Hepatitis B Virus Genotypes in the Kingdom of Bahrain: Prevalence, Gender Distribution and Impact on Hepatic Biomarkers
Abstract
:1. Introduction
2. Method
2.1. Setting
2.2. Study Design
2.3. Sample Collection, HBV DNA Extraction and Genotyping
2.4. Statistical Analysis
2.5. Ethical Approval
3. Results
3.1. Demographic Characteristics of Patients
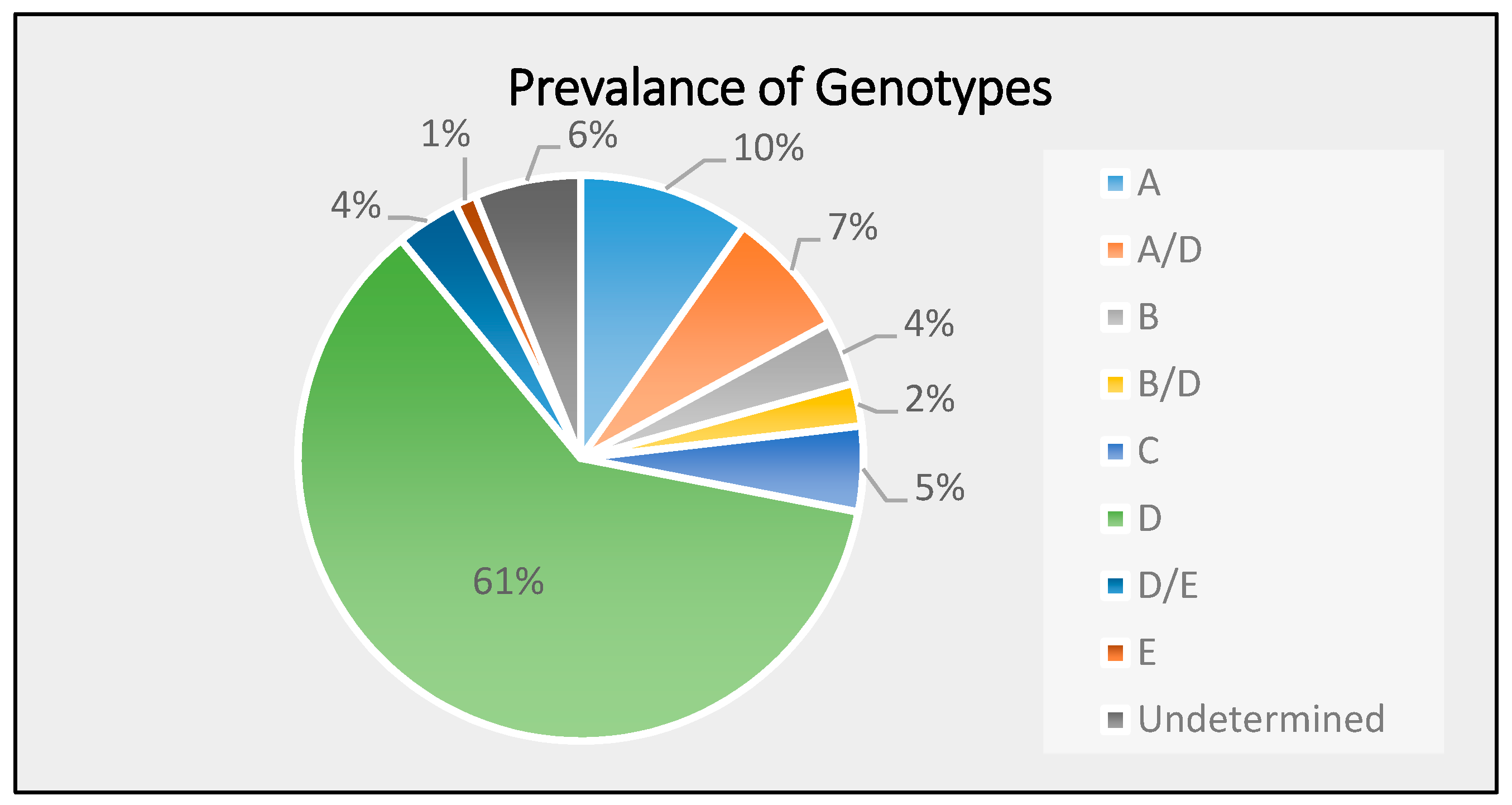
3.2. Distribution of HBV Genotypes in the Study Population
3.3. Genotype and Sociodemographic Data
3.4. HBV Genotype Relationship with Mode of Transmission and Liver Cirrhosis Complications
3.5. HBV Genotype and Liver Function Test
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kao, J.; Chen, P.; Lai, M.; Chen, D. Genotypes and Clinical Phenotypes of Hepatitis B Virus in Patients with Chronic Hepatitis B Virus Infection. J. Clin. Microbiol. 2002, 40, 1207–1209. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chan, H.; Wong, M.; Hui, A.; Hung, L.; Chan, F.; Sung, J. Hepatitis B Virus Genotype C Takes a More Aggressive Disease Course Than Hepatitis B Virus Genotype B in Hepatitis B e Antigen-Positive Patients. J. Clin. Microbiol. 2003, 41, 1277–1279. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ogawa, M. Clinical features and viral sequences of various genotypes of hepatitis B virus compared among patients with acute hepatitis B. Hepatol. Res. 2002, 23, 167–177. [Google Scholar] [CrossRef]
- Lavanchy, D. Hepatitis B virus epidemiology, disease burden, treatment, and current and emerging prevention and control measures. J. Viral Hepat. 2004, 11, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Nicoletta, P.; Daniel, L. Hepatitis B.; World Health Organization: Geneva, Switzerland, 2002. [Google Scholar]
- Zhang, X.; Zoulim, F.; Habersetzer, F.; Xiong, S.; Trépo, C. Analysis of hepatitis B virus genotypes and pre-core region variability during interferon treatment of HBe antigen negative chronic hepatitis B. J. Med Virol. 1996, 48, 8–16. [Google Scholar] [CrossRef]
- André, F. Hepatitis B epidemiology in Asia, the Middle East and Africa. Vaccine 2000, 18, S20–S22. [Google Scholar] [CrossRef]
- Jazayeri, M.; Basuni, A.; Sran, N.; Gish, R.; Cooksley, G.; Locarnini, S.; Carman, W. HBV core sequence: Definition of genotype-specific variability and correlation with geographical origin. J. Viral Hepat. 2004, 11, 488–501. [Google Scholar] [CrossRef]
- Stuyver, L.; Rossau, R.; Zoulim, F.; Fried, M.; De Gendt, S.; Schinazi, R.; Van Geyt, C. A new genotype of hepatitis B virus: Complete genome and phylogenetic relatedness. J. Gen. Virol. 2000, 81, 67–74. [Google Scholar] [CrossRef]
