Evaluation of Zosteric Acid for Mitigating Biofilm Formation of Pseudomonas putida Isolated from a Membrane Bioreactor System
Abstract
:1. Introduction
2. Results
2.1. Confocal Laser Scanning Microscopy (CLSM) Analyses of Fouled Filtration Membranes
2.2. Denaturing Gradient Gel Electrophoresis (DGGE), Sequencing and Community Profiles Analysis

| Source | BlastN reference strains | RDP tassonomic classifier | |||
|---|---|---|---|---|---|
| Closest relative strain | Similarity (%) | GenBank accession number | Most probabile taxon | Similarity (%) | |
| Sludge | Paracoccus sp. | 92 | AY646160 | Alphaproteobacteria | 99 |
| Novosphingobium sp. | 99 | EU430056 | Alphaproteobacteria | 100 | |
| Novosphingobium aromaticivorans | 93 | JF459981 | Alphaproteobacteria | 100 | |
| Novosphingobium sp. | 96 | FM886872 | Alphaproteobacteria | 100 | |
| Alpha proteobacterium | 91 | AB578881 | Alphaproteobacteria | 100 | |
| Sphingomonas sp. | 99 | AJ617690 | Alphaproteobacteria | 100 | |
| Sphingomonas sp. | 89 | GQ181132 | Alphaproteobacteria | 97 | |
| Sphingobacteriaceae bacterium | 97 | FJ386545 | Bacteroidetes | 100 | |
| uncultured Niastella sp. | 98 | GU326307 | Bacteroidetes | 100 | |
| Flavobacteriales bacterium | 95 | EF636194 | Bacteroidetes | 100 | |
| uncultured Oxalobacteraceae bacterium | 91 | HM141157 | Betaproteobacteria | 100 | |
| Fouled membranes | uncultured Xanthomonadalesbacterium | 89 | EF073609 | Gammaproteobacteria | 99 |
| uncultured Gammaproteobacteria bacterium | 99 | CU926678 | Gammaproteobacteria | 100 | |
| Rhodanobacter sp. | 99 | EU876661 | Gammaproteobacteria | 100 | |
| Rhodanobacter sp. | 97 | EU876661 | Gammaproteobacteria | 100 | |
| uncultured Betaproteobacterium | 97 | JF703550 | Betaproteobacteria | 100 | |
| uncultured Comamonas sp. | 89 | JF808874 | Betaproteobacteria | 99 | |
| uncultured bacterium | 98 | AB241559 | Bacteroidetes | 100 | |
| uncultured Acidobacteria bacterium | 98 | HM062044 | Acidobacteria | 100 | |
| uncultured Betaproteobacterium | 89 | FJ184029 | unclassified bacteria | 98 | |

2.3. Bacterial Strains Isolated from Fouled Filtration Membrane
| Closest relative strain number | Cultural media | Taxa | Similarity (%) | GenBank accession |
|---|---|---|---|---|
| Pseudomonas putida | PCA | Gammaproteobacteria | 99% | JF703662 |
| Pseudomonas sp. | PCA | Gammaproteobacteria | 99% | HQ718413 |
| Pseudomonas sp. | SWA | Gammaproteobacteria | 97% | AM689975 |
| Pseudomonas sp. | SWA | Gammaproteobacteria | 99% | HQ588845 |
| Shewanella xiamenensis | PCA | Gammaproteobacteria | 99% | HQ418493 |
| Enterobacter cloacae | PCA | Gammaproteobacteria | 99% | HQ231214 |
| Bacillus arvi | PCA | Firmicutes | 99% | AM260979.1 |
| Lysinibacillus fusiformis | PCA | Firmicutes | 99% | GQ844965 |
| Bacillus cereus | PCA | Firmicutes | 100% | HQ333012 |
| Rhodococcus sp. | PCA | Actinobacteria | 96% | EU293153 |
| Chitinimonas sp. | SWA | Betaproteobacteria | 97% | GQ354569 |
2.4. Zosteric Acid Was not a Carbon and Energy Source for the Membrane Colonizer P. putida but Affected Cell Adhesion

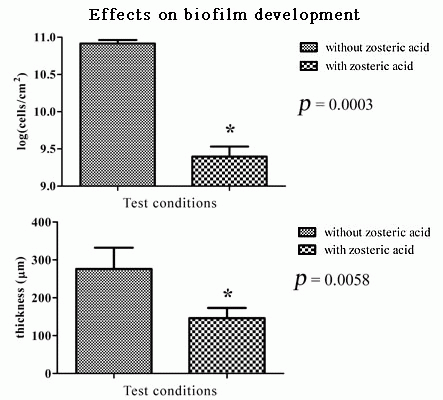
