Graphene Nanosheets Based Cathodes for Lithium-Oxygen Batteries
Abstract
:1. Introduction
2. Results and Discussion
2.1. Microstructure of GNSs

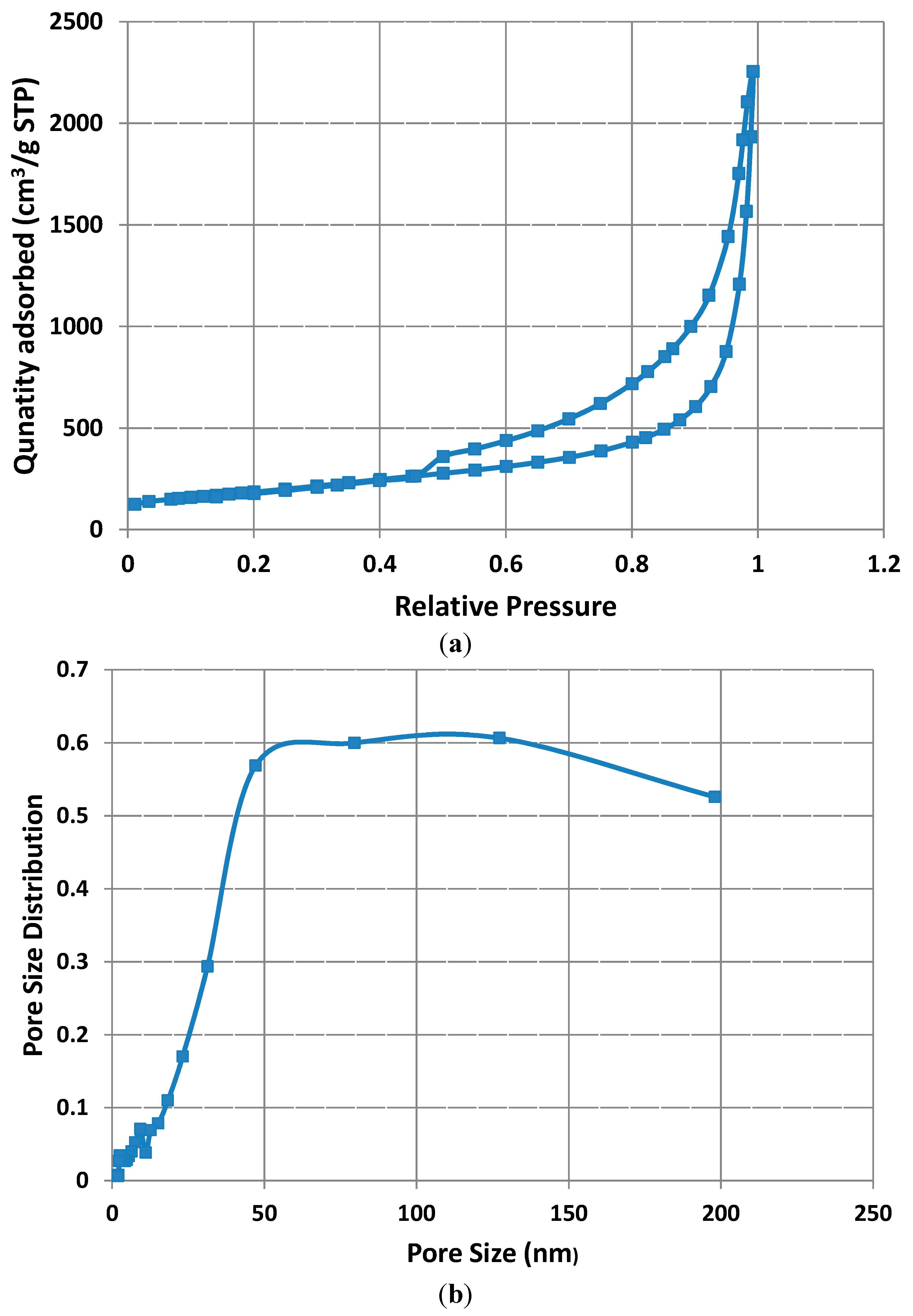
2.2. Nitrogen Adsorption-Desorption Isotherm
| Cathode Materials | N-C Blend | GNSs | N-GNSs | Ex-N-GNSs | Hybrid 1 |
|---|---|---|---|---|---|
| Surface Area (m2/g) | 1385 | 482 | 581.6 | 686.6 | 900 |
| Pore volume (cm3/g) | 1.8 | 1.3 | 1.72 | 1.86 | 1.9 |
| Average Pore Size (nm) | 6.3 | 10.96 | 11.18 | 11.36 | 11.86 |
| Porosity (%) | 42 | 8.76 | 21.88 | 24.5 | 30 |
| Oxygen Reduction Potential (V vs. SCE) | −0.45 | −0.389 | −0.466 | −0.434 | −0.596 |
| Open Circuit voltage (V) | 3.18 | 3.4 | 3.6 | 3.5 | 3.25 |
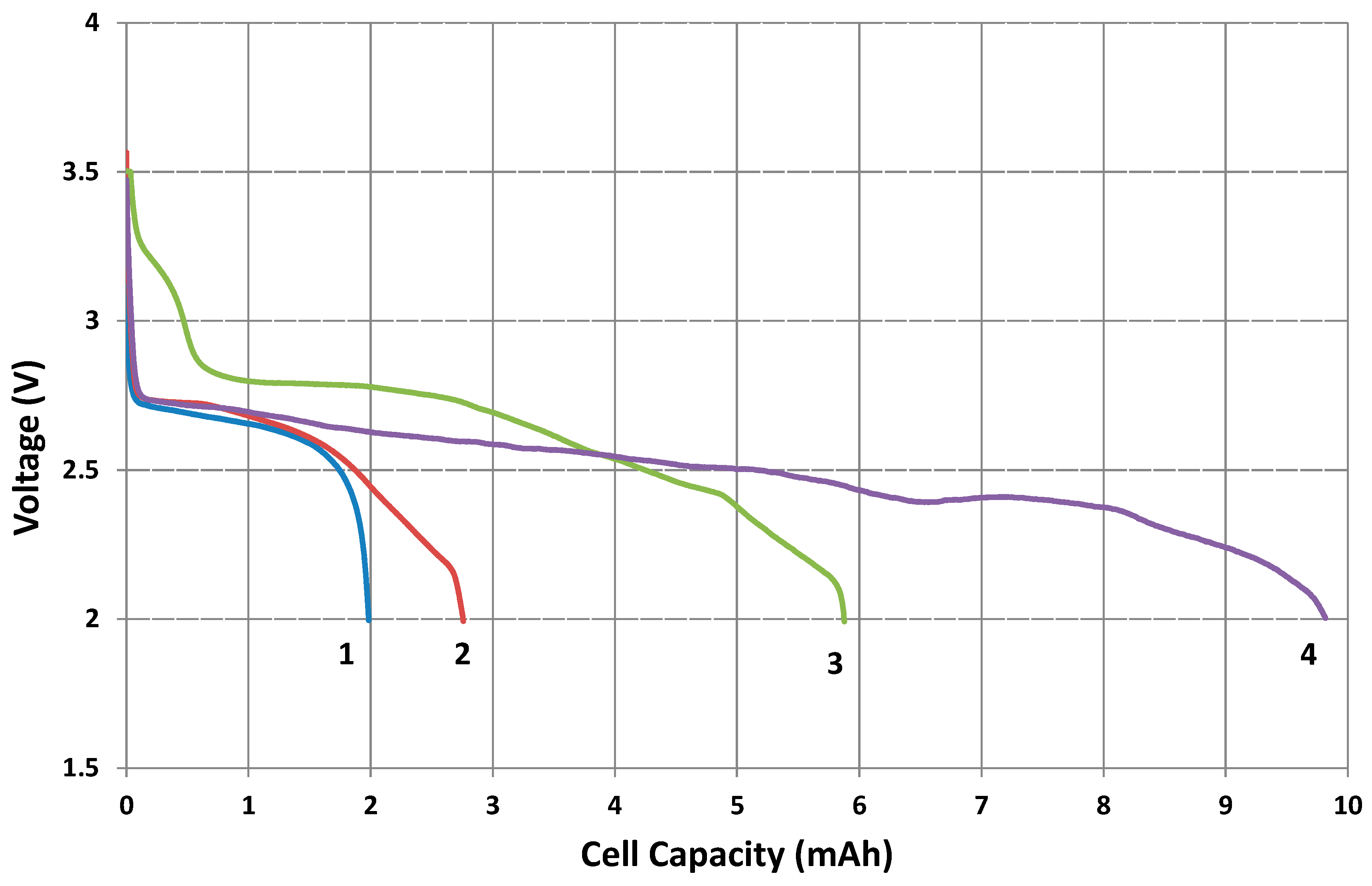
| Discharge cell capacity (mAh) | 1.44 | 1.98 | 2.75 | 5.87 | 9.82 |
| Specific Capacity (mAh/g Graphene and/or Carbon) | 167 | 330 | 456.8 | 1028 | 1687.3 |
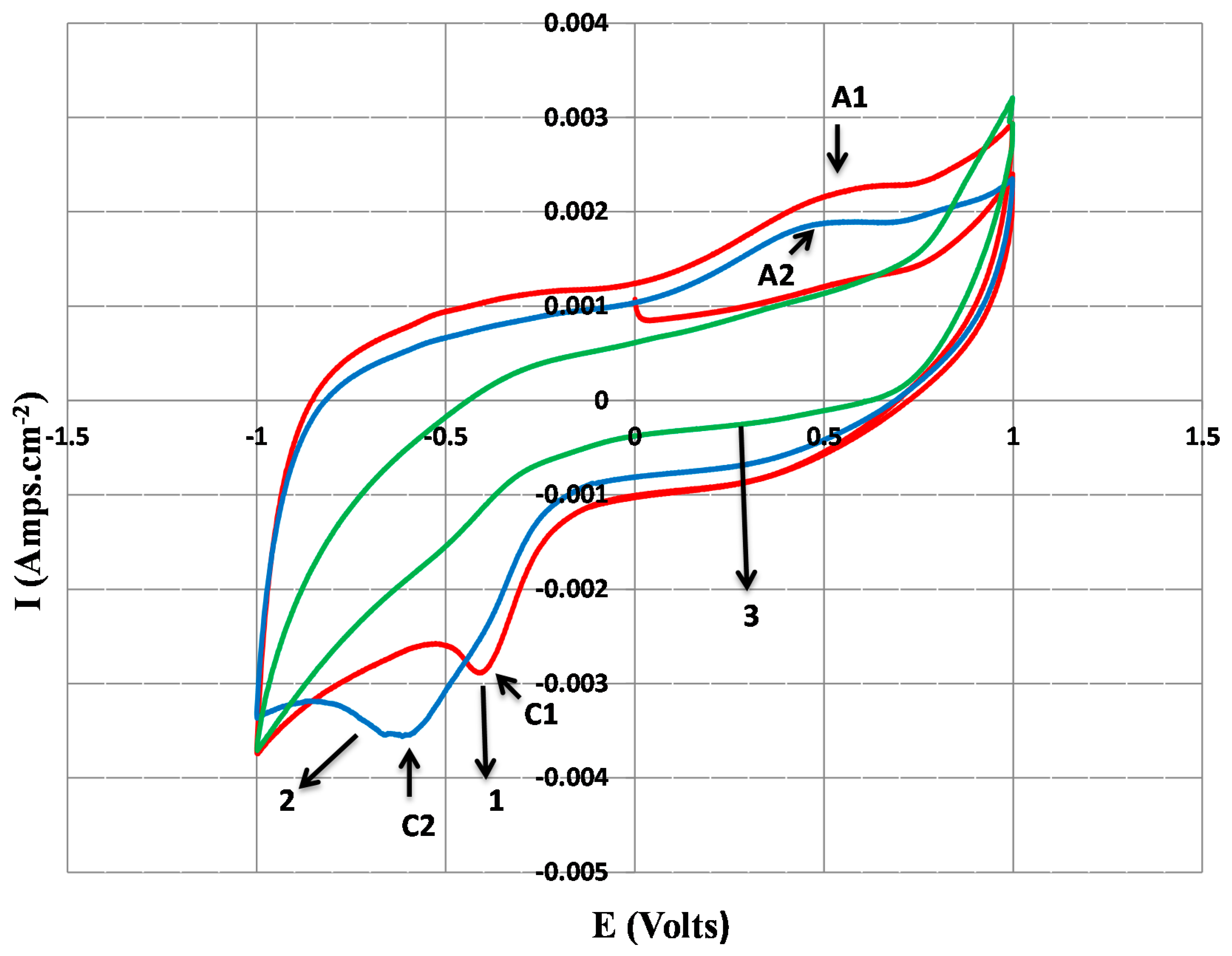
2.3. Cyclic Voltammetry
2.4. Discharge Profile of Cathodes in Lithium-Oxygen Battery
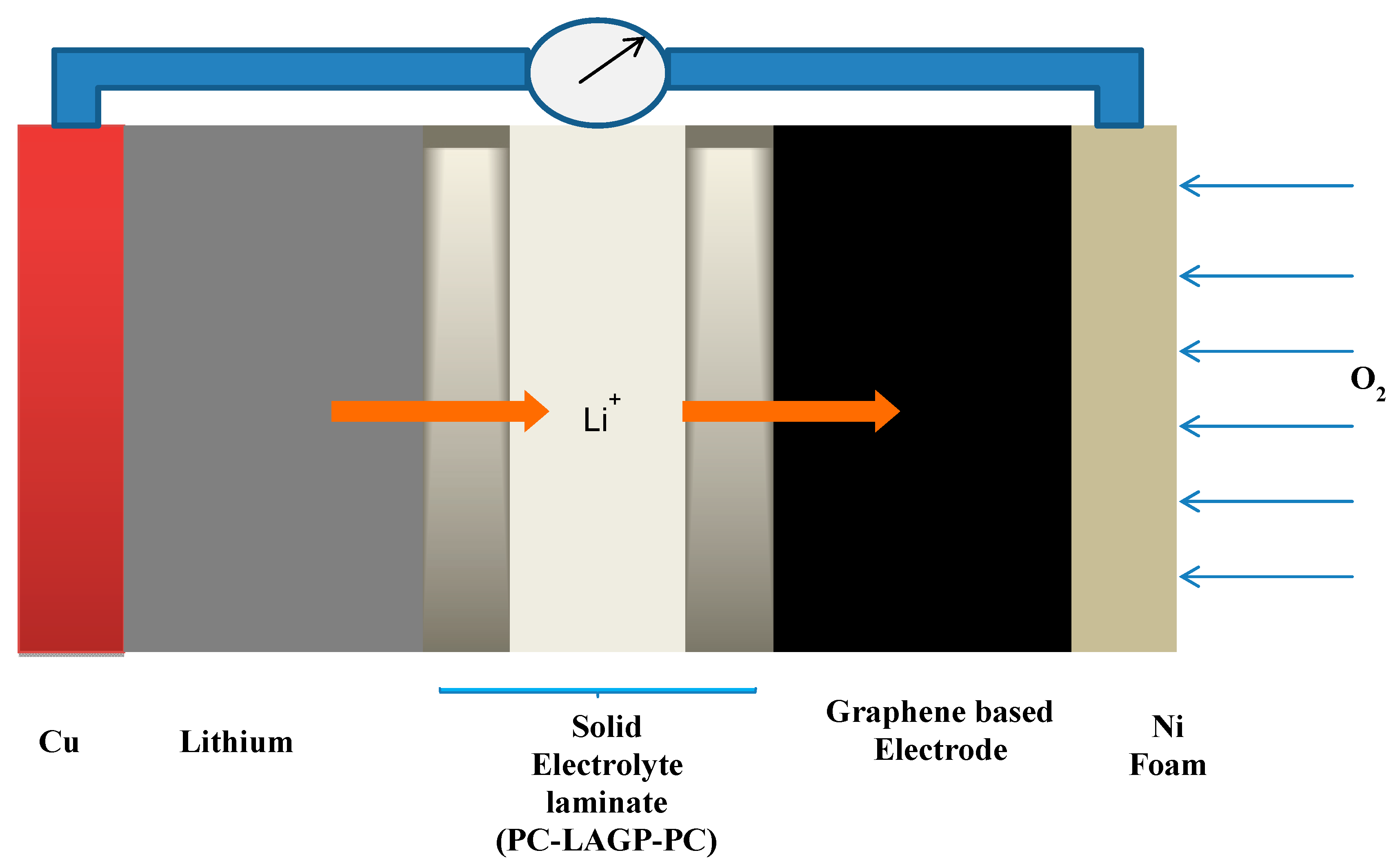
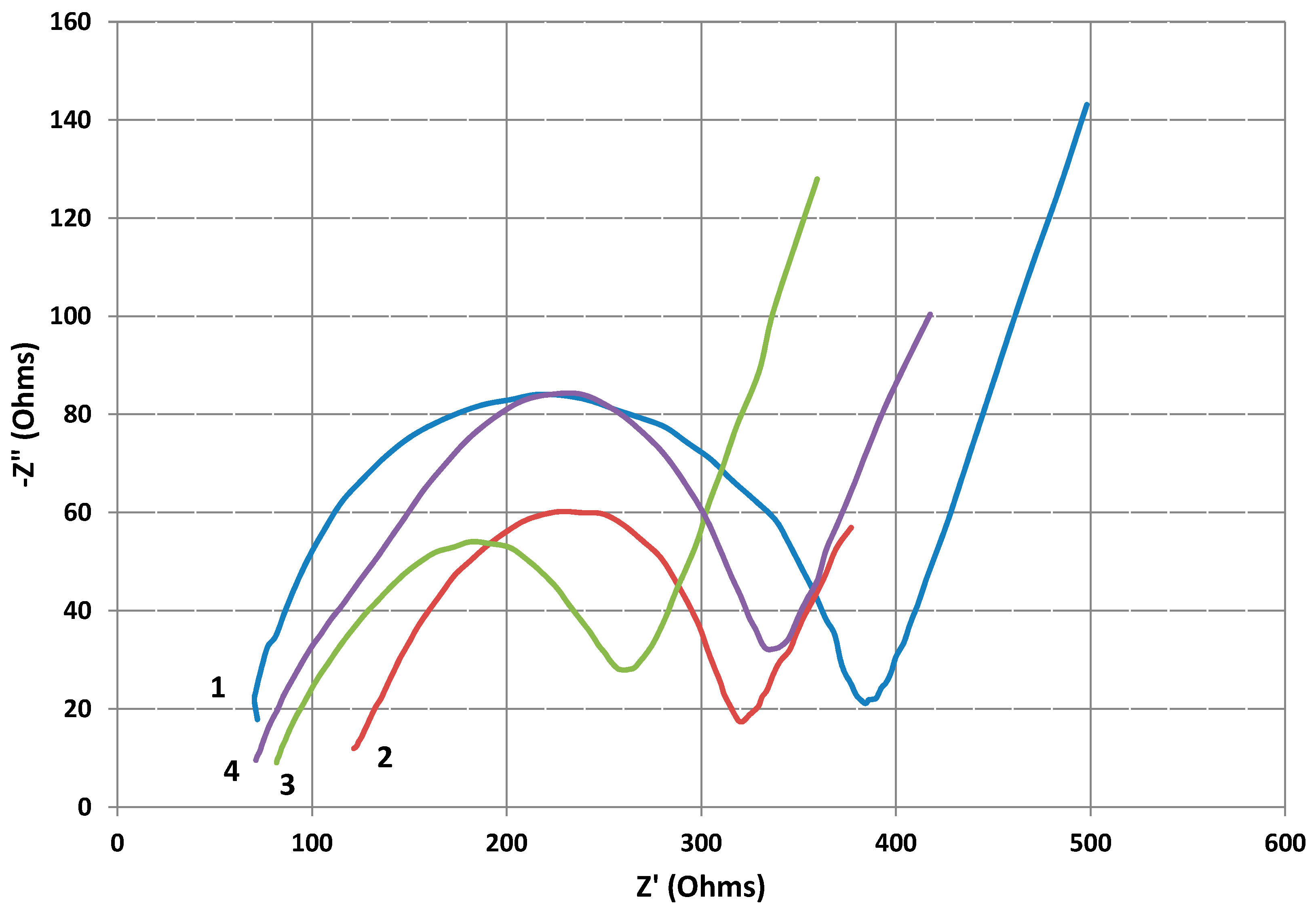
2.5. Electrochemical Impedance Spectroscopy of Cathodes
3. Experimental Section
3.1. Preparation of Cathode Material
3.2. Characterization of Cathode Materials
3.3. Fabrication and Electrochemical Performance of Cathode
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Larcher, D.; Tarascon, J.M. Towards greener and more sustainable batteries for electrical energy storage. Nat. Chem. 2015, 7, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Van Noorden, R. A better battery. Nature 2014, 507, 26–28. [Google Scholar] [CrossRef] [PubMed]
- Bruce, P.G.; Freunberger, S.A.; Hardwick, L.J.; Tarascon, J.M. Li-O2 and Li-S batteries with high energy storage. Nat. Mater. 2012, 1, 19–29. [Google Scholar]
- Linden, D. Handbook of Batteries and Fuel Cells, 2nd ed.; McGraw-Hill: New York, NY, USA, 1984. [Google Scholar]
