Programmed Death Ligand 1 (PD-L1) Tumor Expression Is Associated with a Better Prognosis and Diabetic Disease in Triple Negative Breast Cancer Patients
Abstract
:1. Introduction
2. Results
2.1. Clinical-Pathological Characteristics and Follow Up Data of Triple Negative Breast Cancers (TNBC) Patients
2.2. Association of the PD-L1 Expression with Clinical-Pathological Data
2.3. PD-L1 Relation with Metabolic Features of TNBC Patients
2.4. PD-L1 Expression Association with Survival of TNBC Patients
3. Discussion
4. Materials and Methods
4.1. Patients and Specimens
4.2. TMA Building
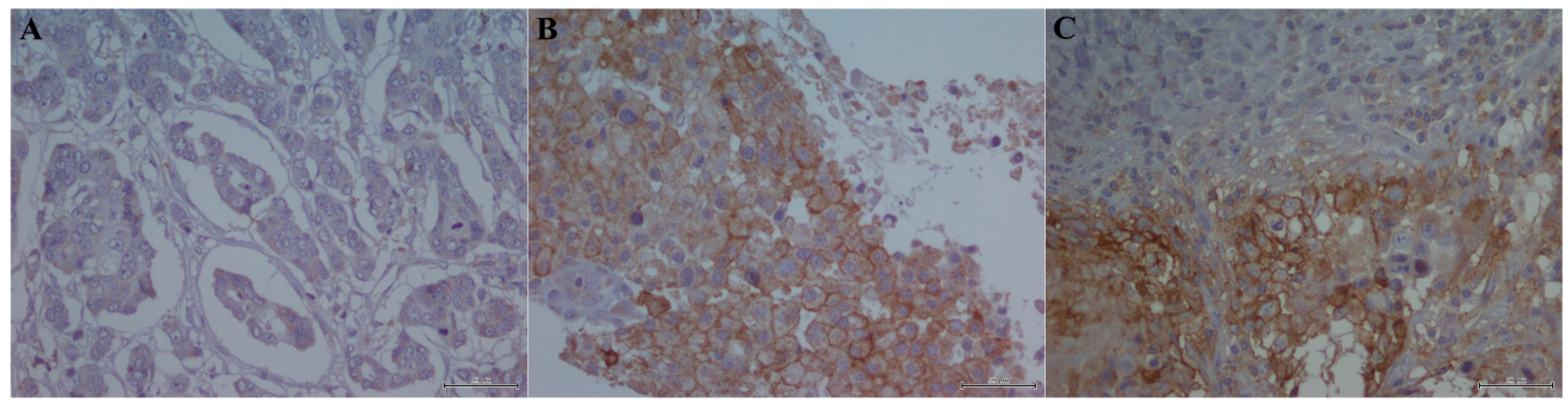
4.3. Immunohistochemistry Analysis
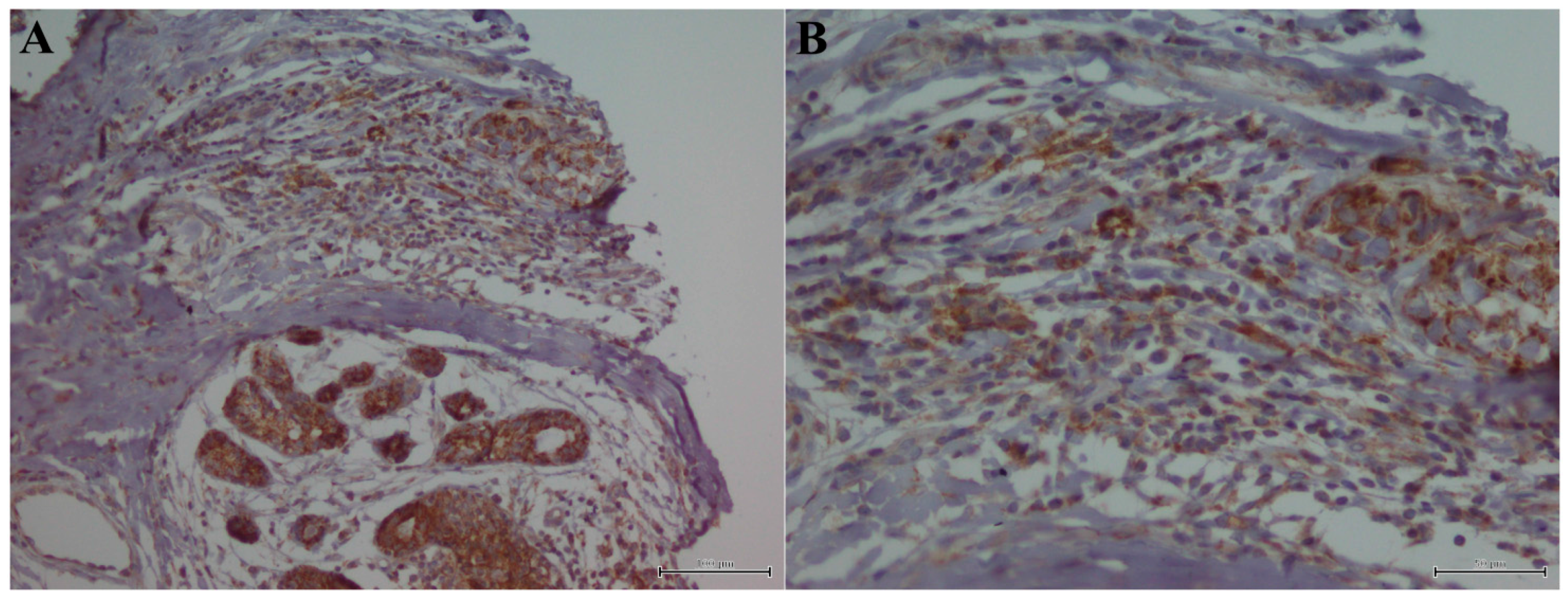
4.4. Evaluation of Immunohistochemistry on TIL and Tumor Cells
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Elsamany, S.; Abdullah, S. Triple-negative breast cancer: Future prospects in diagnosis and management. Med. Oncol. 2014, 31, 834. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, B.D.; Bauer, J.A.; Chen, X.; Sanders, M.E.; Chakravarthy, A.B.; Shyr, Y.; Pietenpol, J.A. Identification of human triple-negative breast cancer subtypes and preclinical models for selection of targeted therapies. J. Clin. Investig. 2011, 121, 2750–2767. [Google Scholar] [CrossRef] [PubMed]
- Le Du, F.; Eckhardt, B.L.; Lim, B.; Litton, J.K.; Moulder, S.; Meric-Bernstam, F.; Gonzalez-Angulo, A.M.; Ueno, N.T. Is the future of personalized therapy in triple-negative breast cancer based on molecular subtype? Oncotarget 2015, 6, 12890–12908. [Google Scholar] [PubMed]
- Butte, M.J.; Peña-Cruz, V.; Kim, M.J.; Freeman, G.J.; Sharpe, A.H. Interaction of human PD-L1 and B7-1. Mol. Immunol. 2008, 45, 3567–3572. [Google Scholar] [CrossRef] [PubMed]
- Ilie, M.; Hofman, V.; Dietel, M.; Soria, J.; Hofman, P. Assessment of the PD-L1 status by immunohistochemistry: Challenges and perspectives for therapeutic strategies in lung cancer patients. Virchows Arch. 2016, 468, 511–525. [Google Scholar] [CrossRef] [PubMed]
