Design, Synthesis and Biological Evaluation of 1,4-Disubstituted-3,4-dihydroisoquinoline Compounds as New Tubulin Polymerization Inhibitors
Abstract
:1. Introduction


2. Results and Discussion
2.1. Chemistry

| Compound | R1 | R2 | IC50 (μM) |
|---|---|---|---|
| CEM | |||
| 20 | 3',4'-OCH3 |  | 36.29 |
| 21 | 3'-NH2-4'-OCH3 |  | 4.10 |
| 22 | 3'-NHCOCH3-4'-OCH3 |  | 24.32 |
| 23 | 4'-OCH3 |  | 17.76 |
| 24 | 4'-OH |  | 40.33 |
| 25 | 4'-OCOCH3 |  | >100 |
| 26 | 4'-F |  | 44.58 |
| 27 | 4'-CH3 |  | >100 |
| 28 | 3'-CH3 |  | 44.07 |
| 29 | 3'-NH2-4'-OCH3 |  | 46.11 |
| 30 | 4'-OH |  | 32.48 |
| 31 | 2',4'-F |  | 3.08 |
| 32 | 2',4'-F |  | 0.64 |
| 2a | 3'-NO2-4'-OCH3 |  | 29.25 |
| 2b | 2',4'-F |  | 15.21 |
| Colchicine | / | / | 0.004 |
2.2. In Vitro Cytotoxic Activity
2.3. In Vitro Tubulin Polymerization Inhibitory Activity

