The M2a Macrophage Phenotype Accompanies Pulmonary Granuloma Resolution in Mmp12 Knock-Out Mice Instilled with Multiwall Carbon Nanotubes
Abstract
:1. Introduction
2. Results
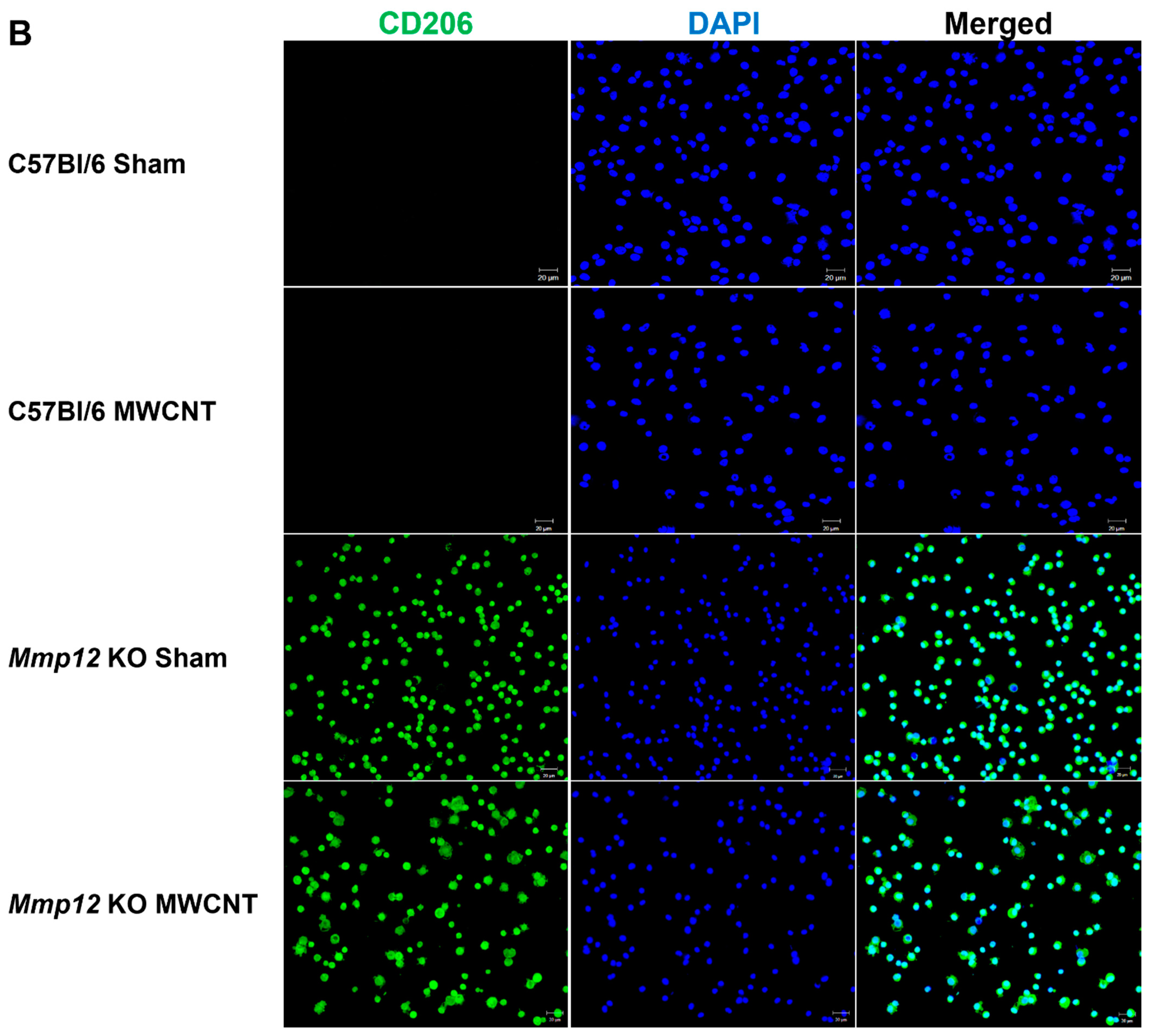
2.1. Alveolar Macrophages from Mmp12 KO Mice Express M2 Marker CD 206 (MRC1)
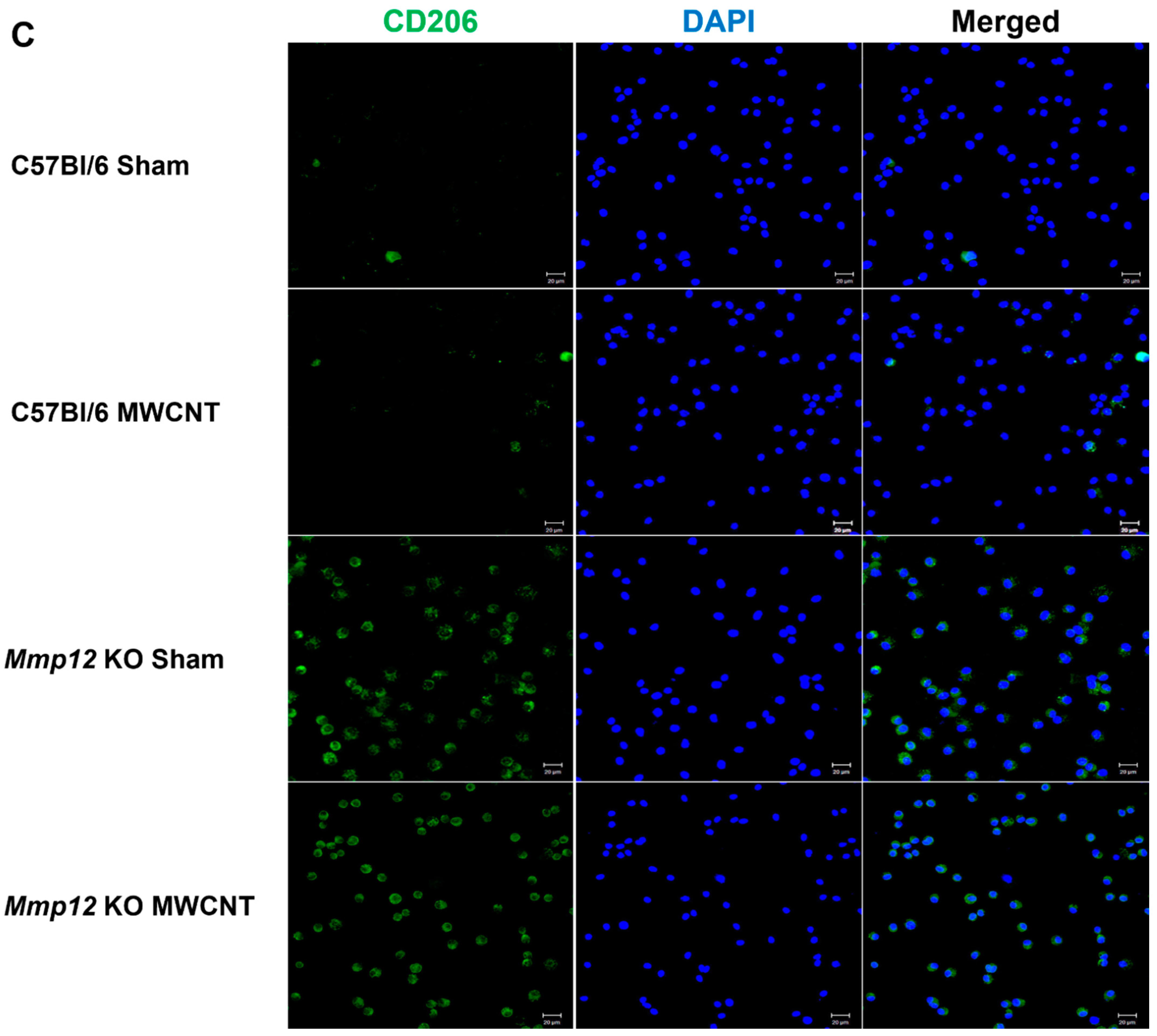
2.2. Mmp12 KO MWCNT Alveolar Macrophages Express M2c Markers at 10 Days and Convert to M2a by 60 Days
2.3. IL-13 Gene and Protein Expression Are Elevated in BALF from Mmp12 KO Mice Instilled with MWCNT at 60 Days
2.4. ApoE Is Upregulated in MWCNT-Instilled Mmp12 KO at 60 Days
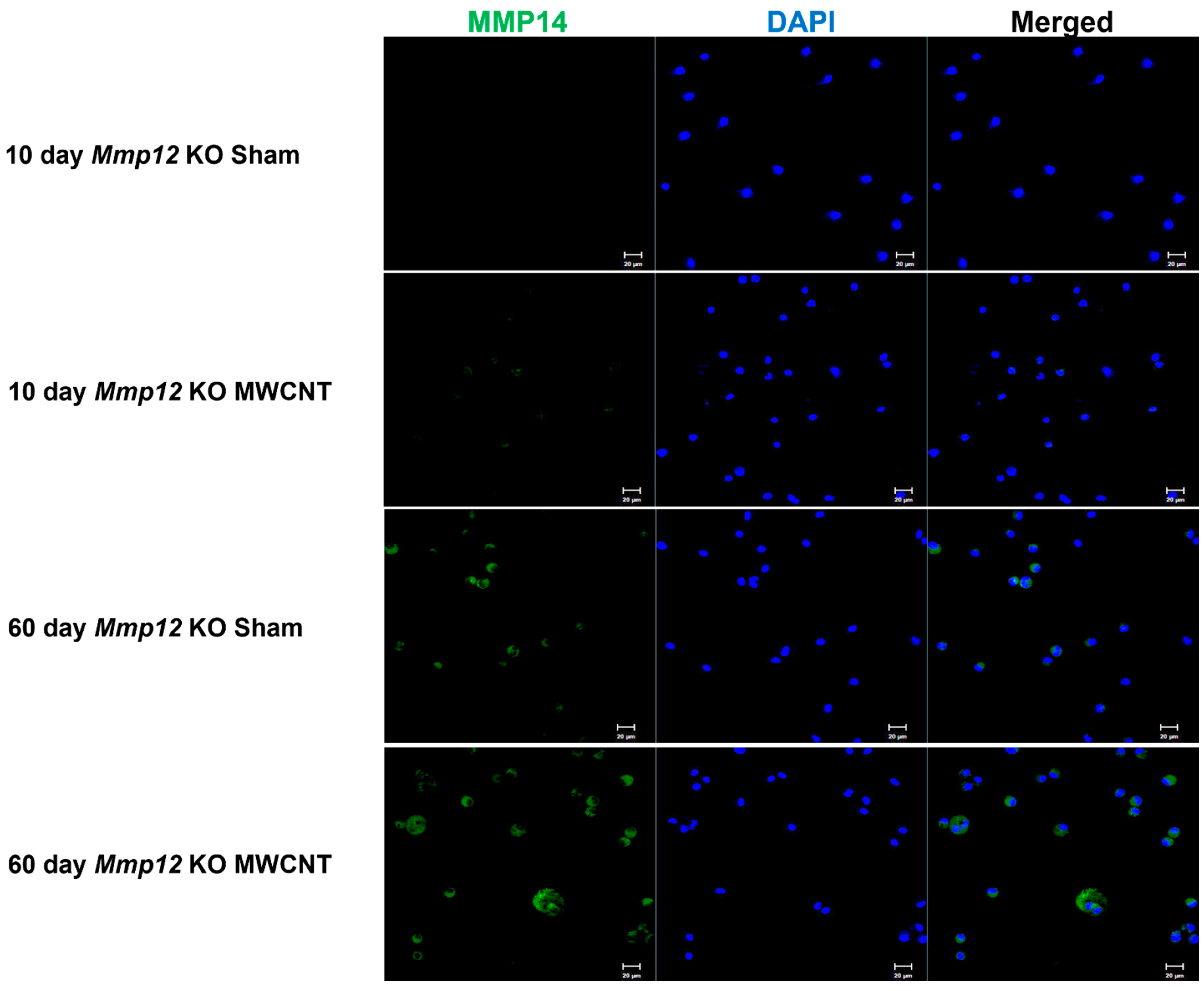
2.5. Surface Expression of MMP14 Is Increased in the Absence of MMP12 at 60 Days
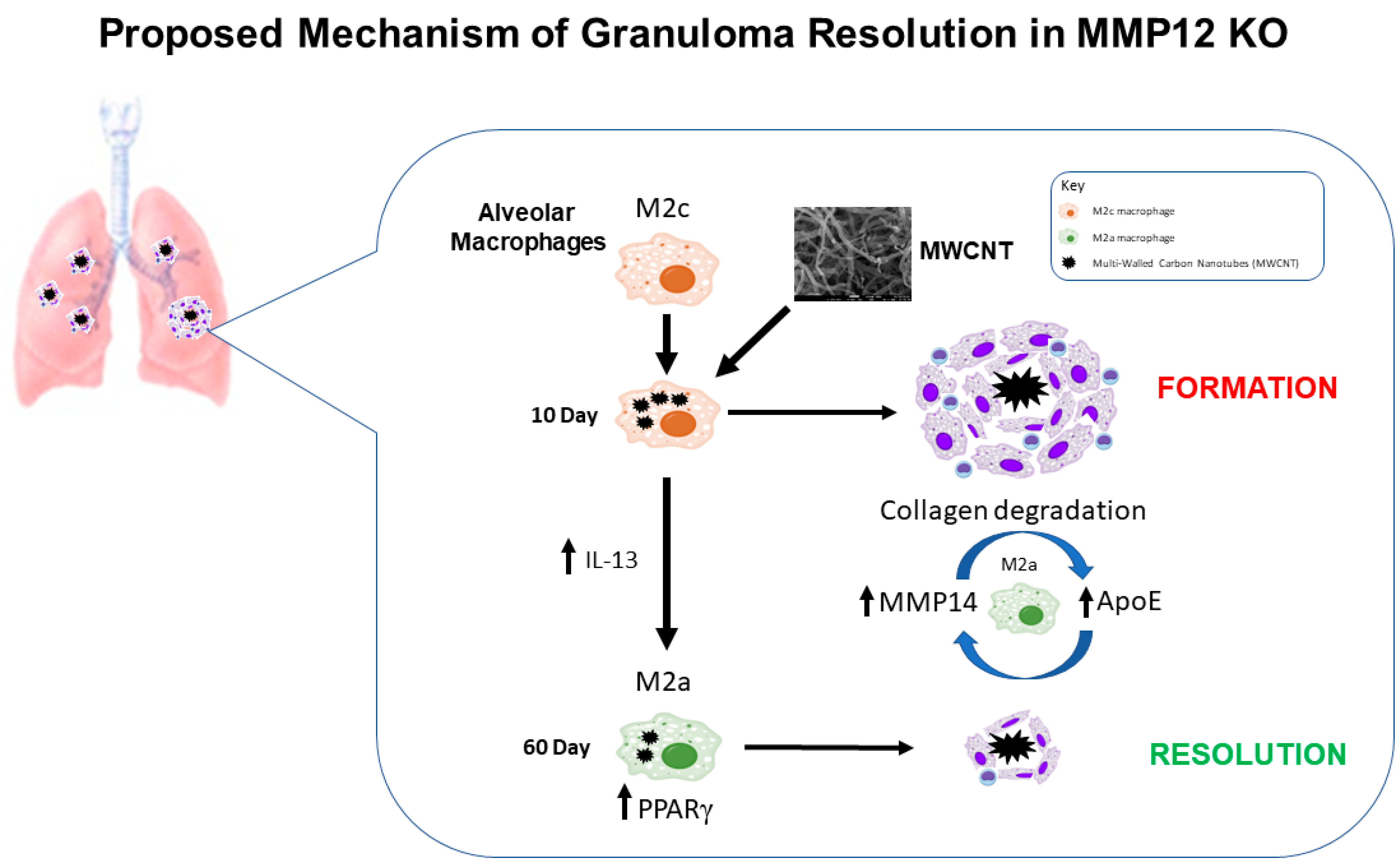
3. Discussion
4. Materials and Methods
4.1. Murine MWCNT Model
4.2. Characterization of Bronchoalveolar Lavage (BAL) Cells
4.3. Immunocytochemistry
4.4. RNA Purification and Gene Expression from BAL Cells
4.5. Protein Analysis of Bronchoalveolar Lavage Fluids (BALF)
4.6. Statistical Analysis
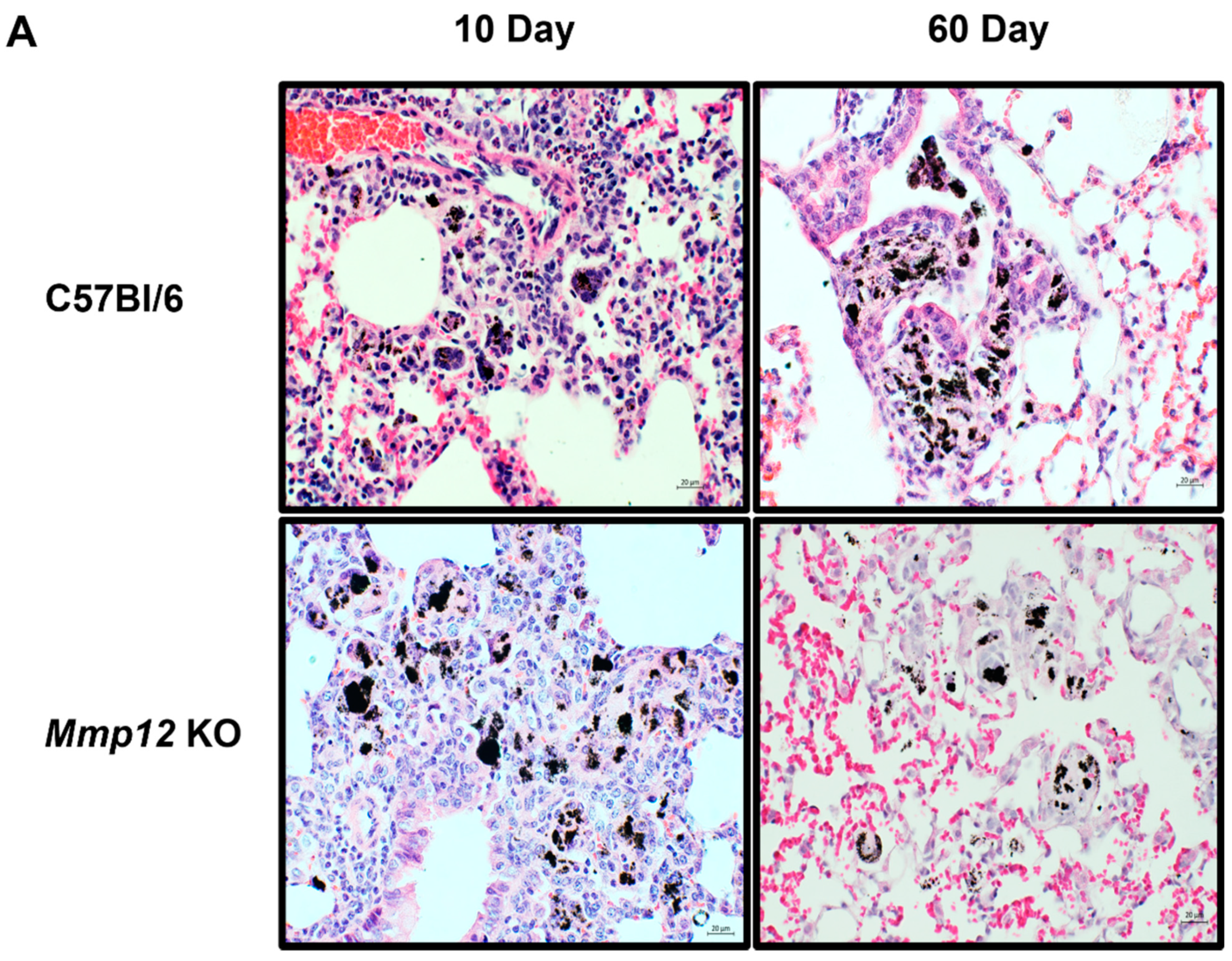
4.7. Histological Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Culver, D.A.; Judson, M.A. New advances in the management of pulmonary sarcoidosis. BMJ 2019, 367, l5553. [Google Scholar] [CrossRef] [Green Version]
- Denning, D.W.; Pleuvry, A.; Cole, D. Global burden of chronic pulmonary aspergillosis complicating sarcoidosis. Eur. Respir. J. 2012, 41, 621–626. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baughman, R.P.; Field, S.; Costabel, U.; Crystal, R.G.; Culver, D.A.; Drent, M.; Judson, M.A.; Wolff, G. Sarcoidosis in America. Analysis Based on Health Care Use. Ann. Am. Thorac. Soc. 2016, 13, 1244–1252. [Google Scholar] [CrossRef] [PubMed]