- Lu, Y.-C.; Gasteiger, H.A.; Parent, M.C.; Chiloyan, V.; Shao-Horn, Y. The influence of catalysts on discharge and charge voltages of rechargeable Li-oxygen batteries. Electrochemical. Solid State Lett. 2010, 13, A69–A72. [Google Scholar] [CrossRef]
- Gallagher, K.G.; Goebel, S.; Greszler, T.; Mathias, M.; Oelerich, W.; Eroglu, D.; Srinivasan, V. Quantifying the promise of lithium-air batteries for electric vehicles. Energy Environ. Sci. 2014, 7, 1555–1563. [Google Scholar] [CrossRef]
- Luntz, A.C.; McCloskey, B.D. Nonaqueous Li-air batteries: A status report. Chem. Rev. 2014, 114, 11721–11750. [Google Scholar] [CrossRef] [PubMed]
- Girishkumar, G.; McCloskey, B.; Luntz, A.C.; Swanson, S.D.; Wilcke, W.J. Lithium-air battery: Promise and challenges. J. Phys. Chem. Lett. 2010, 1, 2193–2203. [Google Scholar] [CrossRef]
- Lu, J.; Park, J.-B.; Sun, Y.-K.; Wu, F.; Amine, K. Aprotic and aqueous Li-O2 batteries. Chem. Rev. 2014, 114, 5611–5640. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Zhou, H.B. A reversible long-life lithium-air battery in ambient air. Nat. Commun. 2013. [Google Scholar] [CrossRef] [PubMed]
- Oh, D.; Qi, J.; Lu, Y.C.; Zhang, Y.; Shao-Horn, Y.; Belcher, A.M. Biologically enhanced cathode design for improved capacity and cycle life for lithium-oxygen batteries. Nat. Commun. 2013. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.D.; Song, H.; Kim, J.; Gwon, H.; Bae, Y.; Park, K.Y.; Hong, J.; Kim, H.; Kim, T.; Kim, Y.H.; et al. Superior rechargeability and efficiency of lithium-oxygen batteries: Hierarchical air electrode architecture combined with a soluble catalyst. Angew. Chem. Int. Ed. 2014, 53, 3926–3931. [Google Scholar] [CrossRef] [PubMed]
- Grande, L.; Paillard, E.; Hassoun, J.; Park, J.-B.; Lee, Y.-J.; Sun, Y.-K.; Passerini, S.; Scrosati, B. The lithium/air battery: Still an emerging system or a practical reality? Adv. Mater. 2015, 27, 784–800. [Google Scholar] [CrossRef] [PubMed]