- Huy, T.; Abe, K. Molecular epidemiology of hepatitis B and C virus infections in Asia. Pediatr. Int. 2004, 46, 223–230. [Google Scholar] [CrossRef]
- Van Geyt, C.; De Gendt, S.; Rombout, A.; Wyseur, A.; Maertens, G.; Bartholomeusz, A.; Schinazi, R.F.; Locarnini, S.A. Significance of mutations in the hepatitis B virus polymerase selected by nucleoside analogues and implicationfor controlling chronic disease. Viral Hepat. Rev. 1998, 4, 167–187. [Google Scholar]
- Ding, X.; Mizokami, M.; Yao, G.; Xu, B.; Orito, E.; Ueda, R.; Nakanishi, M. Hepatitis B Virus Genotype Distribution among Chronic Hepatitis B Virus Carriers in Shanghai, China. Intervirology 2001, 44, 43–47. [Google Scholar] [CrossRef] [PubMed]
- Sugauchi, F.; Chutaputti, A.; Orito, E.; Kato, H.; Suzuki, S.; Ueda, R.; Mizokami, M. Hepatitis B virus genotypes and clinical manifestation among hepatitis B carriers in Thailand. J. Gastroenterol. Hepatol. 2002, 17, 671–676. [Google Scholar] [CrossRef] [PubMed]
- Magnius, L.; Norder, H. Subtypes, Genotypes and Molecular Epidemiology of the Hepatitis B Virus as Reflected by Sequence Variability of the S-Gene. Intervirology 1995, 38, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Arauz-Ruiz, P.; Norder, H.; Robertson, B.H.; Magnius, L.O. Genotype H: A new Amerindian genotype of hepatitis B virus revealed in Central America. J. Gen. Virol. 2002, 83, 2059–2073. [Google Scholar] [CrossRef] [PubMed]
- Liaw, Y.; Brunetto, M.; Hadziyannis, S. The natural history of chronic HBV infection and geographical differences. Antivir. Ther. 2010, 15, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Kramvis, A. Genotypes and Genetic Variability of Hepatitis B Virus. Intervirology 2014, 57, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Cabinet Affairs, Kingdom of Bahrain. Basic Results Population, Housing, Buildings and Establishment Census; Ministry of Cabinet Affairs, Kingdom of Bahrain: Manama, Bahrain, 2010. [Google Scholar]
- Miyakawa, Y.; Mizokami, M. Classifying Hepatitis B Virus Genotypes. Intervirology 2003, 46, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Qirbi, N.; Hall, A.J. Epidemiology of hepatitis B virus infection in the Middle East: A Review. La Revue de Sante de la Mediterranee Orientale 2001, 7, 1034–1045. [Google Scholar]
- Janahi, E. Prevalence and Risk Factors of Hepatitis B Virus Infection in Bahrain, 2000 through 2010. PLoS ONE 2014, 9, e87599. [Google Scholar] [CrossRef]
- Hui, C.; Lau, E.; Monto, A.; Kim, M.; Luk, J.; Poon, R.; Wright, T.L. Natural History of Patients with Recurrent Chronic Hepatitis C Virus and Occult Hepatitis B Co-Infection after Liver Transplantation. Am. J. Transplant. 2006, 6, 1600–1608. [Google Scholar] [CrossRef]
