Response of Cladocera Fauna to Heavy Metal Pollution, Based on Sediments from Subsidence Ponds Downstream of a Mine Discharge (S. Poland)
Abstract
:1. Introduction
2. Materials and Methods
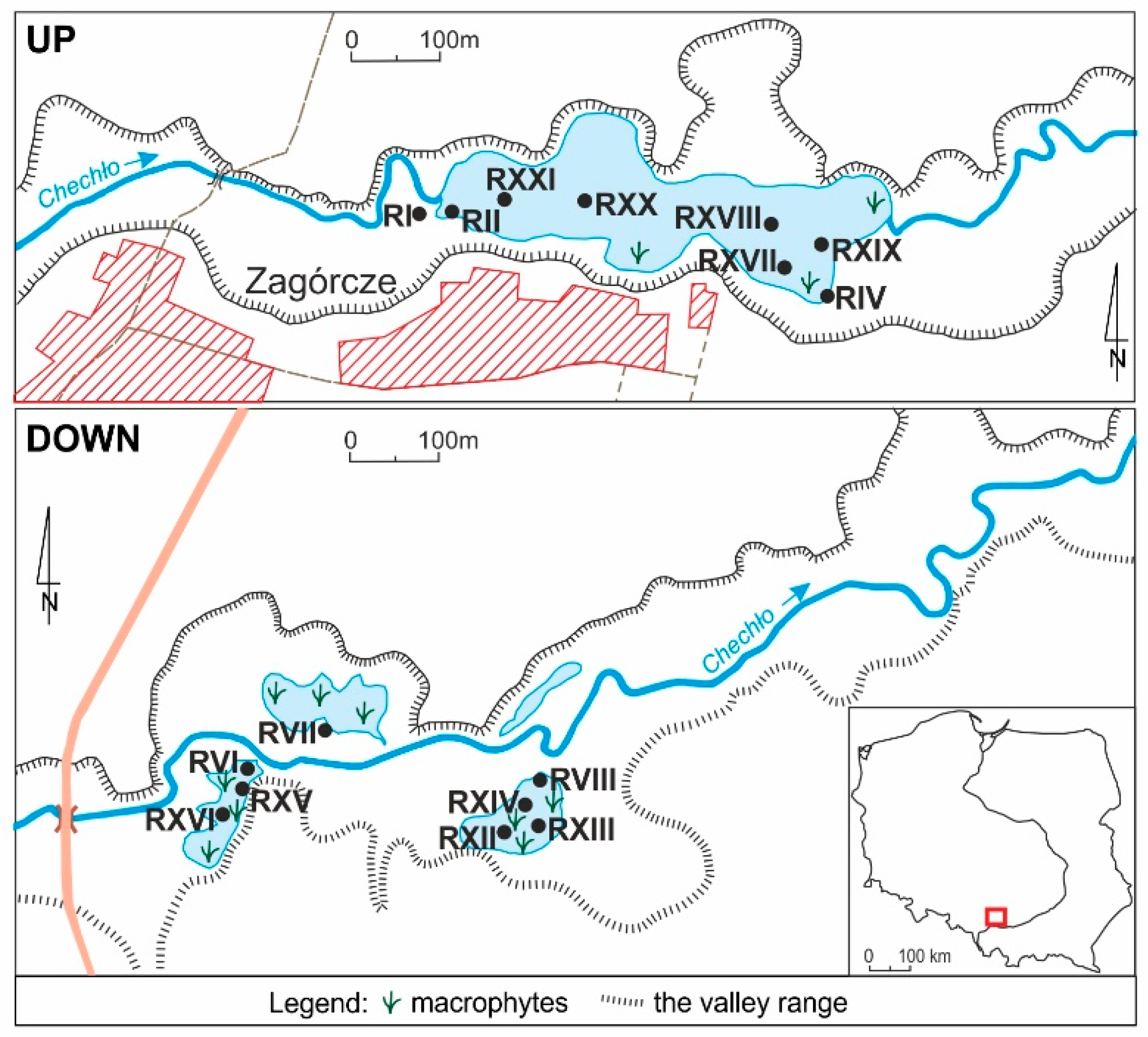
2.1. Study Area
2.2. Sampling and Measurements
2.3. Statistical Analyses
3. Results
3.1. Sediment—Heavy Metals Contamination Analysis
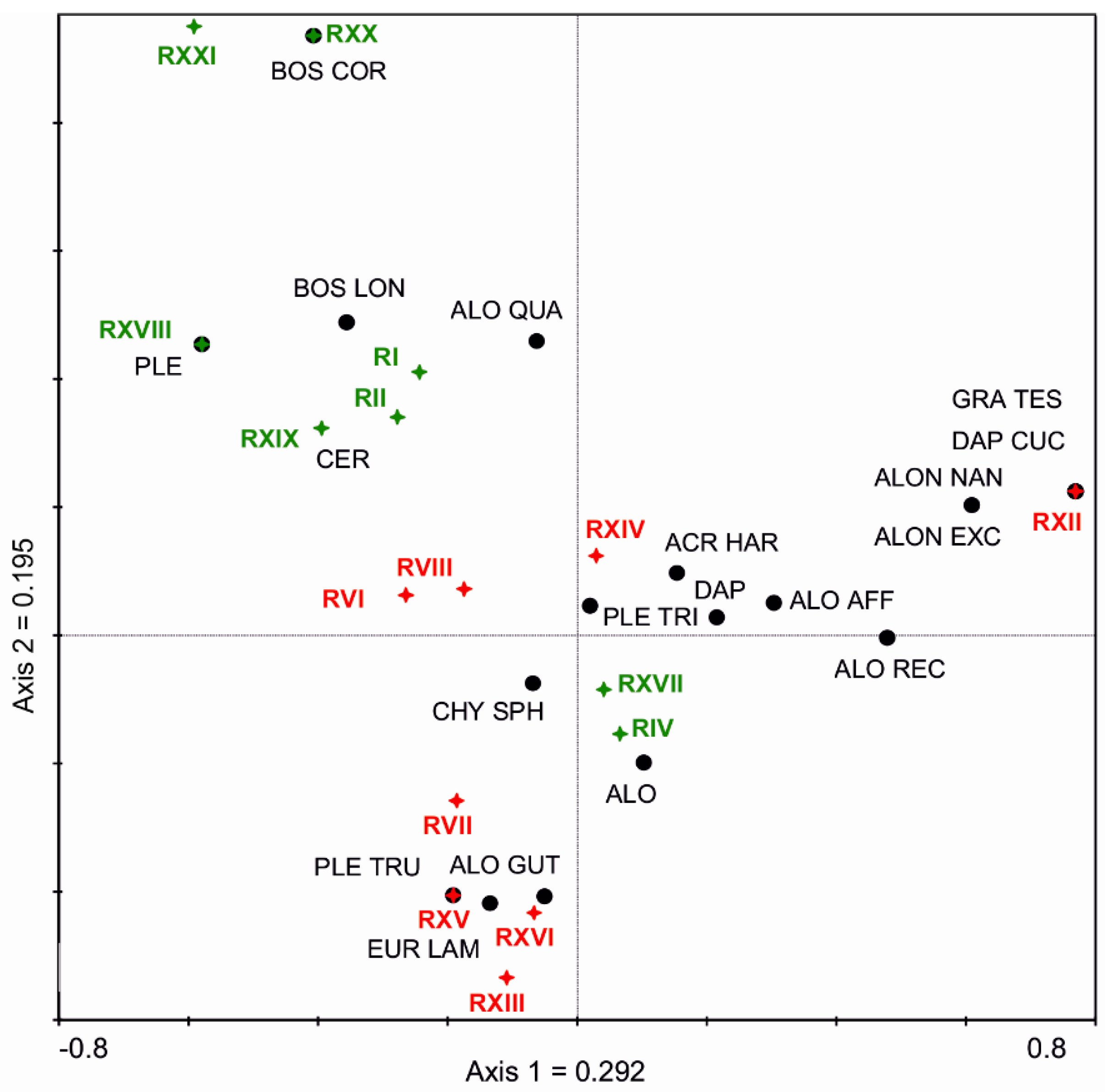
3.2. Subfossil Cladocera Analysis
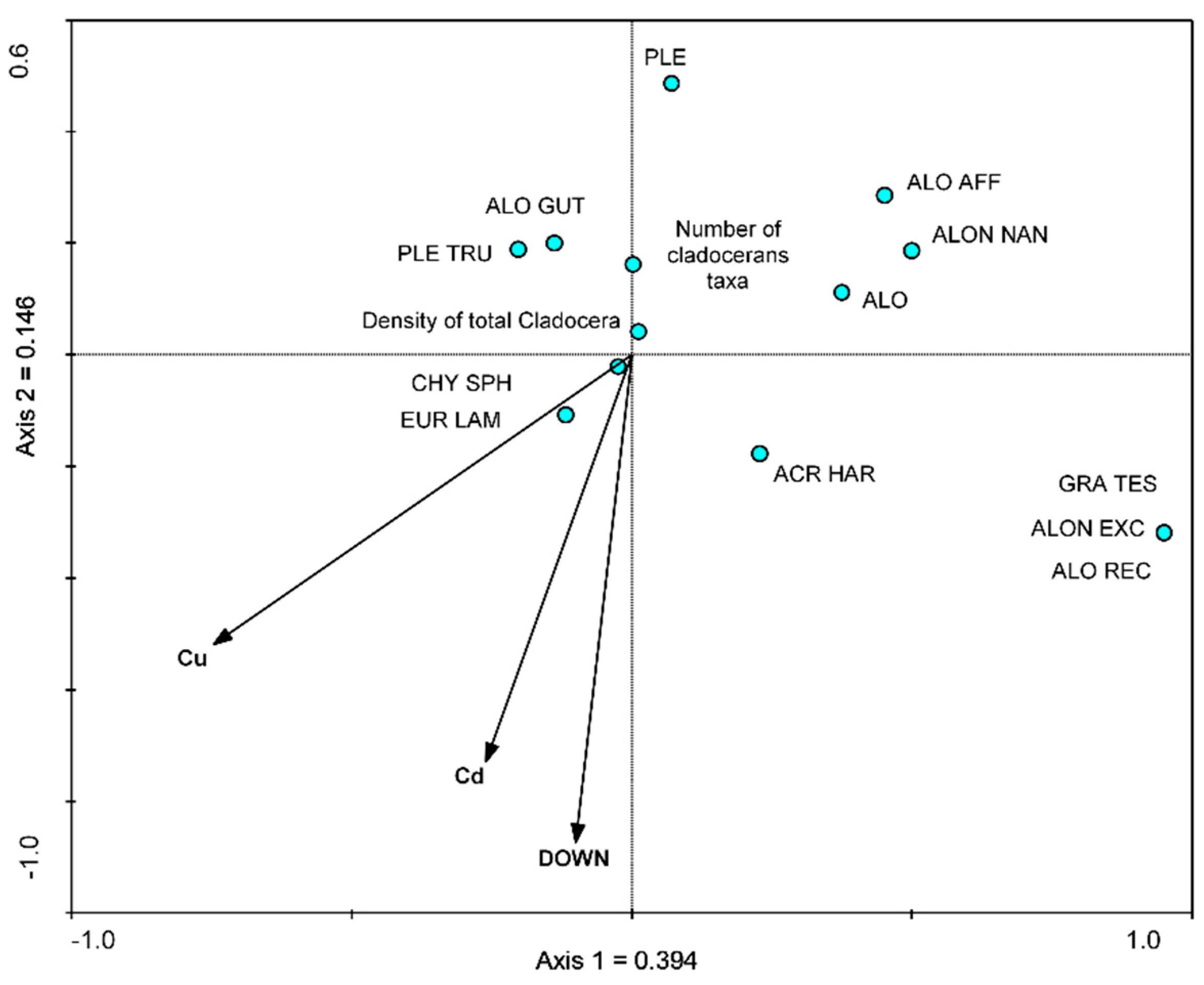
3.3. Relationship between Cladocera and Heavy Metal Concentration
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chen, G.; Dalton, C.; Taylor, D. Cladocera as indicators of trophic state in Irish lakes. J. Paleolimnol. 2010, 44, 465–481. [Google Scholar] [CrossRef]
- Rumes, B.; Eggermont, H.; Verschuren, D. Distribution and faunal richness of cladocera in western Uganda crater lakes. Hydrobiologia 2011, 676, 39–56. [Google Scholar] [CrossRef]
