Plummer-Vinson Syndrome: A Time for Redefinition and New Perspectives
Abstract
:1. Introduction
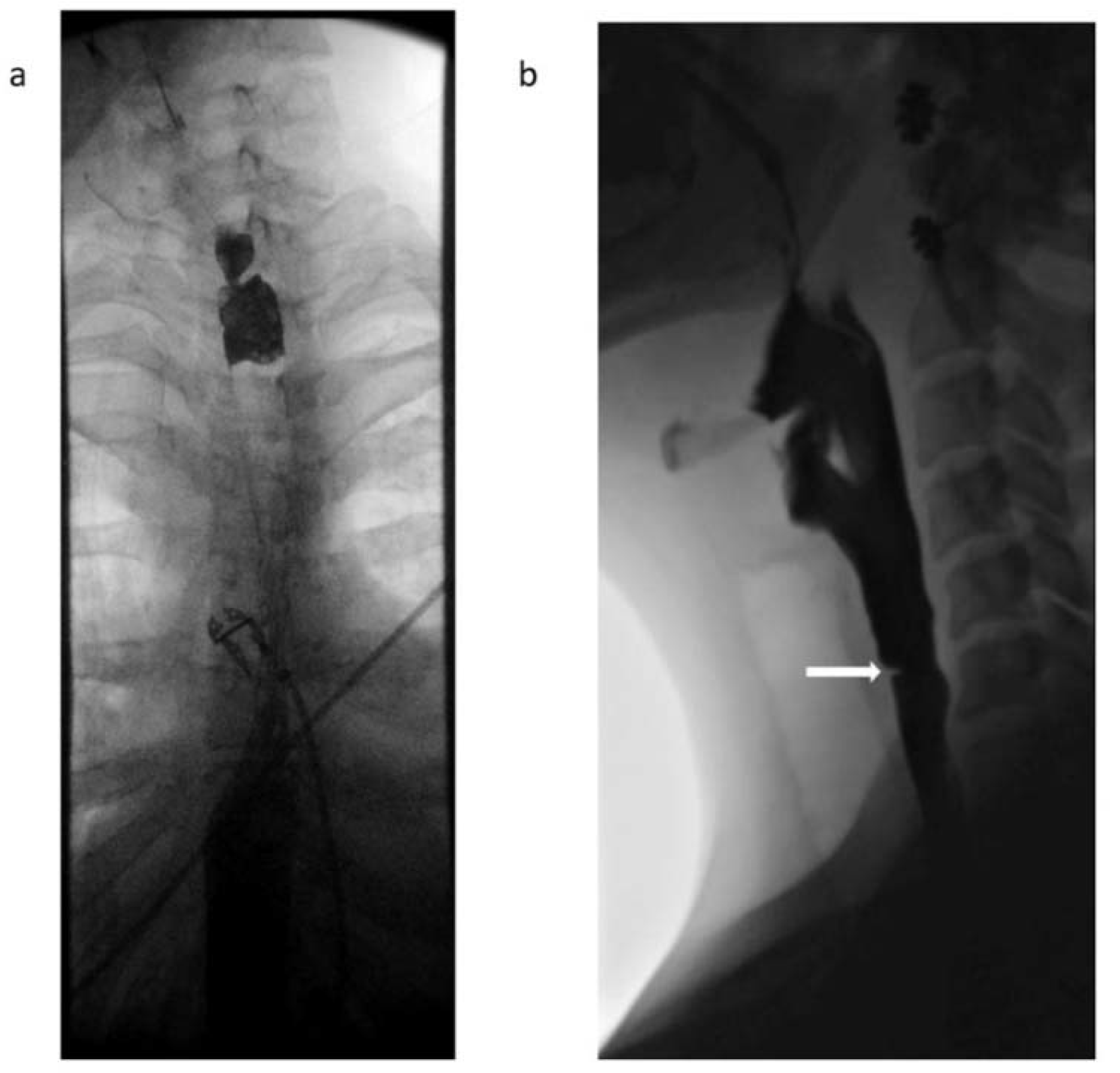
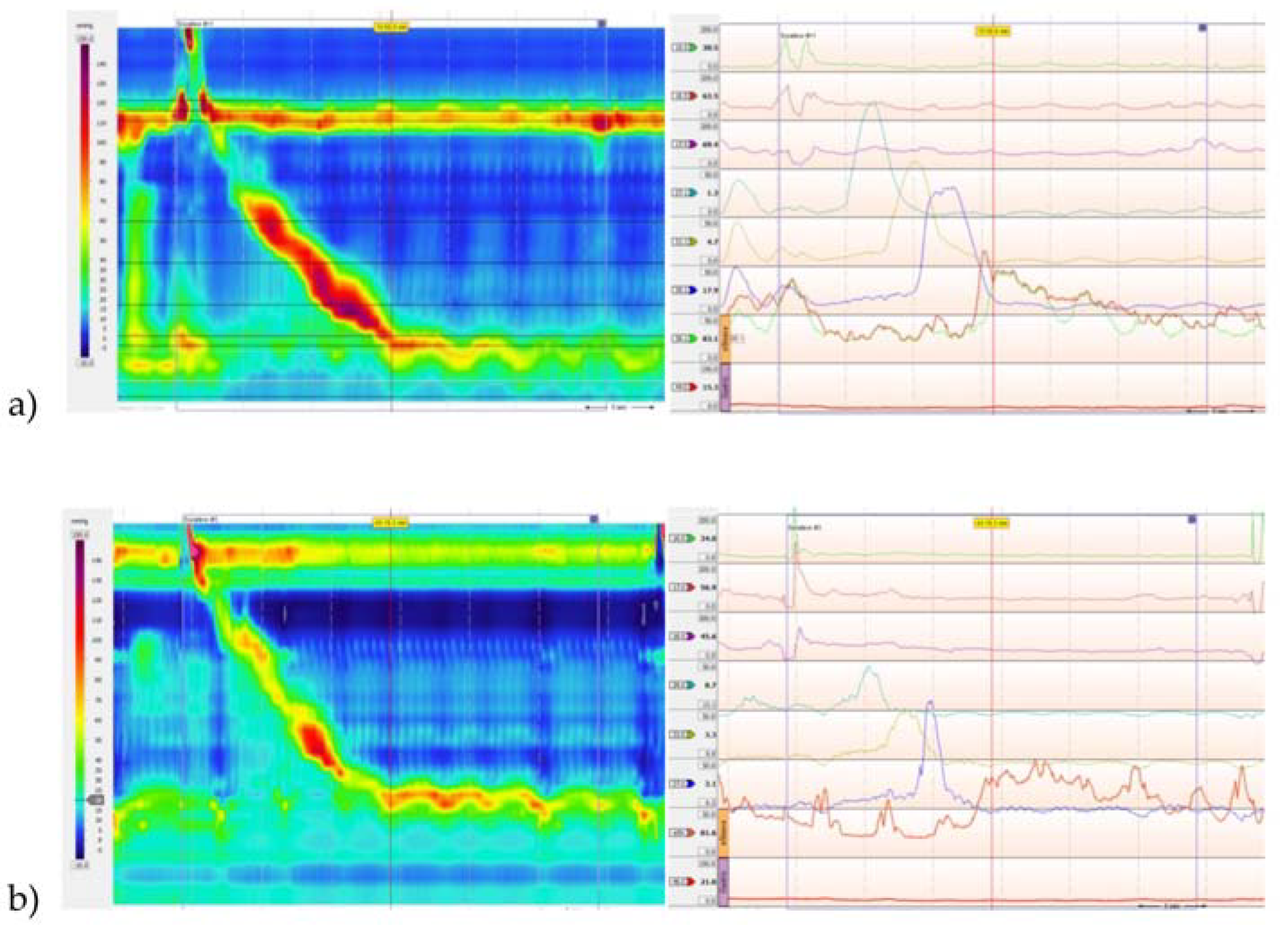
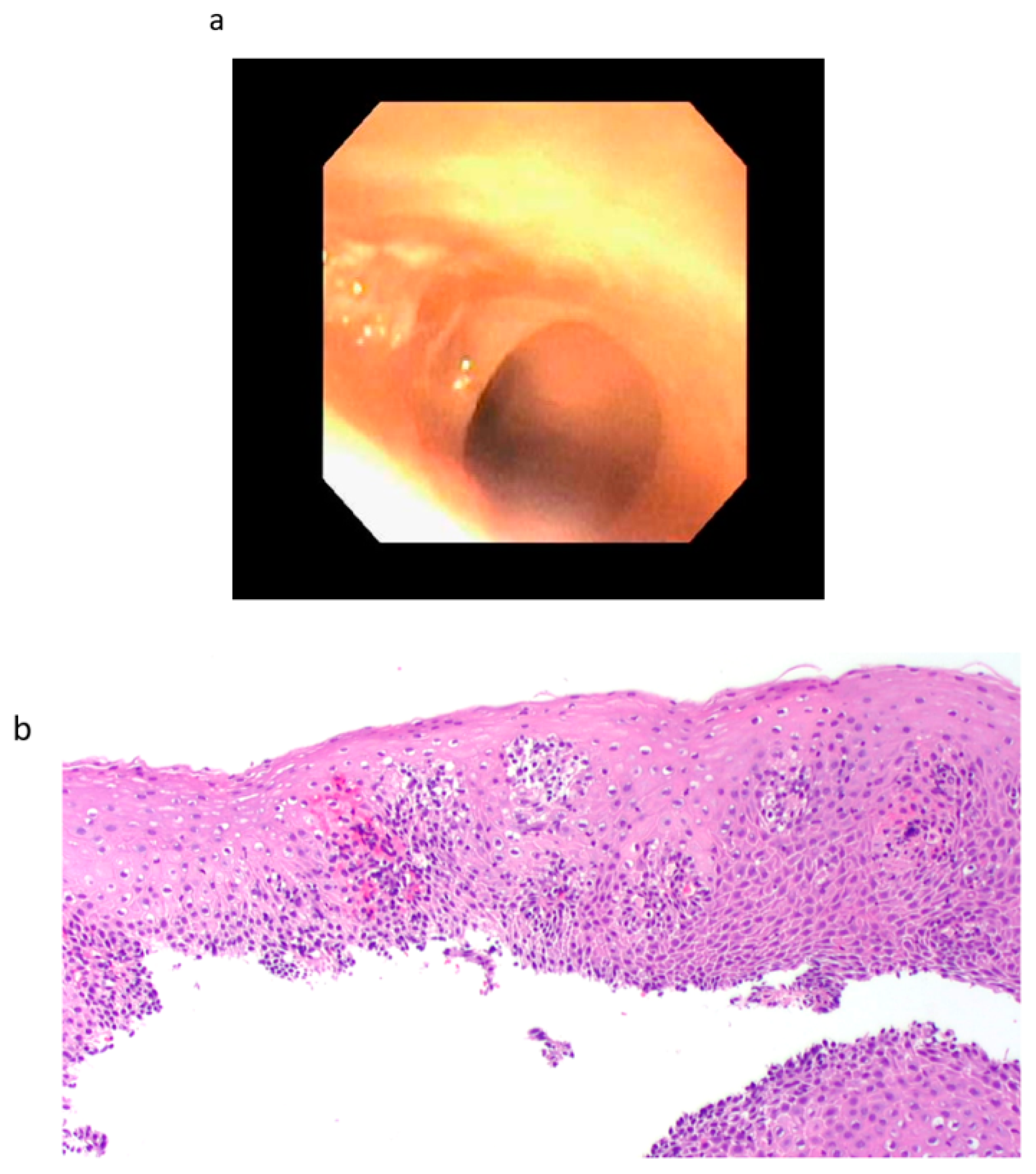
2. Results
3. Discussion
4. Materials and Methods
Author Contributions
Funding
Conflicts of Interest
References
- Novacek, G. Plummer-Vinson syndrome. Orphanet J. Rare Dis. 2006, 1, 36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Plummer, H.S. Diffuse dilation of the oesophagus without anatomical stenosis (cardiospasm) a report of 91 cases. JAMA 1912, 58, 2013–2015. [Google Scholar] [CrossRef] [Green Version]
- Vinson, P.P. A case of cardiospasm with dilation and angulation of oesophagus. Med. Clin. N. Am. 1919, 3, 623–627. [Google Scholar]
- Kelly, A.B. Spasm at the entrance of the oesophagus. J. Laryngol. Rhinol. Otol. 1919, 34, 285–289. [Google Scholar] [CrossRef] [Green Version]
- Patterson, D.R. A clinical type of dysphagia. J. Laryngol. Rhinol. Otol. 1919, 34, 289–291. [Google Scholar] [CrossRef] [Green Version]
- Chisholm, M. The association between webs, iron and post-cricoid carcinoma. Postgrad. Med. J. 1974, 50, 215–219. [Google Scholar] [CrossRef]
- Gude, D.; Bansal, D.; Malu, A. Revisiting Plummer Vinson syndrome. Ann. Med. Health Sci. Res. 2013, 3, 119–121. [Google Scholar] [CrossRef] [Green Version]
