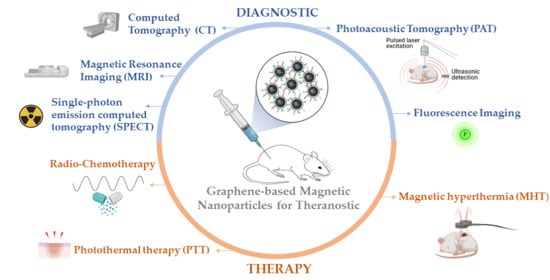
Graphene-Based Magnetic Nanoparticles for Theranostics: An Overview for Their Potential in Clinical Application
Abstract
:1. Introduction
1.1. Graphene-Based Magnetic Nanoparticles Configurations
1.2. Preparation and Synthesis of Graphene-Based Magnetic Nanoparticles Hybrids
2. Graphene-Based Magnetic Nanoparticles for Theranostics
2.1. Graphene-Based Magnetic Nanoparticles in Diagnostic
2.2. Graphene-Based Magnetic Nanoparticles in Therapy
2.3. Biocompatibility and Toxicity of Graphene-Based Magnetic Nanoparticles
2.4. Limitations of Graphene-Based Magnetic Nanoparticles for Potencial Clinical Applications
3. Future Perspective for the Advance of GbMNPs
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Broza, Y.Y.; Mochalski, P.; Ruzsanyi, V.; Amann, A.; Haick, H. Hybrid volatolomics and disease detection. Angew. Chem. Int. Ed. 2015, 54, 11036–11048. [Google Scholar] [CrossRef]
- Nakhleh, M.; Haick, H. Detecting active pulmonary tuberculosis with a breath test using nanomaterial- based sensors. Eur. Respir. J. 2014, 43, 1519–1522. [Google Scholar] [CrossRef] [Green Version]
- Bhattacharya, K.; Mukherjee, S.P.; Gallud, A.; Burkert, S.C.; Bistarelli, S.; Bellucci, S.; Bottini, M.; Star, A.; Fadeel, B. Biological interactions of carbon-based nanomaterials: From coronation to degradation. Nanomed. Nanotechnol. Biol. Med. 2016, 12, 333–351. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bañobre-López, M.; Teijeiro, A.; Rivas, J. Magnetic nanoparticle-based hyperthermia for cancer treatment. Rep. Pract. Oncol. Radiother. 2013, 18, 397–400. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pankhurst, Q.; Connolly, J.; Jones, S.; Dobson, J. Applications of magnetic nanoparticles in biomedicine: Biomedical applications of magnetic nanoparticles. J. Phys. D Appl. Phys. 2003, 36, R167. [Google Scholar] [CrossRef] [Green Version]
- Zhu, K.; Ju, Y.; Xu, J.; Yang, Z.; Gao, S.; Hou, Y. Magnetic Nanomaterials: Chemical design, synthesis, and potential applications. Acc. Chem. Res. 2018, 51, 404–413. [Google Scholar] [CrossRef]
- Dobson, J. Remote control of cellular behaviour with magnetic nanoparticles. Nat. Nanotechnol. 2008, 3, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Kozissnik, B.; Bohorquez, A.C.; Dobson, J.; Rinaldi, C. Magnetic fluid hyperthermia: Advances, challenges, and opportunity. Int. J. Hyperth. 2013, 29, 706–714. [Google Scholar] [CrossRef]
- Marincek, B. Diagnostic improvement in MRI of gynecological neoplasms. J. Belg. Radiol. 1996, 79, 13–17. [Google Scholar]
- Reimer, P. Ferucarbotran (Resovist): A new clinically approved RES-specific contrast agent for contrast-enhanced MRI of the liver: Properties, clinical development, and applications. Eur. Radiol. 2003, 13, 1266–1276. [Google Scholar] [CrossRef]
