A Molecular Perspective on the Potential Benefits of Metformin for the Treatment of Inflammatory Skin Disorders
Abstract
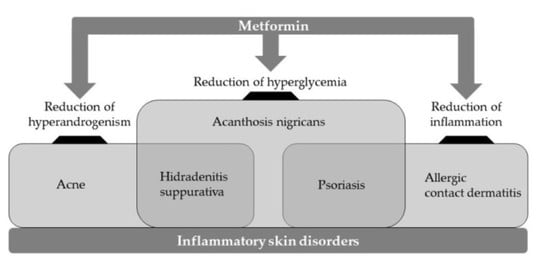
:1. Introduction
2. Potential Molecular Mechanisms of Action for Metformin
3. Inflammatory Skin Disorders
4. Psoriasis
5. Acanthosis Nigricans
6. Acne
7. Hidradenitis Suppurativa
8. Allergic Contact Dermatitis
9. Conclusion and Future Perspectives
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AMPK | 5′-adenosine monophosphate-activated protein kinase |
| GLP-1 | glucagon-like peptide-1 |
| DPP-4 | dipeptidyl peptidase-4 |
| mTOR | mammalian target of the rapamycin |
| GLUT4 | glucose transporter type 4 |
| PCOS | polycystic ovary syndrome |
| IGF-1 | insulin-like growth factor-1 |
| HS | hidradenitis suppurativa |
| ACD | allergic contact dermatitis |
| NLRP3 | NOD-like receptor pyrin domain-containing protein 3 |
| NF-κB | nuclear factor kappa-light-chain-enhancer of activated B cells |
| 11β-HSD1 | 11β-hydroxysteroid dehydrogenase type 1 |
| PARP-1 | poly [ADP-ribose] polymerase 1 |
| SIRT1 | NAD-dependent deacetylase sirtuin-1 |
References
- He, L. Metformin and Systemic Metabolism. Trends Pharmacol. Sci. 2020, 41, 868–881. [Google Scholar] [CrossRef]
- Viollet, B.; Guigas, B.; Sanz Garcia, N.; Leclerc, J.; Foretz, M.; Andreelli, F. Cellular and molecular mechanisms of metformin: An overview. Clin. Sci. 2012, 122, 253–270. [Google Scholar] [CrossRef] [Green Version]
- Deng, M.; Su, D.; Xu, S.; Little, P.J.; Feng, X.; Tang, L.; Shen, A. Metformin and Vascular Diseases: A Focused Review on Smooth Muscle Cell Function. Front. Pharmacol. 2020, 11, 635. [Google Scholar] [CrossRef] [PubMed]
- Solymár, M.; Ivic, I.; Pótó, L.; Hegyi, P.; Garami, A.; Hartmann, P.; Pétervári, E.; Czopf, L.; Hussain, A.; Gyöngyi, Z.; et al. Metformin induces significant reduction of body weight, total cholesterol and LDL levels in the elderly—A meta-analysis. PLoS ONE 2018, 13. [Google Scholar] [CrossRef] [PubMed]
- Del Barco, S.; Vazquez-Martin, A.; Cufí, S.; Oliveras-Ferraros, C.; Bosch-Barrera, J.; Joven, J.; Martin-Castillo, B.; Menendez, J.A. Metformin: Multi-faceted protection against cancer. Oncotarget 2011, 2, 896–917. [Google Scholar] [CrossRef] [Green Version]
- Bo, S.; Benso, A.; Durazzo, M.; Ghigo, E. Does use ofmetformin protect against cancer in type 2 diabetesmellitus? J. Endocrinol. Investig. 2012, 35, 231–235. [Google Scholar] [CrossRef] [PubMed]
- Theut Riis, P.; Thorlacius, L.R.; Jemec, G.B. Investigational drugs in clinical trials for Hidradenitis Suppurativa. Expert Opin. Investig. Drugs 2018, 27, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Patel, N.U.; Roach, C.; Alinia, H.; Huang, W.W.; Feldman, S.R. Current treatment options for acanthosis nigricans. Clin. Cosmet. Investig. Dermatol. 2018, 11, 407–413. [Google Scholar] [CrossRef] [Green Version]
- Robinson, S.; Kwan, Z.; Tang, M.M. Metformin as an adjunct therapy for the treatment of moderate to severe acne vulgaris: A randomized open-labeled study. Dermatol. Ther. 2019, 32. [Google Scholar] [CrossRef]
- Chung, M.M.; Nicol, C.J.; Cheng, Y.C.; Lin, K.H.; Chen, Y.L.; Pei, D.; Lin, C.H.; Shih, Y.N.; Yen, C.H.; Chen, S.J.; et al. Metformin activation of AMPK suppresses AGE-induced inflammatory response in hNSCs. Exp. Cell Res. 2017, 352, 75–83. [Google Scholar] [CrossRef]