- Balaish, M.; Peled, E.; Golodnitsky, D.; Ein-Eli, Y. Liquid-free lithium-oxygen batteries. Angew. Chem. Int. Ed. 2015, 54, 436–440. [Google Scholar] [CrossRef]
- Abraham, K.M.; Jiang, Z. A polymer electrolyte-based rechargeable lithium/oxygen battery. J. Electrochem. Soc. 1996, 143, 1–5. [Google Scholar] [CrossRef]
- Zhang, S.S.; Foster, D.; Read, J. Discharge characteristics of a non-aqueous electrolyte Li/O2 battery. J. Power Sources 2010, 195, 1235–1240. [Google Scholar] [CrossRef]
- Beattie, S.D.; Manolescu, D.M.; Blair, S.L. High-capacity lithium-air cathodes. J. Electrochem. Soc. 2009, 156, A44–A47. [Google Scholar] [CrossRef]
- Wang, D.; Xiao, J.; Xu, W.; Zhang, J.-G. High capacity pouch-type Li-air batteries. J. Electrochem. Soc. 2010, 157, A760–A764. [Google Scholar] [CrossRef]
- Eswaran, M.; Munichandraiah, N.; Scalon, L.G. High capacity Li-O2 cell and electrochemical impedance spectroscopy. Electrochem. Solid State Lett. 2010, 13, A121–A124. [Google Scholar] [CrossRef]
- Trahey, L.; Johnson, C.S.; Vaughey, J.T.; Kang, S.H.; Hardwick, L.J.; Freunberger, S.A.; Bruce, P.G.; Thackeray, M.M. Activated lithium-metal-oxides as catalytic electrodes for Li-O2 cells. Electrochem. Solid State Lett. 2011, 14, A64–A66. [Google Scholar] [CrossRef]
- Shui, J.; Du, F.; Xue, C.; Li, Q.; Dai, L. Vertically aligned N-doped coral-like carbon fiber arrays as efficient air electrodes for high-performance nonaqueous Li-O2 batteries. ACS Nano 2014, 8, 3015–3022. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.C.; Xu, Z.; Gasteiger, H.A.; Chen, S.; Hamad-Schifferli, K.; Shao-Horn, Y. Platinum-gold nanoparticles: A highly active bifunctional electrocatalyst for rechargeable lithium-air batteries. J. Am. Chem. Soc. 2010, 132, 12170–12171. [Google Scholar] [CrossRef] [PubMed]
- Ren, X.; Zhang, S.S.; Tran, D.T.; Read, J. Oxygen reduction reaction catalyst on lithium/air battery discharge performance. J. Mater. Chem. 2011, 21, 10118–10125. [Google Scholar] [CrossRef]
