Antioxidant Activity of Resveratrol Diastereomeric Forms Assayed in Fluorescent-Engineered Human Keratinocytes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cells and Medium
2.2. Fluorescent-Engineered Human Keratinocytes
2.3. Immunofluorescence Technique
2.4. Cell Quantification
2.5. Resveratrol
2.6. Determining Antioxidant Capacity
2.7. Cytotoxicity Assay
2.8. Cell Counting and Proliferation
2.9. Statistical Analysis
3. Results
3.1. Cell Clones
3.2. Cytological Characterization
3.3. Determining Antioxidant Capacity
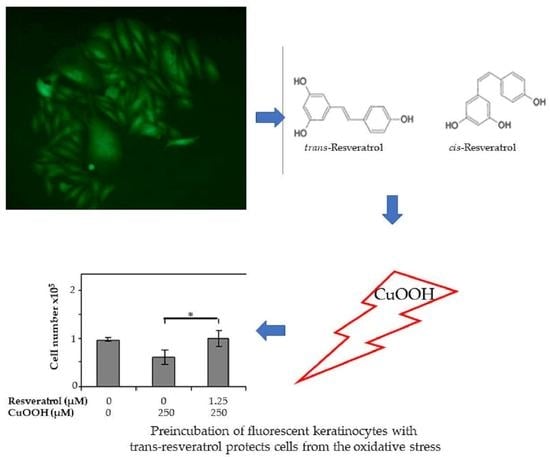
3.4. Cell Number Counting
3.5. Effect of CuOOH on the Human Keratinocyte NCTC/GFP Cell Line
3.6. Resveratrols
3.7. Effect of Resveratrol on the Cell Numbers
3.8. Effect of Resveratrol on Cell Proliferation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Comino-Sanz, I.M.; López-Franco, M.D.; Castro, B.; Pancorbo-Hidalgo, P.L. The Role of Antioxidants on Wound Healing: A Review of the Current Evidence. J. Clin. Med. 2021, 10, 3558. [Google Scholar] [CrossRef] [PubMed]
- Khan, T.A.; Hassan, I.; Ahmad, A.; Perveen, A.; Aman, S.; Quddusi, S.; Alhazza, I.M.; Ashraf, G.M.; Aliev, G. Recent updates on the dynamic association between oxidative stress and neurodegenerative disorders. CNS Neurol. Disord.-Drug Targets 2016, 15, 1. [Google Scholar] [CrossRef] [PubMed]
- Finkel, T. Signal transduction by reactive oxygen species. J. Cell Biol. 2011, 194, 7–15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hemerková, P.; Vališ, M. Role of Oxidative Stress in the Pathogenesis of Amyotrophic Lateral Sclerosis: Antioxidant Metalloenzymes and Therapeutic Strategies. Biomolecules 2021, 11, 437. [Google Scholar] [CrossRef]
- Georgieva, E.; Ivanova, D.; Zhelev, Z.; Bakalova, R.; Gulubova, M.; Aoki, I. Mitochondrial Dysfunction and Redox Imbalance as a Diagnostic Marker of “Free Radical Diseases”. Anticancer. Res. 2017, 37, 5373–5381. [Google Scholar] [CrossRef] [Green Version]
- Carrizzo, A.; Forte, M.; Damato, A.; Trimarco, V.; Salzano, F.; Bartolo, M.; Maciag, A.; Puca, A.A.; Vecchione, C. Antioxidant effects of resveratrol in cardiovascular, cerebral and metabolic diseases. Food Chem. Toxicol. 2013, 61, 215–226. [Google Scholar] [CrossRef]
- Belchí-Navarro, S.; Almagro, L.; Sabater-Jara, A.B.; Fernández-Pérez, F.; Bru, R.; Pedreno, M.A. Induction of trans-resveratrol and extracellular pathogenesis-related proteins in elicited suspension cultured cells of Vitis vinifera cv Monastrell. J. Plant Physiol. 2013, 170, 258–264. [Google Scholar] [CrossRef]
- Pugajeva, I.; Pērkons, I.; Górnaś, P. Identification and determination of stilbenes by Q-TOF in grape skins, seeds, juice and stems. J. Food Compos. Anal. 2018, 74, 44–52. [Google Scholar] [CrossRef]
- Orallo, F. Trans-resveratrol: A magical elixir of eternal youth? Curr. Med. Chem. 2008, 15, 1887–1898. [Google Scholar] [CrossRef]
- Frémont, L. Biological effects of resveratrol. Life Sci. 2000, 66, 663–673. [Google Scholar] [CrossRef]
- Keylor, M.H.; Matsuura, B.S.; Stephenson, C.R.J. Chemistry and Biology of Resveratrol-Derived Natural Products. Chem. Rev. 2015, 115, 8976–9027. [Google Scholar] [CrossRef] [PubMed]
