2. Materials and Methods
Electrospinning was performed with a wire-based electrospinning machine “Nanospider Lab” (Elmarco, Liberec, Czech Republic). The spinning parameters were as follows: voltage 80 kV, resulting current ~ 0.1 mA, nozzle diameter 0.9 mm, electrode-substrate distance 240 mm, carriage speed 100 mm/s, substrate speed 0 mm/min, relative humidity 32%, and temperature in the spinning chamber 22–23 °C. These parameters are identical to those used in [
27] to enable comparison. The spinning duration is usually 30 min, if not defined differently.
For the spinning solution, 16 wt% X-PAN (copolymer with 6% methyl methacrylate, X-PAN, Dralon, Dormagen, Germany) were dissolved in DMSO (min. 99.9%, S3 chemicals, Bad Oeynhausen, Germany) by stirring for 2 h at room temperature. Again, this solution was identical to the previous one from Ref. [
27].
The following substrates were glued via double-sided adhesive tape (Tesa SE, Norderstedt, Germany) onto the polypropylene (PP) nonwoven substrates (Elmarco, Liberec, Czech Republic):
- -
Aluminum foil, thickness 35 µm (Vireo.de, Merseburg, Germany)
- -
Copper foil thin, thickness 35 µm (Blechmaennle-de, Rottenburg, Germany)
- -
Copper foil middle, thickness 51µm (Blechmaennle-de, Rottenburg, Germany)
- -
Copper foil thick, thickness 100 µm (Blechmaennle-de, Rottenburg, Germany)
- -
Copper foil tin-coated, thickness 35 µm (CMC Klebetechnik GmbH, Frankenthal, Germany)
- -
Titanium sheet, thickness 100 µm (Evek GmbH, Mühlheim an der Ruhr, Germany)
- -
Stainless steel V2a sheet, thickness 500 µm (Kannegiesser GmbH, Vlotho, Germany)
- -
Steel st235jr sheet, thickness 500 µm (Kannegiesser GmbH, Vlotho, Germany)
- -
Silicon (100) wafer, thickness 510 µm (Science Service GmbH, Munich, Germany)
As a reference, a pure PAN nanofiber mat directly electrospun on the PP substrate was used. After electrospinning, the nanofiber mat/substrate composites were stabilized in a muffle oven B150 (Nabertherm, Lilienthal, Germany), approaching a temperature of typically 280 °C with a usual heating rate of 0.25 K/min, followed by isothermal treatment for 1 h. These were the most suitable parameters according to the results of [
27]. Different treatment parameters are mentioned in the respective experiments.
Afterwards, a furnace CTF 12/TZF 12 (Carbolite Gero Ltd., Sheffield, UK) was used for carbonization, approaching temperatures of 500 °C to 1200 °C (max. possible oven temperature) with a heating rate of typically 10 K/min in a nitrogen gas flow of 100 mL/min (STP), again followed by isothermal treatment for 1 h. Typical samples sizes were between 2 cm × 2 cm and 3 cm × 3 cm.
Additional measurements by differential scanning calorimetry (DSC) or thermogravimetric analysis (TGA), as often used to understand the heat treatment procedure better [
3], are here not possible due to the large masses of the substrates which would strongly superpose the temperature-dependent effects in the nanofiber mats.
Photographs of the samples after the different process steps were taken with a smartphone camera Xiaomi Redmi Note 9 Pro. Chemical investigation was performed by a Fourier-transform infrared (FTIR) spectroscope Excalibur 3100 (Varian Inc., Palo Alto, CA, USA) in attenuated total reflection mode (ATR-FTIR), using a wavenumber range from 4000–700 cm−1. The spectra were averaged over 32 scans and corrected for atmospheric noise. Confocal laser scanning microscopy (CLSM) was performed with a VK-9700 (Keyence, Neu-Isenburg, Germany).
SEM images were taken by a scanning electron microscope (SEM) Helios NanoLab DualBeam 600 (FEI Company, Hillsboro, OR, USA) after sputter coating with 10 nm ruthenium, applying a voltage of 5 kV and a beam current of 0.34 nA.
Color investigations of the nanofiber mats after stabilization and incipient carbonization were performed using a spectrophotometer sph900 (ColorLite GmbH, Katlenburg-Lindau, Germany). Comparative measurements were taken against a reference white MA38 BAM-Standard 1E2187c, serial no. 45010.
3. Results and Discussion
Firstly, the influence of the substrate material on the stabilization process was investigated.
Figure 1 shows photographs of PAN nanofiber mats, electrospun for 1 h, after stabilization on different substrates at 280 °C, approached with a heating rate of 0.25 K/min.
