A Review of Ex Vivo X-ray Microfocus Computed Tomography-Based Characterization of the Cardiovascular System
Abstract
:1. Introduction
2. General Description of microCT, CE-CT and PC-CT
3. The Heart
3.1. Anatomical and Morphometric Assessment of the Whole Heart
3.2. Morphometrical Analysis of the Myocardium
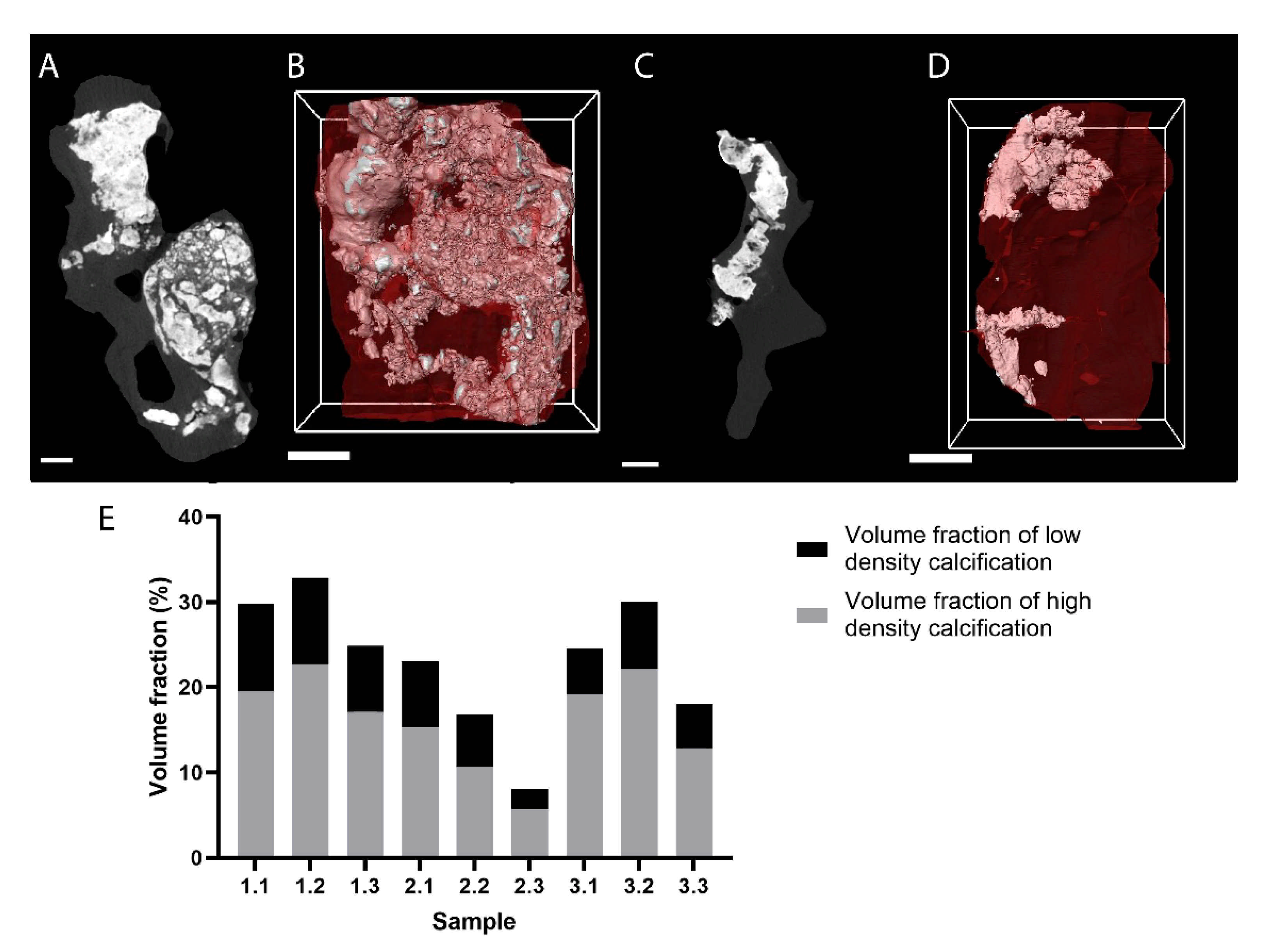
3.3. Measurement of the Calcifications in Heart Valves and Assessment of the Valve Microstructure
4. The Vasculature