- Maier-Salamon, A.; Böhmdorfer, M.; Thalhammer, T.; Szekeres, T.; Jaeger, W. Hepatic Glucuronidation of Resveratrol: Interspecies Comparison of Enzyme Kinetic Profiles in Human, Mouse, Rat, and Dog. Drug Metab. Pharmacokinet. 2011, 26, 364–373. [Google Scholar] [CrossRef] [Green Version]
- Gambini, J.; Ingles, M.; Olaso, G.; Lopez-Grueso, R.; Bonet-Costa, V.; Gimeno-Mallench, L.; Mas-Bargues, C.; Abdelaziz, K.M.; Gomez-Cabrera, M.C.; Vina, J.; et al. Properties of Resveratrol:In VitroandIn VivoStudies about Metabolism, Bioavailability, and Biological Effects in Animal Models and Humans. Oxid. Med. Cell. Longev. 2015, 2015, 837042. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Frombaum, M.; Le Clanche, S.; Bonnefont-Rousselot, D.; Borderie, D. Antioxidant effects of resveratrol and other stilbene derivatives on oxidative stress and NO bioavailability: Potential benefits to cardiovascular diseases. Biochimie 2012, 94, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Cristòfol, R.; Porquet, D.; Corpas, R.; Coto-Montes, A.; Serret, J.; Camins, A.; Pallàs, M.; Sanfeliu, C. Neurons from senescence-accelerated SAMP8 mice are protected against frailty by the sirtuin 1 promoting agents melatonin and resveratrol. J. Pineal Res. 2012, 52, 271–281. [Google Scholar] [CrossRef] [Green Version]
- Galiniak, S.; Aebisher, D.; Bartusik-Aebisher, D. Health benefits of resveratrol administration. Acta Biochim. Pol. 2019, 66, 13–21. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; He, X.-Q.; Huang, X.; Ding, L.; Xu, L.; Shen, Y.-T.; Zhang, F.; Zhu, M.-B.; Xu, B.-H.; Qi, Z.; et al. Resveratrol Protects Mouse Oocytes from Methylglyoxal-Induced Oxidative Damage. PLoS ONE 2013, 8, e77960. [Google Scholar] [CrossRef] [Green Version]
- Liguori, I.; Russo, G.; Curcio, F.; Bulli, G.; Aran, L.; Della-Morte, D.; Gargiulo, G.; Testa, G.; Cacciatore, F.; Bonaduce, D.; et al. Oxidative stress, aging, and diseases. Clin. Interv. Aging 2018, 13, 757–772. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Plauth, A.; Geikowski, A.; Cichon, S.; Wowro, S.; Liedgens, L.; Rousseau, M.; Weidner, C.; Fuhr, L.; Kliem, M.; Jenkins, G.; et al. Hormetic shifting of redox environment by pro-oxidative resveratrol protects cells against stress. Free Radic. Biol. Med. 2016, 99, 608–622. [Google Scholar] [CrossRef] [Green Version]
- Bobermin, L.D.; Souza, D.; Gonçalves, C.-A.; Quincozes-Santos, A. Resveratrol prevents ammonia-induced mitochondrial dysfunction and cellular redox imbalance in C6 astroglial cells. Nutr. Neurosci. 2017, 21, 276–285. [Google Scholar] [CrossRef]
- Wang, N.; Han, Q.; Wang, G.; Ma, W.-P.; Wang, J.; Wu, W.-X.; Guo, Y.; Liu, L.; Jiang, X.-Y.; Xie, X.-L.; et al. Resveratrol Protects Oxidative Stress-Induced Intestinal Epithelial Barrier Dysfunction by Upregulating Heme Oxygenase-1 Expression. Dig. Dis. Sci. 2016, 61, 2522–2534. [Google Scholar] [CrossRef]
- Ginés, C.; Cuesta, S.; Kireev, R.; García, C.; Rancan, L.; Paredes, S.D.; Vara, E.; Tresguerres, J.A. Protective effect of resveratrol against inflammation, oxidative stress and apoptosis in pancreas of aged SAMP8 mice. Exp. Gerontol. 2017, 90, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Hussein, M.M.; Mahfouz, M.K. Effect of resveratrol and rosuvastatin on experimental diabetic nephropathy in rats. Biomed. Pharmacother. 2016, 82, 685–692. [Google Scholar] [CrossRef]