2.5. Zosteric Acid Affected the Morphology, Thickness and Biomass of P. putida Biofilm

2.6. General Features of the Zosteric Acid Behavior in the Environment
| Physico-chemical properties | Value | Source |
|---|---|---|
| Molecular weight (g/mol) | 244 | – |
| Melting point (°C) | 260 | [30] |
| Water solubility (mg/L at 20 °C) | 2.5 × 105 | [21] |
| Vapor pressure (Pa, 25 °C) | 3.81 × 10−9 | Predicted |
| log Kow (octanol water coefficient, 20 °C) | −0.25 | Measured in this work |
| log BCF (bioconcentration factor) | 0.94 | Predicted |
2.7. Ecotoxicity Responses
3. Discussion

4. Experimental Section
4.1. Sample Collection and Preparation
4.2. Analysis of Bacterial Community by Culture Independent Methods
4.3. Isolation and Identification of Bacterial Strains from Fouled Filtration Membrane
4.4. Synthesis of Zosteric Acid
4.5. Cellular Growth with Zosteric Acid
4.6. Microplate-Based Biofilm Assay
4.7. Biofilm Preparation by Colony-Biofilm Method
4.8. Biofilm Sectioning
4.9. Biofilm Imaging by Confocal Laser Scanning Microscopy (CLSM)
4.10. Physicochemical Profiling of Zosteric Acid
4.11. D. magna Acute Ecotoxicity Test
4.12. P. subcapitata Growth Inhibition Ecotoxicity Test
4.13. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Melin, T.; Jefferson, B.; Bixio, D.; Thoeye, C.; de Wilde, W.; de Koning, J.; van der Graaf, J.; Wintgens, T. Membrane bioreactor technology for wastewater treatment and reuse. Desalination 2006, 187, 271–282. [Google Scholar]
- Lee, E.; Shon, H.K.; Cho, J. Biofouling characteristics using flow field-flow fractionation: Effect of bacteria and membrane properties. Bioresour. Technol 2010, 101, 1487–1493. [Google Scholar]
- He, Y.; Bagley, D.M.; Leung, K.T.; Liss, S.N.; Liao, B.-Q. Recent advances in membrane technologies for biorefining and bioenergy production. Biotechnol. Adv 2012, 30, 817–858. [Google Scholar]
- Plakas, K.V.; Karabelas, A.J. Removal of pesticides from water by NF and RO membranes—A review. Desalination 2012, 287, 255–265. [Google Scholar]
- Hoinkis, J.; Deowan, S.A.; Panten, V.; Figoli, A.; Huang, R.R.; Drioli, E. Membrane bioreactor (MBR) technology—A promising approach for industrial water reuse. Procedia Eng 2012, 33, 234–241. [Google Scholar]
- Guo, W.; Ngo, H.-H.; Li, J. A mini-review on membrane fouling. Bioresour. Technol 2012, 122, 27–34. [Google Scholar]
- Mansouri, J.; Harrisson, S.; Chen, V. Strategies for controlling biofouling in membrane filtration systems: Challenges and opportunities. J. Mater. Chem 2010, 20, 4567–4586. [Google Scholar]
- Flemming, H.-C. Biofouling in water systems—Cases, causes and countermeasures. Appl. Microbiol. Biotechnol 2002, 59, 629–640. [Google Scholar]
- Al-Amoudi, A.; Lovitt, R.W. Fouling strategies and the cleaning system of NF membranes and factors affecting cleaning efficiency. J. Membr. Sci 2007, 303, 4–28. [Google Scholar]
- Duan, L.; Moreno-Andrade, I.; Huang, C.-L.; Xia, S.; Hermanowicz, S.W. Effects of short solids retention time on microbial community in a membrane bioreactor. Bioresour. Technol 2009, 100, 3489–3496. [Google Scholar]