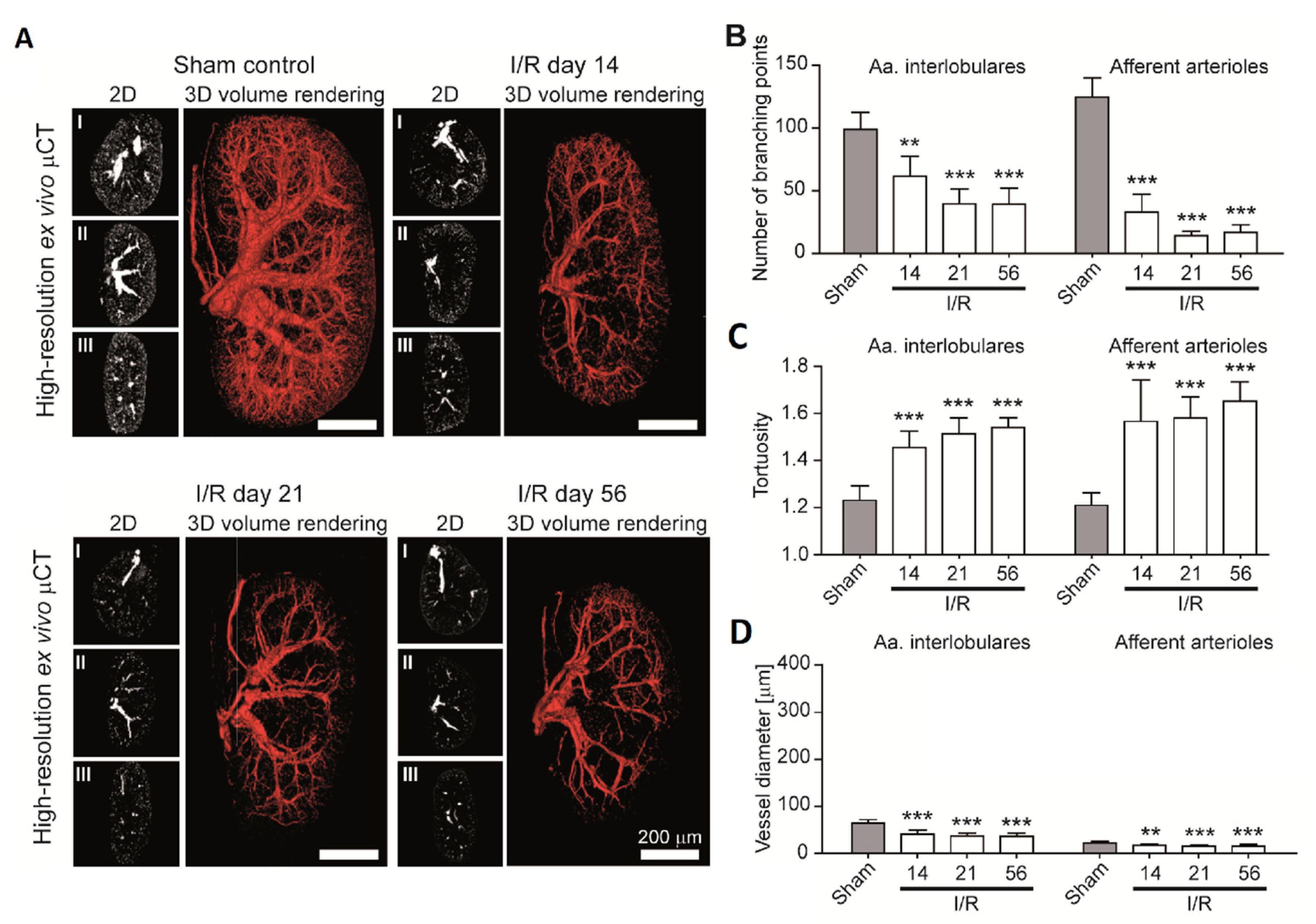
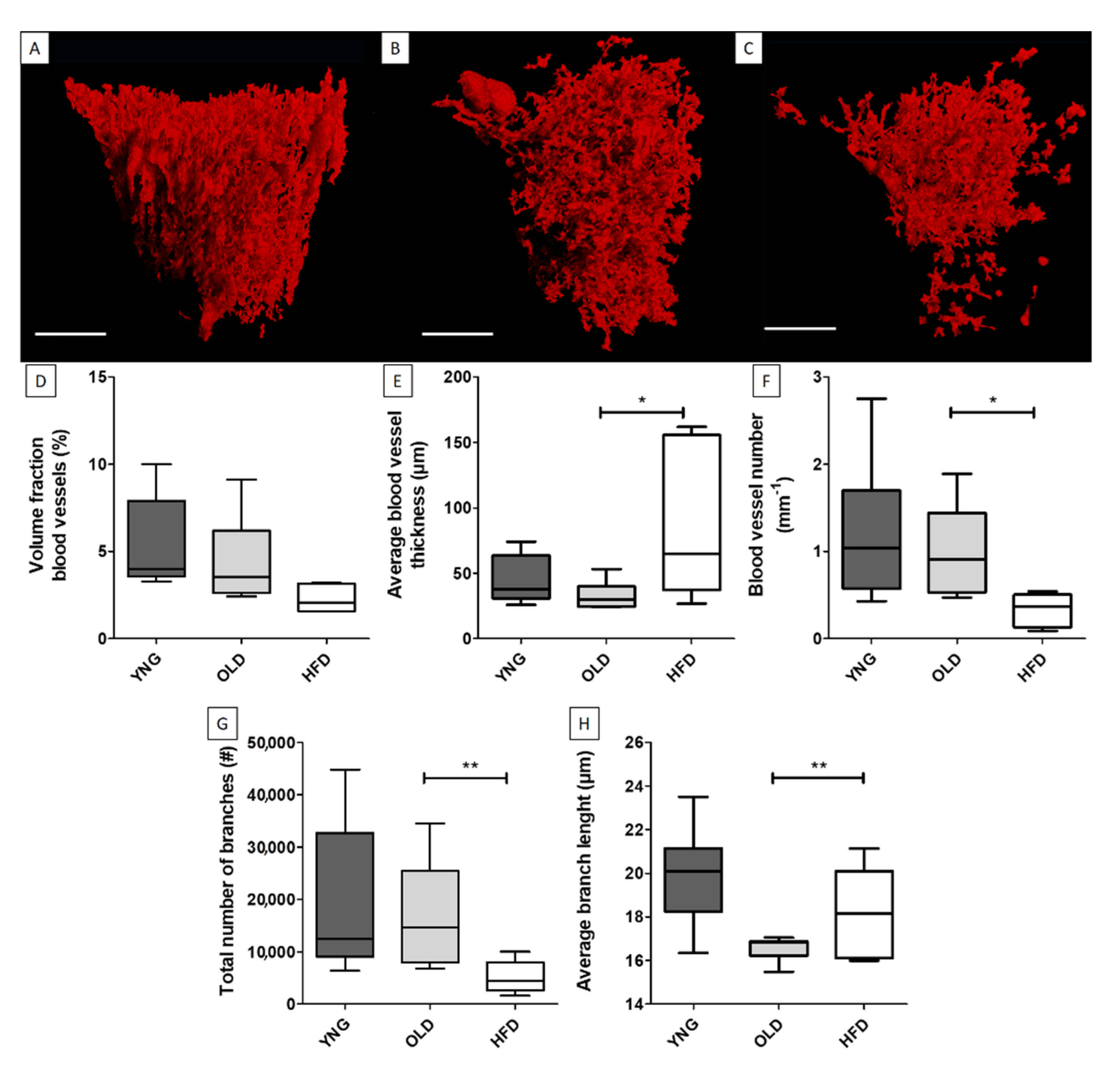
4.1. Spatial Distribution and Morphometrics of the Vascular Tree
4.2. Microstructure of the Vessel Wall
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- Eurostat. Cardiovascular Diseases Statistics—Death from Cardiovascular Diseases. Available online: https://ec.europa.eu/eurostat/statistics-explained/index.php/Cardiovascular_diseases_statistics#Deaths_from_cardiovascular_diseases (accessed on 20 March 2021).