- Zhou, Z.; Tang, Y.; Jin, X.; Chen, C.; Lu, Y.; Liu, L.; Shen, C. Metformin Inhibits Advanced Glycation End Products-Induced Inflammatory Response in Murine Macrophages Partly through AMPK Activation and RAGE/NF B Pathway Suppression. J. Diabetes Res. 2016, 2016. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, S.B.; Park, J.S.; Jung, W.H.; Kim, H.Y.; Kwak, H.J.; Ahn, J.H.; Choi, K.J.; Na, Y.J.; Choi, S.; Dal Rhee, S.; et al. Anti-inflammatory effect of a selective 11β-hydroxysteroid dehydrogenase type 1 inhibitor via the stimulation of heme oxygenase-1 in LPS-activated mice and J774.1 murine macrophages. J. Pharmacol. Sci. 2016, 131, 241–250. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Esteves, C.L.; Kelly, V.; Breton, A.; Taylor, A.I.; West, C.C.; Donadeu, F.X.; Péault, B.; Seckl, J.R.; Chapman, K.E. Proinflammatory cytokine induction of 11β-hydroxysteroid dehydrogenase type 1 (11β-HSD1) in human adipocytes is mediated by MEK, C/EBPβ, and NF-κB/RelA. J. Clin. Endocrinol. Metab. 2014, 99. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Itoi, S.; Terao, M.; Murota, H.; Katayama, I. 11β-Hydroxysteroid dehydrogenase 1 contributes to the pro-inflammatory response of keratinocytes. Biochem. Biophys. Res. Commun. 2013, 440, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Hassa, P.O.; Hottiger, M.O. The functional role of poly(ADP-ribose)polymerase 1 as novel coactivator of NF-κB in inflammatory disorders. Cell. Mol. Life Sci. 2002, 59, 1534–1553. [Google Scholar] [CrossRef]
- Nie, Y.; Nirujogi, T.S.; Ranjan, R.; Reader, B.F.; Chung, S.; Ballinger, M.N.; Englert, J.A.; Christman, J.W.; Karpurapu, M. PolyADP-Ribosylation of NFATc3 and NF-κB Transcription Factors Modulate Macrophage Inflammatory Gene Expression in LPS-Induced Acute Lung Injury. J. Innate Immun. 2020, 1–11. [Google Scholar] [CrossRef]
- Zheng, Z.; Chen, H.; Li, J.; Li, T.; Zheng, B.; Zheng, Y.; Jin, H.; He, Y.; Gu, Q.; Xu, N. Sirtuin 1-mediated cellular metabolic memory of high glucose via the LKB1/AMPK/ROS pathway and therapeutic effects of metformin. Diabetes 2012, 61, 217–228. [Google Scholar] [CrossRef] [Green Version]
- Calixto, M.C.; Lintomen, L.; André, D.M.; Leiria, L.O.; Ferreira, D.; Lellis-Santos, C.; Anhê, G.F.; Bordin, S.; Landgraf, R.G.; Antunes, E. Metformin Attenuates the Exacerbation of the Allergic Eosinophilic Inflammation in High Fat-Diet-Induced Obesity in Mice. PLoS ONE 2013, 8. [Google Scholar] [CrossRef]
- Rogliani, P.; Ora, J.; Di Daniele, N.; Lauro, D. Pleiotropic effects of hypoglycemic agents: Implications in asthma and COPD. Curr. Opin. Pharmacol. 2018, 40, 34–38. [Google Scholar] [CrossRef]
- Yazdi, A.S.; Röcken, M.; Ghoreschi, K. Cutaneous immunology: Basics and new concepts. Semin. Immunopathol. 2016, 38, 3–10. [Google Scholar] [CrossRef]
- Bonilla, F.A.; Oettgen, H.C. Adaptive immunity. J. Allergy Clin. Immunol. 2010, 125. [Google Scholar] [CrossRef] [PubMed]
- Nutman, T.B. Looking beyond the induction of Th2 responses to explain immunomodulation by helminths. Parasite Immunol. 2015, 37, 304–313. [Google Scholar] [CrossRef] [PubMed]