- Beyler, A.R.; Yurdaydin, C.; Bahar, K.; Goren, A.; Soykan, I.; Uzunalimoglu, O. Dilation therapy of upper esophageal webs in two cases of Plummer-Vinson syndrome. Endoscopy 1996, 28, 266–267. [Google Scholar] [CrossRef] [Green Version]
- Enomoto, M.; Kohmoto, M.; Arafa, U.A.; Shiba, M.; Watanabe, T.; Tominaga, K.; Fujiwara, Y.; Saeki, Y.; Higuchi, K.; Nishiguchi, S.; et al. Plummer-Vinson syndrome successfully treated by endoscopic dilatation. J. Gastroenterol. Hepatol. 2007, 22, 2348–2351. [Google Scholar] [CrossRef]
- Patil, M.; Malipatel, R.; Devarbhavi, H. Plummer-Vinson syndrome: A decade’s experience of 132 cases from a single center. J. Gastroenterol. Hepatol 2020. Epub ahead of print. [Google Scholar] [CrossRef]
- Sood, A.; Midha, V.; Sood, N.; Bansal, M. Paterson Kelly syndrome in celiac disease. J. Assoc. Physicians India 2005, 53, 991–992. [Google Scholar]
- Medrano, M. Dysphagia in a patient with rheumatoid arthritis and iron deficiency anemia. MedGenMed 2002, 4, 10. [Google Scholar] [PubMed]
- Okamura, H.; Tsutsumi, S.; Inaki, S.; Mori, T. Esophageal web in Plummer-Vinson syndrome. Laryngoscope 1988, 98, 994–998. [Google Scholar]
