Sitagliptin Potentiates the Anti-Neoplastic Activity of Doxorubicin in Experimentally-Induced Mammary Adenocarcinoma in Mice: Implication of Oxidative Stress, Inflammation, Angiogenesis, and Apoptosis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Drugs and Chemicals
2.2. Cell Line
2.3. Animals
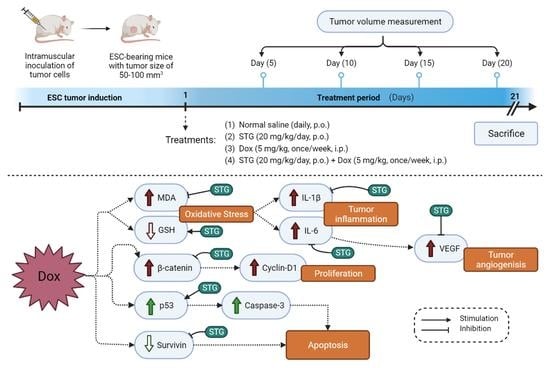
2.4. Experimental Design
- Positive control group: mice daily received normal saline by oral gavage for 21 days;
- Dox group: mice received Dox (5 mg/kg, once/week by i.p.) for 21 days [21]; and
- STG + Dox group: mice received STG (20 mg/kg/day, orally) and Dox (5 mg/kg, once/week by i.p.) for 21 days.
2.5. Tumor Volume and Growth Inhibition Rate
2.6. Tissue Samples Collection
2.7. Assessment of Oxidative Stress Markers
2.8. Enzyme-Linked Immunosorbent Assay (ELISA)
2.9. Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR)
2.10. Histopathological Examination
2.11. Terminal dUTP Nick-End Labeling (TUNEL) Assay
2.12. Immunohistochemical (IHC) Analysis
2.13. Statistical Analysis
3. Results
3.1. Growth Suppressive Effect of STG, Dox, and Their Combination on ESC-Bearing Mice
3.2. Effect of Treatment with STG, Dox, and Their Combination on Oxidative Status and Inflammatory Parameters in Tumor Tissue
3.3. Effect of Treatment with STG, Dox, and Their Combination on the Tumor Tissue Visualized by the Histopathological Examination
3.4. Effect of Treatment with STG, Dox and Their Combination on the Gene Expression of the Angiogenic Marker, VEGF, and the Oncogenic β-Catenin, Cyclin-D1, and Survivin in EAC Tissues
3.5. Effect of Treatment with STG, Dox, and Their Combination on nuclear DNA Fragmentation Illustrated by TUNEL Staining Assay
3.6. Effect of Treatment with STG, Dox, and Their Combination on the Protein Levels of Tumor p53 and Caspase-3
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Feitosa, I.B.; Mori, B.; Teles, C.B.G.; da Costa, A.G. What are the immune responses during the growth of Ehrlich’s tumor in ascitic and solid form? Life Sci. 2020, 264, 118578. [Google Scholar] [CrossRef] [PubMed]
- Bailly, C.; Thuru, X.; Quesnel, B. Combined cytotoxic chemotherapy and immunotherapy of cancer: Modern times. NAR Cancer 2020, 2, zcaa002. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Podyacheva, E.Y.; Kushnareva, E.A.; Karpov, A.A.; Toropova, Y.G. Analysis of Models of Doxorubicin-Induced Cardiomyopathy in Rats and Mice. A Modern View from the Perspective of the Pathophysiologist and the Clinician. Front. Pharmacol. 2021, 12, 670479. [Google Scholar] [CrossRef]
- Christowitz, C.; Davis, T.; Isaacs, A.; van Niekerk, G.; Hattingh, S.; Engelbrecht, A.-M. Mechanisms of doxorubicin-induced drug resistance and drug resistant tumour growth in a murine breast tumour model. BMC Cancer 2019, 19, 757. [Google Scholar] [CrossRef] [Green Version]