- Pallikkuth, S.; Parmigiani, A.; Pahwa, S. Role of IL-21 and IL-21 receptor on B cells in HIV infection. Crit. Rev. Immunol. 2012, 32, 173–195. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jarnicki, A.G.; Lysaght, J.; Todryk, S.; Mills, K.H.G. Suppression of Antitumor Immunity by IL-10 and TGF-β-Producing T Cells Infiltrating the Growing Tumor: Influence of Tumor Environment on the Induction of CD4 + and CD8 + Regulatory T Cells. J. Immunol. 2006, 177, 896–904. [Google Scholar] [CrossRef] [Green Version]
- Sun, Z.; Unutmaz, D.; Zou, Y.R.; Sunshine, M.J.; Pierani, A.; Brenner-Morton, S.; Mebius, R.E.; Littman, D.R. Requirement for RORγ in thymocyte survival and lymphoid organ development. Science 2000, 288, 2369–2373. [Google Scholar] [CrossRef] [Green Version]
- Gatzka, M.; Scharffetter-Kochanek, K. T-cell plasticity in inflammatory skin diseases-the good, the bad, and the chameleons. JDDG J. Ger. Soc. Dermatol. 2015, 13, 647–651. [Google Scholar] [CrossRef]
- Gelfand, J.M.; Troxel, A.B.; Lewis, J.D.; Kurd, S.K.; Shin, D.B.; Wang, X.; Margolis, D.J.; Strom, B.L. The risk of mortality in patients with psoriasis: Results from a population-based study. Arch. Dermatol. 2007, 143, 1493–1499. [Google Scholar] [CrossRef]
- Kolli, S.S.; Kepley, A.L.; Cline, A.; Feldman, S.R. A safety review of recent advancements in the treatment of psoriasis: Analysis of clinical trial safety data. Expert Opin. Drug Saf. 2019, 18, 523–536. [Google Scholar] [CrossRef]
- Menter, A.; Strober, B.E.; Kaplan, D.H.; Kivelevitch, D.; Prater, E.F.; Stoff, B.; Armstrong, A.W.; Connor, C.; Cordoro, K.M.; Davis, D.M.R.; et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with biologics. J. Am. Acad. Dermatol. 2019, 80, 1029–1072. [Google Scholar] [CrossRef] [Green Version]
- Hansel, K.; Zangrilli, A.; Bianchi, L.; Peris, K.; Chiricozzi, A.; Offidani, A.; Diotallevi, F.; Fargnoli, M.C.; Esposito, M.; Amerio, P.; et al. A multicenter study on effectiveness and safety of risankizumab in psoriasis: An Italian 16-week real-life experience during the COVID-19 pandemic. J. Eur. Acad. Dermatol. Venereol. 2020. [Google Scholar] [CrossRef]
- Havnaer, A.; Weinberg, J.M.; Han, G. Systemic therapies in psoriasis: An update on newly approved and pipeline biologics and oral treatments. Cutis 2019, 104, 17–20. [Google Scholar] [PubMed]
- Hansel, K.; Bianchi, L.; Lanza, F.; Bini, V.; Stingeni, L. Adalimumab dose tapering in psoriasis: Predictive factors for maintenance of complete clearance. Acta Derm. Venereol. 2017, 97, 346–350. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Armstrong, A.W.; Harskamp, C.T.; Armstrong, E.J. Psoriasis and the risk of diabetes mellitus: A systematic review and meta-analysis. JAMA Dermatol. 2013, 149, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Chi, C.C.; Lee, C.Y.; Liu, C.Y.; Wang, S.H.; Tien O’Donnell, F.; Tung, T.H. Effects of antidiabetic drugs on psoriasis: A meta-analysis. Eur. J. Clin. Investig. 2020. [Google Scholar] [CrossRef]
- Nauck, M.A.; Quast, D.R.; Wefers, J.; Meier, J.J. GLP-1 receptor agonists in the treatment of type 2 diabetes–state-of-the-art. Mol. Metab. 2020, 101102. [Google Scholar] [CrossRef]
- Ba, W.; Xu, Y.; Yin, G.; Yang, J.; Wang, R.; Chi, S.; Wang, Y.; Li, C. Metformin inhibits pro-inflammatory responses via targeting nuclear factor-κB in HaCaT cells. Cell Biochem. Funct. 2019, 37, 4–10. [Google Scholar] [CrossRef] [Green Version]