- Atmatzidis, K.; Papaziogas, B.; Pavlidis, T.; Mirelis, C.; Papaziogas, T. Plummer-Vinson syndrome. Dis. Esophgagus 2003, 16, 154–157. [Google Scholar] [CrossRef]
- Goel, A.; Bakshi, S.S.; Soni, N.; Chhavi, N. Iron deficiency anemia and Plummer-Vinson syndrome: Current insights. J. Blood Med. 2017, 8, 175–184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hillemeier, C.; Touloukian, R.; McCallum, R.; Gryboski, J. Esophageal web: A previously unrecognized complication of epidermolysis bullosa. Pediatrics 1981, 67, 678–682. [Google Scholar]
- Howell, J.T.; Monto, R.W. Syndrome of anemia, dysphagia and glossitis (plummer vinson syndrome). N. Eng. J. Med. 1953, 249, 1009–1012. [Google Scholar] [CrossRef]
- Tahara, T.; Shibata, T.; Okubo, M.; Yoshioka, D.; Ishizuka, T.; Sumi, K.; Kawamura, T.; Nagasaka, M.; Nakagawa, Y.; Arisawa, T.; et al. A case of plummer-vinson syndrome showing rapid improvement of Dysphagia and esophageal web after two weeks of iron therapy. Case Rep. Gastroenterol. 2014, 8, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Sanai, F.M.; Mohamed, A.E.; Karawi, M.A. Dysphagia caused by Plummer-Vinson syndrome. Endoscopy 2001, 33, 470. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kitahara, S.; Ohmae, Y.; Ogura, M.; Matumury, Y. The operation of upper esophageal web in Plummer-Vinson syndrome: A case report. Auris Nasus Larynx. 1999, 26, 495–500. [Google Scholar] [CrossRef]