- Muenst, S.; Schaerli, A.R.; Gao, F.; Däster, S.; Trella, E.; Droeser, R.A.; Muraro, M.G.; Zajac, P.; Zanetti, R.; Gillanders, W.E.; et al. Expression of programmed death ligand 1 (PD-L1) is associated with poor prognosis in human breast cancer. Breast Cancer Res. Treat. 2014, 146, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Mittendorf, E.A.; Philips, A.V.; Meric-Bernstam, F.; Qiao, N.; Wu, Y.; Harrington, S.; Su, X.; Wang, Y.; Gonzalez-Angulo, A.M.; Akcakanat, A.; et al. PD-L1 expression in triple-negative breast cancer. Cancer Immunol. Res. 2014, 2, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wetherilt, C.S.; Krishnamurti, U.; Yang, J.; Ma, Y.; Styblo, T.M.; Meisel, J.L.; Peng, L.; Siddiqui, M.T.; Cohen, C.; et al. Stromal PD-L1 expression is associated with better disease-free survival in triple-negative breast cancer. Am. J. Clin. Pathol. 2016, 146, 496–502. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.Y.; Lee, Y.K.; Koo, J.S. Expression of PD-L1 in triple-negative breast cancer based on different immunohistochemical antibodies. Transl. Med. 2016, 14, 173. [Google Scholar] [CrossRef] [PubMed]
- Cimino-Mathews, A.; Thompson, E.; Taube, J.M.; Ye, X.; Lu, Y.; Meeker, A.; Xu, H.; Sharma, R.; Lecksell, K.; Cornish, T.C.; et al. PD-L1 (B7-H1) expression and the immune tumor microenvironment in primary and metastatic breast carcinomas. Hum. Pathol. 2016, 47, 52–63. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, F.R.; McElhinny, A.; Stanforth, D.; Ranger-Moore, J.; Jansson, M.; Kulangara, K.; Richardson, W.; Towne, P.; Hanks, D.; Vennapusa, B.; et al. PD-L1 immunohistochemistry assays for lung cancer: Results from Phase 1 of the “blueprint PD-L1 IHC assay comparison project”. J. Thorac. Oncol. 2017, 12, 208–222. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Li, J.; Gray, W.H.; Lehmann, B.D.; Bauer, J.A.; Shyr, Y.; Pietenpol, J.A. TNBC type: A subtyping tool for triple-negative breast cancer. Cancer Inform. 2012, 11, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Burstein, H.J. Patients with triple negative breast cancer: Is there an optimal adjuvant treatment? Breast 2013, 22, 147–148. [Google Scholar] [CrossRef] [PubMed]
- Topalian, S.; Drake, C.G.; Pardoll, D.M. Targeting the PD-1/B7-H1 (PD-L1) pathway to activate anti-tumor immunity. Curr. Opin. Immunol. 2012, 24, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Hamanishi, J.; Mandai, M.; Matsumura, N.; Abiko, K.; Baba, T.; Konishi, I. PD-1/PD-L1 blockade in cancer treatment: Perspectives and issues. Int. J. Clin. Oncol. 2016, 21, 462–473. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Huang, Z.; Teng, F.; Xing, L.; Yu, J. Predictive biomarkers in PD-1/PD-L1 checkpoint blockade immunotherapy. Cancer Treat. Rev. 2015, 41, 868–876. [Google Scholar] [CrossRef] [PubMed]