- Rodrigues, R.O.; Baldi, G.; Doumett, S.; Garcia-Hevia, L.; Gallo, J.; Bañobre-López, M.; Dražić, G.; Calhelha, R.C.; Ferreira, I.C.F.R.; Lima, R.; et al. Multifunctional graphene-based magnetic nanocarriers for combined hyperthermia and dual stimuli-responsive drug delivery. Mater. Sci. Eng. C 2018, 93, 206–217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Geim, A.K.; Novoselov, K.S. The rise of graphene. Nat. Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef]
- Shen, X.-J.; Zeng, X.-L.; Dang, C.-Y. Handbook of Graphene: Graphene Composites; Scrivener Publishing: Beverly, MA, USA, 2019; ISBN 9781119468554. [Google Scholar]
- Yan, Q.L.; Gozin, M.; Zhao, F.Q.; Cohen, A.; Pang, S.P. Highly energetic compositions based on functionalized carbon nanomaterials. Nanoscale 2016, 8, 4799–4851. [Google Scholar] [CrossRef] [Green Version]
- Yang, G.; Li, L.; Lee, W.B.; Ng, M.C. Structure of graphene and its disorders: A review. Sci. Technol. Adv. Mater. 2018, 19, 613–648. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nehra, M.; Dilbaghi, N.; Hassan, A.A.; Kumar, S. Carbon-Based Nanomaterials for the Development of Sensitive Nanosensor Platforms; Elsevier Inc.: Amsterdam, The Netherlands, 2019; Volume 60, ISBN 9780128174562. [Google Scholar]
- Goenka, S.; Sant, V.; Sant, S. Graphene-based nanomaterials for drug delivery and tissue engineering. J. Control. Release 2014, 173, 75–88. [Google Scholar] [CrossRef] [PubMed]
- Saleem, J.; Wang, L.; Chen, C. Carbon-based nanomaterials for cancer therapy via targeting tumor microenvironment. Adv. Healthc. Mater. 2018, 7, 1800525. [Google Scholar] [CrossRef] [PubMed]
- Dasari Shareena, T.P.; McShan, D.; Dasmahapatra, A.K.; Tchounwou, P.B. A Review on graphene-based nanomaterials in biomedical applications and risks in environment and health. Nano-Micro Lett. 2018, 10, 1–34. [Google Scholar] [CrossRef]
- Ramachandra Kurup Sasikala, A.; Thomas, R.G.; Unnithan, A.R.; Saravanakumar, B.; Jeong, Y.Y.; Park, C.H.; Kim, C.S. Multifunctional nanocarpets for cancer theranostics: Remotely controlled graphene nanoheaters for thermo-chemosensitisation and magnetic resonance imaging. Sci. Rep. 2016, 6, 20543. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Huang, Y.; Huang, K.; Lin, J.; Huang, P. Functional magnetic graphene composites for biosensing. Int. J. Mol. Sci. 2020, 21, 390. [Google Scholar] [CrossRef] [Green Version]
- Albert, E.L.; Sajiman, M.B.; Che Abdullah, C.A. Incorporation of magnetic nanoparticle to graphene oxide via simple emulsion method and their cytotoxicity. Appl. Nanosci. 2019, 9, 43–48. [Google Scholar] [CrossRef]
- Boncel, S.; Herman, A.P.; Walczak, K.Z. Magnetic carbon nanostructures in medicine. J. Mater. Chem. 2012, 22, 31–37. [Google Scholar] [CrossRef]
- Xu, M.; Zhu, J.; Wang, F.; Xiong, Y.; Wu, Y.; Wang, Q.; Weng, J.; Zhang, Z.; Chen, W.; Liu, S. Improved in vitro and in vivo biocompatibility of graphene oxide through surface modification: Poly (acrylic acid)-functionalization is superior to pegylation improved in vitro and in vivo biocompatibility of graphene oxide through surface modificatio. ACS Nano 2016, 10, 3267–3281. [Google Scholar] [CrossRef]
