Differences in Biofilm Formation by Methicillin-Resistant and Methicillin-Susceptible Staphylococcus aureus Strains
Abstract
:1. Introduction
1.1. Differences in Biofilm Formation by MRSA and MSSA
1.2. Biofilm Formation Independent of the icaADBC Operon
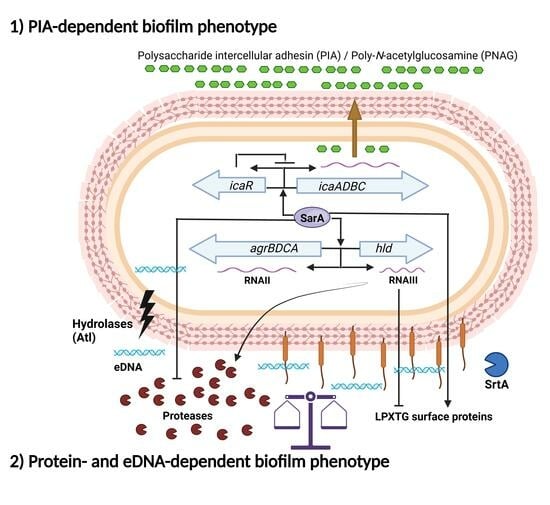
1.3. Main Regulatory Systems in Biofilm Formation by S. aureus
1.4. Concluding Remarks and Future Directions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Peacock, S.J.; Paterson, G.K. Mechanisms of Methicillin Resistance in Staphylococcus aureus. Annu. Rev. Biochem. 2015, 84, 577–601. [Google Scholar] [CrossRef] [PubMed]
- Lakhundi, S.; Zhang, K. Methicillin-Resistant Staphylococcus aureus: Molecular Characterization, Evolution, and Epidemiology. Clin. Microbiol. Rev. 2018, 31, e00020-18. [Google Scholar] [CrossRef] [PubMed]
- Deurenberg, R.H.; Stobberingh, E.E. The evolution of Staphylococcus aureus. Infect. Genet. Evol. 2008, 8, 747–763. [Google Scholar] [CrossRef] [PubMed]
- Gajdács, M. The Continuing Threat of Methicillin-Resistant Staphylococcus aureus. Antibiotics 2019, 8, 52. [Google Scholar] [CrossRef] [PubMed]
- Imani Fooladi, A.A.; Ashrafi, E.; Tazandareh, S.G.; Koosha, R.Z.; Rad, H.S.; Amin, M.; Soori, M.; Larki, R.A.; Choopani, A.; Hosseini, H.M. The distribution of pathogenic and toxigenic genes among MRSA and MSSA clinical isolates. Microb. Pathog. 2015, 81, 60–66. [Google Scholar] [CrossRef]
- Rozgonyi, F.; Kocsis, E.; Kristóf, K.; Nagy, K. Is MRSA more virulent than MSSA? Clin. Microbiol. Infect. 2007, 13, 843–845. [Google Scholar] [CrossRef] [PubMed]
- Gaire, U.; Thapa Shrestha, U.; Adhikari, S.; Adhikari, N.; Bastola, A.; Rijal, K.R.; Ghimire, P.; Banjara, M.R. Antibiotic Susceptibility, Biofilm Production, and Detection of mecA Gene among Staphylococcus aureus Isolates from Different Clinical Specimens. Diseases 2021, 9, 80. [Google Scholar] [CrossRef]
- Goudarzi, M.; Mohammadi, A.; Amirpour, A.; Fazeli, M.; Nasiri, M.J.; Hashemi, A.; Goudarzi, H. Genetic diversity and biofilm formation analysis of Staphylococcus aureus causing urinary tract infections in Tehran, Iran. J. Infect. Dev. Ctries. 2019, 13, 777–785. [Google Scholar] [CrossRef]
- Kadkhoda, H.; Ghalavand, Z.; Nikmanesh, B.; Kodori, M.; Houri, H.; Taghizadeh Maleki, D.; Karimi Bavandpour, A.; Eslami, G. Characterization of biofilm formation and virulence factors of Staphylococcus aureus isolates from paediatric patients in Tehran, Iran. Iran. J. Basic Med. Sci. 2020, 23, 691–698. [Google Scholar] [CrossRef] [PubMed]
- Leshem, T.; Schnall, B.S.; Azrad, M.; Baum, M.; Rokney, A.; Peretz, A. Incidence of biofilm formation among MRSA and MSSA clinical isolates from hospitalized patients in Israel. J. Appl. Microbiol. 2022, 133, 922–929. [Google Scholar] [CrossRef]
