Intrauterine Nitric Oxide Deficiency Weakens Differentiation of Vascular Smooth Muscle in Newborn Rats
Abstract
:1. Introduction
2. Results
2.1. Characteristics of Females
2.2. Gross Characteristics of the Offspring
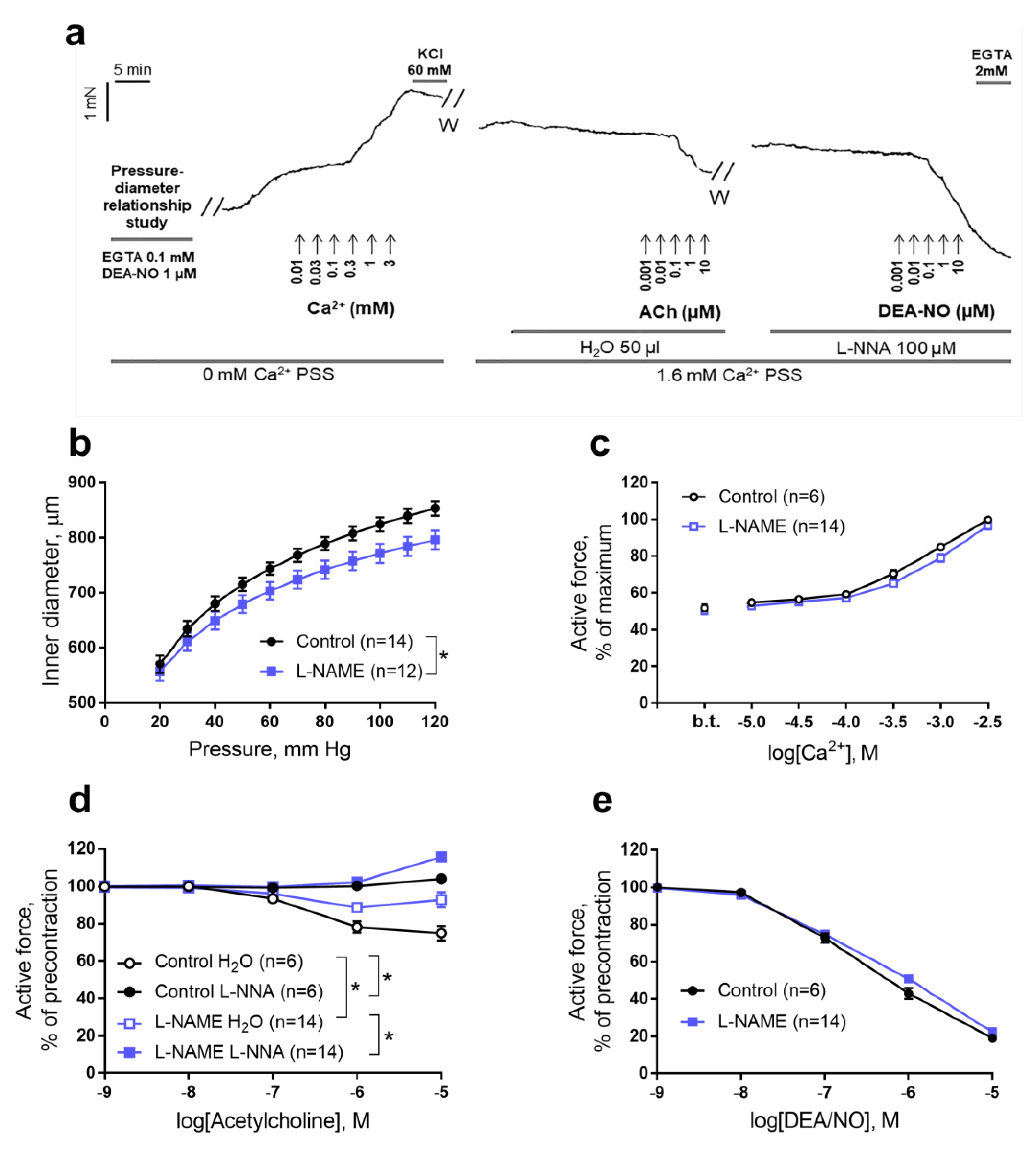
2.3. Functional Studies of the Aorta of Rat Pups
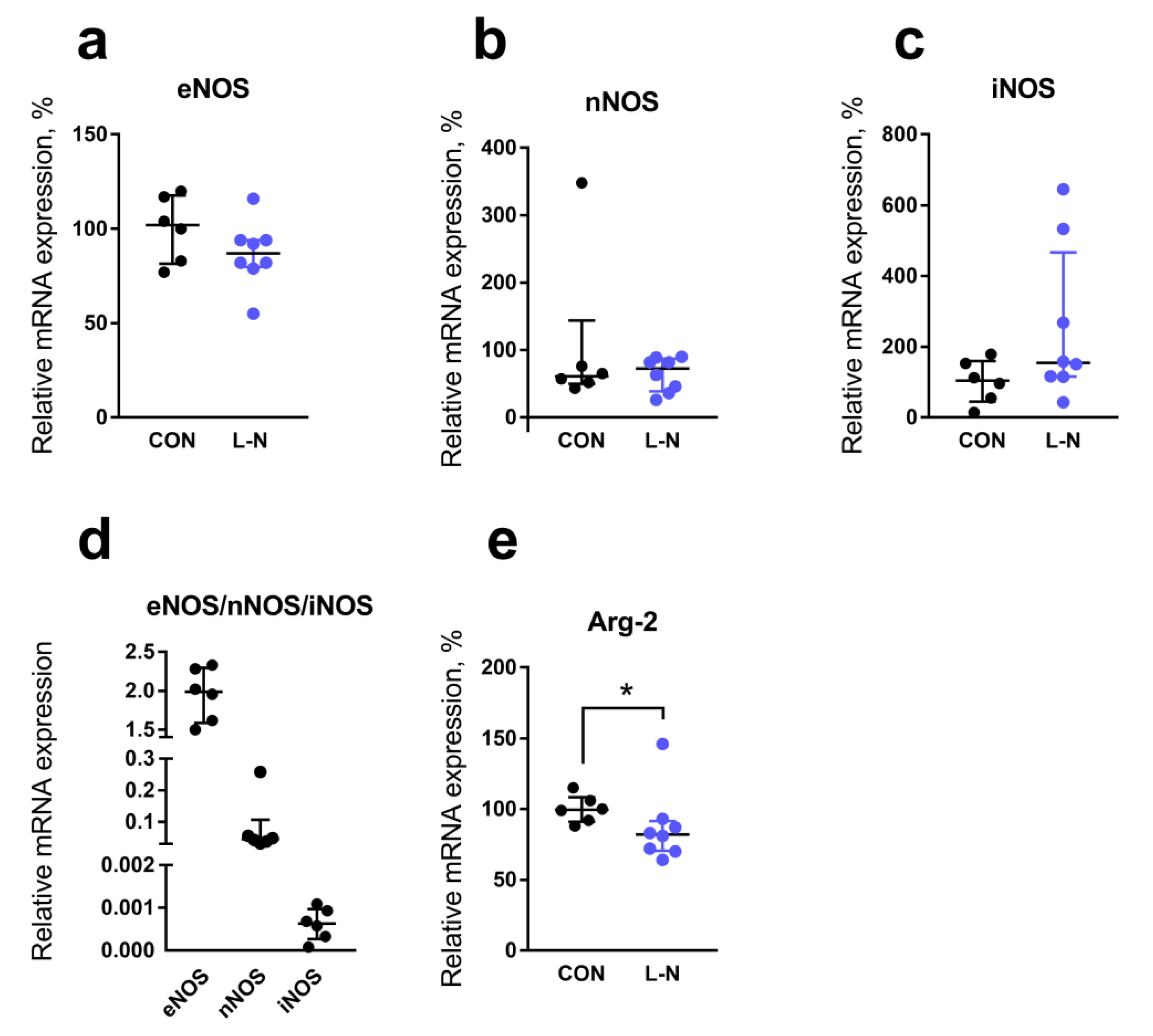
2.4. Comparison of mRNA Expression Levels in the Aorta of Rat Pups from the Control and L-NAME Groups
3. Discussion
3.1. Maternal L-NAME Treatment Provides Fetal NO Deficiency in the Rat
3.2. Maternal L-NAME Treatment Reduces NO Bioavailability in the Vasculature of Newborn Offspring
3.3. Reduced NO Bioavailability in the Vasculature of Newborn Offspring Is Associated with Altered Smooth Muscle Cell Properties
4. Materials and Methods
4.1. Model of Fetal NO Deficiency in the Rat
4.2. Measurement of NO Metabolites in Blood Serum
4.3. Determination of the Relative Content of mRNA in the Aorta of Rat Pups
4.4. Functional Experiments on the Isolated Aorta
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gaynullina, D.K.; Schubert, R.; Tarasova, O.S. Changes in endothelial nitric oxide production in systemic vessels during early ontogenesis—A key mechanism for the perinatal adaptation of the circulatory system. Int. J. Mol. Sci. 2019, 20, 1421. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vanhoutte, P.M.; Zhao, Y.; Xu, A.; Leung, S.W.S. Thirty years of saying NO: Sources, fate, actions, and misfortunes of the endothelium-derived vasodilator mediator. Circ. Res. 2016, 119, 375–396. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sofronova, S.I.; Borzykh, A.A.; Gaynullina, D.K.; Kuzmin, I.V.; Shvetsova, A.A.; Lukoshkova, E.V.; Tarasova, O.S. Endothelial nitric oxide weakens arterial contractile responses and reduces blood pressure during early postnatal development in rats. Nitric Oxide Biol. Chem. 2016, 55, 1–9. [Google Scholar] [CrossRef]
- Kent, A.L.; Kecskes, Z.; Shadbolt, B.; Falk, M.C. Blood pressure in the first year of life in healthy infants born at term. Pediatr. Nephrol. 2007, 22, 1743–1749. [Google Scholar] [CrossRef]
- Tanner, F.C.; Meier, P.; Greutert, H.; Champion, C.; Nabel, E.G.; Lüscher, T.F. Nitric oxide modulates expression of cell cycle regulatory proteins: A cytostatic strategy for inhibition of human vascular smooth muscle cell proliferation. Circulation 2000, 101, 1982–1989. [Google Scholar] [CrossRef] [Green Version]