- Su, Y.J.; Chen, T.H.; Hsu, C.Y.; Chiu, W.T.; Lin, Y.S.; Chi, C.C. Safety of Metformin in Psoriasis Patients with Diabetes Mellitus: A 17-Year Population-Based Real-World Cohort Study. J. Clin. Endocrinol. Metab. 2019, 104, 3279–3286. [Google Scholar] [CrossRef]
- Chang, G.; Wang, J.; Song, J.; Zhang, Z.; Zhang, L. Efficacy and safety of pioglitazone for treatment of plaque psoriasis: A systematic review and meta-analysis of randomized controlled trials. J. Dermatolog. Treat. 2020, 31, 680–686. [Google Scholar] [CrossRef]
- Chen, P.; Chen, X.; Lei, L.; Zhang, Y.; Xiang, J.; Zhou, J.; Lv, J. The efficacy and safety of pioglitazone in psoriasis vulgaris: A meta-analysis of randomized controlled trials. Medicine 2020, 99, e21549. [Google Scholar] [CrossRef]
- Lynch, M.; Ahern, T.B.; Timoney, I.; Sweeney, C.; Kelly, G.; Hughes, R.; Tobin, A.M.; O’Shea, D.; Kirby, B. Dipeptidyl peptidase-4 inhibition and narrow-band ultraviolet-B light in psoriasis (DINUP): Study protocol for a randomised controlled trial. Trials 2016, 17. [Google Scholar] [CrossRef] [Green Version]
- Wu, C.Y.; Shieh, J.J.; Shen, J.L.; Liu, Y.Y.; Chang, Y.T.; Chen, Y.J. Association between antidiabetic drugs and psoriasis risk in diabetic patients: Results from a nationwide nested case-control study in Taiwan. J. Am. Acad. Dermatol. 2015, 72, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Bhansali, A. Randomized placebo control study of insulin sensitizers (Metformin and Pioglitazone) in psoriasis patients with metabolic syndrome (Topical Treatment Cohort). BMC Dermatol. 2016, 16. [Google Scholar] [CrossRef] [Green Version]
- Sag, D.; Carling, D.; Stout, R.D.; Suttles, J. Adenosine 5′-Monophosphate-Activated Protein Kinase Promotes Macrophage Polarization to an Anti-Inflammatory Functional Phenotype. J. Immunol. 2008, 181, 8633–8641. [Google Scholar] [CrossRef]
- Tsuji, G.; Hashimoto-Hachiya, A.; Yen, V.H.; Takemura, M.; Yumine, A.; Furue, K.; Furue, M.; Nakahara, T. Metformin inhibits IL-1β secretion via impairment of NLRP3 inflammasome in keratinocytes: Implications for preventing the development of psoriasis. Cell Death Discov. 2020, 6, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, W.; Ma, W.; Zhong, H.; Liu, W.; Sun, Q. Metformin inhibits proliferation of human keratinocytes through a mechanism associated with activation of the MAPK signaling pathway. Exp. Ther. Med. 2014, 7, 389–392. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; Yang, F.; Ma, W.; Sun, Q. Metformin inhibits proliferation and proinflammatory cytokines of human keratinocytes in vitro via mTOR-signaling pathway. Pharm. Biol. 2016, 54, 1173–1178. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, Y.; Tian, T.; Gao, J.; Liu, X.; Hou, H.; Cao, R.; Li, B.; Quan, M.; Guo, L. Metformin ameliorates the development of experimental autoimmune encephalomyelitis by regulating T helper 17 and regulatory T cells in mice. J. Neuroimmunol. 2016, 292, 58–67. [Google Scholar] [CrossRef]
- Demirbaş, A.; Faruk Elmas, Ö.; Kutlu, Ö.; Kilitçi, A.; Atasoy, M. Utility of dermatoscopy in the diagnosis of acanthosis nigricans. J. Cosmet. Dermatol. 2020. [Google Scholar] [CrossRef]
- Das, A.; Datta, D.; Kassir, M.; Wollina, U.; Galadari, H.; Lotti, T.; Jafferany, M.; Grabbe, S.; Goldust, M. Acanthosis nigricans: A review. J. Cosmet. Dermatol. 2020, 19, 1857–1865. [Google Scholar] [CrossRef]