- Dolk, H.; Loane, M.; Garne, E. Congenital Heart Defects in Europe: Prevalence and Perinatal Mortality, 2000 to 2005. Circulation 2011, 123, 841–849. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huntley, G.D.; Thaden, J.J.; Nkomo, V.T. Chapter 3—Epidemiology of Heart Valve Disease. In Principles of Heart Valve Engineering; Kheradvar, A., Ed.; Academic Press: Cambridge, MA, USA, 2019; pp. 41–62. [Google Scholar] [CrossRef]
- López-Guimet, J.; Peña-Pérez, L.; Bradley, R.S.; García-Canadilla, P.; Disney, C.; Geng, H.; Bodey, A.J.; Withers, P.J.; Bijnens, B.; Sherratt, M.J.; et al. MicroCT Imaging Reveals Differential 3D Micro-Scale Remodelling of the Murine Aorta in Ageing and Marfan Syndrome. Theranostics 2018, 8, 6038–6052. [Google Scholar] [CrossRef] [PubMed]
- Ehling, J.; Bábícková, J.; Gremse, F.; Klinkhammer, B.M.; Baetke, S.; Knuechel, R.; Kiessling, F.; Floege, J.; Lammers, T.; Boor, P. Quantitative Micro-Computed Tomography Imaging of Vascular Dysfunction in Progressive Kidney Diseases. J. Am. Soc. Nephrol. 2016, 27, 520–532. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, M.C.; García-Sanz, A.; Bentley, M.D.; Fortepiani, L.A.; García-Estañ, J.; Ritman, E.L.; Romero, J.C.; Juncos, L.A. Microcomputed Tomography of Kidneys Following Chronic Bile Duct Ligation. Kidney Int. 2000, 58, 1632–1640. [Google Scholar] [CrossRef]
- Bentley, M.D.; Ortiz, M.C.; Ritman, E.L.; Romero, J.C.; Michael, D.; Ortiz, M.C.; Ritman, E.L. The Use of Microcomputed Tomography to Study Microvasculature in Small Rodents. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2002. [Google Scholar] [CrossRef]
- Perrien, D.S.; Saleh, M.A.; Takahashi, K.; Madhur, M.S.; Harrison, D.G.; Harris, R.C.; Takahashi, T. Novel Methods for MicroCT-Based Analyses of Vasculature in the Renal Cortex Reveal a Loss of Perfusable Arterioles and Glomeruli in ENOS-/- Mice. BMC Nephrol. 2016, 17, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Savai, R.; Langheinrich, A.C.; Schermuly, R.T.; Pullamsetti, S.S.; Dumitrascu, R.; Traupe, H.; Rau, W.S.; Seeger, W.; Grimminger, F.; Banat, G.A. Evaluation of Angiogenesis Using Micro-Computed Tomography in a Xenograft Mouse Model of Lung Cancer. Neoplasia 2009, 11, 48–56. [Google Scholar] [CrossRef] [Green Version]
- Nyangoga, H.; Mercier, P.; Libouban, H.; Baslé, M.F.; Chappard, D. Three-Dimensional Characterization of the Vascular Bed in Bone Metastasis of the Rat by Microcomputed Tomography (MicroCT). PLoS ONE 2011, 6, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Pratt, R.; Hutchinson, J.C.; Melbourne, A.; Zuluaga, M.A.; Virasami, A.; Vercauteren, T.; Ourselin, S.; Sebire, N.J.; Arthurs, O.J.; David, A.L. Imaging the Human Placental Microcirculation with Micro-Focus Computed Tomography: Optimisation of Tissue Preparation and Image Acquisition. Placenta 2017, 60, 36–39. [Google Scholar] [CrossRef]
- Rehman, R.; Makaryus, A.N. Cardiac Imaging. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2019. [Google Scholar]
- Pieles, G.; Geyer, S.H.; Szumska, D.; Schneider, J.; Neubauer, S.; Clarke, K.; Dorfmeister, K.; Franklyn, A.; Brown, S.D.; Bhattacharya, S.; et al. ΜMRI-HREM Pipeline for High-Throughput, High-Resolution Phenotyping of Murine Embryos. J. Anat. 2007, 211, 132–137. [Google Scholar] [CrossRef]
- Chen, K.C.; Arad, A.; Song, Z.M.; Croaker, D.G. High-Definition Heart Visualization Using Micro-CT Scanning on Experimental Rats. J. Clin. Exp. Cardiolog. 2018, 9. [Google Scholar] [CrossRef]
- Kleczek, P.; Jaworek-Korjakowska, J.; Gorgon, M. A Novel Method for Tissue Segmentation in High-Resolution H&E-Stained Histopathological Whole-Slide Images. Comput. Med. Imaging Graph. 2020, 79, 101686. [Google Scholar] [CrossRef]
- Gómez-Gaviro, M.V.; Sanderson, D.; Ripoll, J.; Desco, M. Biomedical Applications of Tissue Clearing and Three-Dimensional Imaging in Health and Disease. iScience 2020, 23. [Google Scholar] [CrossRef]
- Chatterjee, S. Artefacts in Histopathology. J. Oral Maxillofac. Pathol. 2014, 18 (Suppl. 1), S111–S116. [Google Scholar] [CrossRef]
- Taqi, S.; Sami, S.; Sami, L.; Zaki, S. A Review of Artifacts in Histopathology. J. Oral Maxillofac. Pathol. 2018, 22, 279. [Google Scholar] [CrossRef]
- Geyer, S.H.; Weninger, W.J. High-Resolution Episcopic Microscopy (HREM): Looking Back on 13 Years of Successful Generation of Digital Volume Data of Organic Material for 3D Visualisation and 3D Display. Appl. Sci. 2019, 9, 3826. [Google Scholar] [CrossRef] [Green Version]
- Vinegoni, C.; Fumene Feruglio, P.; Courties, G.; Schmidt, S.; Hulsmans, M.; Lee, S.; Wang, R.; Sosnovik, D.; Nahrendorf, M.; Weissleder, R. Fluorescence Microscopy Tensor Imaging Representations for Large-Scale Dataset Analysis. Sci. Rep. 2020, 10, 1–15. [Google Scholar] [CrossRef]
- Goergen, C.J.; Chen, H.H.; Sakadžić, S.; Srinivasan, V.J.; Sosnovik, D.E. Microstructural Characterization of Myocardial Infarction with Optical Coherence Tractography and Two-Photon Microscopy. Physiol. Rep. 2016, 4, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Chiu, Y.W.; Lo, M.T.; Tsai, M.R.; Chang, Y.C.; Hsu, R.B.; Yu, H.Y.; Sun, C.K.; Ho, Y.L. Applying Harmonic Optical Microscopy for Spatial Alignment of Atrial Collagen Fibers. PLoS ONE 2010, 5, 1–9. [Google Scholar] [CrossRef]
- Johnson, G.A.; Cofer, G.P.; Fubara, B.; Gewalt, S.L.; Hedlund, L.W.; Maronpot, R.R. Magnetic Resonance Histology for Morphologic Phenotyping. J. Magn. Reson. Imaging 2002, 16, 423–429. [Google Scholar] [CrossRef]
- Tyszka, J.M.; Fraser, S.E.; Jacobs, R.E. Magnetic Resonance Microscopy: Recent Advances and Applications. Curr. Opin. Biotechnol. 2005, 16, 93–99. [Google Scholar] [CrossRef]
- Driehuys, B.; Nouls, J.; Badea, A.; Bucholz, E.; Ghaghada, K.; Petiet, A.; Hedlund, L.W. Small Animal Imaging with Magnetic Resonance Microscopy. ILAR J. 2008, 53, 35–53. [Google Scholar] [CrossRef] [Green Version]