- Nasybulin, E.; Xu, W.; Engelhard, M.H.; Nie, Z.; Burton, S.D.; Cosimbescu, L.; Gross, M.E.; Zhang, J.-G. Effect of electrolyte salts on the performance of Li-O2 batteries. J. Phys. Chem. C 2013, 117, 2635–2645. [Google Scholar] [CrossRef]
- Elia, G.A.; Hassoun, J.; Kwak, W.-J.; Sun, Y.-K.; Scrosati, B.; Mueller, F.; Bresser, D.; Passerini, S.; Oberhumer, P.; Tsiouvara, N.; et al. An advanced lithium-air battery exploiting an ionic liquid-based electrolyte. Nano Lett. 2014, 14, 6572–6577. [Google Scholar] [CrossRef] [PubMed]
- Laoire, C.O.; Mukerjee, S.; Plichta, E.J.; Hendrickson, M.A.; Abraham, K.M. Influence of nonaqueous solvents on the electrochemistry of oxygen in the rechargeable lithium-air battery. J. Phys. Chem. C 2010, 114, 9178–9186. [Google Scholar] [CrossRef]
- Freunberger, S.A.; Chen, Y.; Peng, Z.; Griffin, J.M.; Hardwick, L.J.; Barde, F.; Novak, P.; Bruce, P.G. Reactions in the rechargeable lithium-O2 battery with alkyl carbonate electrolytes. J. Am. Chem. Soc. 2011, 133, 8040–8047. [Google Scholar] [CrossRef] [PubMed]
- Park, H.W.; Lee, D.U.; Nazar, L.F.; Chen, Z. Oxygen reduction reaction using MnO2 nanotubes/nitrogen-doped exfoliated graphene hybrid catalyst for Li-O2 battery applications. J. Electrochem. Soc. 2013, 160, A344–A350. [Google Scholar] [CrossRef]
- Safari, M.; Adams, B.D.; Nazar, L.F. Kinetics of oxygen reduction in aprotic Li-O2 cells: A model-based study. J. Phys. Chem. Lett. 2014, 5, 3486–3491. [Google Scholar] [CrossRef] [PubMed]
- Meini, S.; Piana, M.; Tsiouvaras, N.; Garsuch, A.; Gasteiger, H.A. The effect of water on the discharge capacity of a non-catalyzed carbon cathode for Li-O2 batteries. Electrochem. Solid State Lett. 2012, 15, A45–A48. [Google Scholar] [CrossRef]
- Kim, H.; Lim, H.D.; Kim, J.; Kang, K. Graphene for advanced Li/S and Li/air batteries. J. Mater. Chem. A 2014, 2, 33–47. [Google Scholar] [CrossRef]