- Bomar, L.; Lewallen, R.; Jorizzo, J. Localized acanthosis nigricans at the site of repetitive insulin injections. Cutis 2020, 105, E20–E22. [Google Scholar]
- Álvarez-Villalobos, N.A.; Rodríguez-Gutiérrez, R.; González-Saldivar, G.; Sánchez-García, A.; Gómez-Flores, M.; Quintanilla-Sánchez, C.; Treviño-Álvarez, A.M.; Mancillas-Adame, L.G.; González-González, J.G. Acanthosis nigricans in middle-age adults: A highly prevalent and specific clinical sign of insulin resistance. Int. J. Clin. Pract. 2020, 74. [Google Scholar] [CrossRef] [PubMed]
- Videira-Silva, A.; Albuquerque, C.; Fonseca, H. Acanthosis nigricans as a clinical marker of insulin resistance among overweight adolescents. Ann. Pediatr. Endocrinol. Metab. 2019, 24, 99–103. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Garabadu, D.; Krishnamurthy, S. Metformin attenuates hepatic insulin resistance in type-2 diabetic rats through PI3K/Akt/GLUT-4 signalling independent to bicuculline-sensitive GABAA receptor stimulation. Pharm. Biol. 2017, 55, 722–728. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dash, S.; Sano, H.; Rochford, J.J.; Semple, R.K.; Yeo, G.; Hyden, C.S.S.; Soos, M.A.; Clark, J.; Rodin, A.; Langenberg, C.; et al. A truncation mutation in TBC1D4 in a family with acanthosis nigricans and postprandial hyperinsulinemia. Proc. Natl. Acad. Sci. USA 2009, 106, 9350–9355. [Google Scholar] [CrossRef] [Green Version]
- Mourelatou, R.; Kostopoulou, E.; Rojas-Gil, A.P.; Kehagias, I.; Linos, D.; Kalfarentzos, F.E.; Spiliotis, B.E. Decreased adipocyte glucose transporter 4 (GLUT4) and aquaglyceroporin-7 (AQP7) in adults with morbid obesity: Possible early markers of metabolic dysfunction. Hormones 2019, 18, 297–306. [Google Scholar] [CrossRef]
- Lee, J.O.; Lee, S.K.; Kim, J.H.; Kim, N.; You, G.Y.; Moon, J.W.; Kim, S.J.; Park, S.H.; Kim, H.S. Metformin regulates glucose transporter 4 (GLUT4) translocation through AMP-activated protein kinase (AMPK)-mediated Cbl/CAP signaling in 3T3-L1 preadipocyte cells. J. Biol. Chem. 2012, 287, 44121–44129. [Google Scholar] [CrossRef] [Green Version]
- Hermanns-Lê, T.; Hermanns, J.F.; Piérard, G.E. Juvenile acanthosis nigricans and insulin resistance. Pediatr. Dermatol. 2002, 19, 12–14. [Google Scholar] [CrossRef]
- Bubna, A. Metformin—For the dermatologist. Indian J. Pharmacol. 2016, 48, 4–10. [Google Scholar] [CrossRef] [Green Version]
- Badr, D.; Kurban, M.; Abbas, O. Metformin in dermatology: An overview. J. Eur. Acad. Dermatol. Venereol. 2013, 27, 1329–1335. [Google Scholar] [CrossRef]
- Sharma, S.; Mathur, D.K.; Paliwal, V.; Bhargava, P. Efficacy of metformin in the treatment of acne in women with polycystic ovarian syndrome: A newer approach to acne therapy. J. Clin. Aesthet. Dermatol. 2019, 12, 34–38. [Google Scholar]
- Vora, S.; Ovhal, A.; Jerajani, H.; Nair, N.; Chakrabortty, A. Correlation of facial sebum to serum insulin-like growth factor-1 in patients with acne. Br. J. Dermatol. 2008, 159, 990–991. [Google Scholar] [CrossRef] [PubMed]
- Makrantonaki, E.; Ganceviciene, R.; Zouboulis, C. An update on the role of the sebaceous gland in the pathogenesis of acne. Dermato Endocrinol. 2011, 3, 41–49. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zouboulis, C.C. Acne and sebaceous gland function. Clin. Dermatol. 2004, 22, 360–366. [Google Scholar] [CrossRef] [PubMed]