- Judson, M.A. Environmental Risk Factors for Sarcoidosis. Front. Immunol. 2020, 11, 1340. [Google Scholar] [CrossRef] [PubMed]
- Ungprasert, P.; Carmona, E.M.; Utz, J.P.; Ryu, J.H.; Crowson, C.S.; Matteson, E.L. Epidemiology of Sarcoidosis 1946–2013: A Population-Based Study. Mayo Clin. Proc. 2016, 91, 183–188. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iannuzzi, M.C.; Rybicki, B.A.; Teirstein, A.S. Sarcoidosis. N. Engl. J. Med. 2007, 357, 2153–2165. [Google Scholar] [CrossRef]
- Huizar, I.; Malur, A.; Midgette, Y.A.; Kukoly, C.; Chen, P.; Ke, P.C.; Podila, R.; Rao, A.M.; Wingard, C.; Dobbs, L.; et al. Novel Murine Model of Chronic Granulomatous Lung Inflammation Elicited by Carbon Nanotubes. Am. J. Respir. Cell Mol. Biol. 2011, 45, 858–866. [Google Scholar] [CrossRef] [Green Version]
- Rubins, J.B. Alveolar macrophages: Wielding the double-edged sword of inflammation. Am. J. Respir. Crit. Care Med. 2003, 167, 103–104. [Google Scholar] [CrossRef]
- Ricote, M.; Li, A.C.; Willson, T.M.; Kelly, C.J.; Glass, C.K. The peroxisome proliferator-activated receptor-g is a negative regulator of macrophage activation. Nature 1998, 391, 79–82. [Google Scholar] [CrossRef]
- Culver, D.A.; Barna, B.P.; Raychaudhuri, B.; Bonfield, T.L.; Abraham, S.; Malur, A.; Farver, C.F.; Kavuru, M.S.; Thomassen, M.J. Peroxisome proliferator-activated receptor gamma activity is deficient in alveolar macrophages in pulmonary sarcoidosis. Am. J. Respir. Cell Mol. Biol. 2004, 30, 1–5. [Google Scholar] [CrossRef]