- Mahmoudi, H.; Pourhajibagher, M.; Chiniforush, N.; Soltanian, A.R.; Alikhani, M.Y.; Bahador, A. Biofilm formation and antibiotic resistance in meticillin-resistant and meticillin-sensitive Staphylococcus aureus isolated from burns. J. Wound Care 2019, 28, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Ohadian Moghadam, S.; Pourmand, M.R.; Aminharati, F. Biofilm formation and antimicrobial resistance in methicillin-resistant Staphylococcus aureus isolated from burn patients, Iran. J. Infect. Dev. Ctries. 2014, 8, 1511–1517. [Google Scholar] [CrossRef] [PubMed]
- Piechota, M.; Kot, B.; Frankowska-Maciejewska, A.; Grużewska, A.; Woźniak-Kosek, A. Biofilm Formation by Methicillin-Resistant and Methicillin-Sensitive Staphylococcus aureus Strains from Hospitalized Patients in Poland. BioMed Res. Int. 2018, 2018, 4657396. [Google Scholar] [CrossRef]
- Tabandeh, M.; Kaboosi, H.; Taghizadeh Armaki, M.; Pournajaf, A.; Peyravii Ghadikolaii, F. New update on molecular diversity of clinical Staphylococcus aureus isolates in Iran: Antimicrobial resistance, adhesion and virulence factors, biofilm formation and SCCmec typing. Mol. Biol. Rep. 2022, 49, 3099–3111. [Google Scholar] [CrossRef]
- Ghasemian, A.; Najar Peerayeh, S.; Bakhshi, B.; Mirzaee, M. Comparison of Biofilm Formation between Methicillin-Resistant and Methicillin-Susceptible Isolates of Staphylococcus aureus. Iran. Biomed. J. 2016, 20, 175–181. [Google Scholar] [CrossRef]
- Khasawneh, A.I.; Himsawi, N.; Abu-Raideh, J.; Salameh, M.A.; Al-Tamimi, M.; Al Haj Mahmoud, S.; Saleh, T. Status of Biofilm-Forming Genes among Jordanian Nasal Carriers of Methicillin-Sensitive and Methicillin-Resistant Staphylococcus aureus. Iran. Biomed. J. 2020, 24, 386–398. [Google Scholar] [CrossRef]
- Grinholc, M.; Wegrzyn, G.; Kurlenda, J. Evaluation of biofilm production and prevalence of the icaD gene in methicillin-resistant and methicillin-susceptible Staphylococcus aureus strains isolated from patients with nosocomial infections and carriers. FEMS Immunol. Med. Microbiol. 2007, 50, 375–379. [Google Scholar] [CrossRef] [PubMed]
- Abd El Rahman, A.; El Kholy, Y.; Shash, R.Y. Correlation between mazEF Toxin-Antitoxin System Expression and Methicillin Susceptibility in Staphylococcus aureus and Its Relation to Biofilm-Formation. Microorganisms 2021, 9, 2274. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, E.; Pozzi, C.; Houston, P.; Smyth, D.; Humphreys, H.; Robinson, D.A.; O’Gara, J.P. Association between Methicillin Susceptibility and Biofilm Regulation in Staphylococcus aureus Isolates from Device-Related Infections. J. Clin. Microbiol. 2007, 45, 1379–1388. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, F.; Humphreys, H.; O’Gara, J.P. Environmental regulation of biofilm development in methicillin-resistant and methicillin-susceptible Staphylococcus aureus clinical isolates. J. Hosp. Infect. 2006, 62, 120–122. [Google Scholar] [CrossRef]
- Tahmasebi, H.; Dehbashi, S.; Jahantigh, M.; Arabestani, M.R. Relationship between biofilm gene expression with antimicrobial resistance pattern and clinical specimen type based on sequence types (STs) of methicillin-resistant S. aureus. Mol. Biol. Rep. 2020, 47, 1309–1320. [Google Scholar] [CrossRef] [PubMed]