- Thiboutot, D. Regulation of human sebaceous glands. J. Investig. Dermatol. 2004, 123, 1–12. [Google Scholar] [CrossRef]
- Fan, W.Q.; Yanase, T.; Morinaga, H.; Okabe, T.; Nomura, M.; Daitoku, H.; Fukamizu, A.; Kato, S.; Takayanagi, R.; Nawata, H. Insulin-like growth factor 1/insulin signaling activates androgen signaling through direct interactions of Foxo1 with androgen receptor. J. Biol. Chem. 2007, 282, 7329–7338. [Google Scholar] [CrossRef] [Green Version]
- Cappel, M.; Mauger, D.; Thiboutot, D. Correlation Between Serum Levels of Insulin-like Growth Factor 1, Dehydroepiandrosterone Sulfate, and Dihydrotestosterone and Acne Lesion Counts in Adult Women. Arch. Dermatol. 2005, 141, 333–338. [Google Scholar] [CrossRef]
- Ben-Amitai, D.; Laron, Z. Effect of insulin-like growth factor-1 deficiency or administration on the occurrence of acne. J. Eur. Acad. Dermatol. Venereol. 2011, 25, 950–954. [Google Scholar] [CrossRef]
- Gallitano, S.M.; Berson, D.S. How Acne Bumps Cause the Blues: The Influence of Acne Vulgaris on Self-Esteem. Int. J. Women’s Dermatol. 2018, 4, 12–17. [Google Scholar] [CrossRef]
- Hoon, Y.J.; Jungyoon, M.; Young, Y.J.; Won, K.J.; Sungjun, C.; Ick, C.S.; Ju, H.E.; Hun, S.D. Clinical efficacy of herbal extracts in treatment of mild to moderate acne vulgaris: An 8-week, double-blinded, randomized, controlled trial. J. Dermatolog. Treat. 2019. [Google Scholar] [CrossRef]
- Cristina, I.; Duarte, V. New and emerging drugs for the treatment of acne vulgaris in adolescents. Expert Opin. Pharmacother. 2019, 20, 1–16. [Google Scholar] [CrossRef]
- Gordon, C.M. Menstrual disorders in adolescents: Excess androgens and the polycystic ovary syndrome. Pediatr. Clin. N. Am. 1999, 46, 519–543. [Google Scholar] [CrossRef]
- Kolodziejczyk, B.; Duleba, A.J.; Spaczynski, R.Z.; Pawelczyk, L. Metformin therapy decreases hyperandrogenism and hyperinsulinemia in women with polycystic ovary syndrome. Fertil. Steril. 2000, 73, 1149–1154. [Google Scholar] [CrossRef]
- Behrangi, E.; Sadeghi, S.; Sadeghzadeh-Bazargan, A.; Goodarzi, A.; Ghassemi, M.; Sepasgozar, S.; Rohaninasab, M. The effect of metformin in the treatment of intractable and late onset acne: A comparison with oral isotretinoin. Iran. J. Dermatol. 2019, 22, 47–52. [Google Scholar]
- Fabbrocini, G.; Izzo, R.; Faggiano, A.; Del Prete, M.; Donnarumma, M.; Marasca, C.; Marciello, F.; Savastano, R.; Monfrecola, G.; Colao, A. Low glycaemic diet and metformin therapy: A new approach in male subjects with acne resistant to common treatments. Clin. Exp. Dermatol. 2016, 41, 38–42. [Google Scholar] [CrossRef] [Green Version]
- Verdolini, R.; Clayton, N.; Smith, A.; Alwash, N.; Mannello, B. Metformin for the treatment of hidradenitis suppurativa: A little help along the way. J. Eur. Acad. Dermatol. Venereol. 2013, 27, 1101–1108. [Google Scholar] [CrossRef]
- Fimmel, S.; Zouboulis, C.C. Comorbidities of hidradenitis suppurativa (acne inversa). Dermato Endocrinol. 2010, 2, 9–16. [Google Scholar] [CrossRef] [Green Version]
- Garg, A.; Kirby, J.S.; Lavian, J.; Lin, G.; Strunk, A. Sex- and Age-Adjusted Population Analysis of Prevalence Estimates for Hidradenitis Suppurativa in the United States. JAMA Dermatol. 2017, 153, 760–764. [Google Scholar] [CrossRef]