- Bonfield, T.L.; Farver, C.F.; Barna, B.P.; Malur, A.; Abraham, S.; Raychaudhuri, B.; Kavuru, M.S.; Thomassen, M.J. Peroxisome proliferator-activated receptor-gamma is deficient in alveolar macrophages from patients with alveolar proteinosis. Am. J. Respir. Cell Mol. Biol. 2003, 29, 677–682. [Google Scholar] [CrossRef]
- Huizar, I.; Malur, A.; Patel, J.; McPeek, M.; Dobbs, L.; Wingard, C.; Barna, B.P.; Thomassen, M.J. The role of PPARγ in carbon nanotube-elicited granulomatous lung inflammation. Respir. Res. 2013, 14, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mohan, A.; Malur, A.; McPeek, M.; Barna, B.P.; Schnapp, L.M.; Thomassen, M.J.; Gharib, S.A. Transcriptional survey of alveolar macrophages in a murine model of chronic granulomatous inflammation reveals common themes with human sarcoidosis. Am. J. Physiol. Cell. Mol. Physiol. 2018, 314, L617–L625. [Google Scholar] [CrossRef]
- Abd-Elaziz, K.; Jesenak, M.; Vasakova, M.; Diamant, Z. Revisiting matrix metalloproteinase 12: Its role in pathophysiology of asthma and related pulmonary diseases. Curr. Opin. Pulm. Med. 2021, 27, 54–60. [Google Scholar] [CrossRef]
- Hunninghake, G.M.; Cho, M.H.; Tesfaigzi, Y.; Soto-Quiros, M.E.; Avila, L.; Lasky-Su, J.; Stidley, C.; Melén, E.; Söderhäll, C.; Hallberg, J.; et al. MMP12, Lung Function, and COPD in High-Risk Populations. N. Engl. J. Med. 2009, 361, 2599–2608. [Google Scholar] [CrossRef] [Green Version]
- Crouser, E.D.; Culver, D.A.; Knox, K.S.; Julian, M.W.; Shao, G.; Abraham, S.; Liyanarachchi, S.; Macre, J.E.; Wewers, M.D.; Gavrilin, M.A.; et al. Gene Expression Profiling Identifies MMP-12 and ADAMDEC1 as Potential Pathogenic Mediators of Pulmonary Sarcoidosis. Am. J. Respir. Crit. Care Med. 2009, 179, 929–938. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mohan, A.; Neequaye, N.; Malur, A.; Soliman, E.; McPeek, M.; Leffler, N.; Ogburn, D.; Tokarz, D.A.; Knudson, W.; Gharib, S.A.; et al. Matrix Metalloproteinase-12 Is Required for Granuloma Pro-gression. Front Immunol. 2020, 11, 553949. [Google Scholar] [CrossRef] [PubMed]