- Zhang, T.; Chi, Y.; Kang, Y.; Lu, H.; Niu, H.; Liu, W.; Li, Y. Resveratrol ameliorates podocyte damage in diabetic mice via SIRT1/PGC-1α mediated attenuation of mitochondrial oxidative stress. J. Cell. Physiol. 2019, 234, 5033–5043. [Google Scholar] [CrossRef] [PubMed]
- Khazaei, M.; Karimi, J.; Sheikh, N.; Goodarzi, M.T.; Saidijam, M.; Khodadadi, I.; Moridi, H. Effects of Resveratrol on Receptor for Advanced Glycation End Products (RAGE) Expression and Oxidative Stress in the Liver of Rats with Type 2 Diabetes. Phytotherapy Res. 2016, 30, 66–71. [Google Scholar] [CrossRef]
- Fu, S.; Lv, R.; Wang, L.; Hou, H.; Liu, H.; Shao, S. Resveratrol, an antioxidant, protects spinal cord injury in rats by suppressing MAPK pathway. Saudi J. Biol. Sci. 2018, 25, 259–266. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Corrêa, M.G.; Absy, S.; Tenenbaum, H.; Ribeiro, F.V.; Cirano, F.R.; Casati, M.Z.; Pimentel, S.P. Resveratrol attenuates oxidative stress during experimental periodontitis in rats exposed to cigarette smoke inhalation. J. Periodontal Res. 2018, 54, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.-T.; Wu, Y.-C.; Sun, G.-C.; Ho, C.-Y.; Wong, T.-Y.; Lin, C.-H.; Chen, H.-H.; Yeh, T.-C.; Li, C.-J.; Tseng, C.-J.; et al. Effect of Resveratrol on Reactive Oxygen Species-Induced Cognitive Impairment in Rats with Angiotensin II-Induced Early Alzheimer’s Disease. J. Clin. Med. 2018, 7, 329. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.-L.; Li, T.; Li, J.-H.; Miao, S.-Y.; Xiao, X.-Z. The Effects of Resveratrol on Inflammation and Oxidative Stress in a Rat Model of Chronic Obstructive Pulmonary Disease. Molecules 2017, 22, 1529. [Google Scholar] [CrossRef] [Green Version]
- Hashim, S.N.; Schwarz, L.J.; Boysen, R.I.; Yang, Y.; Danylec, B.; Hearn, M.T. Rapid solid-phase extraction and analysis of resveratrol and other polyphenols in red wine. J. Chromatogr. A 2013, 1313, 284–290. [Google Scholar] [CrossRef] [PubMed]
- Riccio, B.V.F.; Spósito, L.; Carvalho, G.C.; Ferrari, P.C.; Chorilli, M. Resveratrol isoforms and conjugates: A review from biosynthesis in plants to elimination from the human body. Arch. Der Pharm. 2020, 353, 353. [Google Scholar] [CrossRef] [PubMed]
- Orallo, F. Comparative Studies of the Antioxidant Effects of Cis- and Trans- Resveratrol. Curr. Med. Chem. 2006, 13, 87–98. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Bai, Q.; Wang, Q.; Chen, S.; Ma, X.; Cai, C.; Wang, D.; Waqas, A.; Gong, P. Stereoselective Pharmacokinetics and Chiral Inversions of Some Chiral Hydroxy Group Drugs. Curr. Pharm. Biotechnol. 2020, 21, 1632–1644. [Google Scholar] [CrossRef] [PubMed]
- Skalova, L.; Szotakova, B. Chiral Inversion of Drugs: Coincidence or Principle? Curr. Drug Metab. 2004, 5, 517–533. [Google Scholar] [CrossRef] [Green Version]
- Luo, S.; Yin, J.; Peng, Y.; Xie, J.; Wu, H.; He, D.; Li, X.; Cheng, G. Glutathione is Involved in Detoxification of Peroxide and Root Nodule Symbiosis of Mesorhizobium huakuii. Curr. Microbiol. 2020, 77, 1–10. [Google Scholar] [CrossRef]
- Maietti, A.; Menziani, E.; Mazzotta, D. Determinazione del Resveratrolo in Vini del Commercio. In Proceedings of the Atti del IV Congresso di Chimica degli Alimenti, Ferrara, Italy, 28–30 June 2000; pp. 276–281. [Google Scholar]