- Mentoor, I.; Engelbrecht, A.-M.; van Jaarsveld, P.J.; Nell, T. Chemoresistance: Intricate Interplay between Breast Tumor Cells and Adipocytes in the Tumor Microenvironment. Front. Endocrinol. 2018, 9, 758. [Google Scholar] [CrossRef] [Green Version]
- Ji, X.; Lu, Y.; Tian, H.; Meng, X.; Wei, M.; Cho, W.C. Chemoresistance mechanisms of breast cancer and their countermeasures. Biomed. Pharmacother. 2019, 114, 108800. [Google Scholar] [CrossRef]
- Majidpoor, J.; Mortezaee, K. Angiogenesis as a hallmark of solid tumors—Clinical perspectives. Cell Oncol. 2021, 44, 715–737. [Google Scholar] [CrossRef]
- Yazbeck, R.; Jaenisch, S.E.; Abbott, C.A. Dipeptidyl peptidase 4 inhibitors: Applications in innate immunity? Biochem. Pharmacol. 2021, 188, 114517. [Google Scholar] [CrossRef]
- Choi, H.J.; Kim, J.Y.; Lim, S.C.; Kim, G.; Yun, H.J.; Choi, H.S. Dipeptidyl peptidase 4 promotes epithelial cell transformation and breast tumourigenesis via induction of PIN1 gene expression. Br. J. Pharmacol. 2015, 172, 5096–5109. [Google Scholar] [CrossRef]
- Beckenkamp, A.; Davies, S.; Willig, J.B.; Buffon, A. DPPIV/CD26: A tumor suppressor or a marker of malignancy? Tumor Biol. 2016, 37, 7059–7073. [Google Scholar] [CrossRef] [PubMed]
- Qin, C.J.; Zhao, L.H.; Zhou, X.; Zhang, H.L.; Wen, W.; Tang, L.; Zeng, M.; Wang, M.D.; Fu, G.B.; Huang, S.; et al. Inhibition of dipeptidyl peptidase IV prevents high fat diet-induced liver cancer angiogenesis by downregulating chemokine ligand 2. Cancer Lett. 2018, 420, 26–37. [Google Scholar] [CrossRef] [PubMed]
- Kawakita, E.; Koya, D.; Kanasaki, K. CD26/DPP-4: Type 2 Diabetes Drug Target with Potential Influence on Cancer Biology. Cancers 2021, 13, 2191. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Wen, D.; Cheng, Z.; Yang, Y.; Zheng, G.; Yin, F. Effect of sitagliptin, a DPP-4 inhibitor, against DENA-induced liver cancer in rats mediated via NF-κB activation and inflammatory cytokines. J. Biochem. Mol. Toxicol. 2018, 32, e22220. [Google Scholar] [CrossRef]
- Amritha, C.A.; Kumaravelu, P.; Chellathai, D.D. Evaluation of Anti Cancer Effects of DPP-4 Inhibitors in Colon Cancer—An Invitro Study. J. Clin. Diagn. Res. 2015, 9, FC14–FC16. [Google Scholar]
- Trocha, M.; Fleszar, M.G.; Fortuna, P.; Lewandowski, Ł.; Gostomska-Pampuch, K.; Sozański, T.; Merwid-Ląd, A.; Krzystek-Korpacka, M. Sitagliptin Modulates Oxidative, Nitrative and Halogenative Stress and Inflammatory Response in Rat Model of Hepatic Ischemia-Reperfusion. Antioxidants 2021, 10, 1168. [Google Scholar] [CrossRef]
- Louis, K.S.; Siegel, A.C. Cell viability analysis using trypan blue: Manual and automated methods. Methods Mol. Biol. 2011, 740, 7–12. [Google Scholar]
- Vincent, P.C.; Nicholls, A. Comparison of the growth of the Ehrlich ascites tumor in male and female mice. Cancer Res. 1967, 27, 1058–1065. [Google Scholar]
