Antibacterial Activities of Selected Pure Compounds Isolated from Gut Bacteria of Animals Living in Polluted Environments
Abstract
:1. Introduction
2. Results
2.1. Curcumenol, L-Homotyrosine and Docosanedioic Acid Showed Significant Antibacterial Activities Against Gram-Negative and Gram-Positive Pathogenic Bacteria Tested
2.2. Di-Rhamnolipids and N-Tetradecanoyl Homoserine Lactones Showed Promising Antibacterial Activities Against Gram-Positive and Gram-Negative Bacteria
2.3. Pure Compounds Showed Minimal Cytotoxic Effects against Human Cell Lines
3. Discussion
4. Materials and Methods
4.1. Bacterial Cultures
4.2. Preparation of Compounds Stock Solution
4.3. Evaluation of Pure Compounds for Antibacterial Properties
4.4. In Vitro Cytotoxicity Assays
4.5. Statistical Analysis
4.6. Ethical Approval and Consent to Participate
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sprenger, M.; Fukuda, K. New mechanisms, new worries. Science 2016, 351, 1263–1264. [Google Scholar] [CrossRef] [PubMed]
- Brown, E.D.; Wright, G.D. Antibacterial drug discovery in the resistance era. Nature 2016, 529, 336–343. [Google Scholar] [CrossRef] [PubMed]
- Potron, A.; Poirel, L.; Nordmann, P. Emerging broad-spectrum resistance in Pseudomonas aeruginosa and Acinetobacter baumannii: Mechanisms and Epidemiology. Int. J. Antimicrob. Agents 2015, 45, 568–585. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bassetti, M.; Vena, A.; Croxatto, A.; Righi, E.; Guery, B. How to manage Pseudomonas aeruginosa infections. Drugs. Context 2018, 7, 212527. [Google Scholar] [CrossRef]
- Horcajada, J.P.; Montero, M.; Oliver, A.; Sorlí, L.; Luque, S.; Gómez-Zorrilla, S.; Benito, N.; Grau, S. Epidemiology and Treatment of Multidrug-Resistant and Extensively Drug-Resistant Pseudomonas aeruginosa Infections. Clin. Microbiol. Rev. 2019, 32, e00031-19. [Google Scholar] [CrossRef]
- Tacconelli, E.; Carrara, E.; Savoldi, A.; Harbarth, S.; Mendelson, M.; Monnet, D.L.; Pulcini, C.; Kahlmeter, G.; Kluytmans, J.; Carmeli, Y.; et al. Discovery, research, and development of new antibiotics: The WHO priority list of antibiotic-resistant bacteria and tuberculosis. Lancet. Infect. Dis. 2018, 18, 318–327. [Google Scholar] [CrossRef]