- Chawla, A. Control of Macrophage Activation and Function by PPARs. Circ. Res. 2010, 106, 1559–1569. [Google Scholar] [CrossRef]
- Croasdell, A.; Duffney, P.F.; Kim, N.; Lacy, S.; Sime, P.J.; Phipps, R.P. PPARγand the Innate Immune System Mediate the Resolution of Inflammation. PPAR Res. 2015, 2015, 1–20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Odegaard, J.I.; Ricardo-Gonzalez, R.R.; Goforth, M.H.; Morel, C.R.; Subramanian, V.; Mukundan, L.; Eagle, A.R.; Vats, D.; Brombacher, F.; Ferrante, A.W.; et al. Macrophage-specific PPARgamma controls alternative activation and improves insulin resistance. Nature 2007, 447, 1116–1120. [Google Scholar] [CrossRef] [Green Version]
- Ley, K. M1 Means Kill; M2 Means Heal. J. Immunol. 2017, 199, 2191–2193. [Google Scholar] [CrossRef] [PubMed]
- Mills, C.D.; Ley, K. M1 and M2 Macrophages: The Chicken and the Egg of Immunity. J. Innate Immun. 2014, 6, 716–726. [Google Scholar] [CrossRef] [PubMed]
- Chawla, A.; Boisvert, W.A.; Laffitte, B.A.; Barak, Y.; Liao, D.; Nagy, L.; Edwards, P.A. A PPAR gamma-LXR-ABCA1 pathway in macrophages is involved in cholesterol efflux and atherogenesis. Mol. Cell 2001, 7, 161–171. [Google Scholar] [CrossRef]
- Yue, L.; Mazzone, T. Peroxisome proliferator-activated receptor {gamma} stimulation of adipocyte ApoE gene transcription me-diated by the liver receptor X pathway. J. Biol. Chem. 2009, 284, 10453–10461. [Google Scholar] [CrossRef] [Green Version]