- Adami, E.R.; Corso, C.R.; Turin-Oliveira, N.M.; Galindo, C.M.; Milani, L.; Stipp, M.C.; da Silva, L.C.M.; do Nascimento, G.E.; Chaves, P.F.P.; Chequin, A.; et al. Polysaccharides from green sweet pepper increase the antineoplastic effect of methotrexate on mammary tumor cells. Int. J. Biol. Macromol. 2020, 158, 1071–1081. [Google Scholar] [CrossRef]
- Dong, Q.; Teng, S.-W.; Wang, Y.; Qin, F.; Li, Y.; Ai, L.-L.; Yu, H. Sitagliptin protects the cognition function of the Alzheimer’s disease mice through activating glucagon-like peptide-1 and BDNF-TrkB signalings. Neurosci. Lett. 2019, 696, 184–190. [Google Scholar] [CrossRef]
- Rana, T.; Chakrabarti, A.; Freeman, M.; Biswas, S. Doxorubicin-mediated bone loss in breast cancer bone metastases is driven by an interplay between oxidative stress and induction of TGFβ. PLoS ONE 2013, 8, e78043. [Google Scholar] [CrossRef]
- Higuchi, T.; Miyake, K.; Sugisawa, N.; Oshiro, H.; Zhang, Z.; Razmjooei, S.; Yamamoto, N.; Hayashi, K.; Kimura, H.; Miwa, S.; et al. Olaratumab combined with doxorubicin and ifosfamide overcomes individual doxorubicin and olaratumab resistance of an undifferentiated soft-tissue sarcoma in a PDOX mouse model. Cancer Lett. 2019, 451, 122–127. [Google Scholar] [CrossRef] [PubMed]
- Facchini, J.M.; Alves, E.P.; Aguilera, C.; Gern, R.M.; Silveira, M.L.; Wisbeck, E.; Furlan, S.A. Antitumor activity of Pleurotus ostreatus polysaccharide fractions on Ehrlich tumor and Sarcoma 180. Int. J. Biol Macromol. 2014, 68, 72–77. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2−ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Prieto-Callejero, B.; Rivera, F.; Fagundo-Rivera, J.; Romero, A.; Romero-Martín, M.; Gómez-Salgado, J.; Ruiz-Frutos, C. Relationship between chemotherapy-induced adverse reactions and health-related quality of life in patients with breast cancer. Medicine 2020, 99, e21695. [Google Scholar] [CrossRef]
- Abrahamyan, S.; Sahakyan, I.; Tumasyan, N.; Kocharyan, N.; Simonyan, A.; Aroutiounian, R.; Chailyan, G.; Chailyan, S.; Davtyan, T.; Galoian, K. Morpho-functional study of the hypothalamic proline-rich polypeptide apoptotic activity against mouse Ehrlich ascites carcinoma. Oncol. Rep. 2020, 44, 196–212. [Google Scholar] [CrossRef]
- Liu, Q.; Wu, D.; Ma, Y.; Cao, Y.; Pang, Y.; Tang, M.; Pu, Y.; Zhang, T. Intracellular reactive oxygen species trigger mitochondrial dysfunction and apoptosis in cadmium telluride quantum dots-induced liver damage. NanoImpact 2022, 25, 100392. [Google Scholar] [CrossRef]
- Thorn, C.F.; Oshiro, C.; Marsh, S.; Hernandez-Boussard, T.; McLeod, H.; Klein, T.E.; Altman, R.B. Doxorubicin pathways: Pharmacodynamics and adverse effects. Pharmacogenet. Genom. 2011, 21, 440–446. [Google Scholar] [CrossRef]
- Cappetta, D.; De Angelis, A.; Sapio, L.; Prezioso, L.; Illiano, M.; Quaini, F.; Rossi, F.; Berrino, L.; Naviglio, S.; Urbanek, K. Oxidative Stress and Cellular Response to Doxorubicin: A Common Factor in the Complex Milieu of Anthracycline Cardiotoxicity. Oxid. Med. Cell Longev. 2017, 2017, 1521020. [Google Scholar] [CrossRef]