Generally, the brownish color, which can be used to estimate the degree of stabilization [
3], is similar in all cases, but nevertheless shows slight differences. The PAN nanofiber mat stabilized without substrate (
Figure 1j) may show a slightly darker color; this will be investigated later on. One possible explanation is a more pronounced stabilization process; however, previous research [
26] revealed that the darker color here can be explained by the shrinking of the nanofibers which were not fixed during stabilization.
To examine this reliably,
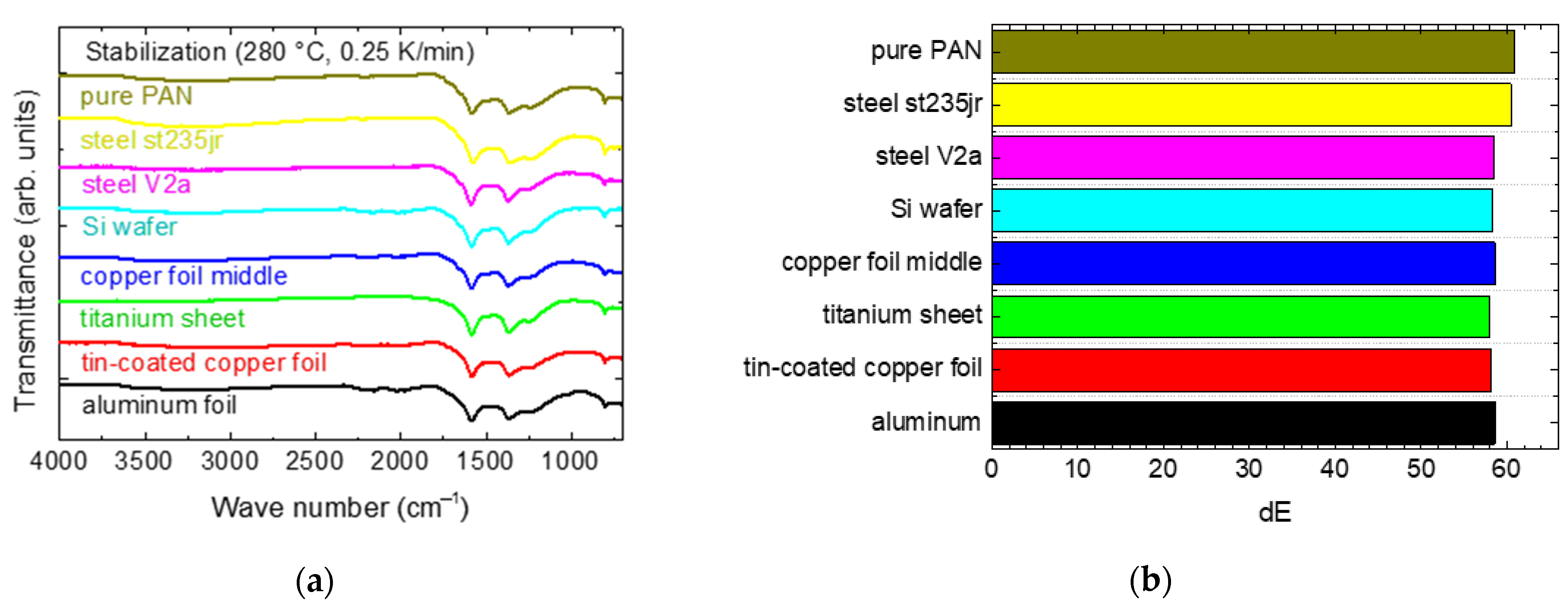
Figure 2a shows FTIR graphs of the nanofiber mats stabilized on different substrates. For the copper substrates, only the middle foil thickness is shown since FTIR images of the others are approximately identical. In all graphs, the typical peaks of stabilized PAN are visible [
3,
18,
26]. The peaks at 1582 cm
−1 and 1660 cm
−1 indicate C=N and C=C stretching vibrations, respectively. The peak at 1360 cm
−1 stems from C–H bending and C–H
2 wagging, while the small peak near 800 cm
−1 can be attributed to aromatic C–H vibrations after the oxidative dehydrogenation aromatization. The oxygen crosslinking between the polymer chains results in C–O and C–O–C vibrations, visible in the ranges between 1230–1250 cm
−1 and 1050–1090 cm
−1.
While these wave number ranges do not show differences for different substrates, the slopes of the curves differ at small wave numbers. This, however, can be attributed to the metal substrates which become visible in this range [
26]. From the FTIR data, it can be concluded that no significant chemical differences are visible for stabilization on different substrates.
The color differences dE, depicted in
Figure 2b, are a good indicator for the stabilization process since samples become darker during stabilization, and at the same time can be expected to be slightly smaller for samples stabilized under identical thermal conditions, but fixed on a substrate, as compared to samples stabilized freely [
3].
Here, however, no significant differences are visible between the samples stabilized on a substrate and the reference sample, stabilized without fixation. This may be due to the relatively small heating rate of 0.25 K/min applied here, as opposed to the heating rate of 1 K/min applied in the previous investigation [
3]. Apparently, morphological differences between samples stabilized freely and fixed are reduced for lower heating rates.
