Intra- and Inter-Annual Variability in the Dissolved Inorganic Nitrogen in an Urbanized River before and after Wastewater Treatment Plant Upgrades: Case Study in the Grand River (Southwestern Ontario)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Site Description
2.2. Sample Collection and Analysis
2.3. Seasonal Demarcation
3. Results and Discussion
3.1. The Impact of the Upgrades at the Kitchener Wastewater Treatment Plant
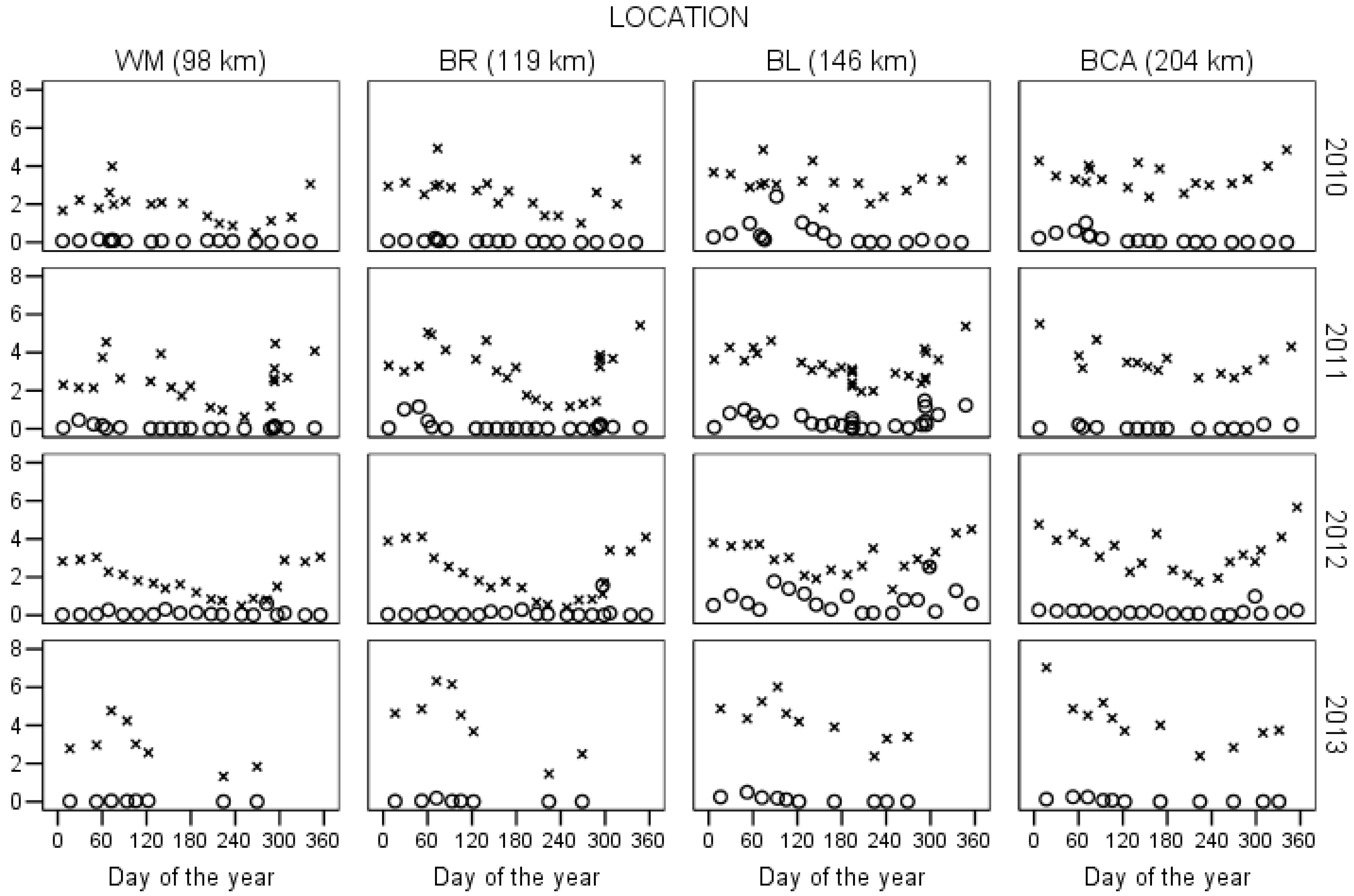
3.2. Intra-Annual Variability of DIN in the Central Grand River
3.3. Inter-Annual Variability of DIN in the Central Grand River
3.4. Loads and Concentrations—A Different Answer to a Different Question
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Temp °C | Historical avg °C | Year Avg °C | Historical Precip. avg [mm] | Total Year Precip. [mm] | Year Snow avg [cm] | Total Year Snow [cm] | Mean Q (m3/s) Water Survey of Canada | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Year | Comments | High | Low | WM | BL | BCA | ||||||
| 2010 | Dry year. 5th warmest year in history, 1.4 °C above average. Warmer spring. One week in July and one in August were notably above average. 11 days over 30 °C. Abundant rain: maximum one day rain: 65 mm. 1 in 10 years precipitation. 46.1 °C highest humidex (July 7th) | 33.1 | −21.5 | 11.89 | 13.11 | 904 | 879.3 | 159.5 | 77.5 | 11.5 | 22 | 45.3 |
| 2011 | Wet year, 4th wettest year since 1914. Wettest April ever. The first half of the year was 1 °C colder than average; the second half was 2.5 °C warmer than average. | 35.7 | −28.8 | 11.89 | 12.6 | 904 | 1146.4 | 159.5 | 165.5 | 17.4 | 38 | 81.6 |
| 2012 | Dry, consistently warm year. Hot March, over 7.5 °C warmer than average. The second warmest year since 1914. July 2011 to June 2012, the warmest 12 month period in the history. A large part of the year was drier than average. 56 mm one-day precipitation event (June 1st). 46.9 °C humidex (July 21st) | 33.5 | −18.3 | 11.89 | 14.31 | 904 | 782.7 | 159.5 | 86.5 | 10.7 | 19.2 | 44 |
| 2013 | Wet year. Year with more precipitation in 99 years record. 94.1 mm in one-day precipitation period (September). February, second snowiest record in the region. Late cold winter. 186 days without frost, longest frost-free season since 1915. 47.6 °C highest humidex (July 17th) | 34.7 | −24 | 11.89 | 11.92 | 904 | 1204.7 | 159.5 | 179 | n.a. | n.a. | n.a. |
References
- Elser, J.J.; Bracken, M.E.; Cleland, E.E.; Gruner, D.S.; Harpole, W.S.; Hillebrand, H.; Ngai, J.T.; Seabloom, E.W.; Shurin, J.B.; Smith, J.E. Global analysis of nitrogen and phosphorus limitation of primary producers in freshwater, marine and terrestrial ecosystems. Ecol. Lett. 2007, 10, 1135–1142. [Google Scholar] [CrossRef] [Green Version]