- Belbase, A.; Pant, N.D.; Nepal, K.; Neupane, B.; Baidhya, R.; Baidya, R.; Lekhak, B. Antibiotic resistance and biofilm production among the strains of Staphylococcus aureus isolated from pus/wound swab samples in a tertiary care hospital in Nepal. Ann. Clin. Microbiol. Antimicrob. 2017, 16, 15. [Google Scholar] [CrossRef] [PubMed]
- Indrawattana, N.; Sungkhachat, O.; Sookrung, N.; Chongsa-nguan, M.; Tungtrongchitr, A.; Voravuthikunchai, S.P.; Kong-ngoen, T.; Kurazono, H.; Chaicumpa, W. Staphylococcus aureus clinical isolates: Antibiotic susceptibility, molecular characteristics, and ability to form biofilm. BioMed Res. Int. 2013, 2013, 314654. [Google Scholar] [CrossRef] [PubMed]
- Maharjan, S.; Ansari, M.; Maharjan, P.; Rai, K.R.; Sabina, K.C.; Kattel, H.P.; Rai, G.; Rai, S.K. Phenotypic detection of methicillin resistance, biofilm production, and inducible clindamycin resistance in Staphylococcus aureus clinical isolates in Kathmandu, Nepal. Trop. Med. Health 2022, 50, 71. [Google Scholar] [CrossRef]
- Saud, B.; Khatri, G.; Amatya, N.; Paudel, G.; Shrestha, V. Methicillin-Resistant and Biofilm-Producing Staphylococcus aureus in Nasal Carriage among Health Care Workers and Medical Students. Can. J. Infect. Dis. Med. Microbiol. 2023, 2023, 8424486. [Google Scholar] [CrossRef] [PubMed]
- Senobar Tahaei, S.A.; Stájer, A.; Barrak, I.; Ostorházi, E.; Szabó, D.; Gajdács, M. Correlation Between Biofilm-Formation and the Antibiotic Resistant Phenotype in Staphylococcus aureus Isolates: A Laboratory-Based Study in Hungary and a Review of the Literature. Infect. Drug Resist. 2021, 14, 1155–1168. [Google Scholar] [CrossRef]
- Kırmusaoğlu, S. The Methods for Detection of Biofilm and Screening Antibiofilm Activity of Agents. In Antimicrobials, Antibiotic Resistance, Antibiofilm Strategies and Activity Methods; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Hryniewicz, M.M.; Garbacz, K. Borderline oxacillin-resistant Staphylococcus aureus (BORSA)—A more common problem than expected? J. Med. Microbiol. 2017, 66, 1367–1373. [Google Scholar] [CrossRef]
- Mlynarczyk-Bonikowska, B.; Kowalewski, C.; Krolak-Ulinska, A.; Marusza, W. Molecular Mechanisms of Drug Resistance in Staphylococcus aureus. Int. J. Mol. Sci. 2022, 23, 8088. [Google Scholar] [CrossRef]
- Kumar, S.; Anwer, R.; Yadav, M.; Sehrawat, N.; Singh, M.; Kumar, V. Molecular Typing and Global Epidemiology of Staphylococcus aureus. Curr. Pharmacol. Rep. 2021, 7, 179–186. [Google Scholar] [CrossRef]
- Uehara, Y. Current Status of Staphylococcal Cassette Chromosome mec (SCCmec). Antibiotics 2022, 11, 86. [Google Scholar] [CrossRef]
- Nagasundaram, N.; Sistla, S. Existence of multiple SCCmec elements in clinical isolates of methicillin-resistant Staphylococcus aureus. J. Med. Microbiol. 2019, 68, 720–727. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.T.T.; Nguyen, T.H.; Otto, M. The staphylococcal exopolysaccharide PIA—Biosynthesis and role in biofilm formation, colonization, and infection. Comput. Struct. Biotechnol. J. 2020, 18, 3324–3334, Erratum in Comput. Struct. Biotechnol. J. 2023, 21, 2035. [Google Scholar] [CrossRef] [PubMed]