- Ali, A.; Zafar, H.; Zia, M.; ul Haq, I.; Phull, A.R.; Ali, J.S.; Hussain, A. Synthesis, characterization, applications, and challenges of iron oxide nanoparticles. Nanotechnol. Sci. Appl. 2016, 9, 49–67. [Google Scholar] [CrossRef] [Green Version]
- Alegret, N.; Criado, A.; Prato, M. Recent advances of graphene-based hybrids with magnetic nanoparticles for biomedical applications. Curr. Med. Chem. 2017, 24, 529–536. [Google Scholar] [CrossRef] [Green Version]
- Huang, X.; Qi, X.; Boey, F.; Zhang, H. Graphene-based composites. Chem. Soc. Rev. 2012, 41, 666–686. [Google Scholar] [CrossRef] [PubMed]
- Pinho, S.L.C.; Pereira, G.A.; Voisin, Ќ.P.; Kassem, J.; Etienne, L.; Peters, J.A.; Carlos, L.; Geraldes, C.F.G.C. Fine tuning of the relaxometry of γ-Fe2O3@SiO2 nanoparticles by tweaking the silica coating thickness. ACS Nano 2010, 4, 5339–5349. [Google Scholar] [CrossRef]
- Mitra, S.; Banerjee, S.; Datta, A.; Chakravorty, D. A brief review on graphene/inorganic nanostructure composites: Materials for the future. Indian J. Phys. 2016, 90, 1019–1032. [Google Scholar] [CrossRef]
- Rodrigues, R.; Baldi, G.; Doumett, S.; Gallo, J.; Bañobre-López, M.; Dražić, G.; Calhelha, R.; Ferreira, I.; Lima, R.; Silva, A.; et al. A tailor-made protocol to synthesize yolk-shell graphene-based magnetic nanoparticles for nanomedicine. C 2018, 4, 55. [Google Scholar] [CrossRef] [Green Version]
- Isıklan, N.; Awel, N.; Türk, M. Colloids and surfaces A: Physicochemical and engineering aspects synthesis and drug delivery performance of gelatin-decorated magnetic graphene oxide nanoplatform. Colloids Surf. A Physicochem. Eng. Asp. 2021, 616, 126256. [Google Scholar] [CrossRef]
- Jusin, J.; Aziz, M.; Sean, G.P.; Jaafar, J. Preparation and characterization of graphene-based magnetic hybrid nanocomposite. Malays. J. Anal. Sci. 2016, 20, 149–156. [Google Scholar] [CrossRef]
- Kui, L.Ü.; Guixia, Z.; Xiangke, W. A brief review of graphene-based material synthesis and its application in environmental pollution management. Chin. Sci. Bull. 2012, 57, 1223–1234. [Google Scholar] [CrossRef] [Green Version]
- Viseu, T.; Lopes, C.M.; Fernandes, E.; Real Oliveira, M.E.C.D.; Lúcio, M. A systematic review and critical analysis of the role of graphene-based nanomaterials in cancer theranostics. Pharmaceutics 2018, 10, 282. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miao, T.; Oldinski, R.A.; Liu, G.; Chen, X. Nanotheranostics for Cancer Applications; Springer International Publishing: Cham, Switzerland, 2019; Volume 5, ISBN 9783030017736. [Google Scholar]
- Singh, D.; Dilnawaz, F.; Sahoo, S.K. Challenges of moving theranostic nanomedicine into the clinic. Nanomedicine 2020, 15, 111–114. [Google Scholar] [CrossRef]
- Tan, Y.Y.; Yap, P.K.; Xin Lim, G.L.; Mehta, M.; Chan, Y.; Ng, S.W.; Kapoor, D.N.; Negi, P.; Anand, K.; Singh, S.K.; et al. Perspectives and advancements in the design of nanomaterials for targeted cancer theranostics. Chem. Biol. Interact. 2020, 329, 109221. [Google Scholar] [CrossRef]