- Lynskey, N.N.; Lawrenson, R.A.; Sriskandan, S. New understandings in Streptococcus pyogenes. Curr. Opin. Infect. Dis. 2011, 24, 196–202. [Google Scholar] [CrossRef]
- Lu, B.; Fang, Y.; Fan, Y.; Chen, X.; Wang, J.; Zeng, J.; Li, Y.; Zhang, Z.; Huang, L.; Li, H.; et al. High prevalence of Macrolide-resistance and molecular characterization of Streptococcus pyogenes isolates circulating in China from 2009 to 2016. Front. Microbiol. 2017, 8, 1–10. [Google Scholar] [CrossRef]
- Akbar, N.; Khan, N.A.; Sagathevan, K.; Iqbal, M.; Tawab, A.; Siddiqui, R. Gut bacteria of Cuora amboinensis (turtle) produce broad-spectrum antibacterial molecules. Sci. Rep. 2019, 9, 1–19. [Google Scholar] [CrossRef] [Green Version]
- Chevrette, M.G.; Carlson, C.M.; Ortega, H.E.; Thomas, C.; Ananiev, G.E.; Barns, K.J.; Book, A.J.; Cagnazzo, J.; Carlos, C.; Flanigan, W.; et al. The antimicrobial potential of Streptomyces from insect microbiomes. Nat. Commun. 2019, 10, 516. [Google Scholar] [CrossRef] [Green Version]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs from 1981 to 2014. J. Nat. Prod 2016, 79, 629–661. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ling, L.L.; Schneider, T.; Peoples, A.J.; Spoering, A.L.; Engels, I.; Conlon, B.P.; Mueller, A.; Schäberle, T.F.; Hughes, D.E.; Epstein, S.; et al. A new antibiotic kills pathogens without detectable resistance. Nature 2015, 517, 455. [Google Scholar] [CrossRef] [PubMed]
- Qin, Z.; Munnoch, J.T.; Devine, R.; Holmes, N.A.; Seipke, R.F.; Wilkinson, K.A.; Wilkinson, B.; Hutchings, M.I. Formicamycins, antibacterial polyketides produced by Streptomyces formicae isolated from African Tetraponera plant-ants. Chem. Sci. 2017, 8, 3218–3227. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Akbar, N.; Siddiqui, R.; Sagathevan, K.A.; Khan, N.A. Gut bacteria of animals/pests living in polluted environments are a potential source of antibacterials. Appl. Microbiol. Biotechnol. 2019, 103, 3955–3964. [Google Scholar] [CrossRef]