- Boerth, N.J.; Dey, N.B.; Cornwell, T.L.; Lincoln, T.M. Cyclic GMP-Dependent protein kinase regulates vascular smooth muscle cell phenotype. J. Vasc. Res. 1997, 34, 245–259. [Google Scholar] [CrossRef]
- Lincoln, T.; Wu, X.; Sellak, H.; Dey, N.; Choi, C.S. Regulation of vascular smooth muscle cell phenotype by cyclic GMP and cyclic GMP-dependent protein kinase. Front. Biosci. 2006, 11, 356–367. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Itoh, S.; Katoh, Y.; Konishi, H.; Takaya, N.; Kimura, T.; Periasamy, M.; Yamaguchi, H. Nitric oxide regulates smooth-muscle-specific myosin heavy chain gene expression at the transcriptional level—Possible role of SRF and YY1 through CArG element. J. Mol. Cell. Cardiol. 2001, 33, 95–107. [Google Scholar] [CrossRef]
- Choi, C.S.; Sellak, H.; Brown, F.M.; Lincoln, T.M. cGMP-dependent protein kinase and the regulation of vascular smooth muscle cell gene expression: Possible involvement of Elk-1 sumoylation. Am. J. Physiol.-Heart Circ. Physiol. 2010, 299, H1660–H1670. [Google Scholar] [CrossRef] [Green Version]
- Rudic, R.D.; Shesely, E.G.; Maeda, N.; Smithies, O.; Segal, S.S.; Sessa, W.C. Direct evidence for the importance of endothelium-derived nitric oxide in vascular remodeling. J. Clin. Investig. 1998, 101, 731–736. [Google Scholar] [CrossRef] [PubMed]
- Ohta, S.; Komori, K.; Yonemitsu, Y.; Onohara, T.; Matsumoto, T.; Sugimachi, K. Intraluminal gene transfer of endothelial cell-nitric oxide synthase suppresses intimai hyperplasia of vein grafts in cholesterol-fed rabbit: A limited biological effect as a result of the loss of medial smooth muscle cells. Surgery 2002, 131, 644–653. [Google Scholar] [CrossRef]
- West, N.E.J.; Qian, H.; Guzik, T.J.; Black, E.; Cai, S.; George, S.E.; Channon, K.M. Nitric oxide synthase (nNOS) gene transfer modifies venous bypass graft remodeling: Effects on vascular smooth muscle cell differentiation and superoxide production. Circulation 2001, 104, 1526–1532. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, G.; Jacquet, L.; Karamariti, E.; Xu, Q. Origin and differentiation of vascular smooth muscle cells. J. Physiol. 2015, 593, 3013–3030. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cook, C.L.; Weiser, M.C.M.; Schwartz, P.E.; Jones, C.L.; Majack, R.A. Developmentally timed expression of an embryonic growth phenotype in vascular smooth muscle cells. Circ. Res. 1994, 74, 189–196. [Google Scholar] [CrossRef] [Green Version]