To investigate this aspect further,
Figure 3 depicts exemplary SEM images of nanofiber mats spun on the pure PP substrate and on a thin copper substrate, taken after electrospinning and after stabilization. Besides some arbitrarily visible thick fibers after electrospinning on the copper substrate (
Figure 3c), comparing both images after stabilization under identical thermal conditions (
Figure 3b,d) shows a much smaller influence of the substrate on the nanofiber morphology than in previous investigations, using different ways of fixation [
3,
26].
This finding supports the importance of carefully choosing the heating rate, besides the possibility to use a metal substrate for fixation of the nanofiber mat during stabilization.
Next,
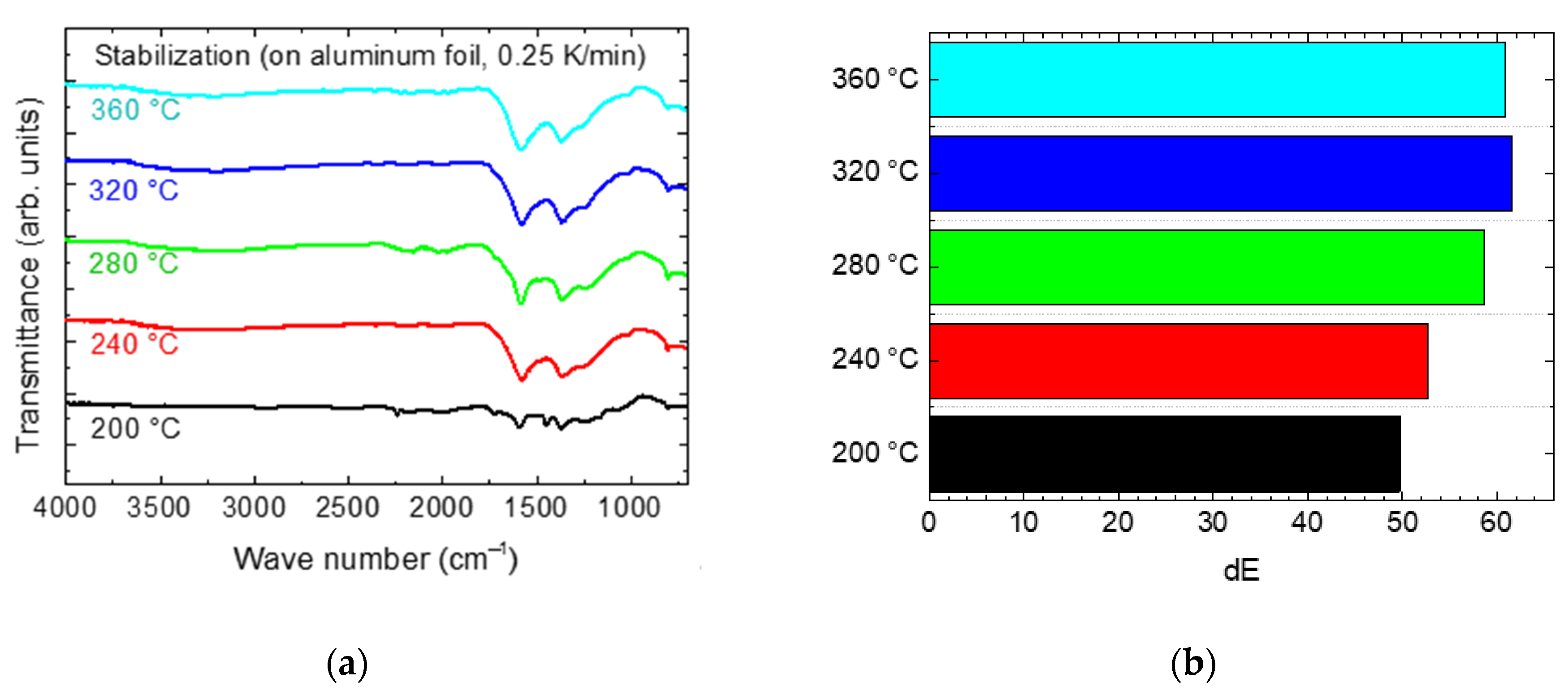
Figure 4 shows series of stabilization tests at different temperatures, approached with a heating rate of 0.25 K/min, on two exemplarily chosen substrates.
For the aluminum foil, the results are similar to the findings in a previous study on pure PAN nanofiber mats [
3]. On the one hand, higher temperatures lead to darker colors, indicating more complete stabilization. On the other hand, less damage occurs at lower temperatures. Apparently, in spite of the mechanically supporting substrate, there is still the need to balance mechanical and chemical properties. On the tin-coated copper foil, similar colors indicate similar grades of stabilization, while the higher stabilization temperatures resulted in detachment of the nanofiber mats.