- Nielles-Vallespin, S.; Khalique, Z.; Ferreira, P.F.; de Silva, R.; Scott, A.D.; Kilner, P.; McGill, L.A.; Giannakidis, A.; Gatehouse, P.D.; Ennis, D.; et al. Assessment of Myocardial Microstructural Dynamics by In Vivo Diffusion Tensor Cardiac Magnetic Resonance. J. Am. Coll. Cardiol. 2017, 69, 661–676. [Google Scholar] [CrossRef]
- Bernus, O.; Radjenovic, A.; Trew, M.L.; Legrice, I.J.; Sands, G.B.; Magee, D.R.; Smaill, B.H.; Gilbert, S.H. Comparison of Diffusion Tensor Imaging by Cardiovascular Magnetic Resonance and Gadolinium Enhanced 3D Image Intensity Approaches to Investigation of Structural Anisotropy in Explanted Rat Hearts. J. Cardiovasc. Magn. Reson. 2015, 17, 1–27. [Google Scholar] [CrossRef] [Green Version]
- Li, W.; Lu, M.; Banerjee, S.; Zhong, J.; Ye, A.; Molter, J.; Yu, X. Ex Vivo Diffusion Tensor MRI Reflects Microscopic Structural Remodeling Associated with Aging and Disease Progression in Normal and Cardiomyopathic Syrian Hamsters. NMR Biomed. 2009, 22, 819–825. [Google Scholar] [CrossRef] [Green Version]
- Papantoniou, I.; Sonnaert, M.; Geris, L.; Luyten, F.P.; Schrooten, J.; Kerckhofs, G. Three-Dimensional Characterization of Tissue-Engineered Constructs by Contrast-Enhanced Nanofocus Computed Tomography. Tissue Eng. Part C Methods 2014, 20, 177–187. [Google Scholar] [CrossRef] [Green Version]
- De Bournonville, S.; Vangrunderbeeck, S.; Kerckhofs, G. Contrast-Enhanced MicroCT for Virtual 3D Anatomical Pathology of Biological Tissues: A Literature Review. Contrast Media Mol. Imaging 2019, 2019. [Google Scholar] [CrossRef] [Green Version]
- Dejea, H.; Garcia-Canadilla, P.; Cook, A.C.; Guasch, E.; Zamora, M.; Crispi, F.; Stampanoni, M.; Bijnens, B.; Bonnin, A. Comprehensive Analysis of Animal Models of Cardiovascular Disease Using Multiscale X-Ray Phase Contrast Tomography. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Logghe, G.; Trachet, B.; Aslanidou, L.; Villaneuva-Perez, P.; De Backer, J.; Stergiopulos, N.; Stampanoni, M.; Aoki, H.; Segers, P. Propagation-Based Phase-Contrast Synchrotron Imaging of Aortic Dissection in Mice: From Individual Elastic Lamella to 3D Analysis. Sci. Rep. 2018, 8, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cloetens, P.; Pateyron-Salomé, M.; Buffière, J.Y.; Peix, G.; Baruchel, J.; Peyrin, F.; Schlenker, M. Observation of Microstructure and Damage in Materials by Phase Sensitive Radiography and Tomography. J. Appl. Phys. 1997, 81, 5878–5886. [Google Scholar] [CrossRef]
- Cloetens, P.; Mache, R.; Schlenker, M.; Lerbs-Mache, S. Quantitative Phase Tomography of Arabidopsis Seeds Reveals Intercellular Void Network. Proc. Natl. Acad. Sci. USA 2006, 103, 14626–14630. [Google Scholar] [CrossRef] [Green Version]
- Baruchel, J.; Buffiere, J.Y.; Maire, E. X-Ray Tomography in Material Science; Hermes Science Publications: Paris, France, 2000. [Google Scholar]
- Langheinrich, A.C.; Vorman, S.; Seidenstücker, J.; Kampschulte, M.; Bohle, R.M.; Wienhard, J.; Zygmunt, M. Quantitative 3D Micro-CT Imaging of the Human Feto-Placental Vasculature in Intrauterine Growth Restriction. Placenta 2008, 29, 937–941. [Google Scholar] [CrossRef]
- Cortell, S. Silicone Rubber for Renal Tubular Injection. J. Appl. Physiol. 1969, 26, 158–159. [Google Scholar] [CrossRef]
- Duvall, C.L.; Taylor, W.R.; Weiss, D.; Guldberg, R.E. Quantitative Microcomputed Tomography Analysis of Collateral Vessel Development after Ischemic Injury. Am. J. Physiol. Hear. Circ. Physiol. 2004, 287, H302–H310. [Google Scholar] [CrossRef]
- Chen, B.; Duan, J.; Chabot-Lecoanet, A.C.; Lu, H.; Tonnelet, R.; Morel, O.; Beaumont, M. Ex Vivo Magnetic Resonance Angiography to Explore Placental Vascular Anatomy. Placenta 2017, 58, 40–45. [Google Scholar] [CrossRef]
- Degenhardt, K.; Wright, A.C.; Horng, D.; Padmanabhan, A.; Epstein, J.A. Rapid 3D Phenotyping of Cardiovascular Development in Mouse Embryos by Micro-CT with Iodine Staining. Circ. Cardiovasc. Imaging 2010, 3, 314–322. [Google Scholar] [CrossRef] [Green Version]
- Gössl, M.; Zamir, M.; Ritman, E.L. Vasa Vasorum Growth in the Coronary Arteries of Newborn Pigs. Anat. Embryol. (Berl.) 2004, 208, 351–357. [Google Scholar] [CrossRef]
- Rennie, M.Y.; Whiteley, K.J.; Kulandavelu, S.; Adamson, S.L.; Sled, J.G. 3D Visualisation and Quantification by Microcomputed Tomography of Late Gestational Changes in the Arterial and Venous Feto-Placental Vasculature of the Mouse. Placenta 2007, 28, 833–840. [Google Scholar] [CrossRef]
- Roche, B.; David, V.; Vanden-Bossche, A.; Peyrin, F.; Malaval, L.; Vico, L.; Lafage-Proust, M.H. Structure and Quantification of Microvascularisation within Mouse Long Bones: What and How Should We Measure? Bone 2012, 50, 390–399. [Google Scholar] [CrossRef]
- Sangaralingham, S.J.; Ritman, E.L.; McKie, P.M.; Ichiki, T.; Lerman, A.; Scott, C.G.; Martin, F.L.; Harders, G.E.; Bellavia, D.; Burnett, J.C. Cardiac Micro-Computed Tomography Imaging of the Aging Coronary Vasculature. Circ. Cardiovasc. Imaging 2012, 5, 518–524. [Google Scholar] [CrossRef] [Green Version]
- Vasquez, S.X.; Gao, F.; Su, F.; Grijalva, V.; Pope, J.; Martin, B.; Stinstra, J.; Masner, M.; Shah, N.; Weinstein, D.M.; et al. Optimization of MicroCT Imaging and Blood Vessel Diameter Quantitation of Preclinical Specimen Vasculature with Radiopaque Polymer Injection Medium. PLoS ONE 2011, 6, 2–7. [Google Scholar] [CrossRef]
- Butcher, J.T.; Sedmera, D.; Guldberg, R.E.; Markwald, R.R. Quantitative Volumetric Analysis of Cardiac Morphogenesis Assessed through Micro-Computed Tomography. Dev. Dyn. 2007, 236, 802–809. [Google Scholar] [CrossRef] [PubMed]
- Ghanavati, S.; Yu, L.X.; Lerch, J.P.; Sled, J.G. A Perfusion Procedure for Imaging of the Mouse Cerebral Vasculature by X-ray Micro-CT. J. Neurosci. Methods 2014, 221, 70–77. [Google Scholar] [CrossRef] [Green Version]