- Sofronova, S.I.; Gaynullina, D.K.; Shvetsova, A.A.; Borzykh, A.A.; Selivanova, E.K.; Kostyunina, D.S.; Sharova, A.P.; Martyanov, A.A.; Tarasova, O.S. Antenatal/early postnatal hypothyroidism alters arterial tone regulation in 2-week-old rats. J. Endocrinol. 2017, 235, 137–151. [Google Scholar] [CrossRef] [PubMed]
- Hamza, R.Z.; Diab, A.A.A.; Zahra, M.H.; Asalah, A.K.; Attia, M.S.; Moursi, S.M.M. Ameliorative effect of apelin-13 against renal complications in L-NAME-induced preeclampsia in rats. PeerJ 2021, 9. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Yang, N.; Wang, B.; Niu, X.; Cai, W.; Li, Y.; Li, Y.; Chen, S. Effect and mechanism of prophylactic use of tadalafil during pregnancy on L-NAME-induced preeclampsia-like rats. Placenta 2020, 99, 35–44. [Google Scholar] [CrossRef]
- Osol, G.; Ko, N.L.; Mandalà, M. Altered endothelial nitric oxide signaling as a paradigm for maternal vascular maladaptation in preeclampsia. Curr. Hypertens. Rep. 2017, 19, 82. [Google Scholar] [CrossRef]
- Hodžić, J.; Izetbegović, S.; Muračević, B.; Iriškić, R.; Jović, H.Š. Biosinteza nitričnog oksida u normalnoj trudnoći i trudnoći kompliciranoj preeklampsijom. Med. Glas. 2017, 14, 211–217. [Google Scholar] [CrossRef]
- Dymara-Konopka, W.; Laskowska, M. The role of nitric oxide, ADMA, and homocysteine in the etiopathogenesis of preeclampsia—Review. Int. J. Mol. Sci. 2019, 20, 2757. [Google Scholar] [CrossRef] [Green Version]
- Fantel, A.G.; Stamps, L.D.; Tran, T.T.; Mackler, B.; Person, R.E.; Nekahi, N. Role of free radicals in the limb teratogenicity of L-NAME (N(G)-nitro-(L)-arginine methyl ester): A new mechanistic model of vascular disruption. Teratology 1999, 60, 151–160. [Google Scholar] [CrossRef]
- Fantel, A.G.; Person, R.E. Further evidence for the role of free radicals in the limb teratogenicity of L-NAME. Teratology 2002, 66, 24–32. [Google Scholar] [CrossRef]
- Hefler, L.A.; Reyes, C.A.; O’Brien, W.E.; Gregg, A.R. Perinatal development of endothelial nitric oxide synthase-deficient mice1. Biol. Reprod. 2001, 64, 666–673. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martínez-Orgado, J.; González, R.; Alonso, M.J.; Salaices, M. Impairment of fetal endothelium-dependent relaxation in a rat model of preeclampsia by chronic nitric oxide synthase inhibition. J. Soc. Gynecol. Investig. 