This is why
Figure 5a depicts FTIR measurements of PAN nanofibers mats stabilized on the aluminum foil at different temperatures for 1 h, approached with a heating rate of 0.25 K/min. By comparing these graphs, it becomes apparent that at 200 °C nearly no typical peaks of stabilized PAN are visible, while already at a temperature of 240 °C, the characteristic double-peak around 1500 cm
−1 can be recognized. Although the shapes of the curves of stabilization temperatures between 240 °C and 360 °C differ slightly, it is not possible to detect a quantitative difference between them.
This contrasts with a former investigation of the stabilization process of pure PAN nanofiber mats, where FTIR graphs clearly showed full stabilization only at 280–300 °C [
3]. This contrast, however, cannot be attributed to the substrates, but is a consequence of the different heating rates. In the previous study [
3], relatively high heating rates of 2 K/min were chosen for the comparison of different stabilization temperatures. The heating rate was found to play a crucial role during stabilization pure nanofiber mats [
3] as well as nanofiber mats on metal substrates [
27].
The color differences dE depicted in
Figure 5b are clearly different for the lower temperatures, but saturate from a stabilization temperature of 280 °C, showing that stabilization is completed at 280 °C.
Comparing both results shows that, for the heating rate chosen here of only 0.25 K/min, a stabilization temperature between 240 and 280 °C seems to be ideal. To enable comparison with previous studies of our group and others, however, most experiments here were conducted with a stabilization temperature of 280 °C which is the typical value often reported in the literature.
The influence of the substrate on the carbonization process was thus investigated, applying the optimum heating rate of 0.25 K/min as well as the typical temperature of 280 °C during stabilization. Carbonization was performed at different temperatures, up to maximum values next to the melting temperature of copper-based metal substrates as well as up to a temperature of 1200 °C on the Si wafer, steel and titanium sheet, respectively.