- Soliman, H.; Khalil, F.; Antonia, S. PD-L1 expression is increased in a subset of basal type breast cancer cells. PLoS ONE 2014, 9, e88557. [Google Scholar] [CrossRef] [PubMed]
- Sabatier, R.; Finetti, P.; Mamessier, E.; Adelaide, J.; Chaffanet, M.; Ali, H.R.; Viens, P.; Caldas, C.; Birnbaum, D.; Bertucci, F. Prognostic and predictive value of PDL1 expression in breast cancer. Oncotarget 2015, 6, 5449–5464. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.R.; Ha, S.J.; Hong, M.H.; Heo, S.J.; Koh, Y.W.; Choi, E.C.; Kim, E.K.; Pyo, K.H.; Jung, I.; Seo, D.; et al. PD-L1 expression on immune cells, but not on tumor cells, is a favorable prognostic factor for head and neck cancer patients. Sci. Rep. 2016, 6, 36956. [Google Scholar] [CrossRef] [PubMed]
- Schalper, K.A.; Velcheti, V.; Carvajal, D.; Wimberly, H.; Brown, J.; Pusztai, L.; Rimm, D.L. In situ tumor PD-L1 mRNA expression is associated with increased TILs and better outcome in breast carcinomas. Clin. Cancer Res. 2014, 20, 2773–2782. [Google Scholar] [CrossRef] [PubMed]
- De Pergola, G.; Silvestris, F. Obesity as a major risk factor for cancer. J. Obes. 2013, 2013, 291546. [Google Scholar] [CrossRef] [PubMed]
- Garg, S.K.; Maurer, H.; Reed, K.; Selagamsetty, R. Diabetes and cancer: Two diseases with obesity as a common risk factor. Diabetes Obes. Metab. 2014, 16, 97–110. [Google Scholar] [CrossRef] [PubMed]
- Crispo, A.; Augustin, L.S.; Grimaldi, M.; Nocerino, F.; Giudice, A.; Cavalcanti, E.; di Bonito, M.; Botti, G.; de Laurentiis, M.; Rinaldo, M.; et al. Risk differences between prediabetes and diabetes according to breast cancer molecular subtypes. J. Cell Physiol. 2016, 232, 1144–1150. [Google Scholar] [CrossRef] [PubMed]
- Reeves, K.W.; McLaughlin, V.; Fredman, L.; Ensrud, K.; Cauley, J.A. Components of metabolic syndrome and risk of breast cancer by prognostic features in the study of osteoporotic fractures cohort. Cancer Cause Control. 2012, 23, 1241–1251. [Google Scholar] [CrossRef] [PubMed]