- Baitsch, D.; Bock, H.H.; Engel, T.; Telgmann, R.; Müller-Tidow, C.; Varga, G.; Bot, M.; Herz, J.; Robenek, H.; von Eckardstein, A.; et al. Apolipoprotein E Induces Antiinflammatory Phenotype in Macrophages. Arter. Thromb. Vasc. Biol. 2011, 31, 1160–1168. [Google Scholar] [CrossRef] [Green Version]
- Rőszer, T. Understanding the Mysterious M2 Macrophage through Activation Markers and Effector Mechanisms. Mediat. Inflamm. 2015, 2015, 816460. [Google Scholar] [CrossRef] [Green Version]
- Lolmede, K.; Campana, L.; Vezzoli, M.; Bosurgi, L.; Tonlorenzi, R.; Clementi, E.; Bianchi, M.E.; Cossu, G.; Manfredi, A.A.; Brunelli, S.; et al. Inflammatory and alternatively activated human macrophages attract vessel-associated stem cells, relying on separate HMGB1- and MMP-9-dependent pathways. J. Leukoc. Biol. 2009, 85, 779–787. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, S.; Wu, H.; Rong, X.; Guo, J. M2b macrophage polarization and its roles in diseases. J. Leukoc. Biol. 2019, 106, 345–358. [Google Scholar] [CrossRef] [Green Version]
- Orekhov, A.N.; Nikiforov, N.G.; Myasoedova, V.A.; Grechko, A.V.; Romanenko, E.B.; Zhang, D.; Chistiakov, D.A. Monocyte differentiation and macrophage polarization. Vessel Plus 2019, 3. [Google Scholar] [CrossRef]
- Nelson, M.P.; Christmann, B.S.; Werner, J.L.; Metz, A.E.; Trevor, J.L.; Lowell, C.A.; Steele, C. IL-33 and M2a alveolar macrophages promote lung defense against the atypical fungal pathogen Pneumocystis murina. J. Immunol. 2011, 186, 2372–2381. [Google Scholar] [CrossRef] [Green Version]
- Yamazaki, Y.; Painter, M.M.; Bu, G.; Kanekiyo, T. Apolipoprotein E as a Therapeutic Target in Alzheimer’s Disease: A Review of Basic Research and Clinical Evidence. CNS Drugs 2016, 30, 773–789. [Google Scholar] [CrossRef] [PubMed]