- Dodds, W.K.; Smith, V.H. Nitrogen, phosphorus, and eutrophication in streams. Inland Waters 2016, 6, 155–164. [Google Scholar] [CrossRef]
- Blaas, H.; Kroeze, C. Excessive nitrogen and phosphorus in European rivers: 2000–2050. Ecol. Indic. 2016, 67, 328–337. [Google Scholar] [CrossRef]
- Burson, A.; Stomp, M.; Akil, L.; Brussaard, C.P.D.; Huisman, J. Unbalanced reduction of nutrient loads has created an offshore gradient from phosphorus to nitrogen limitation in the North Sea. Limnol. Oceanogr. 2016, 61, 869–888. [Google Scholar] [CrossRef] [Green Version]
- Schindler, D.W. The dilemma of controlling cultural eutrophication of lakes. Proc. R. Soc. B. Biol. Sci. 2012, 279, 4322–4333. [Google Scholar] [CrossRef] [Green Version]
- Durand, P.; Breuer, L.; Johnes, P.J.; Billen, G.; Butturini, A.; Pinay, G.; Van Grinsven, H.; Garnier, J.; Rivett, M.; Reay, D.S.; et al. Nitrogen processes in aquatic ecosystems. In European Nitrogen Assessment; Sutton, M.A., Howard, C.M., Erisman, J.W., Billen, G., Bleeker, A., Grennfelt, P., van Grinsven, H., Grizzetti, B., Eds.; Cambridge University Press: Cambridge, UK, 2011; pp. 126–146. [Google Scholar]
- Meybeck, M. C, N, P and S in rivers: From sources to global inputs. In Interactions of C, N, P and S Biogeochemical Cycles and Global Change; Wollast, R., Mackenzie, F.T., Chou, L., Eds.; Springer: Berlin, Germany, 1993; pp. 163–193. [Google Scholar]
- Turner, R.E.; Rabalais, N.N.; Justic, D.; Dortch, Q. Global patterns of dissolved N, P and Si in large rivers. Biogeochemistry 2003, 64, 297–317. [Google Scholar] [CrossRef]
- Puckett, L.J. Identifying the Major Sources of Nutrient Water Pollution. Environ. Sci. Technol. 1995, 29, 408–414. [Google Scholar] [CrossRef]
- Falkowski, P.; Scholes, R.J.; Boyle, E.E.A.; Canadell, J.; Canfield, D.; Elser, J.; Gruber, N.; Hibbard, K.; Högberg, P.; Linder, S.; et al. The Global Carbon Cycle: A Test of Our Knowledge of Earth as a System. Science 2000, 290, 291–296. [Google Scholar] [CrossRef] [Green Version]
- Schlesinger, W.H. On the fate of anthropogenic nitrogen. Proc. Natl. Acad. Sci. USA 2009, 106, 203–208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Allan, J.D.; Castillo, M.M. Stream Ecology: Structure and Function of Running Waters; Springer Science & Business: Dordrecht, The Netherlands, 2007. [Google Scholar]
- Wollheim, W.M.; Peterson, B.J.; Deegan, L.A.; Hobbie, J.E.; Hooker, B.; Bowden, W.B.; Edwardson, K.J.; Arscott, D.B.; Hershey, A.E.; Finlay, J. Influence of stream size on ammonium and suspended particulate nitrogen processing. Limnol. Oceanogr. 2001, 46, 1–13. [Google Scholar] [CrossRef]
