Pretreatment of Loblolly Pine Tree Needles Using Deep Eutectic Solvents
Abstract
:1. Introduction
2. Materials and Methods
2.1. Raw Materials and Chemicals
2.2. Synthesis of DES
2.3. Preparation of Biomass for Pretreatment
2.4. Pretreatment of Pine Needles Using DES
2.5. Separation of Biomass and DES
2.5.1. Filtration
2.5.2. Lignin Precipitation
2.6. Fourier Transform Infrared Spectroscopy (FTIR)
2.7. Enzymatic Hydrolysis
2.8. Carbohydrate Analysis Using High-Performance Liquid Chromatography (HPLC)
2.9. Acid Soluble Lignin Analysis
2.10. Soxhlet Extraction
3. Results and Discussion
3.1. Mass Yield from Deep Eutectic Solvents (DES) Pretreatment
3.2. Fourier Transform Infrared (FTIR) Analysis of Pretreated Loblolly Pine Needles and Precipitate from Deionized Water
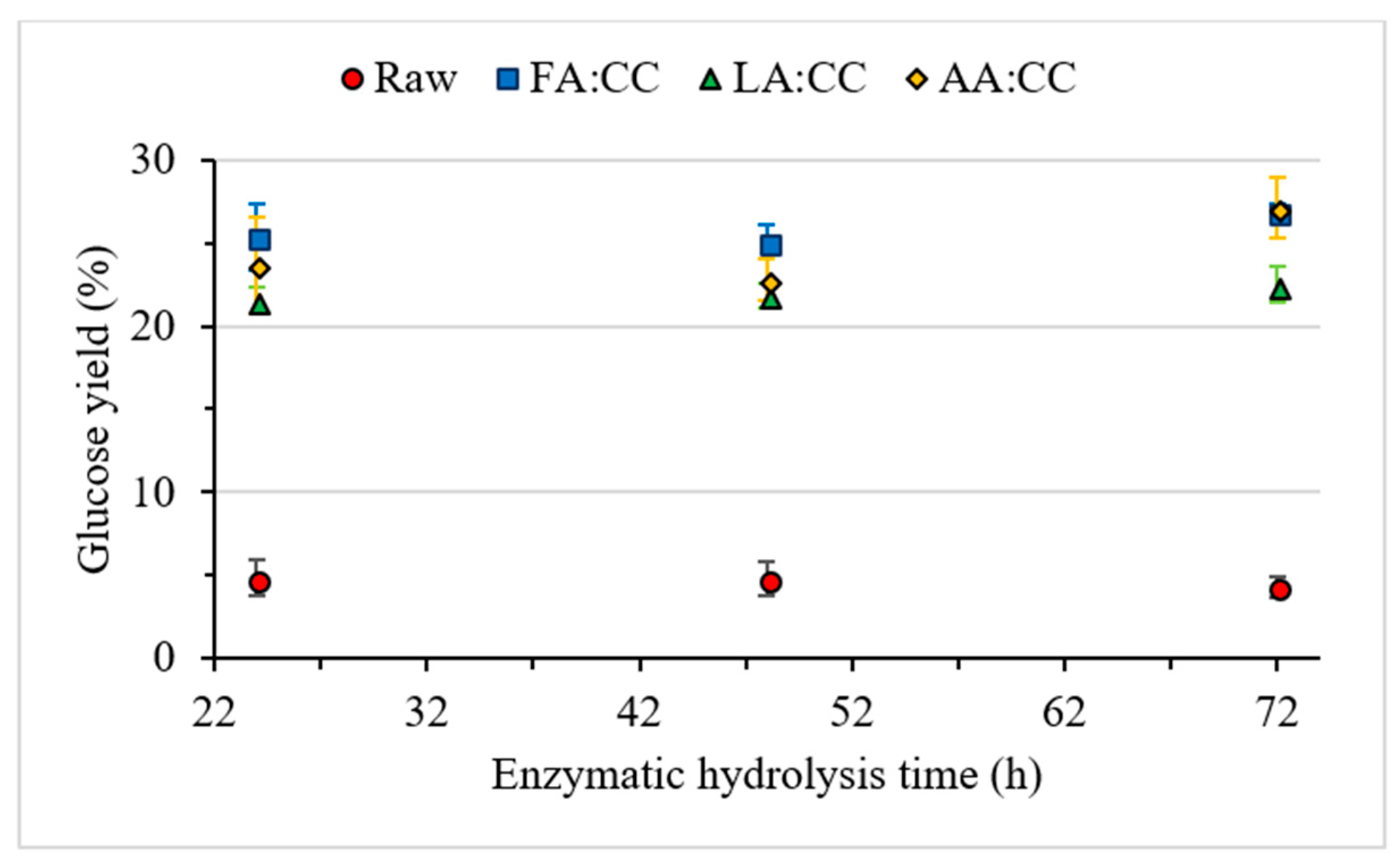
3.3. Enzymatic Hydrolysis
3.4. Fiber Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Malaeke, H.; Housaindokht, M.R.; Monhemi, H.; Izadyar, M. Deep eutectic solvent as an efficient molecular liquid for lignin solubilization and wood delignification. J. Mol. Liq. 2018, 263, 193–199. [Google Scholar] [CrossRef]
- Lee, J. Biological conversion of lignocellulosic biomass to ethanol. J. Biotechnol. 1997, 56, 1–24. [Google Scholar] [CrossRef]
- Panwar, N.L.; Kaushik, S.C.; Kothari, S. Role of renewable energy sources in environmental protection: A review. Renew. Sustain. Energy Rev. 2011, 15, 1513–1524. [Google Scholar] [CrossRef]
- Figueiredo, P.; Lintinen, K.; Hirvonen, J.T.; Kostiainen, M.A.; Santos, H.A. Properties and chemical modifications of lignin: Towards lignin-based nanomaterials for biomedical applications. Prog. Mater. Sci. 2018, 93, 233–269. [Google Scholar] [CrossRef]
- Nix, S. Loblolly Pine, an Important Tree in North America. Available online: https://www.thoughtco.com/loblolly-pine-important-tree-north-america-1342788 (accessed on 23 November 2020).
- Lange, B.M.; Catherine, L.; Heinrich Sandermann, J. Elicitor-Induced Spruce Stress Lignin: Structural Similarity to Early Developmental Lignins. Plant Physiol. 1995, 108, 1277–1287. [Google Scholar] [CrossRef] [Green Version]
- Slathia, P.S.; Raina, N.; Kiran, A.; Kour, R.; Bhagat, D.; Sharma, P. Dilute acid pretreatment of pine needles of Pinus roxburghii by response surface methodology for bioethanol production by separate hydrolysis and fermentation. Biomass Convers. Biorefinery 2020, 10, 95–106. [Google Scholar] [CrossRef]