Figure 6 depicts the results of incipient carbonization at a temperature of 500 °C for 1 h, approached with a typical heating rate of 10 K/min. These images reveal already large differences between the different substrates. On the one hand, some of the nanofiber mats are still partly or even fully attached, while others are completely detached. On the other hand, the colors are quite different. As explained above, it could be expected that the pure nanofiber mat (
Figure 6j) shows the darkest color after incipient carbonization since it shrank strongest during stabilization. Nevertheless, especially the nanofiber mat on the Si wafer shows a similarly dark color. On the thick copper foil (
Figure 6d), most of the nanofiber mat is already detached, and only parts of the nanofiber mat are still stuck on the substrate. On the middle and thin copper foils (
Figure 6e,f), the nanofiber mats are fully detached, but not torn, as it is the case for the thick substrate.
To investigate these findings in more detail,
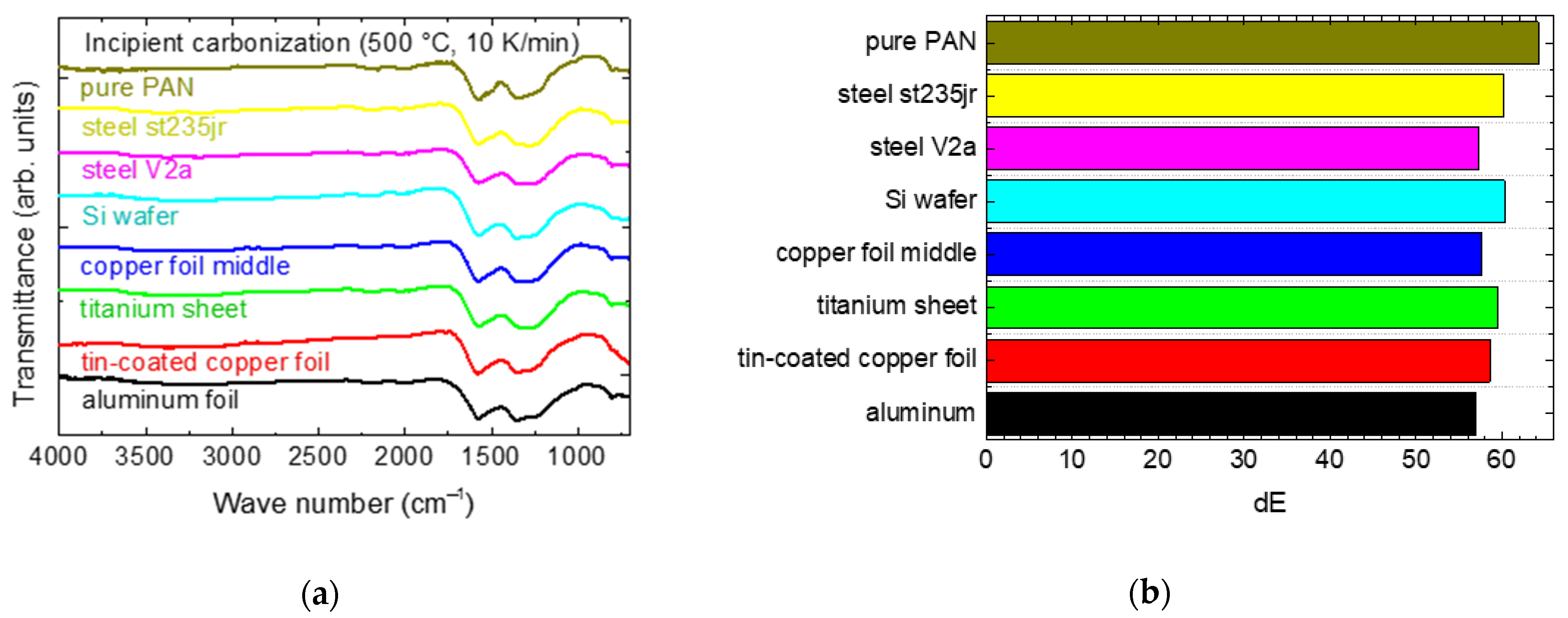
Figure 7a depicts FTIR graphs of these samples. Again, FTIR images of the copper substrates are approximately identical. At first glance, these graphs look quite similar to
Figure 2, measured after stabilization. Indeed, it is well-known that carbonization is only started at a temperature of 500 °C [
27]. Only the C–O vibrations in the wave number range of 1230–1250 cm
−1 are more pronounced now, broadening the maximum at 1360 cm
−1. Generally, no chemical differences are visible, comparing the different substrates.
Similar to
Figure 2b, the colors of the incipiently carbonized nanofiber mats with or without substrates are nearly identical, as depicted in
Figure 7b. The slight differences can be assumed to be not significant; for a statistically relevant comparison, more or larger samples would be necessary to enable measuring on several samples or at diverse positions, respectively.
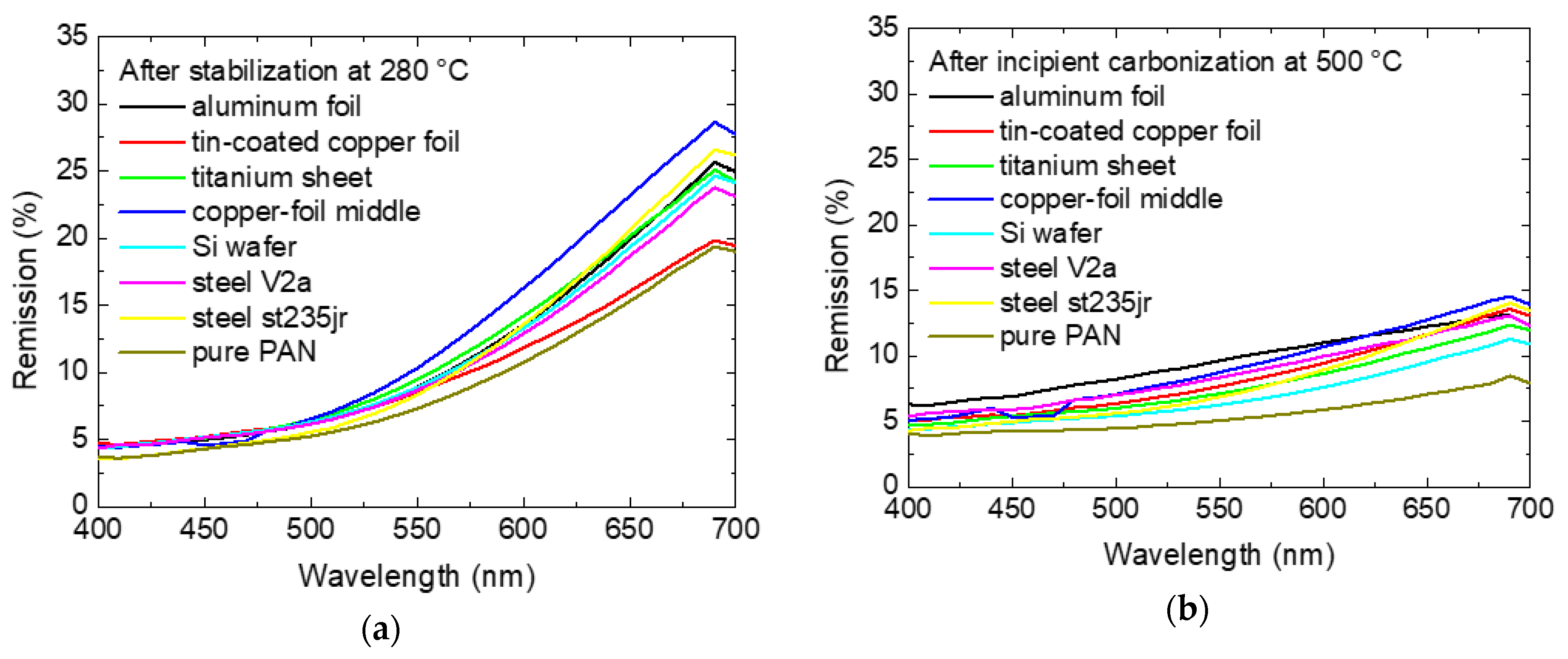
Since the dE values only show color differences to the white reference as scalars, it is not possible to distinguish between two dark colors using this number. Thus,
Figure 8 depicts the measured spectra for samples after stabilization (cf.