- Marques, C.; Gonçalves, A.; Pereira, P.M.R.; Almeida, D.; Martins, B.; Fontes-Ribeiro, C.; Reis, F.; Fernandes, R. The dipeptidyl peptidase 4 inhibitor sitagliptin improves oxidative stress and ameliorates glomerular lesions in a rat model of type 1 diabetes. Life Sci. 2019, 234, 116738. [Google Scholar] [CrossRef]
- Li, Y.; Tian, Q.; Li, Z.; Dang, M.; Lin, Y.; Hou, X. Activation of Nrf2 signaling by sitagliptin and quercetin combination against β-amyloid induced Alzheimer’s disease in rats. Drug Dev. Res. 2019, 80, 837–845. [Google Scholar] [CrossRef]
- Zhang, Y.Y.; Yi, M.; Huang, Y.P. Oxymatrine Ameliorates Doxorubicin-Induced Cardiotoxicity in Rats. Cell Physiol. Biochem. 2017, 43, 626–635. [Google Scholar] [CrossRef] [PubMed]
- Reuter, S.; Gupta, S.C.; Chaturvedi, M.M.; Aggarwal, B.B. Oxidative stress, inflammation, and cancer: How are they linked? Free Radic. Biol. Med. 2010, 49, 1603–1616. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumari, N.; Dwarakanath, B.S.; Das, A.; Bhatt, A.N. Role of interleukin-6 in cancer progression and therapeutic resistance. Tumor Biol. 2016, 37, 11553–11572. [Google Scholar] [CrossRef] [PubMed]
- Rébé, C.; Ghiringhelli, F. Interleukin-1β and Cancer. Cancers 2020, 12, 1791. [Google Scholar] [CrossRef] [PubMed]
- Guo, R.M.; Xu, W.M.; Lin, J.C.; Mo, L.Q.; Hua, X.X.; Chen, P.X.; Wu, K.; Zheng, D.D.; Feng, J.Q. Activation of the p38 MAPK/NF-κB pathway contributes to doxorubicin-induced inflammation and cytotoxicity in H9c2 cardiac cells. Mol. Med. Rep. 2013, 8, 603–608. [Google Scholar] [CrossRef] [Green Version]
- Hajra, S.; Patra, A.R.; Basu, A.; Saha, P.; Bhattacharya, S. Indole-3-Carbinol (I3C) enhances the sensitivity of murine breast adenocarcinoma cells to doxorubicin (DOX) through inhibition of NF-κβ, blocking angiogenesis and regulation of mitochondrial apoptotic pathway. Chem. Biol. Interact. 2018, 290, 19–36. [Google Scholar] [CrossRef]
- Arunachalam, S.; Nagoor Meeran, M.F.; Azimullah, S.; Sharma, C.; Goyal, S.N.; Ojha, S. Nerolidol Attenuates Oxidative Stress, Inflammation, and Apoptosis by Modulating Nrf2/MAPK Signaling Pathways in Doxorubicin-Induced Acute Cardiotoxicity in Rats. Antioxidants 2021, 10, 984. [Google Scholar] [CrossRef]
- He, Y.; Yang, G.; Yao, F.; Xian, Y.; Wang, G.; Chen, L.; Lv, X.; Gao, H.; Zheng, Z.; Sun, L.; et al. Sitagliptin inhibits vascular inflammation via the SIRT6-dependent signaling pathway. Int. Immunopharmacol. 2019, 75, 105805. [Google Scholar] [CrossRef]
- Syukri, A.; Hatta, M.; Amir, M.; Rohman, M.S.; Mappangara, I.; Kaelan, C.; Wahyuni, S.; Bukhari, A.; Junita, A.R.; Primaguna, M.R.; et al. Doxorubicin induced immune abnormalities and inflammatory responses via HMGB1, HIF1-α and VEGF pathway in progressive of cardiovascular damage. Ann. Med. Surg. 2022, 76, 103501. [Google Scholar] [CrossRef]