- Anderson, S.R.; Sinacori, J.T. Plummer-Vinson syndrome heralded by postcricoid carcinoma. Am. J. Otolaryngol. 2007, 28, 22–24. [Google Scholar] [CrossRef]
- Watts, J.M. The importance of plummer-vinson syndrome in the aetiology of carcinoma of the upper gastrointestinal tract. Postgrad. Med. J. 1961, 37, 523–533. [Google Scholar] [CrossRef] [Green Version]

| Demographics of all Patients Undergoing EGD between 4/2019 and 6/2020 | |
|---|---|
| Average Age | 52.9 years |
| Gender | Male: 44% (59/134) Female: 56% (75/134) |
| Ethnicity | Hispanic: 72% Non-Hispanic White: 23% African American: 5 % |
| Initial Hemoglobin (g/dL) | Iron Level (mcg/dL) | Dose of Iron Replacement | Autoimmune Marker | |
|---|---|---|---|---|
| Patient 1 | 12.8 * | N/A | Ferrous Sulfate 325 (65 Fe) mg | None |
| Patient 2 | 3.0 | 16 | Ferrous Sulfate 325 (65 Fe) mg | Free T4: <0.7; TSH: 111; Anti parietal cell antibody: 36.6 (<20 = normal); TTG IgA: 30 (<4 = normal) |
| Patient 3 | 12.9 * | N/A | Ferrous Sulfate 325 (65 Fe) mg | ANA: positive; SSA: positive; SSB: positive |
| Initial EGD Finding | |
|---|---|
| Patient 1 | Single benign appearing intrinsic stenosis located 15 mm from the UES dilated with a TTS balloon dilator to 20 mm, gastric mucosal atrophy |