Figure 1) and after incipient carbonization (cf.
Figure 6). For a fully black sample, the spectra could be expected to be approximately parallel to the x-axis, while colored samples should show maxima and/or minima. This is indeed visible in
Figure 8. While the brownish stabilized samples show a clear remission maximum in the red range (
Figure 8a), the slopes of the curves are much flatter after incipient carbonization (
Figure 8b), quantifying the difference in the color after stabilization and incipient carbonization.
To investigate the influence of the substrate on the morphology of the incipiently carbonized nanofiber mats,
Figure 9 depicts SEM images of samples carbonized at 500 °C without substrate or on thin copper foil.
Here, opposite to the previous images after stabilization, clear differences are visible. While the nanofiber mat on the thin copper foil looks unchanged after incipient carbonization, the pure nanofiber mat (
Figure 8a) shows several broken and conglutinated fibers, indicating that carbonization on a metal substrate is indeed supportive to maintain the desired morphology.
In analogy to
Figure 4,
Figure 10 shows the influence of the stabilization temperature on the carbonization process. The sample stabilized at 360 °C was not further used since there was no difference in the FTIR spectra of the samples stabilized at the highest temperatures. The other three samples (on tin-coated copper foil) do not show significant optical differences; color measurements did not reveal significant differences, either.
To investigate the chemical properties,
Figure 11 shows the FTIR graphs of these samples and the sample carbonized after stabilization at 280 °C (
Figure 7a). Comparing these graphs leads to an unexpected finding. As
Figure 7a revealed, more pronounced C–O vibrations in the wave number range of 1230–1250 cm
−1 show beginning carbonization, as opposed to pure stabilization.
Figure 11 clearly shows that this peak is more pronounced for incipient carbonization after stabilization at relatively low temperatures, while it is clearly reduced for incipient carbonization after stabilization at 320 °C.
Combined with
Figure 5, this finding suggests using a reduced stabilization temperature between 240 and 280 °C in combination with a heating rate of 0.25 K/min to reach this temperature, to achieve optimum carbonization results. This will be investigated in more detail in the near future, with a finer temperature resolution and also on other substrates.
Next, the influence of the heating rate applied to reach the temperature of incipient carbonization was investigated.
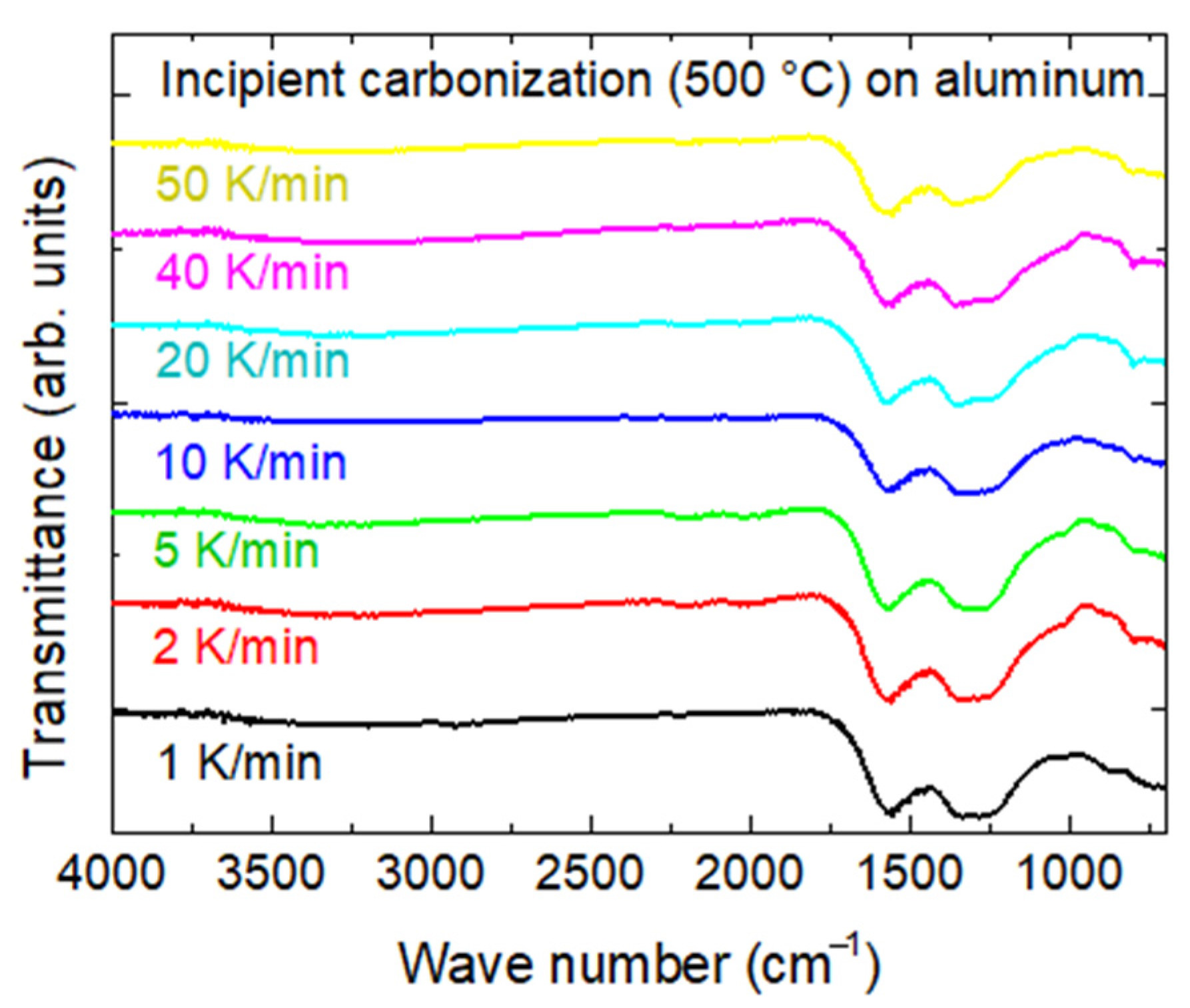
Figure 12 depicts photographic images of the samples carbonized with different heating rates between 1 K/min and 50 K/min (the maximum possible heating rate). Generally, the smaller heating rates seem to lead to a less brownish and more greyish color. This is, however, not displayed in the dE values which do not show significant differences. For a heating rate of 20 K/min (