- Setrerrahmane, S.; Xu, H. Tumor-related interleukins: Old validated targets for new anti-cancer drug development. Mol. Cancer 2017, 16, 153. [Google Scholar] [CrossRef]
- Yang, X.; Zhang, X.; Wu, R.; Huang, Q.; Jiang, Y.; Qin, J.; Yao, F.; Jin, G.; Zhang, Y. DPPIV promotes endometrial carcinoma cell proliferation, invasion and tumorigenesis. Oncotarget 2017, 8, 8679–8692. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Azbazdar, Y.; Karabicici, M.; Erdal, E.; Ozhan, G. Regulation of Wnt Signaling Pathways at the Plasma Membrane and Their Misregulation in Cancer. Front. Cell Dev. Biol. 2021, 9, 631623. [Google Scholar] [CrossRef] [PubMed]
- Lecarpentier, Y.; Schussler, O.; Hébert, J.-L.; Vallée, A. Multiple Targets of the Canonical WNT/β-Catenin Signaling in Cancers. Front. Oncol. 2019, 9, 1248. [Google Scholar] [CrossRef] [PubMed]
- Ji, Y.; Lv, J.; Sun, D.; Huang, Y. Therapeutic strategies targeting Wnt/β-catenin signaling for colorectal cancer (Review). Int. J. Mol. Med. 2022, 49, 1. [Google Scholar] [CrossRef]
- Ren, R.; Yu, J.; Zhang, Y.; Wang, S.F.; Guo, X.; Shen, M.; Xu, M.D.; Jiang, M.; Zhi, Q.; Chen, K.; et al. Inflammation Promotes Progression of Pancreatic Cancer through WNT/β-Catenin Pathway-Dependent Manner. Pancreas 2019, 48, 1003–1014. [Google Scholar] [CrossRef]
- Zhou, Y.; Liang, C.; Xue, F.; Chen, W.; Zhi, X.; Feng, X.; Bai, X.; Liang, T. Salinomycin decreases doxorubicin resistance in hepatocellular carcinoma cells by inhibiting the β-catenin/TCF complex association via FOXO3a activation. Oncotarget 2015, 6, 10350–10365. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Z.; Sun, Y.; Qu, M.; Wan, H.; Cai, F.; Zhang, P. Inhibiting the MNK-eIF4E-β-catenin axis increases the responsiveness of aggressive breast cancer cells to chemotherapy. Oncotarget 2017, 8, 2906–2915. [Google Scholar] [CrossRef] [Green Version]
- Zuryń, A.; Litwiniec, A.; Klimaszewska-Wiśniewska, A.; Nowak, J.M.; Gackowska, L.; Myśliwiec, B.J.; Pawlik, A.; Grzanka, A. Expression of cyclin D1 after treatment with doxorubicin in the HL-60 cell line. Cell Biol. Int. 2014, 38, 857–867. [Google Scholar] [CrossRef]
- Pistritto, G.; Trisciuoglio, D.; Ceci, C.; Garufi, A.; D’Orazi, G. Apoptosis as anticancer mechanism: Function and dysfunction of its modulators and targeted therapeutic strategies. Aging 2016, 8, 603–619. [Google Scholar] [CrossRef] [Green Version]