- Mack, D.; Nedelmann, M.; Krokotsch, A.; Schwarzkopf, A.; Heesemann, J.; Laufs, R. Characterization of transposon mutants of biofilm-producing Staphylococcus epidermidis impaired in the accumulative phase of biofilm production: Genetic identification of a hexosamine-containing polysaccharide intercellular adhesin. Infect. Immun. 1994, 62, 3244–3253. [Google Scholar] [CrossRef]
- Cramton, S.E.; Gerke, C.; Schnell, N.F.; Nichols, W.W.; Goetz, F. The Intercellular Adhesion (ica) Locus Is Present in Staphylococcus aureus and Is Required for Biofilm Formation. Infect. Immun. 1999, 67, 5427–5433. [Google Scholar] [CrossRef]
- Knobloch, J.K.; Horstkotte, M.A.; Rohde, H.; Mack, D. Evaluation of different detection methods of biofilm formation in Staphylococcus aureus. Med. Microbiol. Immunol. 2002, 191, 101–106. [Google Scholar] [CrossRef]
- Fitzpatrick, F.; Humphreys, H.; O’Gara, J.P. Evidence for icaADBC-Independent Biofilm Development Mechanism in Methicillin-Resistant Staphylococcus aureus Clinical Isolates. J. Clin. Microbiol. 2005, 43, 1973–1976. [Google Scholar] [CrossRef] [PubMed]
- Mempel, M.; Feucht, H.; Ziebuhr, W.; Endres, M.; Laufs, R.; Grüter, L. Lack of mecA transcription in slime-negative phase variants of methicillin-resistant Staphylococcus epidermidis. Antimicrob. Agents Chemother. 1994, 38, 1251–1255. [Google Scholar] [CrossRef] [PubMed]
- Mempel, M.; Müller, E.; Hoffmann, R.; Feucht, H.; Laufs, R.; Grüter, L. Variable degree of slime production is linked to different levels of beta-lactam susceptibility in Staphylococcus epidermidis phase variants. Med. Microbiol. Immunol. 1995, 184, 109–113. [Google Scholar] [CrossRef]
- Conlon, K.M.; Humphreys, H.; O Gara, J.P. icaR Encodes a Transcriptional Repressor Involved in Environmental Regulation of ica Operon Expression and Biofilm Formation in Staphylococcus epidermidis. J. Bacteriol. 2002, 184, 4400–4408. [Google Scholar] [CrossRef]
- Rachid, S.; Ohlsen, K.; Witte, W.; Hacker, J.; Ziebuhr, W. Effect of subinhibitory antibiotic concentrations on polysaccharide intercellular adhesin expression in biofilm-forming Staphylococcus epidermidis. Antimicrob. Agents Chemother. 2000, 44, 3357–3363. [Google Scholar] [CrossRef]
- McCarthy, H.; Rudkin, J.K.; Black, N.S.; Gallagher, L.; O’Neill, E.; O’Gara, J.P. Methicillin resistance and the biofilm phenotype in Staphylococcus aureus. Front. Cell. Infect. Microbiol. 2015, 5, 1. [Google Scholar] [CrossRef]
- Beenken, K.E.; Dunman, P.M.; McAleese, F.; Macapagal, D.; Murphy, E.; Projan, S.J.; Blevins, J.S.; Smeltzer, M.S. Global Gene Expression in Staphylococcus aureus Biofilms. J. Bacteriol. 2004, 186, 4665–4684. [Google Scholar] [CrossRef]
- Lim, Y.; Jana, M.; Luong, T.T.; Lee, C.Y. Control of Glucose- and NaCl-Induced Biofilm Formation by rbf in Staphylococcus aureus. J. Bacteriol. 2004, 186, 722–729. [Google Scholar] [CrossRef]