- Akbar, N.; Siddiqui, R.; Iqbal, M.; Sagathevan, K.; Khan, N.A. Gut bacteria of cockroaches are a potential source of antibacterial compound (s). Lett. Appl. Microbiol. 2018, 66, 416–426. [Google Scholar] [CrossRef]
- Ichikawa, S.; Yamaguchi, M.; Matsuda, A. Antibacterial nucleoside natural products inhibiting phospho-MurNAc-pentapeptide translocase; Chemistry and structure-activity relationship. Curr. Med. Chem. 2015, 22, 3951–3979. [Google Scholar] [CrossRef]
- Hartmann, M.; Berditsch, M.; Hawecker, J.; Ardakani, M.F.; Gerthsen, D.; Ulrich, A.S. Damage of the bacterial cell envelope by antimicrobial peptides gramicidin S and PGLa as revealed by transmission and scanning electron microscopy. Antimicrob. Agents Chemother. 2010, 54, 3132–3142. [Google Scholar] [CrossRef] [Green Version]
- Kohanski, M.A.; Dwyer, D.J.; Collins, J.J. How antibiotics kill bacteria: From targets to networks. Nat. Rev. Microbiol. 2010, 8, 423. [Google Scholar] [CrossRef] [Green Version]
- Miller, I.; Chevrette, M.; Kwan, J. Interpreting microbial biosynthesis in the genomic age: Biological and practical considerations. Mar. Drugs 2017, 15, 165. [Google Scholar] [CrossRef]
- Kostylev, M.; Kim, D.Y.; Smalley, N.E.; Salukhe, I.; Greenberg, E.P.; Dandekar, A.A. Evolution of the Pseudomonas aeruginosa quorum-sensing hierarchy. Proc. Natl. Acad. Sci. USA 2019, 116, 7027–7032. [Google Scholar] [CrossRef] [Green Version]
- Abdelrhman, K.F.A.; BaccI, G.; Mancusi, C.; Mengoni, A.; Serena, F.; Ugolini, A. A First Insight into the Gut Microbiota of the Sea Turtle Caretta caretta. Front. Microbiol 2016, 7, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Arizza, V.; VecchionI, L.; Caracappa, S.; Sciurba, G.; Berlinghieri, F.; Gentile, A.; Persichetti, M.F.; Arculeo, M.; Alduina, R. New insights into the gut microbiome in loggerhead sea turtles Caretta caretta stranded on the Mediterranean coast. PLoS ONE 2019, 14, e0220329. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Akbar, N.; Siddiqui, R.; Sagathevan, K.; Iqbal, M.; Khan, N.A. Gut Bacteria of Water Monitor Lizard (Varanus salvator) Are a Potential Source of Antibacterial Compound (s). Antibiotics 2019, 8, 164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wenzel, R.P.; Edmond, M.B. Managing antibiotic resistance. N. Engl. J. Med. 2000, 343, 1961–1963. [Google Scholar] [CrossRef]