- Marais, A.D. Apolipoprotein E in lipoprotein metabolism, health and cardiovascular disease. Pathology 2019, 51, 165–176. [Google Scholar] [CrossRef] [PubMed]
- Ali, K.; Middleton, M.; Pure, E.; Rader, D.J. Apolipoprotein E suppresses the type I inflammatory response in vivo. Circ. Res. 2005, 97, 922–927. [Google Scholar] [CrossRef] [PubMed]
- Bont, D.; Der Meer, V. Apolipoprotein E-deficient mice have an impaired immune response to Klebsiella pneumoniae. Eur. J. Clin. Investig. 2000, 30, 818–822. [Google Scholar] [CrossRef]
- Samokhin, A.O.; Buhling, F.; Theissig, F.; Bromme, D. ApoE-deficient mice on cholate-containing high-fat diet reveal a pa-thology similar to lung sarcoidosis. Am. J. Pathol. 2010, 176, 1148–1156. [Google Scholar] [CrossRef] [Green Version]
- Cui, H.; Jiang, D.; Banerjee, S.; Xie, N.; Kulkarni, T.; Liu, R.-M.; Duncan, S.R.; Liu, G. Monocyte-derived alveolar macrophage apolipoprotein E participates in pulmonary fibrosis resolution. JCI Insight 2020, 5, e134539. [Google Scholar] [CrossRef]
- Atabai, K.; Yang, C.D.; Podolsky, M.J. You Say You Want a Resolution (of Fibrosis). Am. J. Respir. Cell Mol. Biol. 2020, 63, 424–435. [Google Scholar] [CrossRef]
- Yao, Y.; Xu, X.-H.; Jin, L. Macrophage Polarization in Physiological and Pathological Pregnancy. Front. Immunol. 2019, 10, 792. [Google Scholar] [CrossRef]
- Stawowczyk, M.; Wellenstein, M.D.; Lee, S.B.; Yomtoubian, S.; Durrans, A.; Choi, H.; Narula, N.; Altorki, N.K.; Gao, D.; Mittal, V. Matrix Metalloproteinase 14 promotes lung cancer by cleavage of Heparin-Binding EGF-like Growth Factor. Neoplasia 2017, 19, 55–64. [Google Scholar] [CrossRef]