- Wiseman, M.C. Hidradenitis suppurativa: A review. Dermatol. Ther. 2004, 17, 50–54. [Google Scholar] [CrossRef]
- Sung, C.T.; Chao, T.; Lee, A.; Foulad, D.P.; Choi, F.; Juhasz, M.; Dobry, A.; Mesinkovska, N.A. Oral Metformin for Treating Dermatological Diseases: A Systematic Review. J. Drugs Dermatol. 2020, 19, 713–720. [Google Scholar] [CrossRef]
- Deckers, I.E.; Prens, E.P. An Update on Medical Treatment Options for Hidradenitis Suppurativa. Drugs 2016, 76, 215–229. [Google Scholar] [CrossRef]
- Swadi, A.; Jabur, A.H. The Value of Metformin in the Treatment of Hidradenitis Suppurativa in a Cohort of Iraqi Patients. Indian J. Public Health Res. Dev. 2019, 10, 2752–2756. [Google Scholar] [CrossRef]
- Garg, A.; Neuren, E.; Strunk, A. Hidradenitis Suppurativa Is Associated with Polycystic Ovary Syndrome: A Population-Based Analysis in the United States. J. Investig. Dermatol. 2018, 138, 1288–1292. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ingram, J.R.; Jenkins-Jones, S.; Knipe, D.W.; Morgan, C.L.I.; Cannings-John, R.; Piguet, V. Population-based Clinical Practice Research Datalink study using algorithm modelling to identify the true burden of hidradenitis suppurativa *. Br. J. Dermatol. 2018, 178, 917–924. [Google Scholar] [CrossRef] [PubMed]
- Kimball, A.B.; Sundaram, M.; Gauthier, G.; Rakesh, P.; Arijit, S. The Comorbidity Burden of Hidradenitis Suppurativa in the United States: A Claims Data Analysis. Dermatol. Ther. 2018, 8, 557–569. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shlyankevich, J.; Chen, A.J.; Kim, G.E.; Kimball, A.B. Hidradenitis suppurativa is a systemic disease with substantial comorbidity burden: A chart-verified case-control analysis. J. Am. Dermatol. 2014, 71, 1144–1150. [Google Scholar] [CrossRef] [PubMed]
- Phan, K.; Charlton, O.; Smith, S.D. Hidradenitis suppurativa and polycystic ovarian syndrome: Systematic review and meta-analysis. Australas. J. Dermatol. 2020, 61, e28–e33. [Google Scholar] [CrossRef]
- Diamanti-Kandarakis, E.; Kouli, C.; Tsianateli, T.; Bergiele, A. Therapeutic effects of metformin on insulin resistance and hyperandrogenism in polycystic ovary syndrome. Eur. J. Endocrinol. 1998, 138, 269–274. [Google Scholar] [CrossRef] [Green Version]
- Arun, B.; Loffeld, A. Long-standing hidradenitis suppurativa treated effectively with metformin. Clin. Exp. Dermatol. 2009, 34, 920–921. [Google Scholar] [CrossRef]
- Jennings, L.; Hambly, R.; Hughes, R.; Moriarty, B.; Kirby, B. Metformin use in hidradenitis suppurativa. J. Dermatolog. Treat. 2020, 31, 261–263. [Google Scholar] [CrossRef]
- Khandalavala, B.N. A Disease-Modifying Approach for Advanced Hidradenitis Suppurativa (Regimen with Metformin, Liraglutide, Dapsone, and Finasteride): A Case Report. Case Rep. Dermatol. 2017, 9, 70–78. [Google Scholar] [CrossRef]
- Khan, K.; Kumar, P. Effects of metformin use in pregnant patients with polycystic ovary syndrome. J. Hum. Reprod. Sci. 2012, 5, 166–169. [Google Scholar]
- Nawaz, F.H.; Khalid, R.; Naru, T.; Rizvi, J. Does continuous use of metformin throughout pregnancy improve pregnancy outcomes in women with polycystic ovarian syndrome? J. Obstet. Gynaecol. Res. 2008, 34, 832–837. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perng, P.; Zampella, J.G.; Okoye, G.A. Management of hidradenitis suppurativa in pregnancy. J. Am. Acad. Dermatol. 2017, 76, 979–989. [Google Scholar] [CrossRef] [PubMed]