- Li, Y.; Wang, J.; Li, X.; Geng, D.; Li, R.; Sun, X. Superior energy capacity of graphene nanosheets for a nonaqueous lithium-oxygen battery. Chem. Commun. 2011, 47, 9438–9440. [Google Scholar] [CrossRef] [PubMed]
- Yoo, E.; Nakamura, J.; Zhou, H. N-doped graphene nanosheets for Li-air fuel cells under acidic conditions. Energy. Environ. Sci. 2012, 5, 6928–6932. [Google Scholar] [CrossRef]
- Wang, H.; Yang, Y.; Liang, Y.; Zheng, G.; Li, Y.; Cui, Y.; Dai, H. Rechargeable Li-O2 batteries with a covalently coupled MnCo2O4-graphene hybrid as an oxygen cathode catalyst. Energy Environ. Sci. 2012, 5, 7931–7933. [Google Scholar] [CrossRef]
- Wang, Z.-L.; Xu, D.; Xu, J.-J.; Zhang, L.-L.; Zhang, X.-B. Graphene oxide gel-derived, free-standing, hierarchically porous carbon for high-capacity and high-rate rechargeable Li-O2 batteries. Adv. Funct. Mater. 2012, 22, 3699–3705. [Google Scholar] [CrossRef]
- Xiao, J.; Mei, D.; Li, X.; Xu, W.; Wang, D.; Graff, G.L.; Bennett, W.D.; Nie, Z.; Saraf, L.V.; Aksay, I.A.; et al. Hierarchically porous graphene as a lithium-air battery electrode. Nano Lett. 2011, 11, 5071–5078. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Mack, N.H.; Gao, W.; Ma, S.; Zhong, R.; Han, J.; Baldwin, J.K.; Zelenay, P. Nitrogen-doped graphene-rich catalysts derived from heteroatom polymers for oxygen reduction in nonaquous lithium-oxygen battery cathodes. ACS Nano 2012, 6, 9764–9776. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, J.; Li, X.; Geng, D.; Banis, M.N.; Li, R.; Sun, X. Nitrogen-doped graphene nanosheets as cathode materials with excellent electrocatalytic activity for high capacity lithium-oxygen batteries. Electrochem. Commun. 2012, 18, 12–15. [Google Scholar] [CrossRef]
- Kumar, B.; Kumar, J.; Leese, R.; Fellner, J.P.; Rodrigues, S.J.; Abraham, K.M. A solid-state, rechargeable, long cycle life lithium-air battery. J. Electrochem. Soc. 2010, 157, A50–A54. [Google Scholar] [CrossRef]