- Camacho, L.; Dasgupta, A.; Jiralerspong, S. Metformin in breast cancer—An evolving mystery. Breast Cancer Res. 2015, 17, 88. [Google Scholar] [CrossRef] [PubMed]
- Zeng, L.; Zielinska, H.A.; Arshad, A.; Shield, J.P.; Bahl, A.; Holly, J.M.; Perks, C.M. Hyperglycaemia-induced chemoresistance in breast cancer cells: Role of the estrogen receptor. Endocr. Relat. Cancer 2016, 23, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Scognamiglio, G.; Cantile, M.; Scala, S.; Cecere, S.; Russo, F.; Collina, F.; Marra, L.; Sabbatino, F.; Botti, G.; Franco, R. Tissue micro arrays for immunohistochemical detection of inflammatory infiltrates in renal cell carcinoma. Int. J. Clin. Exp. Pathol. 2014, 7, 1175–1179. [Google Scholar]
- Scognamiglio, G.; de Chiara, A.; di Bonito, M.; Tatangelo, F.; Losito, N.S.; Anniciello, A.; de Cecio, R.; D’Alterio, C.; Scala, S.; Cantile, M.; et al. Variability in immunohistochemical detection of programmed death ligand 1 (PD-L1) in cancer tissue types. Int. J. Mol. Sci. 2016, 17, 790. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Fan, Y.; Lang, R.; Gu, F.; Chen, L.; Cui, L.; Pringle, G.A.; Zhang, X.; Fu, L. Tumor infiltrating lymphocytes differ in invasive micropapillary carcinoma and medullary carcinoma of breast. Mod. Pathol. 2008, 21, 1101–1107. [Google Scholar] [CrossRef] [PubMed]
- Collina, F.; Cerrone, M.; Peluso, V.; Laurentiis, M.D.; Caputo, R.; Cecio, R.D.; Liguori, G.; Botti, G.; Cantile, M.; di Bonito, M. Downregulation of androgen receptor is strongly associated with diabetes in triple negative breast cancer patients. Am. J. Transl. Res. 2016, 8, 3530–3539. [Google Scholar] [PubMed]

| Clinical Pathological Features | TUMOR PD-L1 | p-Value | R Pearson | |
|---|---|---|---|---|
| Low | High | |||
| Age | ||||
| <40 | 15 (60%) | 10 (40%) | 0.129 | −0.118 |
| ≥40 and ≤60 | 56 (58.3%) | 40 (41.7%) | ||
| >60 | 69 (71.9%) | 27 (28.1%) | ||
| Histotype | ||||
| Ductal | 120 (62.9) | 71 (37.1) | 0.103 | −0.110 |
| Non Ductal | 22 (78.6) | 6 (21.4) | ||
| Menopausal State | ||||
| Pre | 41 (52.6%) | 37 (47.4) | 0.006 * | −0.187 |
| Post | 99 (71.2%) | 40 (28.8%) | ||
| Tumor Size (cm) | ||||
| ≤2 | 63 (64.9%) | 34 (35.1%) | 0.769 | −0.018 |
| >2 and ≤5 | 60 (63.2%) | 35 (36.8%) | ||
| >5 | 13 (72.2%) | 5 (27.8%) | ||
| LNM | ||||