- McPeek, M.; Malur, A.; Tokarz, D.A.; Murray, G.; Barna, B.P.; Thomassen, M.J. PPAR-gamma pathways attenuate pulmonary granuloma formation in a carbon nanotube induced murine model of sarcoidosis. Biochem. Biophys. Res. Commun. 2018, 503, 684–690. [Google Scholar] [CrossRef]
- Shamaei, M.; Mortaz, E.; Pourabdollah, M.; Garssen, J.; Tabarsi, P.; Velayati, A.; Adcock, I.M. Evidence for M2 mac-rophages in granulomas from pulmonary sarcoidosis: A new aspect of macrophage heterogeneity. Hum. Immunol. 2018, 79, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Cinetto, F.; Scarpa, R.; Dell’Edera, A.; Jones, M.G. Immunology of sarcoidosis: Old companions, new relationships. Curr. Opin. Pulm. Med. 2020, 26, 535–543. [Google Scholar] [CrossRef]
- Malur, A.; Mohan, A.; Barrington, R.A.; Leffler, N.; Malur, A.; Muller-Borer, B.; Murray, G.; Kew, K.; Zhou, C.; Russell, J.; et al. PPARgamma Deficiency Exacerbates Fibrotic Response to Mycobacteria Peptide in Murine Sarcoidosis Model. Am. J. Respir. Cell Mol. Biol. 2019, 11. [Google Scholar]
- Malur, A.; Mccoy, A.J.; Arce, S.; Barna, B.P.; Kavuru, M.S.; Malur, A.G.; Thomassen, M.J. Deletion of PPARg in alveolar macrophages is associated with a Th-1 pulmonary inflammatory response. J. Immunol. 2009, 182, 5816–5822. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]

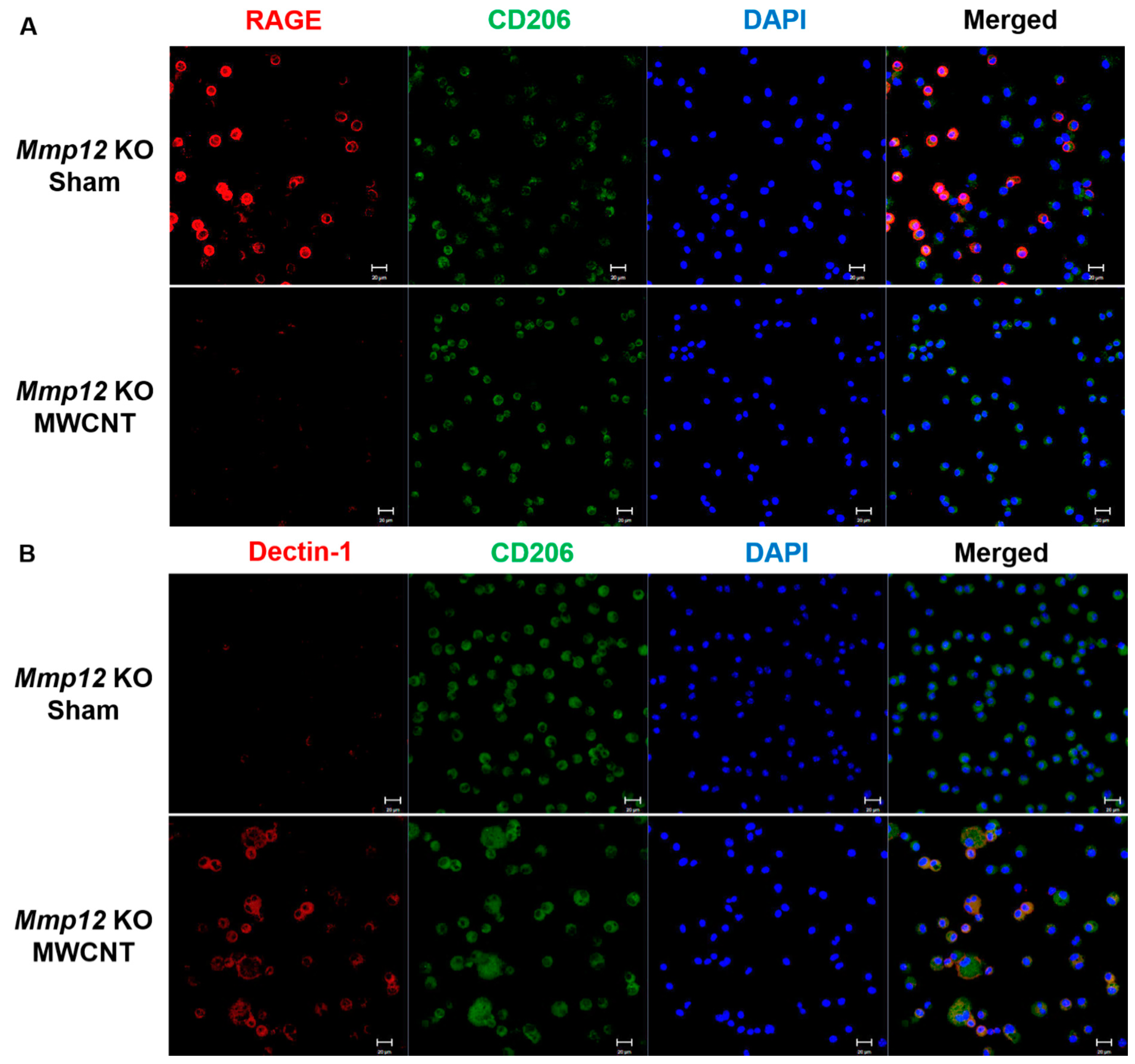


| Mmp12 KO | CD 206 (M2) | RAGE (M2c) | Dectin-1 (M2a) |
|---|---|---|---|
| 10 day Sham | + | + | − |
| 10 day MWCNT | + | + | − |
| 60 day Sham | + | + | − |
| 60 day MWCNT | + | − | + |
| Treatment | N | Total Cell Count (×105) | AM (%) | LYM (%) | PMN (%) | |
|---|---|---|---|---|---|---|
| 10 day | ||||||
| C57Bl/6 | Sham | 10 | 10.0 ± 4.3 | 99.3 ± 1.0 | 0.7 ± 0.9 | 0.0 ± 0.0 |