- Kaushal, S.S.; Delaney-Newcomb, K.; Findlay, S.E.G.; Newcomer, T.A.; Duan, S.; Pennino, M.J.; Sivirichi, G.M.; Sides-Raley, A.M.; Walbridge, M.R.; Belt, K.T. Longitudinal patterns in carbon and nitrogen fluxes and stream metabolism along an urban watershed continuum. Biogeochemistry 2014, 121, 23–44. [Google Scholar] [CrossRef]
- Zhang, X.; Wu, Y.; Gu, B. Urban rivers as hotspots of regional nitrogen pollution. Environ. Pollut. 2015, 205, 139–144. [Google Scholar] [CrossRef]
- Pearce, N.J.; Yates, A.G. Intra-annual variation of the association between agricultural best management practices and stream nutrient concentrations. Sci. Total Environ. 2017, 586, 1124–1134. [Google Scholar] [CrossRef]
- Walsh, P.J.; Milon, J.W. Nutrient Standards, Water Quality Indicators, and Economic Benefits from Water Quality Regulations. Environ. Resour. Econ. 2015, 64, 643–661. [Google Scholar] [CrossRef]
- Loucks, D.P.; van Beek, E. Water Resource Systems Planning and Management: An Introduction to Methods, Models, and Applications; Springer: Cham, Switzerland, 2017. [Google Scholar] [CrossRef]
- Wijesiri, B.; Deilami, K.; Goonetilleke, A. Evaluating the relationship between temporal changes in land use and resulting water quality. Environ. Pollut. 2018, 234, 480–486. [Google Scholar] [CrossRef] [PubMed]
- Ballinger, A.; Lake, P.S. Energy and nutrient fluxes from rivers and streams into terrestrial food webs. Mar. Freshw. Res. 2006, 57, 15–28. [Google Scholar] [CrossRef]
- Artioli, Y.; Friedrich, J.; Gilbert, A.J.; McQuatters-Gollop, A.; Mee, L.D.; Vermaat, J.E.; Wulff, F.; Humborg, C.; Palmeri, L.; Pollehne, F. Nutrient budgets for European seas: A measure of the effectiveness of nutrient reduction policies. Mar. Pollut. Bull. 2008, 56, 1609–1617. [Google Scholar] [CrossRef] [PubMed]
- Grand River Conservation Authority. Grand River Water Management Plan—Water Quality Targets to Support Healthy and Resilient Aquatic Ecosystems in the Grand River Watershed; Prepared by the Water Quality Working Group; Grand River Conservation Authority: Cambridge, ON, Canada, 2013. [Google Scholar]
- MacCrimmon, H.R.; Kelso, J.R.M. Seasonal Variation in Selected Nutrients of a River System. J. Fish. Res. Board Can. 1970, 27, 837–846. [Google Scholar] [CrossRef]
- Barlow-Busch, L.; Baulch, H.M.; Taylor, W.D. Phosphate uptake by seston and epilithon in the Grand River, southern Ontario. Aquat. Sci. 2006, 68, 181–192. [Google Scholar] [CrossRef]
- Rosamond, M.S.; Thuss, S.J.; Schiff, S.L.; Elgood, R.J. Coupled Cycles of Dissolved Oxygen and Nitrous Oxide in Rivers along a Trophic Gradient in Southern Ontario, Canada. J. Environ. Qual. 2011, 40, 256–270. [Google Scholar] [CrossRef]
- Hood, J.L.; Taylor, W.D.; Schiff, S.L. Examining the fate of WWTP effluent nitrogen using δ15N–NH4+, δ15N–NO3- and δ 15N of submersed macrophytes. Aquat. Sci. 2014, 76, 243–258. [Google Scholar] [CrossRef]
