNeil, C.J.; Rawson, K.; Nilsson, O. Disposable noncompetitive immunosensor for free and total prostate-specific antigen based on capacitance measurement. Anal. Chem. 2004, 76, 5649–5656. [Google Scholar] [CrossRef] [PubMed]
- Elkjaer, M.; Burisch, J.; Hansen, V.V.; Kristensen, B.D.; Jensen, K.S.; Munkholm, P. A new rapid home test for faecal calprotectin in ulcerative colitis. Ailm. Pharmacol. Ther. 2010, 31, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Laderman, E.I.; Whitworth, E.; Dumaual, E.; Jones, M.; Hudak, A.; Hogrefe, W.; Carney, J.; Groen, J. Rapid sensitive, and specific lateral-flow immunochromatographic point-of-care device for detection of herpes simplex virus type 2-specific immunoglobulin G antibodies in serum and whole blood. Clin. Vaccine Immunol. 2008, 15, 159–163. [Google Scholar] [CrossRef] [PubMed]
- Cui, S.; Chen, C.; Tong, G. A simple and rapid immuno-chromatographic strip test for monitoring antibodies to H5 subtype avian influenza virus. J. Virol. Methods 2008, 152, 102–105. [Google Scholar] [CrossRef] [PubMed]
- Khreich, N.; Lamourette, P.; Boutal, H.; Devilliers, K.; Creminon, C.; Volland, H. Detection of Staphylococcus enterotoxin B using fluorescent immunoliposomes as label for immunochromatographic testing. Anal. Biochem. 2008, 377, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Lu, Y.T.; Zhu, J.; Hong, J.B.; Li, B.; Zhou, J.; Gong, D.; Montoya, A. Nanocolloidal gold-based immunoassay for the detection of the N-methylcarbamate pesticide carbofuran. J. Agric. Food Chem. 2004, 52, 4355–4359. [Google Scholar] [CrossRef] [PubMed]
- Delmulle, B.S.; Saeger, S.D.; Sibanda, L.; Barna-Vetro, I.; Peteghem, C.H.V. Development of an immunoassay-based lateral flow dipstick for the rapid detection of aflatoxin B1 in pig feed. J. Agric. Food Chem. 2005, 53, 3364–3368. [Google Scholar] [CrossRef] [PubMed]
- Molinelli, A.; Grossalber, K.; Führer, M.; Baumgartner, S.; Sulyok, M.; Krska, R. Development of qualitative and semiquantitative immunoassay-based rapid strip tests for the detection of T-2 toxin in wheat and oat. J. Agric. Food Chem. 2008, 56, 2589–2594. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Chen, W.; Lu, Y.; Cheng, G. Development of an immunochromatographic for the rapid detection of bromoxynil in water. Environ. Pollut. 2008, 156, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Ørskov, I.; Ørskov, F.; Jann, B.; Jann, K. Serology, chemistry and genetics of O and K antigens of Escherichia coli. Bacteriol. Rev. 1977, 41, 667–710. [Google Scholar] [PubMed]


| Serial # | Bacteria | ID#/ECRC # | Serotype | stx1 | stx2 (Type) | Expression of Stx |
|---|---|---|---|---|---|---|
| 1 | E. coli | 0.1304 | O157:H7 | + | − | + |
| 2 | E. coli | 90.2281 | O157:H7 | + | − | + |
| 3 | E. coli | 96.0428 | O157:H7 | + | − | + |
| 4 | E. coli | 0.1288 | O157:H7 | + | + (a,c) | + |
| 5 | E. coli | 0.1292 | O157:H7 | + | + (a) | + |
| 6 | E. coli | 3.0190 | O157:H7 | + | − | + |
| 7 | E. coli | 0.1302 | O26:NM | + | − | + |
| 8 | E. coli | 5.2217 | O26:H11 | + | − | + |
| 9 | E. coli | 7.3964 | O26:H11 | + | − | + |
| 10 | E. coli | 8.0176 | O26:H30 | + | − | + |
| 11 | E. coli | 77.0044 | O26:HNM | + | − | + |
| 12 | E. coli | 10.2529 | O103:H2 | + | − | + |
| 13 | E. coli | 6.1623 | O103:H36 | + | + (a,c) | + |
| 14 | E. coli | 3.2605 | O103:H2 | + | − | + |
| 15 | E. coli | 9.0108 | O103:H2 | + | − | + |
| 16 | E. coli | 85.1983 | O103:H21 | + | − | + |
| 17 | E. coli | 0.0320 | O111:H? | + | − | + |
| 18 | E. coli | 0.1079 | O111:NM | + | − | + |
| 19 | E. coli | 0.1481 | O111:H? | + | − | + |
| 20 | E. coli | 5.0959 | O121:H19 | − | + (a) | + |
| 21 | E. coli | 7.1636 | O121:H19 | − | + (a) | + |
| 22 | E. coli | 6.1598 | O145:H+ | − | + (a) | + |
| 23 | E. coli | 95.1167 | O145:HNM | − | + (c) | + |
| 24 | E. coli | K12 | − | − | − | |
| 25 | Citrobacter freundii | ATCC 8090 | − | − | − | |
| 26 | Enterobacter cloacae | 15.0057 | − | − | − | |
| 27 | Hafnia alvei | ATCC 29926 | − | − | − | |
| 28 | Klebsiella pneumoniae | ATCC 27736 | − | − | − | |
| 29 | Proteus vulgaris | 15.0061 | − | − | − | |
| 30 | Salmonella enterica sv Enteritidis | 15.0060 | − | − | − | |
| 31 | Salmonella enterica sv Typhi | 15.0056 | − | − | − | |
| 32 | Serratia marcescens | ATCC 13880 | − | − | − | |
| 33 | Shigella boydii | 15.0055 | − | − | − | |
| 34 | Shigella flexneri | 15.0059 | − | − | − |
| Assay | Status | Reference Specimen of Known Status | Sensitivity | Specificity | |
|---|---|---|---|---|---|
| Positive (n) | Negative (n) | ||||
| Stx1 and Stx2 | Positive | 23 | 0 | 23/23 = 100% | 11/11 = 100% |
| Negative | 0 | 11 | |||
| O157 | Positive | 10 | 0 | 10/10 = 100% | 184/184 = 100% |
| Negative | 0 | 184 | |||
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, J.; Katani, R.; Li, L.; Hegde, N.; Roberts, E.L.; Kapur, V.; DebRoy, C. Rapid Detection of Escherichia coli O157 and Shiga Toxins by Lateral Flow Immunoassays. Toxins 2016, 8, 92. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins8040092
Wang J, Katani R, Li L, Hegde N, Roberts EL, Kapur V, DebRoy C. Rapid Detection of Escherichia coli O157 and Shiga Toxins by Lateral Flow Immunoassays. Toxins. 2016; 8(4):92. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins8040092
Chicago/Turabian StyleWang, Jinliang, Robab Katani, Lingling Li, Narasimha Hegde, Elisabeth L. Roberts, Vivek Kapur, and Chitrita DebRoy. 2016. "Rapid Detection of Escherichia coli O157 and Shiga Toxins by Lateral Flow Immunoassays" Toxins 8, no. 4: 92. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins8040092