- Cue, D.; Lei, M.G.; Luong, T.T.; Kuechenmeister, L.; Dunman, P.M.; O’Donnell, S.; Rowe, S.; O’Gara, J.P.; Lee, C.Y. Rbf promotes biofilm formation by Staphylococcus aureus via repression of icaR, a negative regulator of icaADBC. J. Bacteriol. 2009, 191, 6363–6373. [Google Scholar] [CrossRef]
- Cucarella, C.; Solano, C.; Valle, J.; Amorena, B.; Lasa, I.; Penades, J.R. Bap, a Staphylococcus aureus Surface Protein Involved in Biofilm Formation. J. Bacteriol. 2001, 183, 2888–2896. [Google Scholar] [CrossRef] [PubMed]
- Gill, S.R.; Fouts, D.E.; Archer, G.L.; Mongodin, E.F.; Deboy, R.T.; Ravel, J.; Paulsen, I.T.; Kolonay, J.F.; Brinkac, L.; Beanan, M.; et al. Insights on evolution of virulence and resistance from the complete genome analysis of an early methicillin-resistant Staphylococcus aureus strain and a biofilm-producing methicillin-resistant Staphylococcus epidermidis strain. J. Bacteriol. 2005, 187, 2426–2438. [Google Scholar] [CrossRef]
- Merino, N.; Toledo-Arana, A.; Vergara-Irigaray, M.; Valle, J.; Solano, C.; Calvo, E.; Lopez, J.A.; Foster, T.J.; Penadés, J.R.; Lasa, I. Protein A-mediated multicellular behavior in Staphylococcus aureus. J. Bacteriol. 2009, 191, 832–843. [Google Scholar] [CrossRef]
- O’Neill, E.; Pozzi, C.; Houston, P.; Humphreys, H.; Robinson, D.A.; Loughman, A.; Foster, T.J.; O’Gara, J.P. A novel Staphylococcus aureus biofilm phenotype mediated by the fibronectin-binding proteins, FnBPA and FnBPB. J. Bacteriol. 2008, 190, 3835–3850. [Google Scholar] [CrossRef] [PubMed]
- Geoghegan, J.A.; Corrigan, R.M.; Gruszka, D.T.; Speziale, P.; O’Gara, J.P.; Potts, J.R.; Foster, T.J. Role of surface protein SasG in biofilm formation by Staphylococcus aureus. J. Bacteriol. 2010, 192, 5663–5673. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, K.; Jularic, M.; Horsburgh, S.M.; Hirschhausen, N.; Neumann, C.; Bertling, A.; Schulte, A.; Foster, S.; Kehrel, B.E.; Peters, G.; et al. Molecular characterization of a novel Staphylococcus aureus surface protein (SasC) involved in cell aggregation and biofilm accumulation. PLoS ONE 2009, 4, e7567. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, E.; Humphreys, H.; O’Gara, J.P. Carriage of both the fnbA and fnbB genes and growth at 37 degrees C promote FnBP-mediated biofilm development in meticillin-resistant Staphylococcus aureus clinical isolates. J. Med. Microbiol. 2009, 58 Pt 4, 399–402. [Google Scholar] [CrossRef]
- Izano, E.A.; Amarante, M.A.; Kher, W.B.; Kaplan, J.B. Differential roles of poly-N-acetylglucosamine surface polysaccharide and extracellular DNA in Staphylococcus aureus and Staphylococcus epidermidis biofilms. Appl. Environ. Microbiol. 2008, 74, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Rice, K.C.; Mann, E.E.; Endres, J.L.; Weiss, E.C.; Cassat, J.E.; Smeltzer, M.S.; Bayles, K.W. The cidA murein hydrolase regulator contributes to DNA release and biofilm development in Staphylococcus aureus. Proc. Natl. Acad. Sci. USA 2007, 104, 8113–8118. [Google Scholar] [CrossRef]