- Shaker, S. Historical Trends In Water Quality in the Grand River Ontario: Reconstruction Of Phosphorus Loadings. Master’s Thesis, University of Waterloo, Waterloo, ON, Canada, 2014. [Google Scholar]
- Cejudo, E.; Hood, J.L.; Schiff, S.L.; Aravena, R.O. Using the δ15N of submerged biomass for assessing changes in the nitrogen cycling in a river receiving wastewater treated effluent. Ecol. Indic. 2018, 95, 645–653. [Google Scholar] [CrossRef]
- Venkiteswaran, J.J.; Schiff, S.L.; Ingalls, B.P. Quantifying the fate of wastewater nitrogen discharged to a Canadian river. Facets 2019, 4, 315–335. [Google Scholar] [CrossRef] [Green Version]
- Bicudo, J.R.; Brown, T.; Waller, M.; Saint, W.; Summach, D. Addressing ammonia levels in the Grand River through nitrification upgrades at the Kitchener WWTP. Influents 2016, 11, 54–57. [Google Scholar]
- Region of Waterloo. Wastewater Treatment Master Plan Final Report; Project No. 78148; Region of Waterloo: Kitchener, ON, Canada, 2007. [Google Scholar]
- Nelson, J.G.; Porter, J.; Farassoglou, C.; Gardiner, S.; Guthrie, C.; Beck, C.; Lemieux, C. The Grand River Watershed: A Heritage Landscape Guide; Heritage Landscape Guide Series, 2; University of Waterloo: Waterloo, ON, Canada, 2003. [Google Scholar]
- Strickland, J.D.; Parsons, T.R. A Practical Handbook of Seawater Analysis; Fisheries Research Board of Canada: Ottawa, ON, Canada, 1972. [Google Scholar]
- Region of Waterloo. Performance of a New Centrate Treatment System At the Kitchener Wastewater Treatment Plant; Region of Waterloo: Kitchener, ON, Canada, 2014. [Google Scholar]
- Mallin, M.A.; McIver, M.R.; Wells, H.A.; Parsons, D.C.; Johnson, V.L. Reversal of eutrophication following sewage treatment upgrades in the New River Estuary, North Carolina. Estuaries 2005, 28, 750–760. [Google Scholar] [CrossRef]
- Chanat, J.G.; Rice, K.C.; Hornberger, G.M. Consistency of patterns in concentration-discharge plots. Water Resour. Res. 2002, 38, 22-1–22-10. [Google Scholar] [CrossRef]
- Duncan, J.M.; Welty, C.; Kemper, J.T.; Groffman, P.M.; Band, L.E. Dynamics of nitrate concentration-discharge patterns in an urban watershed. Water Resour. Res. 2017, 53, 7349–7365. [Google Scholar] [CrossRef]
- Reay, D.S.; Nedwell, D.B.; Priddle, J.; Ellis-Evans, J.C. Temperature Dependence of Inorganic Nitrogen Uptake: Reduced Affinity for Nitrate at Suboptimal Temperatures in Both Algae and Bacteria. Appl. Environ. Microbiol. 1999, 65, 2577–2584. [Google Scholar] [CrossRef] [Green Version]
- Charley, R.C.; Hooper, D.G.; McLee, A.G. Nitrification kinetics in activated sludge at various temperatures and dissolved oxygen concentrations. Water Res. 1980, 14, 1387–1396. [Google Scholar] [CrossRef]