- Goegan, P.; Johnson, G.; Vincent, R. Effects of serum protein and colloid on the alamarBlue assay in cell cultures. Toxicol. Vitr. 1995, 9, 257–266. [Google Scholar] [CrossRef]
- Ahmed, S.A.; Gogal, R.M.; Walsh, J.E. A new rapid and simple non-radioactive assay to monitor and determine the proliferation of lymphocytes: An alternative to [3H]thymidine incorporation assay. J. Immunol. Methods 1994, 170, 211–224. [Google Scholar] [CrossRef]
- Chen, X.; He, H.; Wang, G.; Yang, B.; Ren, W.; Ma, L.; Yu, Q. Stereospecific determination ofcis- andtrans-resveratrol in rat plasma by HPLC: Application to pharmacokinetic studies. Biomed. Chromatogr. 2007, 21, 257–265. [Google Scholar] [CrossRef]
- Brandolini, V.; Maietti, A.; Tedeschi, P.; Durini, E.; Vertuani, A.S.; Manfredini, S. Capillary Electrophoresis Determination, Synthesis, and Stability of Resveratrol and Related 3-O-β-d-Glucopyranosides. J. Agric. Food Chem. 2002, 50, 7407–7411. [Google Scholar] [CrossRef]
- Bergoglio, V.; Larcher, F.; Chevallier-Lagente, O.; Bernheim, A.; Danos, O.; Sarasin, A.; del Rio, M.; Magnaldo, T. Safe Selection of Genetically Manipulated Human Primary Keratinocytes with Very High Growth Potential Using CD24. Mol. Ther. 2007, 15, 2186–2193. [Google Scholar] [CrossRef]
- Cinelli, R.A.G.; Ferrari, A.; Pellegrini, V.; Tyagi, M.; Giacca, M.; Beltram, F. The Enhanced Green Fluorescent Protein as a Tool for the Analysis of Protein Dynamics and Localization: Local Fluorescence Study at the Single-Molecule Level. Photochem. Photobiol. 2000, 71, 771. [Google Scholar] [CrossRef]
- Van Overstraeten-Schlögel, N.; Ho-Shim, Y.; Tevel, V.; Bontems, S.; Dubois, P.; Raes, M. Transfection of Immortalized Keratinocytes by Low Toxic Poly(2-(Dimethylamino)Ethyl Methacrylate)-Based Polymers. J. Biomater. Sci. Polym. Ed. 2012, 23, 739–761. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; Chan, F.; Sun, H.; Yan, J.; Fan, D.; Zhao, D.; An, J.; Zhou, D. Resveratrol protects human keratinocytes HaCaT cells from UVA-induced oxidative stress damage by downregulating Keap1 expression. Eur. J. Pharmacol. 2011, 650, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Moldzio, R.; Radad, K.; Krewenka, C.; Kranner, B.; Duvigneau, J.C.; Rausch, W.-D. Protective effects of resveratrol on glutamate-induced damages in murine brain cultures. J. Neural Transm. 2013, 120, 1271–1280. [Google Scholar] [CrossRef] [PubMed]
- Belguendouz, L.; Fremont, L.; Linard, A. Resveratrol inhibits metal ion-dependent and independent peroxidation of porcine low-density lipoproteins. Biochem. Pharmacol. 1997, 53, 1347–1355. [Google Scholar] [CrossRef]
- Yáñez, M.; Fraiz, N.; Cano, E.; Orallo, F. Inhibitory effects of cis- and trans-resveratrol on noradrenaline and 5-hydroxytryptamine uptake and on monoamine oxidase activity. Biochem. Biophys. Res. Commun. 2006, 344, 688–695. [Google Scholar] [CrossRef] [PubMed]
- Leiro, J.; Álvarez, E.; Arranz, J.A.; Laguna, R.; Uriarte, E.; Orallo, F. Effects of cis -resveratrol on inflammatory murine macrophages: Antioxidant activity and down-regulation of inflammatory genes. J. Leukoc. Biol. 2004, 75, 1156–1165. [Google Scholar] [CrossRef] [Green Version]
- Rius, C.; Abu-Taha, M.; Hermenegildo, C.; Piqueras, L.; Cerda-Nicolas, J.-M.; Issekutz, A.C.; Estañ, L.; Cortijo, J.; Morcillo, E.J.; Orallo, F.; et al. Trans- but Not Cis-Resveratrol Impairs Angiotensin-II–Mediated Vascular Inflammation through Inhibition of NF-κB Activation and Peroxisome Proliferator-Activated Receptor-γ Upregulation. J. Immunol. 2010, 185, 3718–3727. [Google Scholar] [CrossRef] [Green Version]