| Patient 2 | Web/stenosis at the cricopharyngeus unable to pass with neonatal scope due to resistance, guideware placed under fluoroscopic guidance and dilation performed with Savary dilator to 8 mm |
| Patient 3 | Two benign appearing intrinsic stenoses were found at the circopharyngeus at 15 cm and 20 cm from the incisors that could not be traversed with the neonatal scope, dilation performed with Savary dilators to 11 mm |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Herlihy, J.D.; Badhiwala, V.; Padilla, O.; Diaz, J.; McCallum, R. Plummer-Vinson Syndrome: A Time for Redefinition and New Perspectives. Gastrointest. Disord. 2020, 2, 408-414. https://0-doi-org.brum.beds.ac.uk/10.3390/gidisord2040037
Herlihy JD, Badhiwala V, Padilla O, Diaz J, McCallum R. Plummer-Vinson Syndrome: A Time for Redefinition and New Perspectives. Gastrointestinal Disorders. 2020; 2(4):408-414. https://0-doi-org.brum.beds.ac.uk/10.3390/gidisord2040037
Chicago/Turabian StyleHerlihy, J. Daniel, Vismay Badhiwala, Osvaldo Padilla, Jesus Diaz, and Richard McCallum. 2020. "Plummer-Vinson Syndrome: A Time for Redefinition and New Perspectives" Gastrointestinal Disorders 2, no. 4: 408-414. https://0-doi-org.brum.beds.ac.uk/10.3390/gidisord2040037