- Kichambare, P.; Kumar, J.; Rodrigues, S.; Kumar, B. Electrochemical performance of highly mesoporous nitrogen doped carbon cathode in lithium-oxygen batteries. J. Power Sources 2011, 196, 3310–3316. [Google Scholar] [CrossRef]
- Kichambare, P.; Rodrigues, S.; Kumar, J. Mesoporous nitrogen-doped carbon-glass ceramic cathodes for solid-state lithium-oxygen batteries. ACS Appl. Mater. Interfaces 2012, 4, 49–52. [Google Scholar] [CrossRef] [PubMed]
- Kichambare, P.; Rodrigues, S. Mesoporous nitrogen-doped carbon-LiSICON glass ceramics as high performance cathodes in solid-state lithium-oxygen batteries. Energy Technol. 2013, 1, 209–211. [Google Scholar] [CrossRef]
- Wang, X.; Li, X.; Zhang, L.; Yoon, Y.; Weber, P.K.; Wang, H.; Guo, J.; Dai, H. N-doping of graphene through electrochemical reactions with ammonia. Science 2009, 324, 768–771. [Google Scholar] [CrossRef] [PubMed]
- Ozaki, J.; Kimura, N.; Anahara, T.; Oya, A. Preparation and oxygen reduction activity of BN-doped carbons. Carbon 2007, 45, 1847–1853. [Google Scholar] [CrossRef]
- Allen, B.L.; Kichambare, P.D.; Star, A. Synthesis, characterization, and manipulation of nitrogen-doped carbon nanotubes cups. ACS Nano 2008, 2, 1914–1920. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Allen, B.L.; Kauffman, D.R.; Star, A. Electrocatalytic activity of nitrogen-doped carbon nanotube cups. J. Am. Chem. Soc. 2009, 131, 13200–13201. [Google Scholar] [CrossRef] [PubMed]
- Gong, K.; Du, F.; Xia, Z.; Dustock, M.; Dai, L. Nitrogen-doped carbon nanotube arrays with high electrocatalytic activity for oxygen reduction. Science 2009, 323, 760–764. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.T.; Zhao, Z.H.; Xia, Z.H.; Dai, L.M. A metal-free bifunctional electrocatalyst for oxygen reduction and oxygen evolution reactions. Nat. Nanotechnol. 2015, 10, 444–452. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, H,; Robinson, J.T.; Sanchez, H.; Diankov, G.; Dai, H. Simultaneous nitrogen doping and reduction of graphene oxide. J. Am. Chem. Soc. 2009, 131, 15939–15944. [Google Scholar] [CrossRef] [PubMed]