- Pfenning, K.S.; McMahon, P.B. Effect of nitrate, organic carbon, and temperature on potential denitrification rates in nitrate-rich riverbed sediments. J. Hydrol. 1997, 187, 283–295. [Google Scholar] [CrossRef] [Green Version]
- Westberg, R.E. The Use of Temperature and Environmental Isotopes as Tools to Characterize Groundwater Discharge to the Grand River, Ontario, Canada. Master’s Thesis, University of Waterloo, Waterloo, ON, Canada, 2013. [Google Scholar]
- Grand River Conservation Authority. Assessment of Future Water Quality Conditions in the Grand and Speed Rivers; Water Management Plant Assimilative Capacity Working Group Report; Grand River Conservation Authority: Cambridge, ON, Canada, 2012. [Google Scholar]
- Canadian Council of Ministers of the Environment. Canadian Water Quality Guidelines for the Protection of Aquatic Life: Nitrate Ion; Canadian Council of Ministers of the Environment: Winnipeg, MB, Canada, 2010.
- Venkiteswaran, J.J.; Schiff, S.L.; Taylor, W.D. Linking aquatic metabolism, gas exchange, and hypoxia to impacts along the 300 km Grand River, Canada. Freshw. Sci. 2015, 34, 1216–1232. [Google Scholar] [CrossRef]
- Neff, J.C.; Holland, E.A.; Dentener, F.J.; McDowell, W.H.; Russell, K.M. The origin, composition and rates of organic nitrogen deposition: A missing piece of the nitrogen cycle? Biogeochemistry 2002, 57, 99–136. [Google Scholar] [CrossRef]
- Van Kessel, C.; Clough, T.; Van Groenigen, J.W. Dissolved Organic Nitrogen: An Overlooked Pathway of Nitrogen Loss from Agricultural Systems? J. Environ. Qual. 2009, 38, 393–401. [Google Scholar] [CrossRef] [Green Version]
- Liu, H.; Jeong, J.; Gray, H.; Smith, S.; Sedlak, D.L. Algal Uptake of Hydrophobic and Hydrophilic Dissolved Organic Nitrogen in Effluent from Biological Nutrient Removal Municipal Wastewater Treatment Systems. Environ. Sci. Technol. 2011, 46, 713–721. [Google Scholar] [CrossRef]
- Qin, C.; Liu, H.; Liu, L.; Smith, S.; Sedlak, D.L.; Gu, A.Z. Bioavailability and characterization of dissolved organic nitrogen and dissolved organic phosphorus in wastewater effluents. Sci. Total Environ. 2015, 511, 47–53. [Google Scholar] [CrossRef]
- Kaushal, S.S.; Lewis, W.M. Fate and transport of organic nitrogen in minimally disturbed montane streams of Colorado, USA. Biogeochemistry 2005, 74, 303–321. [Google Scholar] [CrossRef]
- Cummings, T.F. Assessment of Nitrate Export In Agricultural Sub-catchments of the Grand River Watershed: An Isotope Approach. Master’s Thesis, University of Waterloo, Waterloo, ON, Canada, 2015. [Google Scholar]
- de Wit, M.; Bendoricchio, G. Nutrient fluxes in the Po basin. Sci. Total Environ. 2001, 273, 147–161. [Google Scholar] [CrossRef]