- Kim, D.; Shin, K.; Kwon, S.G.; Hyeon, T. Synthesis and biomedical applications of multifunctional nanoparticles. Adv. Mater. 2018, 30, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Qian, R.; Maiti, D.; Zhong, J.; Xiong, S.S.; Zhou, H.; Zhu, R.; Wan, J.; Yang, K. T1/T2-weighted magnetic resonance imaging and SPECT imaging guided combined radioisotope therapy and chemotherapy using functionalized reduced graphene oxide-manganese ferrite nanocomposites. Carbon N. Y. 2019, 149, 55–62. [Google Scholar] [CrossRef]
- Wang, G.; Ma, Y.; Wei, Z.; Qi, M. Development of multifunctional cobalt ferrite/graphene oxide nanocomposites for magnetic resonance imaging and controlled drug delivery. Chem. Eng. J. 2016, 289, 150–160. [Google Scholar] [CrossRef]
- Baktash, M.S.; Zarrabi, A.; Avazverdi, E.; Reis, N.M. Development and optimization of a new hybrid chitosan-grafted graphene oxide/magnetic nanoparticle system for theranostic applications. J. Mol. Liq. 2020, 322, 114515. [Google Scholar] [CrossRef]
- Hsu, Y.H.; Hsieh, H.L.; Viswanathan, G.; Voon, S.H.; Kue, C.S.; Saw, W.S.; Yeong, C.H.; Azlan, C.A.; Imae, T.; Kiew, L.V.; et al. Multifunctional carbon-coated magnetic sensing graphene oxide-cyclodextrin nanohybrid for potential cancer theranosis. J. Nanopart. Res. 2017, 19, 359. [Google Scholar] [CrossRef]
- Shirvalilou, S.; Khoei, S.; Khoee, S.; Raoufi, N.J.; Karimi, M.R.; Shakeri-Zadeh, A. Development of a magnetic nano-graphene oxide carrier for improved glioma-targeted drug delivery and imaging: In vitro and in vivo evaluations. Chem. Biol. Interact. 2018, 295, 97–108. [Google Scholar] [CrossRef]
- Bi, H.; He, F.; Dai, Y.; Xu, J.; Dong, Y.; Yang, D.; Gai, S.; Li, L.; Li, C.; Yang, P. Quad-model imaging-guided high-efficiency phototherapy based on upconversion nanoparticles and ZnFe2O4 integrated graphene oxide. Inorg. Chem. 2018, 57, 9988–9998. [Google Scholar] [CrossRef]
- Badrigilan, S.; Shaabani, B.; Gharehaghaji, N.; Mesbahi, A. Iron oxide/bismuth oxide nanocomposites coated by graphene quantum dots: “Three-in-one” theranostic agents for simultaneous CT/MR imaging-guided in vitro photothermal therapy. Photodiagnosis Photodyn. Ther. 2019, 25, 504–514. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Tang, Y.; Liu, T.; Chen, Q.; Zhou, X.; Wang, N.; Ma, M.; Cheng, Y.; Chen, H. Engineering graphene oxide with ultrasmall SPIONs and smart drug release for cancer theranostics. Chem. Commun. 2019, 55, 1963–1966. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Rodriguez, R.; Campbell, E.; Naumov, A. Multifunctional graphene oxide/iron oxide nanoparticles for magnetic targeted drug delivery dual magnetic resonance/fluorescence imaging and cancer sensing. PLoS ONE 2019, 14, e0217072. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zheng, L.; Xiao, L.; Wang, L.; Cui, J.; Sha, D.; Liu, C. Eco-friendly development of ultrasmall ionp-loaded nanoplatform for bimodal imaging-guided cancer theranostics yulin. Biomater. Sci. 2020, 8, 6375–6386. [Google Scholar] [CrossRef] [PubMed]