- Hlushchuk, R.; Haberthür, D.; Djonov, V. Ex Vivo MicroangioCT: Advances in Microvascular Imaging. Vascul. Pharmacol. 2019, 112, 2–7. [Google Scholar] [CrossRef] [Green Version]
- Schaad, L.; Hlushchuk, R.; Barré, S.; Gianni-Barrera, R.; Haberthür, D.; Banfi, A.; Djonov, V. Correlative Imaging of the Murine Hind Limb Vasculature and Muscle Tissue by MicroCT and Light Microscopy. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Hlushchuk, R.; Haberthür, D.; Soukup, P.; Barré, S.F.; Khoma, O.Z.; Schittny, J.; Haghayegh Jahromi, N.; Bouchet, A.; Engelhardt, B.; Djonov, V. Innovative High-Resolution MicroCT Imaging of Animal Brain Vasculature. Brain Struct. Funct. 2020, 225, 2885–2895. [Google Scholar] [CrossRef] [PubMed]
- Blery, P.; Pilet, P.; Vanden-Bossche, A.; Thery, A.; Guicheux, J.; Amouriq, Y.; Espitalier, F.; Mathieu, N.; Weiss, P. Vascular Imaging with Contrast Agent in Hard and Soft Tissues Using Microcomputed-Tomography. J. Microsc. 2016, 262, 40–49. [Google Scholar] [CrossRef]
- Deng, Y.; Rowe, K.J.; Chaudhary, K.R.; Yang, A.; Mei, S.H.J.; Stewart, D.J. Optimizing Imaging of the Rat Pulmonary Microvasculature by Micro-Computed Tomography. Pulm. Circ. 2019, 9. [Google Scholar] [CrossRef]
- Qiu, X.; Shi, X.; Ouyang, J.; Xu, D.; Zhao, D. A Method to Quantify and Visualize Femoral Head Intraosseous Arteries by Micro-CT. J. Anat. 2016, 229, 326–333. [Google Scholar] [CrossRef]
- Vickerton, P.; Jarvis, J.; Jeffery, N. Concentration-Dependent Specimen Shrinkage in Iodine-Enhanced MicroCT. J. Anat. 2013, 223, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Novo Matos, J.; Garcia-Canadilla, P.; Simcock, I.C.; Hutchinson, J.C.; Dobromylskyj, M.; Guy, A.; Arthurs, O.J.; Cook, A.C.; Luis Fuentes, V. Micro-Computed Tomography (Micro-CT) for the Assessment of Myocardial Disarray, Fibrosis and Ventricular Mass in a Feline Model of Hypertrophic Cardiomyopathy. Sci. Rep. 2020, 10, 20169. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, C.M.; Zambelli, V.; Botta, G.; Moltrasio, F.; Cattoretti, G.; Lucchini, V.; Fesslova, V.; Cuttin, M.S. Postmortem Microcomputed Tomography (Micro-CT) of Small Fetuses and Hearts. Ultrasound Obstet. Gynecol. Off. J. Int. Soc. Ultrasound Obstet. Gynecol. 2014, 44, 600–609. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.J.; Francis, R.; Liu, X.; Devine, W.A.; Ramirez, R.; Anderton, S.J.; Wong, L.Y.; Faruque, F.; Gabriel, G.C.; Chung, W.; et al. Microcomputed Tomography Provides High Accuracy Congenital Heart Disease Diagnosis in Neonatal and Fetal Mice. Circ. Cardiovasc. Imaging 2013, 6, 551–559. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hutchinson, J.C.; Arthurs, O.J.; Ashworth, M.T.; Ramsey, A.T.; Mifsud, W.; Lombardi, C.M.; Sebire, N.J. Clinical Utility of Postmortem Microcomputed Tomography of the Fetal Heart: Diagnostic Imaging vs Macroscopic Dissection. Ultrasound Obstet. Gynecol. 2016, 47, 58–64. [Google Scholar] [CrossRef]
- Dunmore-Buyze, P.J.; Tate, E.; Xiang, F.L.; Detombe, S.A.; Nong, Z.; Pickering, J.G.; Drangova, M. Three-Dimensional Imaging of the Mouse Heart and Vasculature Using Micro-CT and Whole-Body Perfusion of Iodine or Phosphotungstic Acid. Contrast Media Mol. Imaging 2014, 9, 383–390. [Google Scholar] [CrossRef]
- Doost, A.; Rangel, A.; Nguyen, Q.; Morahan, G.; Arnolda, L. Micro-CT Scan with Virtual Dissection of Left Ventricle Is a Non-Destructive, Reproducible Alternative to Dissection and Weighing for Left Ventricular Size. Sci. Rep. 2020, 10, 13853. [Google Scholar] [CrossRef]
- Butters, T.D.; Castro, S.J.; Lowe, T.; Zhang, Y.; Lei, M.; Withers, P.J.; Zhang, H. Optimal Iodine Staining of Cardiac Tissue for X-Ray Computed Tomography. PLoS ONE 2014, 9, e105552. [Google Scholar] [CrossRef]
- Aslanidi, O.V.; Nikolaidou, T.; Zhao, J.; Smaill, B.H.; Gilbert, S.H.; Holden, A.V.; Lowe, T.; Withers, P.J.; Stephenson, R.S.; Jarvis, J.C.; et al. Application of Micro-Computed Tomography with Iodine Staining to Cardiac Imaging, Segmentation, and Computational Model Development. IEEE Trans. Med. Imaging 2013, 32, 8–17. [Google Scholar] [CrossRef]
- Stephenson, R.S.; Boyett, M.R.; Hart, G.; Nikolaidou, T.; Cai, X.; Corno, A.F.; Alphonso, N.; Jeffery, N.; Jarvis, J.C. Contrast Enhanced Micro-Computed Tomography Resolves the 3-Dimensional Morphology of the Cardiac Conduction System in Mammalian Hearts. PLoS ONE 2012, 7, 1–11. [Google Scholar] [CrossRef]
- Stephenson, R.S.; Atkinson, A.; Kottas, P.; Perde, F.; Jafarzadeh, F.; Bateman, M.; Iaizzo, P.A.; Zhao, J.; Zhang, H.; Anderson, R.H.; et al. High Resolution 3-Dimensional Imaging of the Human Cardiac Conduction System from Microanatomy to Mathematical Modeling. Sci. Rep. 2017, 7, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Stephenson, R.S.; Rowley-Nobel, J.; Jones, C.B.; Guerrero, R.; Lowe, T.; Zhao, J.; Zhang, H.; Jarvis, J.C. Morphological Substrates for Atrial Arrhythmogenesis in a Heart With Atrioventricular Septal Defect. Front. Physiol. 2018, 9, 1071. [Google Scholar] [CrossRef]
- Nierenberger, M.; Rémond, Y.; Ahzi, S.; Choquet, P. Assessing the Three-Dimensional Collagen Network in Soft Tissues Using Contrast Agents and High Resolution Micro-CT: Application to Porcine Iliac Veins. Comptes Rendus Biol. 2015, 338, 425–433. [Google Scholar] [CrossRef]
- Simcock, I.C.; Hutchinson, J.C.; Shelmerdine, S.C.; Matos, J.N.; Sebire, N.J.; Fuentes, V.L.; Arthurs, O.J. Investigation of Optimal Sample Preparation Conditions with Potassium Triiodide and Optimal Imaging Settings for Microfocus Computed Tomography of Excised Cat Hearts. Am. J. Vet. Res. 2020, 81, 326–333. [Google Scholar] [CrossRef]