- Bose, J.L.; Lehman, M.K.; Fey, P.D.; Bayles, K.W. Contribution of the Staphylococcus aureus Atl AM and GL murein hydrolase activities in cell division, autolysis, and biofilm formation. PLoS ONE 2012, 7, e42244. [Google Scholar] [CrossRef]
- Houston, P.; Rowe, S.E.; Pozzi, C.; Waters, E.M.; O’Gara, J.P. Essential role for the major autolysin in the fibronectin-binding protein-mediated Staphylococcus aureus biofilm phenotype. Infect. Immun. 2011, 79, 1153–1165. [Google Scholar] [CrossRef]
- Liu, Q.; Wang, X.; Qin, J.; Cheng, S.; Yeo, W.S.; He, L.; Ma, X.; Liu, X.; Li, M.; Bae, T. The ATP-Dependent Protease ClpP Inhibits Biofilm Formation by Regulating Agr and Cell Wall Hydrolase Sle1 in Staphylococcus aureus. Front. Cell. Infect. Microbiol. 2017, 7, 181. [Google Scholar] [CrossRef] [PubMed]
- Vergara-Irigaray, M.; Valle, J.; Merino, N.; Latasa, C.; García, B.; Ruiz de Los Mozos, I.; Solano, C.; Toledo-Arana, A.; Penadés, J.R.; Lasa, I. Relevant role of fibronectin-binding proteins in Staphylococcus aureus biofilm-associated foreign-body infections. Infect. Immun. 2009, 77, 3978–3991. [Google Scholar] [CrossRef]
- Vuong, C.; Saenz, H.L.; Götz, F.; Otto, M. Impact of the agr quorum-sensing system on adherence to polystyrene in Staphylococcus aureus. J. Infect. Dis. 2000, 182, 1688–1693. [Google Scholar] [CrossRef]
- Jenul, C.; Horswill, A.R. Regulation of Staphylococcus aureus Virulence. Microbiol. Spectr. 2019, 7. [Google Scholar] [CrossRef] [PubMed]
- Boles, B.R.; Horswill, A.R. Agr-mediated dispersal of Staphylococcus aureus biofilms. PLoS Pathog. 2008, 4, e1000052. [Google Scholar] [CrossRef] [PubMed]
- Dunman, P.M.; Murphy, E.; Haney, S.; Palacios, D.; Tucker-Kellogg, G.; Wu, S.; Brown, E.L.; Zagursky, R.J.; Shlaes, D.; Projan, S.J. Transcription profiling-based identification of Staphylococcus aureus genes regulated by the agr and/or sarA loci. J. Bacteriol. 2001, 183, 7341–7353. [Google Scholar] [CrossRef] [PubMed]
- Beenken, K.E.; Blevins, J.S.; Smeltzer, M.S. Mutation of sarA in Staphylococcus aureus Limits Biofilm Formation. Infect. Immun. 2003, 71, 4206–4211. [Google Scholar] [CrossRef] [PubMed]
- Chien, Y.; Manna, A.C.; Cheung, A.L. SarA level is a determinant of agr activation in Staphylococcus aureus. Mol. Microbiol. 1998, 30, 991–1001. [Google Scholar] [CrossRef] [PubMed]
- Blevins, J.S.; Beenken, K.E.; Elasri, M.O.; Hurlburt, B.K.; Smeltzer, M.S. Strain-dependent differences in the regulatory roles of sarA and agr in Staphylococcus aureus. Infect. Immun. 2002, 70, 470–480. [Google Scholar] [CrossRef] [PubMed]
- Trotonda, M.P.; Manna, A.C.; Cheung, A.L.; Lasa, I.; Penadés, J.R. SarA positively controls bap-dependent biofilm formation in Staphylococcus aureus. J. Bacteriol. 2005, 187, 5790–5798. [Google Scholar] [CrossRef]
- Wolz, C.; Pöhlmann-Dietze, P.; Steinhuber, A.; Chien, Y.T.; Manna, A.; van Wamel, W.; Cheung, A. Agr-independent regulation of fibronectin-binding protein(s) by the regulatory locus sar in Staphylococcus aureus. Mol. Microbiol. 2000, 36, 230–243. [Google Scholar] [CrossRef]