Figure 12d), an unusually bright color is visible, which may be attributed to this nanofiber mat being attached nearly fully to the substrate after incipient carbonization, i.e., to the better retained morphology of the nanofiber mat, as discussed before.
It must also be mentioned that some of the nanofiber mats are partly or even fully detached from the substrates after incipient carbonization. This finding must be investigated in detail in the future to establish a reliable process to either separate both parts or to form composites.
The chemical investigation of these samples by FTIR (
Figure 13) indeed reveals small modifications of the peak near 1230–1250 cm
−1, showing a trend to pronounced C–O vibrations and thus possibly a slightly higher degree of carbonization for the samples with smaller heating rates. However, these findings cannot be quantified from the FTIR graphs and are thus not significant without further investigation.
The main aim of this study, however, was the investigation whether carbonization at high temperatures is enabled by leaving the nanofiber mats attached to a substrate. Previous experiments often revealed the problem that the fine nanofiber mats were moved away by the constant gas flow. On the other hand, many studies discuss the necessity of a relatively high gas flow to ensure full carbonization, i.e., to prevent contamination by air and to provide removal of emitted off-gas during carbonization [
28]. Thus, fixation on a substrate may help solving this problem.
Figure 14 shows carbonization results at higher temperatures on suitable substrates, i.e., copper foils. Carbonization on the Si wafer and on the titanium sheet led to a loss of the sample which was detached during the process.
On the copper foils, there is still a slightly greyish color visible instead of the expected black tone. The FTIR measurements performed for the chemical evaluation revealed nearly no more peaks, as usual for carbon [
25]. However, due to the problem to distinguish between vanishing peaks due to full carbonization or due to a lost nanofiber mat, these FTIR results are not taken into consideration here. Instead, microscopic images will be used next to evaluate this question.
The main aim of stabilizing and carbonizing PAN nanofiber mats on substrates is the possibility to unambiguously fix the sample dimensions during thermal treatment. This is especially important since most applications of carbon nanofibers or nanofiber mats, such as electrodes in pseudo- or supercapacitors, batteries or fuel cells [
29,
30,
31,
32,
33,
34,
35], necessitate intact, strong fibers without undesired modifications of their morphology by shrinking, bending or even breaking.
To investigate this aspect, CLSM images are used, offering high enough resolution to evaluate the nanofibers without the danger to see too small areas only [
35] and at the same time allowing seeing colors and thus obtaining an impression of the spatially resolved stabilization effort.