- Rachakonda, S.; Cartee, L. Challenges in antimicrobial drug discovery and the potential of nucleoside antibiotics. Curr. Med. Chem. 2004, 11, 775–793. [Google Scholar] [CrossRef]
- Von Nussbaum, F.; Brands, M.; Hinzen, B.; Weigand, S.; Häbich, D. Antibacterial natural products in medicinal chemistry-exodus or revival? Angew. Chem. Int. Ed. 2006, 45, 5072–5129. [Google Scholar] [CrossRef]
- Ruiz, B.; Chávez, A.; Forero, A.; García-Huante, Y.; Romero, A.; Sánchez, M.; Rocha, D.; Sánchez, B.; Rodríguez-Sanoja, R.; Sánchez, S.; et al. Production of microbial secondary metabolites: Regulation by the carbon source. Crit. Rev. Microbiol. 2010, 36, 146–167. [Google Scholar] [CrossRef]
- Demain, A.L.; Fang, A. The natural functions of secondary metabolites. Adv. Biochem. Eng. Biotechnol. 2000, 69, 1–39. [Google Scholar]
- Schneider, T.; Sahl, H.G. An oldie but a goodie–cell wall biosynthesis as antibiotic target pathway. Int. J. Med. Microbiol. 2010, 300, 161–169. [Google Scholar] [CrossRef]
- Rahayu, D.U.C.; Sugita, P. Antibacterial activity of curcumenol from rhizomes of Indonesian Curcuma aeruginosa (Zingiberaceae). Rasayan J. Chem. 2018, 11, 762–765. [Google Scholar] [CrossRef]
- Diastuti, H.; Syah, Y.M.; Juliawaty, L.D.; Singgih, M. Antibacterial Activity of Germacrane Type Sesquiterpenes from Curcuma heyneana Rhizomes. Indones. J. Chem. 2014, 14, 32–36. [Google Scholar] [CrossRef] [Green Version]
- Kacem, N.; Roumy, V.; Duhal, N.; Merouane, F.; Neut, C.; Christen, P.; Hostettmann, K.; Rhouati, S. Chemical composition of the essential oil from Algerian Genista quadriflora Munby and determination of its antibacterial and antifungal activities. Ind Crops Prod 2016, 90, 87–93. [Google Scholar] [CrossRef]
- Pascal, R.A., Jr.; Oliver, M.A.; Chen, Y.J. Alternate substrates and inhibitors of bacterial 4-hydroxyphenylpyruvate dioxygenase. Biochemistry 1985, 24, 3158–3165. [Google Scholar] [CrossRef] [PubMed]