- Sandrini, C.; Rossetti, L.; Zambelli, V.; Zanarotti, R.; Bettinazzi, F.; Soldá, R.; Di Pace, C.; Hoxha, S.; Ribichini, F.L.; Faggian, G.; et al. Accuracy of Micro-Computed Tomography in Post-Mortem Evaluation of Fetal Congenital Heart Disease. Comparison Between Post-Mortem Micro-CT and Conventional Autopsy. Front. Pediatr. 2019, 7, 92. [Google Scholar] [CrossRef]
- Midgett, M.; Thornburg, K.; Rugonyi, S. Blood Flow Patterns Underlie Developmental Heart Defects. Am. J. Physiol. Heart Circ. Physiol. 2017, 312, H632–H642. [Google Scholar] [CrossRef]
- Merchant, S.S.; Kosaka, Y.; Yost, H.J.; Hsu, E.W.; Brunelli, L. Micro-Computed Tomography for the Quantitative 3-Dimensional Assessment of the Compact Myocardium in the Mouse Embryo. Circ. J. 2016, 80, 1795–1803. [Google Scholar] [CrossRef] [Green Version]
- Rykiel, G.; López, C.S.; Riesterer, J.L.; Fries, I.; Deosthali, S.; Courchaine, K.; Maloyan, A.; Thornburg, K.; Rugonyi, S. Multiscale Cardiac Imaging Spanning the Whole Heart and Its Internal Cellular Architecture in a Small Animal Model. Elife 2020, 9. [Google Scholar] [CrossRef]
- Pai, V.M.; Kozlowski, M.; Donahue, D.; Miller, E.; Xiao, X.; Chen, M.Y.; Yu, Z.X.; Connelly, P.; Jeffries, K.; Wen, H. Coronary Artery Wall Imaging in Mice Using Osmium Tetroxide and Micro-Computed Tomography (Micro-CT). J. Anat. 2012, 220, 514–524. [Google Scholar] [CrossRef]
- Helfenstein-Didier, C.; Taïnoff, D.; Viville, J.; Adrien, J.; Maire, É.; Badel, P. Tensile Rupture of Medial Arterial Tissue Studied by X-Ray Micro-Tomography on Stained Samples. J. Mech. Behav. Biomed. Mater. 2018, 78, 362–368. [Google Scholar] [CrossRef]
- Brunet, J.; Pierrat, B.; Adrien, J.; Maire, E.; Curt, N.; Badel, P. A Novel Method for In Vitro 3D Imaging of Dissecting Pressurized Arterial Segments Using X-Ray Microtomography. Exp. Mech. 2020, 147–157. [Google Scholar] [CrossRef]
- Dunmore-Buyze, P.J.; Cruje, C.; Nong, Z.; Lee, J.J.; Kiernan, J.A.; Pickering, J.G.; Drangova, M. 3D Vessel-Wall Virtual Histology of Whole-Body Perfused Mice Using a Novel Heavy Element Stain. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Dullin, C.; Ufartes, R.; Larsson, E.; Martin, S.; Lazzarini, M.; Tromba, G.; Missbach-Guentner, J.; Pinkert-Leetsch, D.; Katschinski, D.M.; Alves, F. ΜCT of Ex-Vivo Stained Mouse Hearts and Embryos Enables a Precise Match between 3D Virtual Histology, Classical Histology and Immunochemistry. PLoS ONE 2017, 12. [Google Scholar] [CrossRef] [PubMed]
- De Clercq, K.; Persoons, E.; Napso, T.; Luyten, C.; Parac-Vogt, T.N.; Sferruzzi-Perri, A.N.; Kerckhofs, G.; Vriens, J. High-Resolution Contrast-Enhanced MicroCT Reveals the True Three-Dimensional Morphology of the Murine Placenta. Proc. Natl. Acad. Sci. USA 2019, 116, 13927–13936. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kerckhofs, G.; Stegen, S.; van Gastel, N.; Sap, A.; Falgayrac, G.; Penel, G.; Durand, M.; Luyten, F.P.; Geris, L.; Vandamme, K.; et al. Simultaneous Three-Dimensional Visualization of Mineralized and Soft Skeletal Tissues by a Novel MicroCT Contrast Agent with Polyoxometalate Structure. Biomaterials 2018, 159, 1–12. [Google Scholar] [CrossRef]
- De Bournonville, S.; Vangrunderbeeck, S.; Ly, H.G.T.; Geeroms, C.; De Borggraeve, W.M.; Parac-Vogt, T.N.; Kerckhofs, G. Exploring Polyoxometalates as Non-Destructive Staining Agents for Contrast-Enhanced Microfocus Computed Tomography of Biological Tissues. Acta Biomater. 2020, 105, 253–262. [Google Scholar] [CrossRef]
- Gonzalez-Tendero, A.; Zhang, C.; Balicevic, V.; Cárdenes, R.; Loncaric, S.; Butakoff, C.; Paun, B.; Bonnin, A.; Garcia-Cañadilla, P.; Muñoz-Moreno, E.; et al. Whole Heart Detailed and Quantitative Anatomy, Myofibre Structure and Vasculature from X-Ray Phase-Contrast Synchrotron Radiation-Based Micro Computed Tomography. Eur. Heart J. Cardiovasc. Imaging 2017, 18, 732–741. [Google Scholar] [CrossRef]
- Shinohara, G.; Morita, K.; Hoshino, M.; Ko, Y.; Tsukube, T.; Kaneko, Y.; Morishita, H.; Oshima, Y.; Matsuhisa, H.; Iwaki, R.; et al. Three Dimensional Visualization of Human Cardiac Conduction Tissue in Whole Heart Specimens by High-Resolution Phase-Contrast CT Imaging Using Synchrotron Radiation. World J. Pediatr. Congenit. Heart Surg. 2016, 7, 700–705. [Google Scholar] [CrossRef]
- Kaneko, Y.; Shinohara, G.; Hoshino, M.; Morishita, H.; Morita, K.; Oshima, Y.; Takahashi, M.; Yagi, N.; Okita, Y.; Tsukube, T. Intact Imaging of Human Heart Structure Using X-Ray Phase-Contrast Tomography. Pediatr. Cardiol. 2017, 38, 390–393. [Google Scholar] [CrossRef] [Green Version]
- Garcia-Canadilla, P.; Dejea, H.; Bonnin, A.; Balicevic, V.; Loncaric, S.; Zhang, C.; Butakoff, C.; Aguado-Sierra, J.; Vázquez, M.; Jackson, L.H.; et al. Complex Congenital Heart Disease Associated With Disordered Myocardial Architecture in a Midtrimester Human Fetus. Circ. Cardiovasc. Imaging 2018, 11, e007753. [Google Scholar] [CrossRef] [Green Version]
- Dejea, H.; Bonnin, A.; Cook, A.C.; Garcia-Canadilla, P. Cardiac Multi-Scale Investigation of the Right and Left Ventricle Ex Vivo: A Review. Cardiovasc. Diagn. Ther. 2020, 10, 1701–1717. [Google Scholar] [CrossRef]
- Zach, B.; Hofer, E.; Asslaber, M.; Ahammer, H. Automated Texture Analysis and Determination of Fibre Orientation of Heart Tissue: A Morphometric Study. PLoS ONE 2016, 11, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Mirea, I.; Varray, F.; Zhu, Y.M.; Fanton, L.; Langer, M.; Jouk, P.S.; Michalowicz, G.; Usson, Y.; Magnin, I.E. Very High-Resolution Imaging of Post-Mortem Human Cardiac Tissue Using X-Ray Phase Contrast Tomography BT—Functional Imaging and Modeling of the Heart; Van Assen, H., Bovendeerd, P., Delhaas, T., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 172–179. [Google Scholar]
- Reichardt, M.; Töpperwien, M.; Khan, A.; Alves, F.; Salditt, T. Fiber Orientation in a Whole Mouse Heart Reconstructed by Laboratory Phase-Contrast Micro-CT. J. Med. Imaging 2020, 7, 1. [Google Scholar] [CrossRef] [Green Version]