- Rankinen, K.; Keinänen, H.; Bernal, J.E.C. Influence of climate and land use changes on nutrient fluxes from Finnish rivers to the Baltic Sea. Agric. Ecosyst. Environ. 2016, 216, 100–115. [Google Scholar] [CrossRef]
- Hanief, A.; Laursen, A.E. SWAT modeling of hydrology, sediment and nutrients from the Grand River, Ontario. Water Qual. Res. J. 2017, 52, 243–257. [Google Scholar] [CrossRef]
- Ludwig, W.; Dumont, E.; Meybeck, M.; Heussner, S. River discharges of water and nutrients to the Mediterranean and Black Sea: Major drivers for ecosystem changes during past and future decades? Prog. Oceanogr. 2009, 80, 199–217. [Google Scholar] [CrossRef]
- Van Cappellen, P.; Maavara, T. Rivers in the Anthropocene: Global scale modifications of riverine nutrient fluxes by damming. Ecohydrol. Hydrobiol. 2016, 16, 106–111. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Yang, W.; Leon, L.; Wong, I.; McCrimmon, C.; Dove, A.; Fong, P. Hydrologic modeling and evaluation of Best Management Practice scenarios for the Grand River watershed in Southern Ontario. J. Great Lakes Res. 2016, 42, 1289–1301. [Google Scholar] [CrossRef]
- Malagó, A.; Bouraoui, F.; Vigiak, O.; Grizzetti, B.; Pastori, M. Modelling water and nutrient fluxes in the Danube River Basin with SWAT. Sci. Total Environ. 2017, 603, 196–218. [Google Scholar] [CrossRef] [PubMed]





| Season | Start Day | End Day | Duration (Days) | Flow Regime |
|---|---|---|---|---|
| Winter | 320 | 45 | 91 | Average flow |
| Spring–Spring melt | 46 | 134 | 89 | High flow |
| Summer–Fall | 135 | 319 | 185 | Low flow |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cejudo, E.; Rosamond, M.S.; Elgood, R.J.; Schiff, S.L. Intra- and Inter-Annual Variability in the Dissolved Inorganic Nitrogen in an Urbanized River before and after Wastewater Treatment Plant Upgrades: Case Study in the Grand River (Southwestern Ontario). Nitrogen 2021, 2, 139-154. https://0-doi-org.brum.beds.ac.uk/10.3390/nitrogen2020010
Cejudo E, Rosamond MS, Elgood RJ, Schiff SL. Intra- and Inter-Annual Variability in the Dissolved Inorganic Nitrogen in an Urbanized River before and after Wastewater Treatment Plant Upgrades: Case Study in the Grand River (Southwestern Ontario). Nitrogen. 2021; 2(2):139-154. https://0-doi-org.brum.beds.ac.uk/10.3390/nitrogen2020010
Chicago/Turabian StyleCejudo, Eduardo, Madeline S. Rosamond, Richard J. Elgood, and Sherry L. Schiff. 2021. "Intra- and Inter-Annual Variability in the Dissolved Inorganic Nitrogen in an Urbanized River before and after Wastewater Treatment Plant Upgrades: Case Study in the Grand River (Southwestern Ontario)" Nitrogen 2, no. 2: 139-154. https://0-doi-org.brum.beds.ac.uk/10.3390/nitrogen2020010