- Al Ashgar, H.; Peedikayil, M.; Imambaccus, H.; Althawadi, S. Prevalence of hepatitis B virus genotype in Saudi Arabia: A preliminary report. Indian J. Gastroenterol. 2008, 2008. [Google Scholar]
- Alfaresi, M.; Elkoush, A.; Alshehhi, H.; Alzaabi, A.; Islam, A. Hepatitis B virus genotypes and precore and core mutants in UAE patients. Virol. J. 2010, 7. [Google Scholar] [CrossRef] [PubMed]
- Alavian, S.M.; Fallahian, F.; Lankarani, K.B. The changing epidemiology of viral hepatitis B in Iran. J. Gastrointest. Liver Dis. 2007, 16, 403–406. [Google Scholar]
- Ghazzawi, I.; Hamoudi, M.; Hamoudi, W. Hepatitis B Genotypic and Serologic Characteristics in Jordan. J. R. Med. Serv. 2016, 23, 17–22. [Google Scholar] [CrossRef]
- Fung, S.K.; Wong, F.S.; Wong, D.K.; Hussain, M.T.; Lok, A.S. Hepatitis B virus genotypes, precore and core promoter variants among predominantly Asian patients with chronic HBV infection in a Canadian center. Liver Int. 2006, 26, 796–804. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McMahon, B. The influence of hepatitis B virus genotype and subgenotype on the natural history of chronic hepatitis B. Hepatol. Int. 2008, 3, 334–342. [Google Scholar] [CrossRef] [Green Version]
- Chen, C.; Hung, C.; Lee, C.; Hu, T.; Wang, J.H.; Wang, J.C.; Changchien, C.S. Pre-S Deletion and Complex Mutations of Hepatitis B Virus Related to Advanced Liver Disease in HBeAg-Negative Patients. Gastroenterology 2007, 133, 1466–1474. [Google Scholar] [CrossRef] [PubMed]
- Hadziyannis, S.J.; Papatheodoridis, G.V. Hepatitis B e Antigen-Negative Chronic Hepatitis B: Natural History and Treatment. Semin. Liver Dis. 2006, 26, 130–141. [Google Scholar] [CrossRef]
- Limdi, J.K.; Hyde, G.M. Evaluation of abnormal liver function tests. Postgrad. Med. J. 2003, 79, 307–312. [Google Scholar] [CrossRef] [Green Version]
- Giboney, P.T. Mildly elevated liver transaminase levels in the asymptomatic patient. Am. Fam. Phys. 2005, 71, 1105–1110. [Google Scholar]
| Variables | No. of Isolation 1 | p-Value |
|---|---|---|
| Gender | 0.447 | |
| Male | 58.5 (48) | |
| Female | 41.5 (34) | |
| Nationality | 0.44 | |
| Bahraini | 53.7 (44) | |
| Non-Bahraini | 46.3 (38) | |
| Age-group | 0.409 | |
| <21 | 4.9 (4) | |
| 21–30 | 28 (23) | |
| 31–40 | 25.6 (21) | |
| 41–50 | 13.4(11) | |
| 51–60 | 15.9(13) | |
| >61 | 12.2(10) |
| Gender | Nationality | Age-Group | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Genotype | Male (48) | Female (34) | Bahraini (44) | Non-Bahraini (38) | <20 (4) | 21–30 (23) | 31–40 (21) | 41–50 (11) | 51–60 (13) | >61 (10) |