- Mandal, S.; Bhattacharya, T.K.; Haydary, J.; Husar, J.; Tanna, H.R.; Haz, A. Valorization of Pine Needles by Thermal Conversion to Solid, Liquid and Gaseous Fuels in a Screw Reactor. Waste Biomass Valorization 2019, 10, 3587–3599. [Google Scholar] [CrossRef]
- Francisco, M.; van den Bruinhorst, A.; Kroon, M.C. New natural and renewable low transition temperature mixtures (LTTMs): Screening as solvents for lignocellulosic biomass processing. Green Chem. 2012, 14, 2153–2157. [Google Scholar] [CrossRef]
- Lynam, J.G.; Kumar, N.; Wong, M.J. Deep eutectic solvents’ ability to solubilize lignin, cellulose, and hemicellulose; thermal stability; and density. Bioresour. Technol. 2017, 238, 684–689. [Google Scholar] [CrossRef] [Green Version]
- Catalan, L.J.J.; Liang, V.; Jia, C.Q.; Walton, C. Effects of process changes on concentrations of individual malodorous sulphur compounds in ambient air near a Kraft pulp plant in Thunder Bay, Ontario, Canada. Wit Trans. Ecol. Environ. 2007, 101, 437–447. [Google Scholar] [CrossRef]
- Kumar, N.; Gautam, R.; Stallings, J.D.; Coty, G.G.; Lynam, J.G. Secondary Agriculture Residues Pretreatment Using Deep Eutectic Solvents. Waste Biomass Valorization 2020, 12, 2259–2269. [Google Scholar] [CrossRef]
- Shi, J.T.; Li, J. Metabolites and chemical group changes in the wood-forming tissue of pinus koraiensisunder inclided conditions. Bioresources 2012, 7, 3463–3475. [Google Scholar]
- Raj, T.; Kapoor, M.; Gaur, R.; Christopher, J.; Lamba, B.; Tuli, D.K.; Kumar, R. Physical and Chemical Characterization of Various Indian Agriculture Residues for Biofuels Production. Energy Fuels 2015, 29, 3111–3118. [Google Scholar] [CrossRef]
- Selig, M.W.N.; Ji, Y. Enzymatic Saccharification of Lignocellulosic Biomass, LAP-009 NREL Analytical Procedure; Technical Report NREL/TP-510-42629; National Renewable Energy Laboratory (NREL): Golden, CO, USA, 2008. [Google Scholar]
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D.; Crocker, D. Determination of structural carbohydrates and lignin in biomass. Lab. Anal. Proced. 2008, 1617, 1–16. [Google Scholar]
- Kumar, N.; Muley, P.D.; Boldor, D.; Coty, I.V.G.G.; Lynam, J.G. Pretreatment of waste biomass in deep eutectic solvents: Conductive heating versus microwave heating. Ind. Crop. Prod. 2019, 142, 111865. [Google Scholar] [CrossRef]
- Oh, Y.; Park, S.; Jung, D.; Oh, K.K.; Lee, S.H. Effect of hydrogen bond donor on the choline chloride-based deep eutectic solvent-mediated extraction of lignin from pine wood. Int. J. Biol. Macromol. 2020, 165, 187–197. [Google Scholar] [CrossRef]