- Gazestani, A.M.; Khoei, S.; Khoee, S.; Minaei, E.; Motevalian, M. In vivo evaluation of the combination effect of near-infrared laser and 5-fluorouracil-loaded PLGA-coated magnetite nanographene oxide. Artif. Cells Nanomed. Biotechnol. 2018, 46, 25–33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bañobre-López, M.; Bran, C.; Rodríguez-Abreu, C.; Gallo, J.; Vázquez, M.; Rivas, J. A colloidally stable water dispersion of Ni nanowires as an efficient: T2-MRI contrast agent. J. Mater. Chem. B 2017, 5, 3338–3347. [Google Scholar] [CrossRef]
- Na, H.B.; Song, I.C.; Hyeon, T. Inorganic nanoparticles for MRI contrast agents. Adv. Mater. 2009, 21, 2133–2148. [Google Scholar] [CrossRef]
- Rivas, J.; Kolen’ko, Y.V.; Bañobre-López, M. Magnetic Nanocolloids. In Nanocolloids; Elsevier: Amsterdam, The Netherlands, 2016; pp. 75–129. ISBN 9780128015780. [Google Scholar]
- Patanjali, P.; Singh, R.; Kumar, A.; Chaudhary, P. Nanotechnology for Water Treatment: A Green Approach; Elsevier Inc.: Amsterdam, The Netherlands, 2019; ISBN 9780081025796. [Google Scholar]
- De Crozals, G.; Bonnet, R.; Farre, C.; Chaix, C. Nanoparticles with multiple properties for biomedical applications: A strategic guide. Nano Today 2016, 11, 435–463. [Google Scholar] [CrossRef]
- Chatterjee, D.K.; Diagaradjane, P.; Krishnan, S. Nanoparticle-mediated hyperthermia in cancer therapy. Ther. Deliv. 2011, 2, 1001–1014. [Google Scholar] [CrossRef] [Green Version]
- Prigent, P.; Robic, C.; Raynal, I.; Guilbert, I.; Dencausse, A.; Corot, C.; Idée, J.-M.; Raynaud, J.; Port, M.; Robert, P. Superparamagnetic Contrast Agents. In Molecular and Cellular MR Imaging; CRC Press: Boca Raton, FL, USA, 2007; pp. 59–83. ISBN 978-0-8493-7252-0. [Google Scholar]
- Balasubramaniam, S.; Kayandan, S.; Lin, Y.N.; Kelly, D.F.; House, M.J.; Woodward, R.C.; St. Pierre, T.G.; Riffle, J.S.; Davis, R.M. Toward design of magnetic nanoparticle clusters stabilized by biocompatible diblock copolymers for T2-weighted MRI contrast. Langmuir 2014, 30, 1580–1587. [Google Scholar] [CrossRef] [PubMed]
- Garg, B.; Sung, C.H.; Ling, Y.C. Graphene-based nanomaterials as molecular imaging agents. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2015, 7, 737–758. [Google Scholar] [CrossRef]
- Wang, L.V.; Yao, J. A practical guide to photoacoustic tomography in the life sciences. Nat. Methods 2016, 13, 627. [Google Scholar] [CrossRef]
- Zou, L.; Wang, H.; He, B.; Zeng, L.; Tan, T.; Cao, H.; He, X.; Zhang, Z.; Guo, S.; Li, Y. Current approaches of photothermal therapy in treating cancer metastasis with nanotherapeutics. Theranostics 2016, 6, 762–772. [Google Scholar] [CrossRef]
- Chen, Q.; Wen, J.; Li, H.; Xu, Y.; Liu, F.; Sun, S. Recent advances in different modal imaging-guided photothermal therapy. Biomaterials 2016, 106, 144–166. [Google Scholar] [CrossRef]
- Yang, K.; Feng, L.; Shi, X.; Liu, Z. Nano-graphene in biomedicine: Theranostic applications. Chem. Soc. Rev. 2013, 42, 530–547. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, L.; Shi, J. Two-dimensional non-carbonaceous materials-enabled efficient photothermal cancer therapy. Nano Today 2016, 11, 292–308. [Google Scholar] [CrossRef]