- Panchakarla, L.S.; Subrahmanyam, K.S.; Saha, S.K.; Govindaraj, A.; Krishnamurthy, H.R.; Waghmare, U.V.; Rao, C.N.R. Synthesis, structure, and properties of boron- and nitrogen-doped graphene. Adv. Mater. 2009, 21, 4726–4730. [Google Scholar] [CrossRef]
- David, L.; Singh, G. Reduced graphene oxide paper electrode: Opposing effect of thermal annealing on Li and Na cyclability. J. Phys. Chem. C 2014, 118, 28401–28408. [Google Scholar] [CrossRef]
- Sandhu, S.S.; Fellner, J.P.; Brutchen, G.W. Diffusion-limited model for a lithium/air battery with an organic electrolyte. J. Power Sources 2007, 164, 365–371. [Google Scholar] [CrossRef]
- Ambrosi, A.; Chua, C.K.; Bonanni, A.; Pumera, M. Electrochemistry of graphene and related materials. Chem. Rev. 2014, 114, 7150–7188. [Google Scholar] [CrossRef] [PubMed]
- Komarova, N.S.; Krivenko, A.G.; Stenina, E.V.; Sviridova, L.N.; Mironovich, K.V.; Shulga, Y.M.; Krivchenko, V.A. Enhancement of the carbon nanowall film capacitance. Electron transfer kinetics on functionalized surface. Langmuir 2015, 31, 7129–7137. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y. Lithium ion conducting membranes. Nano Energy 2013, 2, 801–816. [Google Scholar] [CrossRef]
- Fu, J. Fast Li+ ion conducting glass-ceramics in the system Li2O-Al2O3-GeO2-P2O5. Solid State Ionics 1997, 104, 191–194. [Google Scholar] [CrossRef]
- Leo, C.J.; Subba Rao, G.V.; Chowdari, B.V.R. Effect of MgO addition on the ionic conductivity of LiGe2(PO4)3 ceramics. Solid State Ionics 2003, 159, 357–367. [Google Scholar] [CrossRef]
- Ding, N.; Chien, S.W.; Andy Hor, T.S.; Lum, R.; Zong, Y.; Liu, Z. Influence of carbon pore size on the discharge capacity of Li-O2 batteries. J. Mater. Chem. A 2014, 2, 12433–12441. [Google Scholar] [CrossRef]