- Molina-Muñoz, M.; Poyatos, J.M.; Sánchez-Peinado, M.; Hontoria, E.; González-López, J.; Rodelas, B. Microbial community structure and dynamics in a pilot-scale submerged membrane bioreactor aerobically treating domestic wastewater under real operation conditions. Sci. Total Environ 2009, 407, 3994–4003. [Google Scholar]
- Yuan, D.; Fan, Y.; Yu, Y.; Xu, G.; Yang, W.; Wu, G. Study of a membrane bioreactor with glass fibre flat grille modules and the modules’ optimization based on the local critical flux theory. Water Res 2010, 44, 997–1005. [Google Scholar]
- Vrouwenvelder, J.S.; van Loosdrecht, M.C.M.; Kruithof, J.C. A novel scenario for biofouling control of spiral wound membrane systems. Water Res 2011, 45, 3890–3898. [Google Scholar]
- Kappelhof, J.W.N.M.; Vrouwenvelder, H.S.; Schaap, M.; Kruithof, J.C.; van der Kooij, D.; Schippers, J.C. An in situ biofouling monitor for membrane systems. Water Sci. Technol 2003, 3, 205–210. [Google Scholar]
- Cappitelli, F.; Sorlini, C. Microorganisms attack synthetic polymers in items representing our cultural heritage. Appl. Environ. Microbiol 2008, 74, 564–569. [Google Scholar]
- Bereschenko, L.; Prummel, H.; Euverink, G.J.W.; Stams, J.M.; van Loosdrecht, M.C.M. Effect of conventional chemical treatment on the microbial population in a biofouling layer of reverse osmosis systems. Water Res 2011, 45, 405–416. [Google Scholar]
- Stewart, P.S.; Franklin, M.J. Physiological heterogeneity in biofilms. Nat. Rev. Microbiol 2008, 6, 199–210. [Google Scholar]
- Høiby, N.; Bjarnsholt, T.; Givskov, M.; Molin, S.; Ciofu, O. Antibiotic resistance of bacterial biofilms. Int. J. Antimicrob. Agents 2010, 35, 322–332. [Google Scholar]
- Pace, J.L.; Rupp, M.E.; Finch, R.G. Biofilms, Infection, and Antimicrobial Therapy; Taylor & Francis: Boca Raton, FL, USA, 2006. [Google Scholar]
- Villa, F.; Albanese, D.; Giussani, B.; Stewart, P.S.; Daffonchio, D.; Cappitelli, F. Hindering biofilm formation with zosteric acid. Biofouling 2010, 26, 739–752. [Google Scholar]
- Villa, F.; Pitts, B.; Stewart, P.S.; Giussani, B.; Roncoroni, S.; Albanese, D.; Giordano, C.; Tunesi, M.; Cappitelli, F. Efficacy of zosteric acid sodium salt on the yeast biofilm model Candida albicans. Microb. Ecol 2011, 62, 584–598. [Google Scholar]
- Dreszer, C.; Flemming, H.-C.; Zwijnenburg, A.; Kruithof, J.C.; Vrouwenvelder, J.S. Impact of biofilm accumulation on transmembrane and feed channel pressure drop: Effects of crossflow velocity, feed spacer and biodegradable nutrient. Water Res 2014, 50, 200–211. [Google Scholar]
- Timmis, K.N. Pseudomonas putida: A cosmopolitan opportunist par excellence. Environ. Microbiol 2002, 4, 779–781. [Google Scholar]
- Tolker-Nielsen, T.; Brinch, U.C.; Ragas, P.C.; Andersen, J.B.; Jacobsen, C.S.; Molin, S. Development and dynamics of Pseudomonas sp. biofilms. J. Bacteriol 2000, 182, 6482–6489. [Google Scholar]
- Gerardi, M.H. Wastewater Bacteria; Gerardi, M.H., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2006; Volume 127. [Google Scholar]
- Dams, R.I.; Biswas, A.; Olesiejuk, A.; Fernandes, T.; Christofi, N. Silver nanotoxicity using a light-emitting biosensor Pseudomonas putida isolated from a wastewater treatment plant. J. Hazard. Mater 2011, 195, 68–72. [Google Scholar]