| Negative | 61 (60.4%) | 40 (39.6%) | 0.026 * | −0.168 |
| Positive | 58 (76.3%) | 18 (23.7%) | ||
| Metastases | ||||
| Negative | 82 (66.7%) | 41 (33.3%) | 0.079 | −0.138 |
| Positive | 31 (81.6%) | 7 (18.4%) | ||
| Grade | ||||
| G1 | 2 (100%) | 0 (0%) | 0.494 | 0.068 |
| G2 | 15 (68.2%) | 7 (31.8%) | ||
| G3 | 116 (62.7%) | 69 (37.3%) | ||
| Ki-67 | ||||
| Low | 31 (83.8%) | 6 (16.2%) | 0.009 * | 0.180 |
| High | 107 (61.1%) | 68 (38.9%) | ||
| BMI | ||||
| <30 | 48 (73.8%) | 17 (26.2%) | 0.962 | −0.005 |
| ≥30 | 26 (74.3%) | 9 (25.7%) | ||
| Diabetes | ||||
| No | 42 (82.4%) | 9 (17.6%) | 0.034 * | 0.248 |
| Yes | 13 (59.1%) | 9 (40.9) | ||
| Glicemya | ||||
| <110 | 43 (82.7%) | 9 (17.3%) | 0.043 * | 0.244 |
| >110 | 10 (58.8%) | 7 (41.2%) | ||
| Status | ||||
| Alive | 66 (66.7%) | 33 (32.3%) | 1 | 0.000 |
| Dead | 10 (66.7%) | 5 (32.3%) | ||
| Relapse | ||||
| No | 47 (61.8%) | 29 (38.2) | 0.008 * | −0.238 |
| Yes | 42 (84%) | 8 (16%) | ||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Botti, G.; Collina, F.; Scognamiglio, G.; Rao, F.; Peluso, V.; De Cecio, R.; Piezzo, M.; Landi, G.; De Laurentiis, M.; Cantile, M.; et al. Programmed Death Ligand 1 (PD-L1) Tumor Expression Is Associated with a Better Prognosis and Diabetic Disease in Triple Negative Breast Cancer Patients. Int. J. Mol. Sci. 2017, 18, 459. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms18020459
Botti G, Collina F, Scognamiglio G, Rao F, Peluso V, De Cecio R, Piezzo M, Landi G, De Laurentiis M, Cantile M, et al. Programmed Death Ligand 1 (PD-L1) Tumor Expression Is Associated with a Better Prognosis and Diabetic Disease in Triple Negative Breast Cancer Patients. International Journal of Molecular Sciences. 2017; 18(2):459. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms18020459
Chicago/Turabian StyleBotti, Gerardo, Francesca Collina, Giosuè Scognamiglio, Federica Rao, Valentina Peluso, Rossella De Cecio, Michela Piezzo, Gabriella Landi, Michelino De Laurentiis, Monica Cantile, and et al. 2017. "Programmed Death Ligand 1 (PD-L1) Tumor Expression Is Associated with a Better Prognosis and Diabetic Disease in Triple Negative Breast Cancer Patients" International Journal of Molecular Sciences 18, no. 2: 459. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms18020459