- Xiao, J.; Wang, D.; Xu, W.; Wang, D.; Williford, R.E.; Liu, J.; Zhang, J.-G. Optimization of air electrode for Li/Air batteries. J. Electrochem. Soc. 2010, 157, A487–A492. [Google Scholar] [CrossRef]
- Yang, X.-H.; He, P.; Xia, Y.-Y. Preparation of mesocellular carbon foam and its application for lithium/oxygen battery. Electrochem. Commun. 2009, 11, 1127–1130. [Google Scholar] [CrossRef]
- Sun, B.; Wang, B.; Su, D.; Xiao, L.; Ahn, H.; Wang, G. Graphene nanosheets as cathode catalysts for lithium-air batteries with an enhanced electrochemical performance. Carbon 2012, 50, 727–733. [Google Scholar] [CrossRef]
- Jiang, D.E.; Sumpter, B.G.; Dai, S. Unique chemical reactivity of a graphene nanoribbon’s zigzag edge. J. Chem. Phys. 2007, 126, 134701–134706. [Google Scholar] [CrossRef] [PubMed]
- Brownson, D.A.C.; Kampouris, D.K.; Banks, C.E. Graphene electrochemistry: Fundamental concepts through to prominent applications. Chem. Soc. Rev. 2012, 41, 6944–6976. [Google Scholar] [CrossRef] [PubMed]
- Trans, C.; Yang, X.Q.; Qu, D. Investigation of the gas-diffusion electrode used as lithium/air cathode in non-aqueous electrolyte and the importance of carbon material porosity. J. Power Sources 2010, 195, 2057–2063. [Google Scholar] [CrossRef]
- Barsoukov, E.; Macdonald, J.R. Impedance Spectroscopy: Theory, Experiment, and Applications, 2nd ed.; John Wiley and Sons: Hoboken, NJ, USA, 2005. [Google Scholar]
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kichambare, P.; Rodrigues, S. Graphene Nanosheets Based Cathodes for Lithium-Oxygen Batteries. C 2015, 1, 27-42. https://0-doi-org.brum.beds.ac.uk/10.3390/c1010027
Kichambare P, Rodrigues S. Graphene Nanosheets Based Cathodes for Lithium-Oxygen Batteries. C. 2015; 1(1):27-42. https://0-doi-org.brum.beds.ac.uk/10.3390/c1010027
Chicago/Turabian StyleKichambare, Padmakar, and Stanley Rodrigues. 2015. "Graphene Nanosheets Based Cathodes for Lithium-Oxygen Batteries" C 1, no. 1: 27-42. https://0-doi-org.brum.beds.ac.uk/10.3390/c1010027