2004, 11, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Owens, G.K.; Kumar, M.S.; Wamhoff, B.R. Molecular regulation of vascular smooth muscle cell differentiation in development and disease. Physiol. Rev. 2004, 84, 767–801. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Qiao, Y.N.; Tao, T.; Zhao, W.; Wei, L.S.; Li, Y.Q.; Wang, W.; Wang, Y.; Zhou, Y.W.; Zheng, Y.Y.; et al. Distinct roles of smooth muscle and non-muscle myosin light chain-mediated smooth muscle contraction. Front. Physiol. 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Mochalov, S.V.; Tarasova, N.V.; Kudryashova, T.V.; Gaynullina, D.K.; Kalenchuk, V.U.; Borovik, A.S.; Vorotnikov, A.V.; Tarasova, O.S.; Schubert, R. Higher Ca2+-sensitivity of arterial contraction in 1-week-old rats is due to a greater Rho-kinase activity. Acta Physiol. 2018, 223, 1–15. [Google Scholar] [CrossRef]
- Guibert, C.; Ducret, T.; Savineau, J. Voltage-independent calcium influx in smooth muscle. Prog. Biophys. Mol. Biol. 2008, 98, 10–23. [Google Scholar] [CrossRef]
- Golovina, V.; Platoshyn, O.; Bailey, C.; Wang, J.; Limsuwan, A.; Sweeney, M.; Rubin, L.; Yuan, J. Upregulated TRP and enhanced capacitative Ca2+ entry in human pulmonary artery myocytes during proliferation. Am. J. Physiol.-Heart Circ. Physiol. 2001, 280, H746–H755. [Google Scholar] [CrossRef] [Green Version]
- Gollasch, M.; Haase, H.; Ried, C.; Lindschau, C.; Morano, I.; Luft, F.C.; Haller, H. L-type calcium channel expression depends on the differentiated state of vascular smooth muscle cells. FASEB J. 1998, 12, 593–601. [Google Scholar] [CrossRef]
- House, S.J.; Potier, M.; Bisaillon, J.; Singer, H.A.; Trebak, M. The non-excitable smooth muscle: Calcium signaling and phenotypic switching during vascular disease. Pflugers Arch. Eur. J. Physiol. 2008, 456, 769–785. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vallot, O.; Combettes, L.; Jourdon, P.; Inamo, J.; Marty, I.; Claret, M.; Lompré, A.M. Intracellular Ca2+ handling in vascular smooth muscle cells is affected by proliferation. Arterioscler. Thromb. Vasc. Biol. 2000, 20, 1225–1235. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sofronova, S.I.; Gaynullina, D.K.; Martyanov, A.A.; Tarasova, O.S. Endogenous oestrogens do not regulate endothelial nitric oxide production in early postnatal rats. Eur. J. Pharmacol. 2015, 765, 598–605. [Google Scholar] [CrossRef] [PubMed]
- Mulvany, M.J.; Halpern, W. Contractile properties of small arterial resistance vessels in spontaneously hypertensive and normotensive rats. Circ. Res. 1977, 41, 19–26. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| Parameters | Control | L-NAME |