- Or, Y.S. Preparation of Tricyclic Erythromycins as Bactericides. International Patent WO1997017356, 15 March 1997. [Google Scholar]
- Klein, L.L.; Li, L.; Chen, H.J.; Curty, C.B.; DeGoey, D.A.; Grampovnik, D.J.; Leone, C.L.; Thomas, S.A.; Yeung, C.M.; Funk, K.W.; et al. Total synthesis and antifungal evaluation of cyclic aminohexapeptides. Bioorg. Med. Chem. 2000, 8, 1677–1696. [Google Scholar] [CrossRef]
- Ali, S.M.; Khan, N.A.; Sagathevan, K.; Anwar, A.; Siddiqui, R. Biologically active metabolite(s) from haemolymph of red-headed centipede Scolopendra subspinipes possess broad spectrum antibacterial activity. AMB Express 2019, 9, 95. [Google Scholar] [CrossRef]
- Patel, N.M.; Moore, J.D.; Blackwell, H.E.; Amador-Noguez, D. Identification of unanticipated and novel N-acyl L-homoserine lactones (AHLs) using a sensitive non- targeted LC-MS/MS method. PLoS ONE 2016, 11, e0163469. [Google Scholar] [CrossRef]
- Kušar, D.; Šrimpf, K.; Isaković, P.; Kalšek, L.; Hosseini, J.; Zdovc, I.; Kotnik, T.; Vengušt, M.; Tavčar-Kalcher, G. Determination of N-acylhomoserine lactones of Pseudomonas aeruginosa in clinical samples from dogs with otitis externa. BMC Vet. Res. 2016, 12, 233. [Google Scholar] [CrossRef] [Green Version]
- Bharali, P.; Saikia, J.P.; Ray, A.; Konwar, B.K. Rhamnolipid (RL) from Pseudomonas aeruginosa OBP1: A novel chemotaxis and antibacterial agent. Colloids Surf B Biointerfaces 2013, 103, 502–509. [Google Scholar] [CrossRef]
- Dusane, D.H.; Nancharaiah, Y.V.; Zinjarde, S.S.; Venugopalan, V.P. Rhamnolipid mediated disruption of marine Bacillus pumilus biofilms. Colloids Surf B Biointerfaces 2010, 81, 242–248. [Google Scholar] [CrossRef]
- Ali, S.M.; Siddiqui, R.; Ong, S.K.; Shah, M.R.; Anwar, A.; Heard, P.J.; Khan, N.A. Identification and characterization of antibacterial compound (s) of cockroaches (Periplaneta americana). Appl. Microbiol. Biotechnol 2017, 101, 253–286. [Google Scholar] [CrossRef] [PubMed] [Green Version]

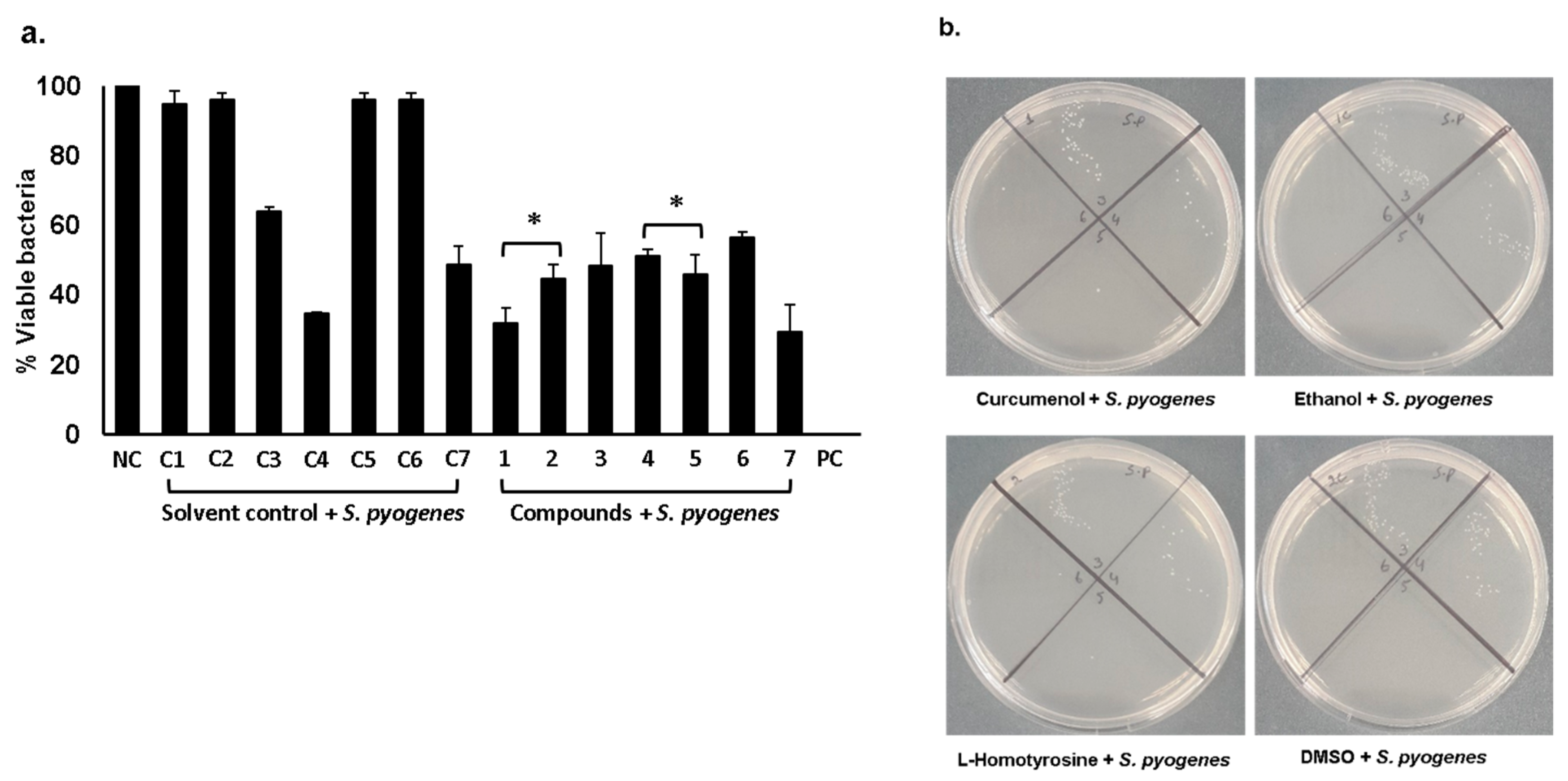

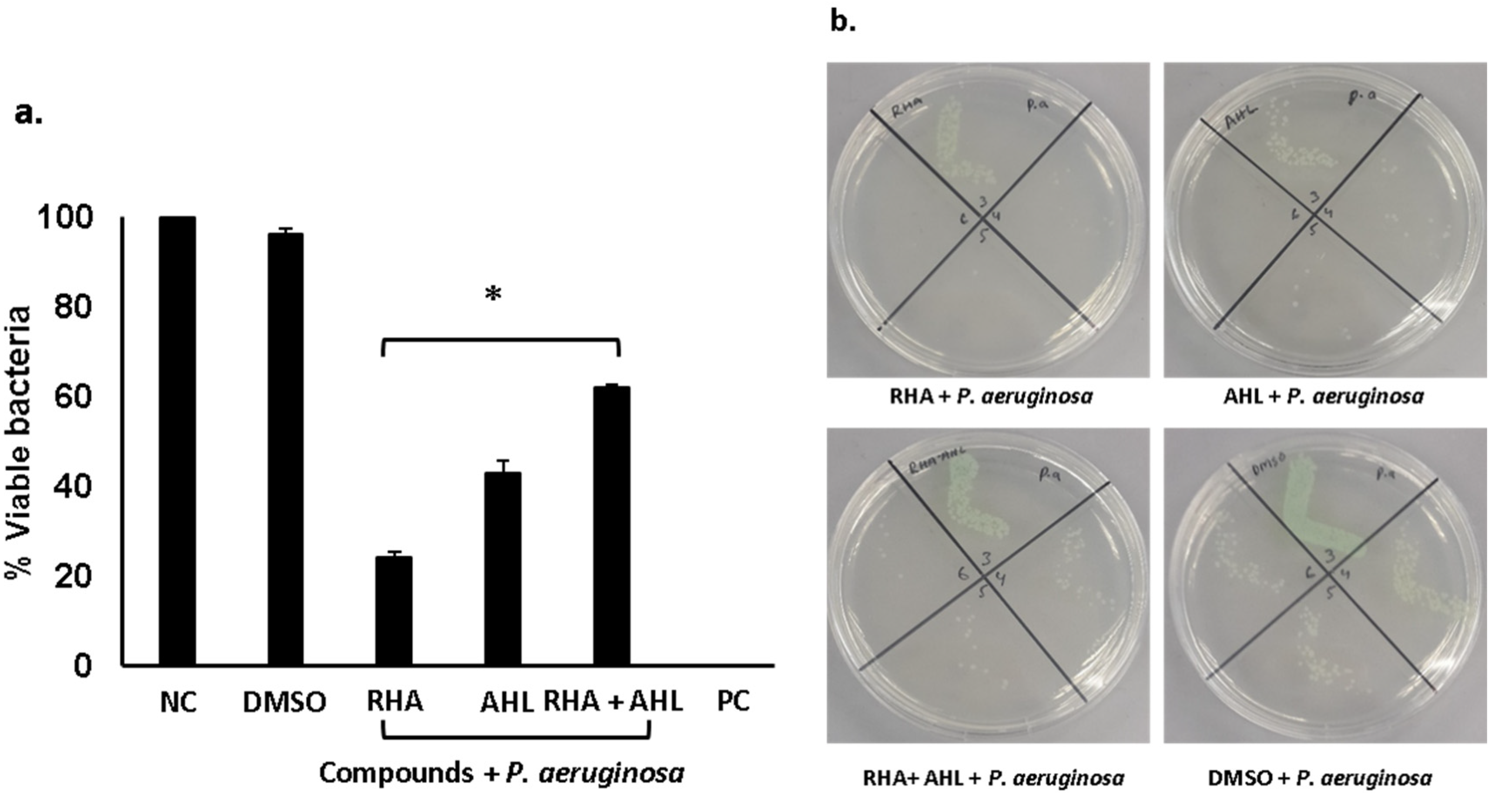


| S. No | Compounds | S. Pyogenes | P. Aeruginosa | ||
|---|---|---|---|---|---|
| MIC50 | MIC90 | MIC50 | MIC90 | ||