- Hildebrandt, B.; Riess, H. The cellular and molecular basis of hyperthermia. Crit. Rev. Oncol. Hematol. 2002, 43, 33–56. [Google Scholar] [CrossRef]
- Laurent, S.; Dutz, S.; Häfeli, U.O.; Mahmoudi, M. Magnetic fluid hyperthermia: Focus on superparamagnetic iron oxide nanoparticles. Adv. Colloid Interface Sci. 2011, 166, 8–23. [Google Scholar] [CrossRef] [PubMed]
- Fan, W.; Yung, B.; Huang, P.; Chen, X. Nanotechnology for Multimodal Synergistic Cancer Therapy. Chem. Rev. 2017, 117, 13566–13638. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Tan, C.; Zhang, H.; Wang, L. As featured in: Biomedical applications. Chem. Soc. Rev. 2015, 44, 2681–2701. [Google Scholar] [CrossRef]
- Ma, L.; Le, P.; Kohli, M.; Smith, A.M. Nanotheranostics for Cancer Applications; Springer: Berlin/Heidelberg, Germany, 2019. [Google Scholar] [CrossRef]
- Augustine, S.; Singh, J.; Srivastava, M.; Sharma, M.; Das, A.; Malhotra, B.D. Recent advances in carbon based nanosystems for cancer theranostics. Biomater. Sci. 2017, 5, 901–952. [Google Scholar] [CrossRef] [PubMed]
- Chang, X.; Zhang, Y.; Xu, P.; Zhang, M.; Wu, H.; Yang, S. Graphene oxide/MnWO4 nanocomposite for magnetic resonance/photoacoustic dual-model imaging and tumor photothermo-chemotherapy. Carbon N. Y. 2018, 138, 397–409. [Google Scholar] [CrossRef]
- Chen, L.; Weiss, L. The role of the sinus wall in the passage of erythrocytes through the spleen. Blood 1972, 60, 714–721. [Google Scholar] [CrossRef]
- Liao, C.; Li, Y.; Tjong, S.C. Graphene Nanomaterials: Synthesis. Int. J. Mol. Sci. 2018, 19, 3564. [Google Scholar] [CrossRef] [Green Version]
- Mahmoudi, M. Debugging nano–bio interfaces: Systematic strategies to accelerate clinical translation of nanotechnologies. Trends Biotechnol. 2018, 36, 755–769. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Xie, J.; Wah, Y.; Wang, C. Effect of PEG conformation and particle size on the cellular uptake efficiency of nanoparticles with the HepG2 cells. J. Control Release 2007, 118, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Monteiro-Riviere, N.A.; Inman, A.O. Challenges for assessing carbon nanomaterial toxicity to the skin. Carbon N. Y. 2006, 44, 1070–1078. [Google Scholar] [CrossRef]
- Bullock, C.J.; Bussy, C. Biocompatibility considerations in the design of graphene biomedical materials. Adv. Mater. Interfaces 2019, 6, 1–15. [Google Scholar] [CrossRef]
- Faria, M.; Björnmalm, M.; Thurecht, K.J.; Kent, S.J.; Parton, R.G.; Kavallaris, M.; Johnston, A.P.R.; Gooding, J.J.; Corrie, S.R.; Boyd, B.J.; et al. Minimum information reporting in bio–nano experimental literature. Nat. Nanotechnol. 2018, 13, 777–785. [Google Scholar] [CrossRef]
- Rodrigues, R.O.; Sousa, P.C.; Gaspar, J.; Bañobre-López, M.; Lima, R.; Minas, G. Organ-on-a-chip: A preclinical microfluidic platform for the progress of nanomedicine. Small 2020, 16, 2003517. [Google Scholar] [CrossRef] [PubMed]