- Paul, D.; Kim, Y.S.; Ponnusamy, K.; Kweon, J.H. Application of quorum quenching to inhibit biofilm formation. Environ. Eng. Sci 2009, 26, 1319–1324. [Google Scholar]
- Dasari, A.; Quirós, J.; Herrero, B.; Boltes, K.; García-Calvo, E.; Rosal, R. Antifouling membranes prepared by electrospinning polylactic acid containing biocidal nanoparticles. J. Membr. Sci 2012, 405–406, 134–140. [Google Scholar]
- Rosas, I.; Collado, S.; Fernández, I.; Gutierrez-Lavin, A.; Diaz, M. Fouling in pleated microfiltration cartridges caused by Pseudomonas putida. J. Membr. Sci 2013, 443, 107–114. [Google Scholar]
- Nakazawa, T.; Ohsawa, K. Metabolites of orally administered Perilla frutescens extract in rats and humans. Biol. Pharm. Bull 2000, 23, 122–127. [Google Scholar]
- Kim, A.S.; Chen, H.; Yuan, R. EPS biofouling in membrane filtration: An analytic modeling study. J. Colloid Interface Sci 2006, 303, 243–249. [Google Scholar]
- Chen, C.-L.; Liu, W.-T.; Chong, M.-L.; Wong, M.-T.; Ong, S.L.; Seah, H.; Ng, W.J. Community structure of microbial biofilms associated with membrane-based water purification processes as revealed using a polyphasic approach. Appl. Microbiol. Biotechnol 2004, 63, 466–473. [Google Scholar]
- Zhang, J.; Chua, H.C.; Zhou, J.; Fane, A.G. Factors affecting the membrane performance in submerged membrane bioreactors. J. Membr. Sci 2006, 284, 54–66. [Google Scholar]
- Ivnitsky, H.; Katz, I.; Minz, D.; Volvovic, G.; Shimoni, E.; Kesselman, E.; Semiat, R.; Dosoretz, C.G. Bacterial community composition and structure of biofilms developing on nanofiltration membranes applied to wastewater treatment. Water Res 2007, 41, 3924–3935. [Google Scholar]
- Huang, L.-N.; de Wever, H.; Diels, L. Diverse and distinct bacterial communities induced biofilm fouling in membrane bioreactors operated under different conditions. Environ. Sci. Technol 2008, 42, 8360–8366. [Google Scholar]
- Xia, S.; Li, J.; He, S.; Xie, K.; Wang, X.; Zhang, Y.; Duan, L.; Zhang, Z. The effect of organic loading on bacterial community composition of membrane biofilms in a submerged polyvinyl chloride membrane bioreactor. Bioresour. Technol 2010, 101, 6601–6609. [Google Scholar]
- Zhang, K.; Choi, H.; Dionysiou, D.D.; Sorial, G.; Oerther, D.B. Identifying pioneer bacterial species responsible for biofouling membrane bioreactors. Environ. Microbiol 2006, 8, 433–440. [Google Scholar]
- Fanget, N.V.J.; Foley, S. Starvation/stationary-phase survival of Rhodococcus erythropolis SQ1: A physiological and genetic analysis. Arch. Microbiol 2011, 193, 1–13. [Google Scholar]
- Huang, J.; Sun, B.; Zhang, X. Shewanella xiamenensis sp. nov., isolated from coastal sea sediment. Int. J. Syst. Evol. Microbiol 2010, 60, 1585–1589. [Google Scholar]
- Potron, A.; Poirel, L.; Nordmann, P. Origin of OXA-181, an emerging carbapenem-hydrolyzing oxacillinase, as a chromosomal gene in Shewanella xiamenensis. Antimicrob. Agents Chemother 2011, 55, 4405–4407. [Google Scholar]
- Heyrman, J.; Rodríguez-Díaz, M.; Devos, J.; Felske, A.; Logan, N.; de Vos, P. Bacillus arenosi sp. nov., Bacillus arvi sp. nov. and Bacillus humi sp. nov., isolated from soil. Int. J. Syst. Evol. Microbiol 2005, 55, 111–117. [Google Scholar]