- Reichardt, M.; Frohn, J.; Khan, A.; Alves, F.; Salditt, T. Multi-Scale X-Ray Phase-Contrast Tomography of Murine Heart Tissue. Biomed. Opt. Express 2020, 11, 2633. [Google Scholar] [CrossRef]
- Varray, F.; Wang, L.; Fanton, L.; Zhu, Y.M.; Magnin, I.E. High Resolution Extraction of Local Human Cardiac Fibre Orientations. In International Conference on Functional Imaging and Modeling of the Heart; Springer: Berlin/Heidelberg, Germany, 2013; Volume 7945, pp. 150–157. [Google Scholar] [CrossRef]
- Varray, F.; Mirea, I.; Langer, M.; Peyrin, F.; Fanton, L.; Magnin, I.E. Extraction of the 3D Local Orientation of Myocytes in Human Cardiac Tissue Using X-Ray Phase-Contrast Micro-Tomography and Multi-Scale Analysis. Med. Image Anal. 2017, 38, 117–132. [Google Scholar] [CrossRef]
- Wang, S.; Mirea, I.; Varray, F.; Liu, W.; Magnin, I. Investigating the 3D Local Myocytes Arrangement in the Human LV Mid-Wall with the Transverse Angle. In International Conference on Functional Imaging and Modeling of the Heart; Springer: Cham, Switzerland, 2019; pp. 208–216. [Google Scholar] [CrossRef]
- Baličević, V.; Lončarić, S.; Cárdenes, R.; Gonzalez-Tendero, A.; Paun, B.; Crispi, F.; Butakoff, C.; Bijnens, B. Assessment of Myofiber Orientation in High Resolution Phase-Contrast CT Images. In Functional Imaging and Modeling of the Heart; Van Assen, H., Bovendeerd, P., Delhaas, T., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 111–119. [Google Scholar]
- Trayanova, N.A. Whole-Heart Modeling: Applications to Cardiac Electrophysiology and Electromechanics. Circ. Res. 2011, 108, 113–128. [Google Scholar] [CrossRef] [Green Version]
- Vadakkumpadan, F.; Arevalo, H.; Prassl, A.J.; Chen, J.; Kickinger, F.; Kohl, P.; Plank, G.; Trayanova, N. Image-Based Models of Cardiac Structure in Health and Disease. WIREs Syst. Biol. Med. 2010, 2, 489–506. [Google Scholar] [CrossRef] [Green Version]
- Creative Commons—Attribution 4.0 International—CC BY 4.0. Available online: https://creativecommons.org/licenses/by/4.0/ (accessed on 26 January 2021).
- Coté, N.; Mahmut, A.; Bosse, Y.; Couture, C.; Pagé, S.; Trahan, S.; Boulanger, M.C.; Fournier, D.; Pibarot, P.; Mathieu, P. Inflammation Is Associated with the Remodeling of Calcific Aortic Valve Disease. Inflammation 2013, 36, 573–581. [Google Scholar] [CrossRef]
- Chitsaz, S.; Gundiah, N.; Blackshear, C.; Tegegn, N.; Yan, K.S.; Azadani, A.N.; Hope, M.; Tseng, E.E. Correlation of Calcification on Excised Aortic Valves by Micro-Computed Tomography with Severity of Aortic Stenosis. J. Heart Valve Dis. 2012, 21, 320–327. [Google Scholar]
- Orzechowska, S.; Wróbel, A.; Goncerz, G.; Podolec, P.; Rokita, E. Physicochemical and Micro-Tomographic Characterization of Inorganic Deposits Associated with Aortic Stenosis. J. Heart Valve Dis. 2014, 23, 40–47. [Google Scholar]
- Mazur, P.; Wypasek, E.; Gawęda, B.; Sobczyk, D.; Kapusta, P.; Natorska, J.; Malinowski, K.P.; Tarasiuk, J.; Bochenek, M.; Wroński, S.; et al. Stenotic Bicuspid and Tricuspid Aortic Valves: Micro-Computed Tomography and Biological Indices of Calcification. Circ. J. 2017, 81, 1043–1050. [Google Scholar] [CrossRef] [Green Version]
- Solache-Berrocal, G.; Barral-Varela, A.M.; Areces-Rodríguez, S.; Junco-Vicente, A.; Vallina-Álvarez, A.; Corte-Torres, M.D.; Valdivielso, J.M.; Llosa, J.C.; Morís, C.; Martín, M.; et al. Correlation of Micro-Computed Tomography Assessment of Valvular Mineralisation with Histopathological and Immunohistochemical Features of Calcific Aortic Valve Disease. J. Clin. Med. 2019, 9, 29. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boulif, J.; Gerber, B.; Slimani, A.; Lazam, S.; de Meester, C.; Piérard, S.; Pasquet, A.; Pouleur, A.C.; Vancraeynest, D.; El Khoury, G.; et al. Assessment of Aortic Valve Calcium Load by Multidetector Computed Tomography. Anatomical Validation, Impact of Scanner Settings and Incremental Diagnostic Value. J. Cardiovasc. Comput. Tomogr. 2017, 11, 360–366. [Google Scholar] [CrossRef] [PubMed]
- Pierce, E.L.; Bloodworth, C.H.; Naran, A.; Easley, T.F.; Jensen, M.O.; Yoganathan, A.P. Novel Method to Track Soft Tissue Deformation by Micro-Computed Tomography: Application to the Mitral Valve. Ann. Biomed. Eng. 2016, 44, 2273–2281. [Google Scholar] [CrossRef] [PubMed]
- Toma, M.; Bloodworth, C.H.; Einstein, D.R.; Pierce, E.L.; Cochran, R.P.; Yoganathan, A.P.; Kunzelman, K.S. High-Resolution Subject-Specific Mitral Valve Imaging and Modeling: Experimental and Computational Methods. Biomech. Model. Mechanobiol. 2016, 15, 1619–1630. [Google Scholar] [CrossRef] [PubMed]
- Bloodworth Iv, C.H.; Pierce, E.L.; Easley, T.F.; Drach, A.; Khalighi, A.H.; Toma, M.; Jensen, M.O.; Sacks, M.S.; Yoganathan, A.P.; Biomed, A.; et al. Ex Vivo Methods for Informing Computational Models of the Mitral Valve HHS Public Access Author Manuscript. Ann. Biomed. Eng. 2017, 45, 496–507. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jorgensen, S.M.; Demirkaya, O.; Ritman, E.L.; Steven, M.; Demirkaya, O.; Erik, L. Special Communication. Spec. Commun. 1998, 275, 1103–1114. [Google Scholar]
- Van den Wijngaard, J.P.H.M.; Schwarz, J.C.V.; van Horssen, P.; van Lier, M.G.J.T.B.; Dobbe, J.G.G.; Spaan, J.A.E.; Siebes, M. 3D Imaging of Vascular Networks for Biophysical Modeling of Perfusion Distribution within the Heart. J. Biomech. 2013, 46, 229–239. [Google Scholar] [CrossRef]
- Heinzer, S.; Krucker, T.; Stampanoni, M.; Abela, R.; Meyer, E.P.; Schuler, A.; Schneider, P.; Müller, R. Hierarchical Microimaging for Multiscale Analysis of Large Vascular Networks. Neuroimage 2006, 32, 626–636. [Google Scholar] [CrossRef]
- Walker, E.J.; Shen, F.; Young, W.L.; Su, H. Cerebrovascular Casting of the Adult Mouse for 3D Imaging and Morphological Analysis. J. Vis. Exp. 2011. [Google Scholar] [CrossRef] [Green Version]