- Cheteh, E.H.; Sarne, V.; Ceder, S.; Bianchi, J.; Augsten, M.; Rundqvist, H.; Egevad, L.; Östman, A.; Wiman, K.G. Interleukin-6 derived from cancer-associated fibroblasts attenuates the p53 response to doxorubicin in prostate cancer cells. Cell Death Discov. 2020, 6, 42. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; He, Z.; Liu, B.; Wang, P.; Chen, Y. Downregulation of Meg3 enhances cisplatin resistance of lung cancer cells through activation of the WNT/β-catenin signaling pathway. Mol. Med. Rep. 2015, 12, 4530–4537. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kelleni, M.T.; Amin, E.F.; Abdelrahman, A.M. Effect of Metformin and Sitagliptin on Doxorubicin-Induced Cardiotoxicity in Rats: Impact of Oxidative Stress, Inflammation, and Apoptosis. J. Toxicol. 2015, 2015, 424813. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, Y.; Guo, Z.; Yan, W.; Wang, W. Cardiovascular effects of sitagliptin—An anti-diabetes medicine. Clin. Exp. Pharmacol. Physiol. 2018, 45, 628–635. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aziz, T.A. Cardioprotective Effect of Quercetin and Sitagliptin in Doxorubicin-Induced Cardiac Toxicity in Rats. Cancer Manag. Res. 2021, 13, 2349–2357. [Google Scholar] [CrossRef]
- Jo, C.H.; Kim, S.; Park, J.S.; Kim, G.H. Anti-Inflammatory Action of Sitagliptin and Linagliptin in Doxorubicin Nephropathy. Kidney Blood Press Res. 2018, 43, 987–999. [Google Scholar] [CrossRef]








| Primer | Sequence | NCBI Reference Sequence | Amplification Size | Annealing Temperature | |
|---|---|---|---|---|---|
| GAPDH | F | 5′-ATGGTGAAGGTCGGTGTGAAC-3′ | NM_008084.3 | 251 | 60 °C |
| R | 5′-TTGATGTTAGTGGGGTCTCGC-3′ | ||||
| VEGF | F | 5′-CACGACAGAAGGAGAGCAGAAG-3′ | NM_001025250.3 | 82 | 60 °C |
| R | 5′-CTCAATCGGACGGCAGTAGC-3′ | ||||
| β-Catenin | F | 5′-GTTCGCCTTCATTATGGACTGCC-3′ | NM_007614.3 | 146 | 60 °C |
| R | 5′-ATAGCACCCTGTTCCCGCAAAG-3′ | ||||
| Cyclin-D1 | F | 5′-AGAAGTGCGAAGAGGAGGTC-3′ | NM_001379248.1 | 157 | 60 °C |
| R | 5′-TTCTCGGCAGTCAAGGGAAT-3′ | ||||
| Survivin | F | 5′-GAATCCTGCGTTTGAGTCGT-3′ | NM_001012273.1 | 207 | 60 °C |
| R | 5′-AATCAGGCTCGTTCTCGGTA-3′ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salama, M.M.; Zaghloul, R.A.; Khalil, R.M.; El-Shishtawy, M.M. Sitagliptin Potentiates the Anti-Neoplastic Activity of Doxorubicin in Experimentally-Induced Mammary Adenocarcinoma in Mice: Implication of Oxidative Stress, Inflammation, Angiogenesis, and Apoptosis. Sci. Pharm. 2022, 90, 42. https://0-doi-org.brum.beds.ac.uk/10.3390/scipharm90030042
Salama MM, Zaghloul RA, Khalil RM, El-Shishtawy MM. Sitagliptin Potentiates the Anti-Neoplastic Activity of Doxorubicin in Experimentally-Induced Mammary Adenocarcinoma in Mice: Implication of Oxidative Stress, Inflammation, Angiogenesis, and Apoptosis. Scientia Pharmaceutica. 2022; 90(3):42. https://0-doi-org.brum.beds.ac.uk/10.3390/scipharm90030042
Chicago/Turabian StyleSalama, Mohamed M., Randa A. Zaghloul, Rania M. Khalil, and Mamdouh M. El-Shishtawy. 2022. "Sitagliptin Potentiates the Anti-Neoplastic Activity of Doxorubicin in Experimentally-Induced Mammary Adenocarcinoma in Mice: Implication of Oxidative Stress, Inflammation, Angiogenesis, and Apoptosis" Scientia Pharmaceutica 90, no. 3: 42. https://0-doi-org.brum.beds.ac.uk/10.3390/scipharm90030042