| 1 | Rhamnolipids | 34.6 µg/mL | 98.36 µg/mL | 44. 06 µg/mL | 86.87 µg/mL |
| 2 | AHL | 74.1 µg/mL | 140.06 µg/mL | 44.53 µg/mL | 87.73 µg/mL |
| 3 | Curcumenol | 82.26 µg/mL | 156 µg/mL | 82.96 µg/mL | 165.69 µg/mL |
| 4 | L-Homotyrosine | 73.27 µg/mL | 141.45 µg/mL | 51.39 µg/mL | 94.25 µg/mL |
| 5 | Docosanedioic acid | 111.1 µg/mL | 204.76 µg/mL | 220.8 µg/mL | 405.48 µg/mL |
| Codes Given | Compounds/Combinations and Their Concentrations |
|---|---|
| 1 | Curcumenol (50 µg/mL) |
| 2 | L-Homotyrosine (50 µg/mL) |
| 3 | Docosanedioic acid (50 µg/mL) |
| 4 | Curcumenol (25 µg/mL) + L-Homotyrosine (25 µg/mL) |
| 5 | Curcumenol (25 µg/mL) + Docosanedioic acid (25 µg/mL) |
| 6 | L-Homotyrosine (25 µg/mL) + Docosanedioic acid (25 µg/mL) |
| 7 | Curcumenol (16.67 µg/mL) + L-Homotyrosine (16.67 µg/mL) + Docosanedioic acid (16.67 µg/mL) |
| C1 | Ethanol |
| C2 | DMSO |
| C3 | n- hexane |
| C4 | Ethanol + DMSO + n- hexane |
| C5 | Ethanol + DMSO |
| C6 | Ethanol + n-hexane |
| C7 | DMSO + n- hexane |
| NC | Bacteria alone |
| PC | Bacteria + Gentamicin |
| RHA | Rhamnolipids (50 µg/mL) |
| AHL | N-Tetradecanonyl homoserine lactone (50 µg/mL) |
| RHA + AHL | Rhamnolipids (25 µg/mL+ N-Tetradecanonyl homoserine lactone (25 µg/mL) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akbar, N.; Siddiqui, R.; Iqbal, M.; Khan, N.A. Antibacterial Activities of Selected Pure Compounds Isolated from Gut Bacteria of Animals Living in Polluted Environments. Antibiotics 2020, 9, 190. https://0-doi-org.brum.beds.ac.uk/10.3390/antibiotics9040190
Akbar N, Siddiqui R, Iqbal M, Khan NA. Antibacterial Activities of Selected Pure Compounds Isolated from Gut Bacteria of Animals Living in Polluted Environments. Antibiotics. 2020; 9(4):190. https://0-doi-org.brum.beds.ac.uk/10.3390/antibiotics9040190
Chicago/Turabian StyleAkbar, Noor, Ruqaiyyah Siddiqui, Mazhar Iqbal, and Naveed Ahmed Khan. 2020. "Antibacterial Activities of Selected Pure Compounds Isolated from Gut Bacteria of Animals Living in Polluted Environments" Antibiotics 9, no. 4: 190. https://0-doi-org.brum.beds.ac.uk/10.3390/antibiotics9040190