- Zawisza, E.; Zawiska, I.; Correa-Metrio, A. Cladocera community composition as a function of physicochemical and morphological parameters of dystrophic lakes in NE Poland. Wetlands 2016, 36, 1131–1142. [Google Scholar] [CrossRef]
- Doig, L.E.; Schiffer, S.T.; Liber, K. Reconstructing the ecological impacts of eight decades of mining, metallurgical, and municipal activities on a small boreal lake in northern Canada. Integr. Environ. Assess. Manag. 2015, 11, 490–501. [Google Scholar] [CrossRef] [PubMed]
- Frey, D.G. Cladocera analysis. In Handbook of Holocene Palaeoecology and Palaeohydrology; Berglund, B.E., Ed.; Wiley: New York, NY, USA, 1986; pp. 667–692. [Google Scholar]
- Ciszewski, D.; Aleksander-Kwaterczak, U.; Pociecha, A.; Szarek-Gwiazda, E.; Waloszek, A.; Wilk-Woźniak, E. Small effects of a large sediments contamination with heavy metals on aquatic organisms in the vicinity of an abandoned lead and zinc mine. Environ. Monit. Assess. 2013, 185, 9825–9842. [Google Scholar] [CrossRef] [PubMed]
- Koivisto, S.; Ketola, M.; Walls, M. Comparison of five cladoceran species in short-and long-term copper exposure. Hydrobiologia 1992, 248, 125–136. [Google Scholar] [CrossRef]
- Ciszewski, D. The past and prognosis of mining cessation impact on river sediment pollution. J. Soils Sediments 2019, 19, 393–402. [Google Scholar] [CrossRef]
- Michailova, P.; Ilkova, J.; Szarek-Gwiazda, E.; Kownacki, A.; Ciszewski, D. Genome instability in Chironomus annularius sensu Strenzke (Diptera, Chironomidae): A biomarker for assessment of the heavy metal contaminants in Poland. J. Limnol. 2018, 7, 15–24. [Google Scholar] [CrossRef]
- Frey, D.G. The taxonomy and biogeography of the Cladocera. Hydrobiologia 1987, 145, 5–17. [Google Scholar] [CrossRef]
- Szeroczyńska, K.; Sarmaja-Korjonen, K. Atlas of Subfossil Cladocera from Central and Northern Europe; Towarzystwo Przyjaciół Dolnej Wisły: Świecie, Polska, 2007; pp. 1–84. [Google Scholar]
- Kovach, W.L. A multivariate statistical package for Windows, version 3.1; Kovach Computing Services: Pentraeth, Wales, UK, 1999; p. 133. [Google Scholar]
- Ter Braak, C.J.F.; Šmilauer, P. CANOCO v. 4.5; Microcomputer Power-Ithaca: New York, NY, USA, 2002. [Google Scholar]
- Forstner, U.; Salomons, W. Trace metal analysis in polluted sediments. Environ. Technol. Lett. 1980, 1, 1–494. [Google Scholar] [CrossRef]
- Byrne, P.; Reid, I.; Wood, P.J. Sediment geochemistry of streams draining abandoned lead/zinc mines in central Wales: The Afon Twymyn. J. Soils Sediments 2010, 10, 683–697. [Google Scholar] [CrossRef]
- EC and MENVIQ (Environment Canada and Ministère de l’Environnement du Québec). Interim Criteria for Quality Assessment of St. Lawrence River Sediment; Environment Canada: Ottawa, ON, Canada, 1992; ISBN 622-19849-2.