Figure 15 gives an overview of the samples stabilized at 280 °C for 1 h, approached by 0.25 K/min, on diverse substrates. Most of the samples look very similar, with similar colors and straight, long fibers. Only the pure nanofiber mat (
Figure 15h) shows a darker color, as already recognized in
Figure 1h, and thicker, more bent fibers, as expected. Besides, all samples show a certain inhomogeneity, as can be recognized by the color modification. Here, the nanofiber mat on the thick copper foil belongs to the apparently more homogeneous samples. It must be mentioned, however, that even with this relatively large field of view, only a small part of a sample is visible. Additional images (not shown here) revealed also inhomogeneous areas on this substrate.
Next,
Figure 16 shows the impact of the heating rate during stabilization, corresponding to the samples depicted in
Figure 4.
While the macroscopic images of the samples in
Figure 4 revealed only slight color differences between both substrates, here these deviations become more pronounced. Especially for stabilization on the aluminum substrate at 200 °C, there are small darker spots in a generally light brown nanofiber mat, indicating probably full stabilization only at these spots. Even for 240 °C, there are still color variations visible, leading to the conclusion that a temperature of 240 °C may be slightly too low for full stabilization, and 250 °C, e.g., should be tested instead.
On the tin-coated copper foil, however, the colors are clearly darker for all temperatures, and no irregularities are visible for the lower temperatures.
This indicates that a more detailed study of stabilization temperatures on all substrates should be carried out, possibly resulting in the finding that this process can be carried out with less energy consumption if an optimum substrate is chosen.
Next, the samples after incipient carbonization at 500 °C are presented in
Figure 17, analogously to
Figure 6. On the one hand, all samples still show a brownish color, underlining that carbonization has only slightly started at this temperature, but is far from being completed. The pure nanofiber mat again shows the darkest color and correspondingly relatively thick, bent nanofibers. Especially on aluminum, the color is less brownish, suggesting that here possibly carbonization has proceeded further. However, these differences are not significant and cannot be used to rate some of these substrates as especially well-suited as substrates for stabilization and carbonization.
To evaluate the influence of the stabilization temperature on subsequent carbonization (cf.
Figure 6 and
Figure 7a) more in detail,
Figure 18 shows CLSM images on the aluminum substrate, carbonized after stabilization at different temperatures. While the macroscopic images (
Figure 6) revealed no significant color differences, here a clear color difference between the carbonized samples stabilized at low temperatures and at 320 °C is visible, indicating that the latter is still more stabilized, while the others are already less brownish and thus slightly more carbonized. This finding is supported by the corresponding FTIR graphs (
Figure 7a) which already indicated more pronounced C–O vibrations, i.e., beginning carbonization for the samples stabilized at lower temperatures.
CLSM images of nanofiber mats incipiently carbonized at a temperature of 500 °C, approached by different heating rates, are depicted in
Figure 19. As already assumed according to
Figure 12 and
Figure 13, the heating rates below 10 K/min result in slightly less brownish, i.e., more carbonized, nanofiber mats. Nevertheless, many more investigations, including color measurements, are necessary to evaluate whether these differences are quantitative.
Finally,
Figure 20a shows a CLSM image of the sample carbonized on thin copper foil at 900 °C. Here, we clearly see that both materials form a metal/carbon composite, in which the carbon nanofiber mat started sinking into the slightly molten copper. Brighter areas show a stronger reflection—as usual for metals—indicating that here, the copper is at the surface. Residual brown–orange colors are typical for incomplete carbonization, showing residues of stabilized PAN.
On both steel sheets, the nanofiber mats are also partly vanished or sunk into the metal surface (cf.
Figure 18b,c). Especially on the V2a steel, the residues of the nanofiber mats look dark red, indicating a possible chemical modification in comparison with the nanofiber mat carbonized on copper at 900 °C. On the steel st235jr, the nanofiber mat looks slightly less reddish, but still quite different from the color of the sample carbonized on copper. These differences have to be investigated by FTIR to evaluate whether there are indeed chemical differences between the different carbon nanofiber mats.
It must be mentioned that these findings are not fully reproducible. On the other copper fabrics, e.g., no residual nanofiber mats were found. Thus, the procedure of preparing nano-composites has to be improved in the future, to prepare such nano-composites in a reliable way. Besides, SEM images of these samples revealed only few fibers along the surface. Future investigations at different carbonization temperatures are necessary to find out whether the missing carbon nanofiber mats vanish due to the nitrogen flow in the carbonization oven after being detached from the substrates, or whether they are fully integrated into the metal surface so that their shapes are nearly vanished, and only the larger-scale CLSM images are still able to find colored nanofiber residues.
Apparently, in this way it is indeed possible to prepare metal/carbon composites with a copper foil, as tested before with aluminum as substrate [
26]. The mechanical properties of such composites will be tested in the near future.