| C57Bl/6 | MWCNT | 10 | 15.4 ± 4.7 a | 87.7 ± 7.2 | 3.8 ± 2.5 | 8.5 ± 6.6 |
| Mmp12 KO | Sham | 10 | 9.6 ± 1.4 | 98.9 ± 1.4 | 1.1 ± 1.4 | 0.0 ± 0.0 |
| Mmp12 KO | MWCNT | 10 | 14.3 ± 6.0 a | 93.4 ± 5.7 | 3.1 ± 2.1 | 3.5 ± 4.3 |
| 60 day | ||||||
| C57Bl/6 | Sham | 9 | 6.6 ± 1.2 | 99.3 ± 1.0 | 0.7 ± 1.0 | 0.0 ± 0.0 |
| C57Bl/6 | MWCNT | 10 | 7.1 ± 1.4 | 98.9 ± 1.3 | 0.9 ± 1.0 | 0.2 ± 0.6 |
| Mmp12 KO | Sham | 10 | 10.1 ± 2.9 | 97.2 ± 4.7 | 1.3 ± 1.3 | 1.5 ± 3.8 |
| Mmp12 KO | MWNCT | 10 | 17.7 ± 9.0 *a | 92.6 ± 4.8 | 5.3 ± 3.5 | 2.1 ± 1.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ogburn, D.; Bhalla, S.; Leffler, N.; Mohan, A.; Malur, A.; Malur, A.G.; McPeek, M.; Barna, B.P.; Thomassen, M.J. The M2a Macrophage Phenotype Accompanies Pulmonary Granuloma Resolution in Mmp12 Knock-Out Mice Instilled with Multiwall Carbon Nanotubes. Int. J. Mol. Sci. 2021, 22, 11019. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms222011019
Ogburn D, Bhalla S, Leffler N, Mohan A, Malur A, Malur AG, McPeek M, Barna BP, Thomassen MJ. The M2a Macrophage Phenotype Accompanies Pulmonary Granuloma Resolution in Mmp12 Knock-Out Mice Instilled with Multiwall Carbon Nanotubes. International Journal of Molecular Sciences. 2021; 22(20):11019. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms222011019
Chicago/Turabian StyleOgburn, David, Sophia Bhalla, Nan Leffler, Arjun Mohan, Anagha Malur, Achut G. Malur, Matthew McPeek, Barbara P. Barna, and Mary Jane Thomassen. 2021. "The M2a Macrophage Phenotype Accompanies Pulmonary Granuloma Resolution in Mmp12 Knock-Out Mice Instilled with Multiwall Carbon Nanotubes" International Journal of Molecular Sciences 22, no. 20: 11019. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms222011019