- Arnesen, S.L.P.; Fagerlund, A.; Granum, P.E. From soil to gut: Bacillus cereus and its food poisoning toxins. FEMS Microbiol. Rev 2008, 32, 579–606. [Google Scholar]
- Guentzel, M.N. Escherichia, Klebsiella, Enterobacter, Serratia, Citrobacter, and Proteus; Barron, S., Ed.; University of Texas Medical Branch at Galveston: Galveston, TX, USA, 1996; pp. 1–14. [Google Scholar]
- Pang, C.M.; Liu, W.-T. Community structure analysis of reverse osmosis membrane biofilms and the significance of Rhizobiales bacteria in biofouling. Environ. Sci. Technol 2007, 41, 4728–4734. [Google Scholar]
- Hörsch, P.; Gorenflo, A.; Fuder, C.; Deleage, A.; Frimmel, F.H. Biofouling of ultra- and nano-filtration membranes fordrinking water treatment characterized by fluorescence in situ hybridization (FISH). Desalination 2005, 172, 41–52. [Google Scholar]
- Geuenich, H.H.; Müller, H.E. Occurrence of Pseudomonas aeruginosa in waste water and its behavior under biological treatment. Zentralbl. Bakteriol. Mikrobiol. Hyg. B 1984, 179, 259–265. [Google Scholar]
- Tchobanoglous, G.; Burton, F.L.; Stensel, H.D. Wastewater Engineering: Treatment and Reuse; McGraw-Hill Co.: New York, NY, USA, 2003. [Google Scholar]
- Rodríguez-Chueca, J.; Morales, M.; Mosteo, R.; Ormad, M.P.; Ovelleiro, J.L. Inactivation of Enterococcus faecalis, Pseudomonas aeruginosa and Escherichia coli present in treated urban wastewater by coagulation–flocculation and photo-Fenton processes. Photochem. Photobiol. Sci 2013, 12, 864–871. [Google Scholar]
- Xu, Q.; Barrios, C.A.; Cutright, T.; Newby, B.Z. Assessment of antifouling effectiveness of two natural product antifoulants by attachment study with freshwater bacteria. Environ. Sci. Pollut. Res. Int 2005, 12, 278–284. [Google Scholar]
- Newby, B.Z.; Cutright, T.; Barrios, C.A.; Xu, Q. Zosteric acid—An effective antifoulant for reducing fresh water bacterial attachment on coatings. J. Coatings Technol. Res 2006, 3, 69–76. [Google Scholar]
- Pitts, B.; Hamilton, M.A.; Zelver, N.; Stewart, P.S. A microtiter-plate screening method for biofilm disinfection and removal. J. Microbiol. Methods 2003, 54, 269–276. [Google Scholar]
- Villa, F.; Remelli, W.; Forlani, F.; Vitali, A.; Cappitelli, F. Altered expression level of Escherichia coli proteins in response to treatment with the antifouling agent zosteric acid sodium salt. Environ. Microbiol 2012, 14, 1753–1761. [Google Scholar]
- Flemming, H.-C. Alternative and conventional anti-fouling strategies. Int. Biodeterior. Biodegrad 2005, 56, 121–134. [Google Scholar]
- Allen, D.T. Evaluating environmental fate: Approaches based on chemical structure. In Green Engineering: Environmentally Conscious Design of Chemical Processes; Allen, D.T., Shonnard, D.R., Eds.; Prentice Hall: Upper Saddle River, NJ, USA, 2001. [Google Scholar]
- United States Environmental Protection Agency (USEPA). Estimation Program Interface Suite for Microsoft Windows 2010. Available online: http://www.epa.gov/opptintr/exposure/pubs/episuite.htm (accessed on 21 April 2014).