- Calmano, W.; Von Der Kammer, F.; Schwartz, R. Characterization of redox conditions in soils and sediments: Heavy metals. In Soil and Sediment Remediation; Lens, P., Grotenhuis, T., Malina, G., Tabak, H., Eds.; IWA Publ.: London, UK, 2005; pp. 102–120. [Google Scholar]
- Szarek-Gwiazda, E.; Ciszewski, D. Variability of heavy metal concentrations in waters of fishponds affected by the former lead and zinc mine in southern Poland. Environ. Prot. Eng. 2017, 43, 121–137. [Google Scholar] [CrossRef]
- Belyaeva, M.; Deneke, R. Colonization of acidic mining lakes: Chydorus sphaericus and other Cladocera within a dynamic horizontal pH gradient (pH 3−7) in Lake Senftenberger See (Germany). Hydrobiologia 2007, 594, 97–108. [Google Scholar] [CrossRef]
- Burton, S.M.; Rundle, S.D.; Jones, M.B. The relationship between trace metal contamination and stream meiofauna. Environ. Pollut. 2001, 111, 159–167. [Google Scholar] [CrossRef]
- Sarma, S.S.S.; Peredo-Alvarez, V.M.; Nandini, S. Comparative study of the sensitivities of neonates and adults of selected cladoceran (Cladocera: Crustacea) species to acute toxicity stress. J. Environ. Sci. Health Part A 2007, 42, 1449–1452. [Google Scholar] [CrossRef] [PubMed]
- Petera-Zganiacz, J.; Dzieduszyńska, D.A.; Twardy, J.; Pawłowski, D.; Płóciennik, M.; Lutyńska, M.; Kittel, P. Younger Dryas flood events: A case study from the middle Warta River valley (Central Poland). Quat. Int. 2015, 386, 55–69. [Google Scholar] [CrossRef]




| Subsidence Ponds Area | Core Number | Number of Taxa in Core | Range (Min.–Max.) of Cladocera Individuals in 1 cm3 of Sediment |
|---|---|---|---|
| UP (subsidence pond formed after the closure of the mine) | RI | 5 | 1–6 |
| RII | 3 | 0–2 | |
| RIV | 7 | 4–22 | |
| RXVII | 5 | 37 | |
| RXVIII | 3 | 1–12 | |
| RXIX | 4 | 0–15 | |
| RXX | 5 | 3–6 | |
| RXXI | 1 | 1 | |
| DOWN (subsidence ponds created during the peak of Zn and Pb ore exploitation) | RVI | 3 | 3–8 |
| RVII | 2 | 1–6 | |
| RVIII | 6 | 1–50 | |
| RXII | 13 | 23–109 | |
| RXIII | 3 | 22–101 | |
| RXIV | 6 | 38–86 | |
| RXV | 6 | 20–93 | |
| RXVI | 4 | 3–76 |
| Core (UP) | Index Value | Core (DOWN) | Index Value |
|---|---|---|---|
| RI | 1.49 | RVI | 0.35 |
| RII | 1.04 | RVII | 0.58 |
| RIV | 1.61 | RVIII | 0.66 |
| RXVII | 1.07 | RXII | 2.12 |
| RXVIII | 0.79 | RXIII | 0.73 |
| RXIX | 0.85 | RXIV | 0.80 |
| RXX | 1.58 | RXV | 0.57 |
| RXXI | 0.00 | RXVI | 0.68 |
| A Variables | Cu | Acroperus harpae | Alona sp. | Alona affinis | Alona quadrangularis | Chydorus sphaericus |