| Graphene-Based Magnetic Nanoparticle | Configuration | Preparation Method | Dh | Dc | Relaxivity (mM−1S−1) | Magnetic Properties | Drug Loading Capacity | pH Sensitive Release | Diagnostic Strategy | Therapeutic Strategy | In Vitro | In Vivo | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| rGO-MnFe2O4-PEG | GbDMNPs | In situ: hydrothermal reaction | - | - | r1= 11.74; r2 = 295.48 (3.0 T) | - | - | MRI; SPECT | RIT; Chemotherapy | x | x | [39] | |
| CoFe2O4/GO | GbDMNPs | In situ: Sonochemical reaction | - | L = 300–2000 nm; D = 5–13 nm | r2 = 92.71 (3.0 T) | Ms = 38.7 emu/g | =1.08 mg/mg | MRI | Chemotherapy | x | - | [40] | |
| GO-IO-DOX | GbDMNPs | Ex situ: hydrophobic–hydrophobic | - | L = n/d ; D = 20 nm | r2 = 84.0 (3.0 T) | Ms = 25.37 emu/g | - | pH 7.4 = 19%; pH 5 = 58% (6 h) | MRI | MFH; Chemotherapy | x | - | [20] |
| MNP/GO/chitosan | GbDMNPs | Ex situ | - | - | r1 = 6.99; r2 = 44.51 (1.5 T) | MRI | Chemotherapy | x | - | [41] | |||
| GO-CD/Fe@C | GbDMNPs | Ex situ | 267–334 nm | - | r2 = 9.37 (3.0 T) | Ms = 2.45 emu/g | =3.50 mg/mg | pH 7 = 20%; pH 5 = 38% (48 h) | MRI | Chemotherapy | x | - | [42] |
| IUdR/NGO/SPION/PLGA | GbEMNPs | Ex situ: emulsion solvent evaporation | 71.8 nm | D = 26.3 nm | - | Ms = 15.98 emu/g | - | pH 7.4 = 67% (8 h) | MRI | Chemotherapy; PTT | x | x | [43] |
| GO/ZnFe2O4/UCNPs | GbDMNPs | Ex situ: electrostatic interactions | 400 nm | L = n/d ; D = 12 nm; T = 1- 2 nm | r2 = 24.84 (1.2 T) | - | - | - | UCL; CT; MRI; PAT | PDT | x | x | [44] |
| GQDs-Fe/Bi | GbEMNPs | Ex situ | >100 nm | D = 64± 5.46 nm | r1 = 2.37; r2 = 62.34 (1.5 T) | Ms = 48.59 emu/g | - | - | CT; MRI | PTT | x | - | [45] |
| CAD-SPIONs@GO | GbDMNPs | In situ | - | L= n/d; D = 5 nm | r1 = 3.17 r2 = 8.36 (0.5 T) | Ms = 10.50 emu/g | - | pH 7.4 = 13.7% (48 h); pH 5.5 = 35.4% (1 h) | MRI | Chemotherapy | x | x | [46] |
| GO-Fe3O4 | GbDMNPs | Ex situ | 76 nm | L = 265 nm | r1 = 6.6 r2 = 71.1 (3.0 T) | - | =0.2 mg/mg | - | MRI; FI | Chemotherapy | x | - | [47] |
| GIPD | GbDMNPs | In situ | 86.7 ± 3.4 nm | L = 150 nm ; D = 8.25 nm | - | - | =0.48 mg/mg | - | MRI | PTT | x | x | [48] |
| NGO-SPION-PLGA-5-Fu | GbDMNPs | Ex situ | L = 72.9 | - | - | - | pH 7.4 = 41.36% (24 h) | MRI | PTT; Chemotherapy | x | x | [49] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lage, T.; Rodrigues, R.O.; Catarino, S.; Gallo, J.; Bañobre-López, M.; Minas, G. Graphene-Based Magnetic Nanoparticles for Theranostics: An Overview for Their Potential in Clinical Application. Nanomaterials 2021, 11, 1073. https://0-doi-org.brum.beds.ac.uk/10.3390/nano11051073
Lage T, Rodrigues RO, Catarino S, Gallo J, Bañobre-López M, Minas G. Graphene-Based Magnetic Nanoparticles for Theranostics: An Overview for Their Potential in Clinical Application. Nanomaterials. 2021; 11(5):1073. https://0-doi-org.brum.beds.ac.uk/10.3390/nano11051073
Chicago/Turabian StyleLage, Teresa, Raquel O. Rodrigues, Susana Catarino, Juan Gallo, Manuel Bañobre-López, and Graça Minas. 2021. "Graphene-Based Magnetic Nanoparticles for Theranostics: An Overview for Their Potential in Clinical Application" Nanomaterials 11, no. 5: 1073. https://0-doi-org.brum.beds.ac.uk/10.3390/nano11051073