|---|---|---|
| BW, g (n = 17;20) | 6.9 ± 0.5 | 5.6 ± 0.7 * |
| Heart weight/BW, % (n = 17;20) | 0.46 ± 0.03 | 0.51 ± 0.04 * |
| Kidney (both) weight/BW, % (n = 17;20) | 0.86 ± 0.10 | 0.87 ± 0.12 |
| NOx, µM (n = 10;10) | 39 ± 11 | 14 ± 8 * |
| Parameters | Control (n = 6) | L-NAME (n = 14) |
|---|---|---|
| Inner diameter d100, µm | 854 ± 39 | 765 ± 119 * |
| Maximum active tension, mN/mm | 1.07 ± 0.18 | 1.14 ± 0.24 |
| Protein | Gene | Forward | Reverse |
|---|---|---|---|
| eNOS | Nos3 | GGATTCTGGCAAGACCGATTAC | GGTGAGGACTTGTCCAAACACT |
| nNOS | Nos1 | GCCATCCAGCGCATAATGACCCAG | GAGGGTGACTCCAAAGATGTCCTC |
| iNOS | Nos2 | AGGCTTGGGTCTTGTTAGCCTAGT | ATTCTGTGCAGTCCCAGTGAGGAA |
| Arginase-2 | Arg2 | CCAGCCTAGCAGTGGATGTGA | CTCTGGAATGCTGTCGTGAA |
| SM-MHC | Myh11 | TTTGCCATTGAGGCCTTAG | GTTCACACGGCTGAGAATCCA |
| α-Actin | Acta2 | CCTGACCCTGAAGTATCCGA | CATCTCCAGAGTCCAGCACA |
| SM22α | Tagln | TTCTGCCTCAACATGGCCAAC | CACCTTCACTGGCTTGGATC |
| Cav1.2 | Cacna1c | CATCTCCATCACCTTCTTCC | AAATACCTGCATCCCAATCAC |
| RyR2 | Ryr2 | GAGAGCCCGGAAGCTCTGAA | GGCAACTCCATGGCACACAC |
| SERCA2A | Atp2a2 | TGAATCTGACCCAGTGGCTGA | ACTCCAGTATTGCAGGCTCCA |
| GAPDH | Gapdh | CACCAGCATCACCCCATTT | CCATCAAGGACCCCTTCATT |
| RPLP0 | Rplp0 | AGGGTCCTGGCTTTGTCTGTGG | AGCTGCAGGAGCAGCAGTGG |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shvetsova, A.A.; Borzykh, A.A.; Selivanova, E.K.; Kiryukhina, O.O.; Gaynullina, D.K.; Tarasova, O.S. Intrauterine Nitric Oxide Deficiency Weakens Differentiation of Vascular Smooth Muscle in Newborn Rats. Int. J. Mol. Sci. 2021, 22, 8003. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms22158003
Shvetsova AA, Borzykh AA, Selivanova EK, Kiryukhina OO, Gaynullina DK, Tarasova OS. Intrauterine Nitric Oxide Deficiency Weakens Differentiation of Vascular Smooth Muscle in Newborn Rats. International Journal of Molecular Sciences. 2021; 22(15):8003. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms22158003
Chicago/Turabian StyleShvetsova, Anastasia A., Anna A. Borzykh, Ekaterina K. Selivanova, Oxana O. Kiryukhina, Dina K. Gaynullina, and Olga S. Tarasova. 2021. "Intrauterine Nitric Oxide Deficiency Weakens Differentiation of Vascular Smooth Muscle in Newborn Rats" International Journal of Molecular Sciences 22, no. 15: 8003. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms22158003