- Cao, Y.; Wu, T.; Yuan, Z.; Li, D.; Ni, S.; Hu, J.; Lu, H. Three-Dimensional Imaging of Microvasculature in the Rat Spinal Cord Following Injury. Sci. Rep. 2015, 5, 1–11. [Google Scholar] [CrossRef]
- Junaid, T.O.; Bradley, R.S.; Lewis, R.M.; Aplin, J.D.; Johnstone, E.D. Whole Organ Vascular Casting and MicroCT Examination of the Human Placental Vascular Tree Reveals Novel Alterations Associated with Pregnancy Disease. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef]
- Giuvǎrǎşteanu, I. Scanning Electron Microscopy of Vascular Corrosion Casts--Standard Method for Studying Microvessels. Rom. J. Morphol. Embryol. 2007, 48, 257–261. [Google Scholar]
- Olianti, C.; Costantini, I.; Giardini, F.; Lazzeri, E.; Crocini, C.; Ferrantini, C.; Pavone, F.S.; Camici, P.G.; Sacconi, L. 3D Imaging and Morphometry of the Heart Capillary System in Spontaneously Hypertensive Rats and Normotensive Controls. Sci. Rep. 2020, 10, 1–9. [Google Scholar] [CrossRef]
- Walton, L.A.; Bradley, R.S.; Withers, P.J.; Newton, V.L.; Watson, R.E.B.; Austin, C.; Sherratt, M.J. Morphological Characterisation of Unstained and Intact Tissue Micro-Architecture by X-Ray Computed Micro- and Nano-Tomography. Sci. Rep. 2015, 5, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Lloyd-Jones, D.; Adams, R.J.; Brown, T.M.; Carnethon, M.; Dai, S.; De Simone, G.; Ferguson, T.B.; Ford, E.; Furie, K.; Gillespie, C.; et al. Executive Summary: Heart Disease and Stroke Statistics-2010 Update: A Report from the American Heart Association. Circulation 2010, 121. [Google Scholar] [CrossRef]
- Postnov, A.A.; D’Haese, P.C.; Neven, E.; De Clerck, N.M.; Persy, V.P. Possibilities and Limits of X-Ray Microtomography for in Vivo and Ex Vivo Detection of Vascular Calcifications. Int. J. Cardiovasc. Imaging 2009, 25, 615–624. [Google Scholar] [CrossRef]
- Holme, M.N.; Schulz, G.; Deyhle, H.; Weitkamp, T.; Beckmann, F.; Lobrinus, J.A.; Rikhtegar, F.; Kurtcuoglu, V.; Zanette, I.; Saxer, T.; et al. Complementary X-Ray Tomography Techniques for Histology-Validated 3D Imaging of Soft and Hard Tissues Using Plaque-Containing Blood Vessels as Examples. Nat. Protoc. 2014, 9, 1401–1415. [Google Scholar] [CrossRef]
- Barrett, H.E.; Mulvihill, J.J.; Cunnane, E.M.; Walsh, M.T. Characterising Human Atherosclerotic Carotid Plaque Tissue Composition and Morphology Using Combined Spectroscopic and Imaging Modalities. Biomed. Eng. Online 2015, 14 (Suppl. 1), S5. [Google Scholar] [CrossRef] [Green Version]
- Self, T.S.; Ginn-Hedman, A.M.; Kaulfus, C.N.; Newell-Fugate, A.E.; Weeks, B.R.; Heaps, C.L. Iodine-Enhanced Micro-Computed Tomography of Atherosclerotic Plaque Morphology Complements Conventional Histology. Atherosclerosis 2020, 313, 43–49. [Google Scholar] [CrossRef]
- Borland, S.J.; Behnsen, J.; Ashton, N.; Francis, S.E.; Brennan, K.; Sherratt, M.J.; Withers, P.J.; Canfield, A.E. X-Ray Micro-Computed Tomography: An Emerging Technology to Analyze Vascular Calcification in Animal Models. Int. J. Mol. Sci. 2020, 21, 4538. [Google Scholar] [CrossRef]





| Technique | Pros | Cons | |
|---|---|---|---|
| MicroCT without contrast- or phase-enhancement | Attenuation of the X-rays by the tissues |
|
|
| Contrast-enhanced microCT (CE-CT) | Attenuation of the X-rays by the tissues and the CAs |
|
|
| Phase-contrast microCT (PC-CT) | Phase shift at interfaces between different tissue constituents |
|
|
| Casting Contrast Agents (CCAs) | |||
|---|---|---|---|
| Contrast Agent | Composition | Characteristics | References |
| Microfil | Lead-containing radiopaque silicone rubber |
| [5,7,8,9,10,11,36,38,41,42,43,44,45,46,47] |
| µAngiofil | Angiofil (liquid radio-contrast agent based on iodine), Orasol Blue dye, polyurethane resin, hardener |
| [48,49,50] |
| Barium sulfate | BaSO4 and gelatin suspension |
| [11,36,38,43,51,52,53] |
| Contrast-Enhancing Staining Agents (CESAs) | |||
| Contrast Agent | Composition | Characteristics | References |
| Lugol’s iodine | I3K (often referred to as I2KI) |
| [40,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70] |
| Osmium tetroxide | OsO4 |
| [71,72] |
| Sodium Polytungstate (SPT) (also referred to as sodium metatungstate) | H2Na6O40W12 |
| [73,74] |
| Modified Verhoef’s stain (Tri-element stain with iodine, aluminum, and iron) | 56% v/v aluminum hematoxylin, 2.2% w/v FeCl3•6H2O, 0.6% w/v KI and 0.3% w/v I2 |
| [75] |
| Phosphotungstic acid (PTA) Keggin polyoxometalate (POM) | H3PW12O40 |
| [59,66,76] |
| Phosphomolybdic acid (PMA) Keggin POM | H3PMo12O40 |
| [66] |
| Zirconium-substituted Keggin POM (Zr-K POM) Keggin POM | (Et2NH2)10[Zr(PW11O39)2] |
| [77] |
| 1:2 Hafnium-substituted Wells-Dawson POM (Hf-WD POM) Wells-Dawson POM | K15H[Hf(α2−P2W17O61)2] • 19 H20 | [78,79] | |
| Monolacunary-Wells-Dawson POM (mono-WD POM) Wells-Dawson POM | α2-K10P2W17O61 • 20H2O | [79] | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leyssens, L.; Pestiaux, C.; Kerckhofs, G. A Review of Ex Vivo X-ray Microfocus Computed Tomography-Based Characterization of the Cardiovascular System. Int. J. Mol. Sci. 2021, 22, 3263. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms22063263
Leyssens L, Pestiaux C, Kerckhofs G. A Review of Ex Vivo X-ray Microfocus Computed Tomography-Based Characterization of the Cardiovascular System. International Journal of Molecular Sciences. 2021; 22(6):3263. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms22063263
Chicago/Turabian StyleLeyssens, Lisa, Camille Pestiaux, and Greet Kerckhofs. 2021. "A Review of Ex Vivo X-ray Microfocus Computed Tomography-Based Characterization of the Cardiovascular System" International Journal of Molecular Sciences 22, no. 6: 3263. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms22063263