- Finlay, B.J.; Esteban, G.F. Freshwater protozoa: Biodiversity and ecological function. Biodivers. Conserv 1998, 7, 1163–1186. [Google Scholar]
- Goulder, R. Observations over 24 hours on the quantity of algae inside grazing ciliated protozoa. Oecologia 1973, 13, 177–182. [Google Scholar]
- Canter, H.M.; Lund, J.W.G. The importance of Protozoa in controlling the abundance of planktonic algae in lakes. Proc. Linn. Soc. Lond 1968, 179, 203–219. [Google Scholar]
- Murray, A.E.; Preston, C.M.; Massana, R.; Taylor, L.T.; Blakis, A.; Wu, K.; DeLong, E.F. Seasonal and spatial variability of bacterial and archaeal assemblages in the coastal waters near Anvers Island, Antarctica. Appl. Environ. Microbiol 1998, 64, 2585–2595. [Google Scholar]
- Polo, A.; Cappitelli, F.; Brusetti, L.; Principi, P.; Villa, F.; Giacomucci, L.; Ranalli, G.; Sorlini, C. Feasibility of removing surface deposits on stone using biological and chemical remediation methods. Microb. Ecol 2010, 60, 1–14. [Google Scholar]
- BLASTN software. Available online: http://0-www-ncbi-nlm-nih-gov.brum.beds.ac.uk/BLAST (accessed on 1 June 2011).
- European Nucleotide Archive (ENA). Available online: http://www.ebi.ac.uk/ena/ (accessed on 2 November 2012).
- ImageJ. Available online: http://rsb.info.nih.gov/ij/ (accessed on 21 April 2014).
- Bundy, J.G.; Paton, G.I.; Campbell, C.D. Combined microbial community level and single species biosensor responses to monitor recovery of oil polluted soil. Soil Biol. Biochem 2004, 36, 1149–1159. [Google Scholar]
- Khan, J.S.; Ilyas, S.; Javid, S.; Visvanathan, C.; Jegatheesan, V. Performance of suspended and attached growth MBR systems in treating high strength synthetic wastewater. Bioresour. Technol 2011, 102, 5331–5336. [Google Scholar]
- Anderl, J.N.; Franklin, M.J.; Stewart, P.S. Role of antibiotic penetration limitation in Klebsiella pneumoniae biofilm resistance to ampicillin and ciprofloxacin. Antimicrob. Agents Chemother 2000, 44, 1818–1824. [Google Scholar]
- Vistoli, G.; Straniero, V.; Pedretti, A.; Fumagalli, L.; Bolchi, C.; Pallavicini, M.; Valoti, E.; Testa, B. Predicting the physicochemical profile of diastereoisomeric histidine-containing dipeptides by property space analysis. Chirality 2012, 24, 566–576. [Google Scholar]
- Organisation for Economic Cooperation and Development (OECD). Guidelines for Testing of Chemicals. No. 202: Daphnia sp. Acute Immobilization Test. Available online: http://0-www-oecd--ilibrary-org.brum.beds.ac.uk/docserver/download/9720201e.pdf?expires=1398063318&id=id&accname=guest&checksum=347E866C55D58C501CF68773EC0E9696 (accessed on 21 April 2014).
- Organisation for Economic Cooperation and Development (OECD). Guidelines for Testing of Chemicals. No. 201: Freshwater Alga and Cyanobacteria, Growth Inihibition Test. Available online: http://0-www-oecd--ilibrary-org.brum.beds.ac.uk/docserver/download/9720101e.pdf?expires=1398063381&id=id&accname=guest&checksum=73B29063EC9A4971F6CF5380437D7FC2 (accessed on 21 April 2014).
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Polo, A.; Foladori, P.; Ponti, B.; Bettinetti, R.; Gambino, M.; Villa, F.; Cappitelli, F. Evaluation of Zosteric Acid for Mitigating Biofilm Formation of Pseudomonas putida Isolated from a Membrane Bioreactor System. Int. J. Mol. Sci. 2014, 15, 9497-9518. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms15069497
Polo A, Foladori P, Ponti B, Bettinetti R, Gambino M, Villa F, Cappitelli F. Evaluation of Zosteric Acid for Mitigating Biofilm Formation of Pseudomonas putida Isolated from a Membrane Bioreactor System. International Journal of Molecular Sciences. 2014; 15(6):9497-9518. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms15069497
Chicago/Turabian StylePolo, Andrea, Paola Foladori, Benedetta Ponti, Roberta Bettinetti, Michela Gambino, Federica Villa, and Francesca Cappitelli. 2014. "Evaluation of Zosteric Acid for Mitigating Biofilm Formation of Pseudomonas putida Isolated from a Membrane Bioreactor System" International Journal of Molecular Sciences 15, no. 6: 9497-9518. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms15069497