- Brandt, A.; Gräsvik, J.; Hallett, J.P.; Welton, T. Deconstruction of lignocellulosic biomass with ionic liquids. Green Chem. 2013, 15, 550–583. [Google Scholar] [CrossRef] [Green Version]
- Okur, M.; Eslek Koyuncu, D.D. Investigation of pretreatment parameters in the delignification of paddy husks with deep eutectic solvents. Biomass Bioenergy 2020, 142, 105811. [Google Scholar] [CrossRef]
- Mamilla, J.L.K.; Novak, U.; Grilc, M.; Likozar, B. Natural deep eutectic solvents (DES) for fractionation of waste lignocellulosic biomass and its cascade conversion to value-added bio-based chemicals. Biomass Bioenergy 2019, 120, 417–425. [Google Scholar] [CrossRef]
- Carrión-Prieto, P.; Hernández-Navarro, S.; Sánchez-Sastre, L.F.; Marcos-Robles, J.L.; Martín-Gil, J.; Martín-Ramos, P. Furfural, 5-HMF, acid-soluble lignin and sugar contents in C. ladanifer and E. arborea lignocellulosic biomass hydrolysates obtained from microwave-assisted treatments in different solvents. Biomass Bioenergy 2018, 119, 135–143. [Google Scholar] [CrossRef] [Green Version]
- Fernandes, C.; Melro, E.; Magalhães, S.; Alves, L.; Craveiro, R.; Filipe, A.; Valente, A.J.M.; Martins, G.; Antunes, F.E.; Romano, A.; et al. New deep eutectic solvent assisted extraction of highly pure lignin from maritime pine sawdust (Pinus pinaster Ait.). Int. J. Biol. Macromol. 2021, 177, 294–305. [Google Scholar] [CrossRef] [PubMed]



| Hydrogen Bond Donor | Hydrogen Bond Acceptor | Molar Ratio of DES | pH of DES | pH of DES Diluted 10% with Water | pH of DES Diluted 90% with Water |
|---|---|---|---|---|---|
| Formic acid | Choline chloride | 2:1 | −0.92 | 0.70 | 1.24 |
| Lactic acid | Choline chloride | 10:1 | −0.60 | 0.54 | 1.91 |
| Acetic acid | Choline chloride | 2:1 | 0.47 | 1.01 | 1.84 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gujjula, P.; Kumar, N.; Lynam, J.G. Pretreatment of Loblolly Pine Tree Needles Using Deep Eutectic Solvents. Biomass 2021, 1, 1-10. https://0-doi-org.brum.beds.ac.uk/10.3390/biomass1010001
Gujjula P, Kumar N, Lynam JG. Pretreatment of Loblolly Pine Tree Needles Using Deep Eutectic Solvents. Biomass. 2021; 1(1):1-10. https://0-doi-org.brum.beds.ac.uk/10.3390/biomass1010001
Chicago/Turabian StyleGujjula, Prathima, Narendra Kumar, and Joan G. Lynam. 2021. "Pretreatment of Loblolly Pine Tree Needles Using Deep Eutectic Solvents" Biomass 1, no. 1: 1-10. https://0-doi-org.brum.beds.ac.uk/10.3390/biomass1010001