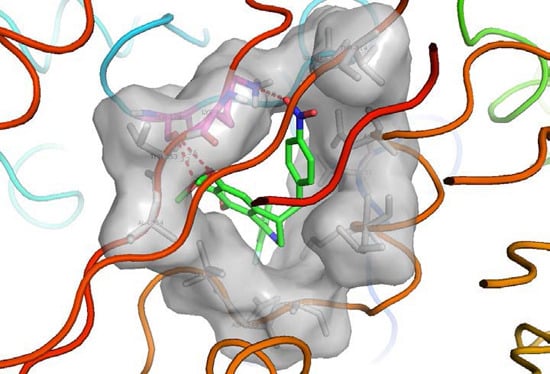
2.4. Molecular Docking Study

3. Experimental Section
3.1. Chemistry
3.2. In Vitro Cytotoxic Activity
3.3. In Vitro Tubulin Polymerization Inhibitory Activity
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Honore, S.; Pasquier, E.; Braguer, D. Understanding microtubule dynamics for improved cancer therapy. Cell. Mol. Life Sci. 2005, 62, 3039–3056. [Google Scholar] [CrossRef] [PubMed]
- Downing, K.H.; Nogales, E. Tublin structure: Insights into microtubule properties and functions. Curr. Opin. Struct. Biol. 1998, 8, 785–791. [Google Scholar] [CrossRef] [PubMed]
- Sorger, P.K.; Dobles, M.; Tournebize, R.; Hyman, A.A. Coupling cell division and cell death to microtuble dynamics. Curr. Opin. Cell Biol. 1997, 9, 807–814. [Google Scholar] [CrossRef] [PubMed]
- Jordan, M.A.; Wilson, L. Microtubles as a target for anticancer drugs. Nat. Rev. Cancer 2004, 4, 253–265. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.W.; Liu, J.; Liu, N.; Shi, D.; Zhou, X.T.; Lv, J.G.; Zhu, J.; Zheng, C.H.; Zhou, Y.J. Imidazolone-amide bridges and their effects on tubulin polymerization in cis-locked vinylogous combretastatin-A4 analogues: Synthesis and biological evaluation. Bioorg. Med. Chem. 2011, 19, 3579–3584. [Google Scholar] [CrossRef] [PubMed]
- Altmann, K.H. Microtubule-stabilizing agents: A growing class of important anticancer drugs. Curr. Opin. Chem. Biol. 2001, 5, 424–431. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.H.; Chen, J.; Liu, J.; Zhou, X.T.; Liu, N.; Shi, D.; Huang, J.J.; Lv, J.G.; Zhu, J.; Zhou, Y.J. Synthesis and biological evaluation of 1-phenyl-1,2,3,4-dihydroisoquinoline compounds as tubulin polymerization Inhibitors. Arch. Pharm. Chem. Life Sci. 2012, 345, 454–462. [Google Scholar] [CrossRef]
- Zhu, W.W.; Song, Y.L.; Zheng, C.H.; Lv, J.G.; Chen, J.; Huang, J.J.; Zhou, Y.J.; Zhu, J. Synthesis, crystal structure and antitumor activity of 6,7,8-trimethoxy-1-(4-methoxy-3-nitrophenyl)-4-(pyridin-4-ylmethyl)-3,4-dihydroisoquinoline. Chin. J. Struct. Chem. 2011, 30, 717–723. [Google Scholar]
- Jetter, M.C.; Youngman, M.A.; McNally, J.J.; McDonnell, M.E.; Zhang, S.P.; Dubin, A.E.; Nasser, N.; Codd, E.E.; Flores, C.M.; Dax, S.L. Heteroaryl β-tetralin ureas as novel antagonists of human TRPV1. Bioorg. Med. Chem. Lett. 2007, 17, 6160–6163. [Google Scholar] [CrossRef] [PubMed]
- Yoon, N.M.; Brown, H.C. Selective reductions. XII. Explorations in some representative applications of aluminum hydride for selective reductions. J. Am. Chem. Soc. 1968, 90, 2927–2938. [Google Scholar] [CrossRef]
- Shan, Y.; Zhang, J.; Liu, Z.; Wang, M.; Dong, Y. Developments of combretastatin A-4 derivatives as anticancer agents. Curr. Med. Chem. 2011, 18, 523–538. [Google Scholar] [CrossRef] [PubMed]
- Bailly, C.; Bal, C.; Barbier, P.; Douillard, S.; McLeer-Florin, A.; Bourgarel-Rey, V.; Pierson, J.T.; Fedorov, A.Y.; Finet, J.P.; Boutonnat, J.; et al. Synthesis and biological evaluation of 4-arylcoumarin analogues of combretastatins. J. Med. Chem. 2003, 46, 5437–5444. [Google Scholar] [CrossRef] [PubMed]
- Hamel, E. Evaluation of antimiotic agents by quantitative comparisons of their effects on the polymerization of purified tubulin. Cell Biochem. Biophys. 2003, 38, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Peng, Y.Y.; Wang, X.I.; Keenan, S.M.; Arora, S.; Welsh, W.J. Highly potent triazole-based tubulin polymerization inhibitors. J. Med. Chem. 2007, 50, 749–754. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, L.; Song, Y.; Huang, J.; Liu, J.; Zhu, W.; Zhou, Y.; Lv, J.; Zheng, C.; Zhu, J. Design, Synthesis and Biological Evaluation of 1,4-Disubstituted-3,4-dihydroisoquinoline Compounds as New Tubulin Polymerization Inhibitors. Int. J. Mol. Sci. 2015, 16, 10173-10184. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms160510173
Zhang L, Song Y, Huang J, Liu J, Zhu W, Zhou Y, Lv J, Zheng C, Zhu J. Design, Synthesis and Biological Evaluation of 1,4-Disubstituted-3,4-dihydroisoquinoline Compounds as New Tubulin Polymerization Inhibitors. International Journal of Molecular Sciences. 2015; 16(5):10173-10184. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms160510173
Chicago/Turabian StyleZhang, Ling, Yunlong Song, Jingjing Huang, Jia Liu, Wenwen Zhu, Youjun Zhou, Jiaguo Lv, Canhui Zheng, and Ju Zhu. 2015. "Design, Synthesis and Biological Evaluation of 1,4-Disubstituted-3,4-dihydroisoquinoline Compounds as New Tubulin Polymerization Inhibitors" International Journal of Molecular Sciences 16, no. 5: 10173-10184. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms160510173