- Campos-Toimil, M.; Elíes, J.; Álvarez, E.; Verde, I.; Orallo, F. Effects of trans- and cis-resveratrol on Ca2+ handling in A7r5 vascular myocytes. Eur. J. Pharmacol. 2007, 577, 91–99. [Google Scholar] [CrossRef]
- VanAntwerp, I.R.; Phelps, L.E.; Peuler, J.D.; Kopf, P.G. Effects of trans- versus cis-resveratrol on adrenergic contractions of the rat tail artery and role of endothelium. Physiol. Rep. 2020, 8, e14666. [Google Scholar] [CrossRef]
- Fabbrocini, G.; Kisslinger, A.; Iannelli, P.; Vitale, N.; Procaccini, C.; Sparaneo, G.; Chieffi, P.; Ayala, F.; Mancini, F.P.; Tramontano, D. Resveratrol regulates p66Shc activation in HaCaT cells. Exp. Dermatol. 2010, 19, 895–903. [Google Scholar] [CrossRef]
- Wen, S.; Zhang, J.; Yang, B.; Elias, P.M.; Man, M.-Q. Role of Resveratrol in Regulating Cutaneous Functions. Evidence-Based Complement. Altern. Med. 2020, 2020, 2416837. [Google Scholar] [CrossRef] [Green Version]
- Olas, B.; Nowak, P.; Ponczek, M.; Wachowicz, B. Resveratrol, a natural phenolic compound may reduce carbonylation proteins induced by peroxynitrite in blood platelets. Gen. Physiol. Biophys. 2006, 25, 215–222. [Google Scholar] [PubMed]
- Liu, Y.; Xiong, W.; Wang, C.-W.; Shi, J.-P.; Shi, Z.-Q.; Zhou, J.-D. Resveratrol promotes skin wound healing by regulating the miR-212/CASP8 axis. Lab. Investig. 2021, 101, 1363–1370. [Google Scholar] [CrossRef]
- Bastianetto, S.; Dumont, Y.; Duranton, A.; Vercauteren, F.; Breton, L.; Quirion, R. Protective Action of Resveratrol in Human Skin: Possible Involvement of Specific Receptor Binding Sites. PLoS ONE 2010, 5, e12935. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iuga, C.; Alvarez-Idaboy, J.R.; Russo, N. Antioxidant Activity of trans-Resveratrol toward Hydroxyl and Hydroperoxyl Radicals: A Quantum Chemical and Computational Kinetics Study. J. Org. Chem. 2012, 77, 3868–3877. [Google Scholar] [CrossRef] [PubMed]
- Mehta, J.; Rayalam, S.; Wang, X. Cytoprotective Effects of Natural Compounds against Oxidative Stress. Antioxidants 2018, 7, 147. [Google Scholar] [CrossRef] [Green Version]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bononi, I.; Tedeschi, P.; Mantovani, V.; Maietti, A.; Mazzoni, E.; Pancaldi, C.; Brandolini, V.; Tognon, M. Antioxidant Activity of Resveratrol Diastereomeric Forms Assayed in Fluorescent-Engineered Human Keratinocytes. Antioxidants 2022, 11, 196. https://0-doi-org.brum.beds.ac.uk/10.3390/antiox11020196
Bononi I, Tedeschi P, Mantovani V, Maietti A, Mazzoni E, Pancaldi C, Brandolini V, Tognon M. Antioxidant Activity of Resveratrol Diastereomeric Forms Assayed in Fluorescent-Engineered Human Keratinocytes. Antioxidants. 2022; 11(2):196. https://0-doi-org.brum.beds.ac.uk/10.3390/antiox11020196
Chicago/Turabian StyleBononi, Ilaria, Paola Tedeschi, Vanessa Mantovani, Annalisa Maietti, Elisa Mazzoni, Cecilia Pancaldi, Vincenzo Brandolini, and Mauro Tognon. 2022. "Antioxidant Activity of Resveratrol Diastereomeric Forms Assayed in Fluorescent-Engineered Human Keratinocytes" Antioxidants 11, no. 2: 196. https://0-doi-org.brum.beds.ac.uk/10.3390/antiox11020196