- Valle, J.; Toledo-Arana, A.; Berasain, C.; Ghigo, J.M.; Amorena, B.; Penadés, J.R.; Lasa, I. SarA and not sigmaB is essential for biofilm development by Staphylococcus aureus. Mol. Microbiol. 2003, 48, 1075–1087. [Google Scholar] [CrossRef]
- Campbell, M.J.; Beenken, K.E.; Ramirez, A.M.; Smeltzer, M.S. The major role of sarA in limiting Staphylococcus aureus extracellular protease production in vitro is correlated with decreased virulence in diverse clinical isolates in osteomyelitis. Virulence 2023, 14, 2175496. [Google Scholar] [CrossRef]
- Ramirez, A.M.; Beenken, K.E.; Byrum, S.D.; Tackett, A.J.; Shaw, L.N.; Gimza, B.D.; Smeltzer, M.S. SarA plays a predominant role in controlling the production of extracellular proteases in the diverse clinical isolates of Staphylococcus aureus LAC and UAMS-1. Virulence 2020, 11, 1738–1762. [Google Scholar] [CrossRef] [PubMed]
- Knobloch, J.K.; Jäger, S.; Horstkotte, M.A.; Rohde, H.; Mack, D. RsbU-dependent regulation of Staphylococcus epidermidis biofilm formation is mediated via the alternative sigma factor sigmaB by repression of the negative regulator gene icaR. Infect. Immun. 2004, 72, 3838–3848. [Google Scholar] [CrossRef]
- Cerca, N.; Brooks, J.L.; Jefferson, K.K. Regulation of the intercellular adhesin locus regulator (icaR) by SarA, sigmaB, and IcaR in Staphylococcus aureus. J. Bacteriol. 2008, 190, 6530–6533. [Google Scholar] [CrossRef] [PubMed]
- Ma, R.; Qiu, S.; Jiang, Q.; Sun, H.; Xue, T.; Cai, G.; Sun, B. AI-2 quorum sensing negatively regulates rbf expression and biofilm formation in Staphylococcus aureus. Int. J. Med. Microbiol. 2017, 307, 257–267. [Google Scholar] [CrossRef] [PubMed]


| MRSA > MSSA | MRSA = MSSA | MRSA < MSSA | |
|---|---|---|---|
| mecA + DDM | 40% (8/20) REF. [7,8,11,14] (Cef), [9,12] (Ox), [10,13] (Nm) | 15% (3/20) REF. [15] (Ox), [16] (Ox/Cef), [19] (Nd) | 5% (1/20) REF. [17] (Nm) |
| DDM | 10% (2/20) REF. [18,21] (Cef) | 30% (6/20) REF. [24,25,26] (Cef), [22] (Ox), [23] (Ox/Cef), [20] (Nm) | 0% (0/20) |
| Total (100%) | 50% | 45% | 5% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hernández-Cuellar, E.; Tsuchiya, K.; Valle-Ríos, R.; Medina-Contreras, O. Differences in Biofilm Formation by Methicillin-Resistant and Methicillin-Susceptible Staphylococcus aureus Strains. Diseases 2023, 11, 160. https://0-doi-org.brum.beds.ac.uk/10.3390/diseases11040160
Hernández-Cuellar E, Tsuchiya K, Valle-Ríos R, Medina-Contreras O. Differences in Biofilm Formation by Methicillin-Resistant and Methicillin-Susceptible Staphylococcus aureus Strains. Diseases. 2023; 11(4):160. https://0-doi-org.brum.beds.ac.uk/10.3390/diseases11040160
Chicago/Turabian StyleHernández-Cuellar, Eduardo, Kohsuke Tsuchiya, Ricardo Valle-Ríos, and Oscar Medina-Contreras. 2023. "Differences in Biofilm Formation by Methicillin-Resistant and Methicillin-Susceptible Staphylococcus aureus Strains" Diseases 11, no. 4: 160. https://0-doi-org.brum.beds.ac.uk/10.3390/diseases11040160