|---|---|---|---|---|---|---|
| Alona affinis | −0.53 p = 0.028 | 0.94 p = 0.000 | ||||
| Alona guttata | −0.60 p = 0.01 | 0.83 p = 0.000 | 0.84 p = 0.000 | |||
| Bosmina coregoni | 0.80 p = 0.000 | |||||
| Chydorus sphaericus | 0.65 p = 0.004 | 0.62 p = 0.008 | 0.57 p = 0.018 | |||
| Daphnia sp. | −0.48 p = 0.049 | |||||
| Pleuroxus truncatus | 0.65 p = 0.005 | 0.82 p = 0.000 |
| B Variables | Zn | Pb | Cu | Alona sp. | Alona affinis | Alonella excisa | Alona quadrangularis | Alona rectangula | Alonella nana | Chydorus sphaericus | Eurycercus lamellatus | Pleuroxus truncatus |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Alona sp. | −0.40 p = 0.042 | −0.41 p = 0.034 | −0.45 p = 0.021 | |||||||||
| Alona affinis | −0.48 p = 0.012 | 0.66 p = 0.00 | ||||||||||
| Alonella excisa | 0.71 p = 0.00 | 0.93 p = 0.00 | ||||||||||
| Alona guttata | 0.64 p = 0.00 | |||||||||||
| Alona quadrangularis | −0.41 p = 0.037 | |||||||||||
| Alona rectangula | 0.73 p = 0.00 | 0.90 p = 0.00 | 0.97 p = 0.00 | |||||||||
| Alonella nana | −0.40 p = 0.041 | 0.99 p = 0.00 | ||||||||||
| Daphnia cucullata | −0.41 p = 0.037 | 0.99 p = 0.00 | ||||||||||
| Eurycercus lamellatus | 0.65 p = 0.000 | |||||||||||
| Graptoleberis testudinaria | −0.41 p = 0.037 | 0.69 p = 0.00 | 0.96 p = 0.00 | 0.98 p = 0.00 | 0.95 p = 0.000 | |||||||
| Pleuroxus truncatus | 0.44 p = 0.023 | 0.41 p = 0.039 | ||||||||||
| Pleuroxus trigonellus | 0.52 p = 0.006 | 0.43 p = 0.029 | 0.72 p = 0.000 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pociecha, A.; Wojtal, A.Z.; Szarek-Gwiazda, E.; Cieplok, A.; Ciszewski, D.; Kownacki, A. Response of Cladocera Fauna to Heavy Metal Pollution, Based on Sediments from Subsidence Ponds Downstream of a Mine Discharge (S. Poland). Water 2019, 11, 810. https://0-doi-org.brum.beds.ac.uk/10.3390/w11040810
Pociecha A, Wojtal AZ, Szarek-Gwiazda E, Cieplok A, Ciszewski D, Kownacki A. Response of Cladocera Fauna to Heavy Metal Pollution, Based on Sediments from Subsidence Ponds Downstream of a Mine Discharge (S. Poland). Water. 2019; 11(4):810. https://0-doi-org.brum.beds.ac.uk/10.3390/w11040810
Chicago/Turabian StylePociecha, Agnieszka, Agata Z. Wojtal, Ewa Szarek-Gwiazda, Anna Cieplok, Dariusz Ciszewski, and Andrzej Kownacki. 2019. "Response of Cladocera Fauna to Heavy Metal Pollution, Based on Sediments from Subsidence Ponds Downstream of a Mine Discharge (S. Poland)" Water 11, no. 4: 810. https://0-doi-org.brum.beds.ac.uk/10.3390/w11040810