- Hale, T.; Kristensen, J.; Hackett, L.; Kohan, R.; Ilett, K. Transfer of metformin into human milk. Diabetologia 2002, 45, 1509–1514. [Google Scholar] [CrossRef] [PubMed]
- Briggs, G.G.; Ambrose, P.J.; Nageotte, M.P.; Padilla, G.; Wan, S. Excretion of metformin into breast milk and the effect on nursing infants. Obstet. Gynecol. 2005, 105, 1437–1441. [Google Scholar] [CrossRef]
- Gorbachev, A.V.; Fairchild, R.L. CD4 + T Cells Regulate CD8 + T Cell-Mediated Cutaneous Immune Responses by Restricting Effector T Cell Development through a Fas Ligand-Dependent Mechanism. J. Immunol. 2004, 172, 2286–2295. [Google Scholar] [CrossRef] [Green Version]
- Brys, A.K.; Rodriguez-Homs, L.G.; Suwanpradid, J.; Atwater, A.R.; MacLeod, A.S. Shifting Paradigms in Allergic Contact Dermatitis: The Role of Innate Immunity. J. Investig. Dermatol. 2020, 140, 21–28. [Google Scholar] [CrossRef] [Green Version]
- Suzuki, K.; Meguro, K.; Nakagomi, D.; Nakajima, H. Roles of alternatively activated M2 macrophages in allergic contact dermatitis. Allergol. Int. 2017, 66, 392–397. [Google Scholar] [CrossRef]
- Wang, M.; Qu, S.; Ma, J.; Wang, X.; Yang, Y. Metformin suppresses LPS-induced inflammatory responses in macrophage and ameliorates allergic contact dermatitis in mice via autophagy. Biol. Pharm. Bull. 2020, 43, 129–137. [Google Scholar] [CrossRef] [Green Version]
- Nakahira, K.; Haspel, J.A.; Rathinam, V.A.K.; Lee, S.J.; Dolinay, T.; Lam, H.C.; Englert, J.A.; Rabinovitch, M.; Cernadas, M.; Kim, H.P.; et al. Autophagy proteins regulate innate immune responses by inhibiting the release of mitochondrial DNA mediated by the NALP3 inflammasome. Nat. Immunol. 2011, 12, 222–230. [Google Scholar] [CrossRef] [Green Version]
| Inflammatory Skin Disorders | Major Action | Potential Mechanism of Action |
|---|---|---|
| Psoriasis | Reduction of hyperglycemia | Improving glucose utilization |
| Decrease of inflammatory cytokines | Activating AMPK | |
| Acanthosis nigricans | Reduction of hyperglycemia | Stimulating GLUT4 translocation |
| Acne | Reduction of hyperandrogenism | Lowering IGF-1 and androgens |
| Hidradenitis suppurativa | Reduction of hyperandrogenism | Reducing androgen overproduction |
| Reduction of hyperglycemia | Improving glucose utilization | |
| Allergic contact dermatitis | Decrease of inflammatory cytokines | Activating AMPK |
| Reduction of NLRP3 inflammasome | Improving autophagy flux |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, J.-E.; Choi, M.S. A Molecular Perspective on the Potential Benefits of Metformin for the Treatment of Inflammatory Skin Disorders. Int. J. Mol. Sci. 2020, 21, 8960. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms21238960
Chang J-E, Choi MS. A Molecular Perspective on the Potential Benefits of Metformin for the Treatment of Inflammatory Skin Disorders. International Journal of Molecular Sciences. 2020; 21(23):8960. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms21238960
Chicago/Turabian StyleChang, Ji-Eun, and Min Sik Choi. 2020. "A Molecular Perspective on the Potential Benefits of Metformin for the Treatment of Inflammatory Skin Disorders" International Journal of Molecular Sciences 21, no. 23: 8960. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms21238960