| A | 62.5% | 37.5% | 12.5% | 87.5% | 0.0% | 25.0% | 25.0% | 25.0% | 12.5% | 12.5% |
| A/D | 66.7% | 33.3% | 66.7% | 33.3% | 0.0% | 83.3% | 16.7% | 0.0% | 0.0% | 0.0% |
| B | 66.7% | 33.3% | 33.3% | 66.7% | 0.0% | 0.0% | 33.3% | 0.0% | 33.3% | 33.3% |
| B/D | 50.0% | 50.0% | 50.0% | 50.0% | 0.0% | 0.0% | 100.0% | 0.0% | 0.0% | 0.0% |
| C | 100.0% | 0.0% | 25.0% | 75.0% | 0.0% | 25.0% | 50.0% | 25.0% | 0.0% | 0.0% |
| D | 54.0% | 46.0% | 64.0% | 36.0% | 8.0% | 22.0% | 20.0% | 14.0% | 22.0% | 14.0% |
| D/E | 100.0% | 0.0% | 0.0% | 100.0% | 0.0% | 100.0% | 0.0% | 0.0% | 0.0% | 0.0% |
| E | 0.0% | 100.0% | 0.0% | 100.0% | 0.0% | 100.0% | 0.0% | 0.0% | 0.0% | 0.0% |
| Undetermined | 40.0% | 60.0% | 80.0% | 20.0% | 0.0% | 0.0% | 60.0% | 20.0% | 0.0% | 20.0% |
| Mode of Transmission | Liver Cirrhosis | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Genotype | Unknown (49) | Blood/Blood Products (16) | Sexual (6) | Maternal/Vertical (6) | Organ Transplant (3) | None (63) | Ascites (13) | Portal HTn (2) | HCC (2) | Hepatic Encephalopathy (2) |
| A | 37.50% | 12.50% | 25.00% | 12.50% | 0.00% | 50.00% | 37.50% | 12.50% | 0.00% | 0.00% |
| A/D | 66.70% | 16.70% | 0.00% | 0.00% | 16.70% | 66.70% | 0.00% | 0.00% | 16.70% | 16.70% |
| B | 0.00% | 66.70% | 0.00% | 0.00% | 33.30% | 100.00% | 0.00% | 0.00% | 0.00% | 0.00% |
| B/D | 50.00% | 0.00% | 50.00% | 0.00% | 0.00% | 50.00% | 50.00% | 0.00% | 0.00% | 0.00% |
| C | 25.00% | 75.00% | 0.00% | 0.00% | 0.00% | 100.00% | 0.00% | 0.00% | 0.00% | 0.00% |
| D | 66.00% | 16.00% | 6.00% | 8.00% | 2.00% | 78.00% | 16.00% | 2.00% | 2.00% | 2.00% |
| D/E | 100.00% | 0.00% | 0.00% | 0.00% | 0.00% | 100.00% | 0.00% | 0.00% | 0.00% | 0.00% |
| E | 0.00% | 0.00% | 0.00% | 100.00% | 0.00% | 100.00% | 0.00% | 0.00% | 0.00% | 0.00% |
| Undetermined | 80.00% | 20.00% | 0.00% | 0.00% | 0.00% | 80.00% | 20.00% | 0.00% | 0.00% | 0.00% |
| Genotypes | Bilirubin (umol/L) | Dbilirubin (umol/L) | AST (lU/L) | ALT (lU/L) | GGT (lU/L) |
|---|---|---|---|---|---|
| A | 14.39 (±3.65) | 6.94 (±2.81) | 44.04 * (±3.78) | 49.54 * (±8.77) | 65.75 * (±23.22) |
| A/D | 25.10 * (±8.48) | 12.78 (±4.79) | 77.43 * (±3.68) | 94.23 * (±8.89) | 65.33 * (±28.07) |
| B | 16.63 (±12.35) | 8.97 (±6.77) | 35.40 (±14.61) | 63.93 * (±38.93) | 60.00 * (±39.68) |
| B/D | 16.35 (±11.05) | 10.55 (±8.35) | 69.40 * (±0.00) | 86.90 * (±69.80) | 76.00 * (±67.00) |
| C | 6.68 (±2.18) | 1.93 (±0.39) | 20.45 (±3.15) | 21.53 (±5.97) | 41.25 (±15.26) |
| D | 13.26 (±1.51) | 6.09 (±0.62) | 31.76 (±3.05) | 45.89 * (±6.60) | 63.36 * (±2.68) |
| D/E | 7.70 (±2.99) | 3.30 (±1.14) | 24.93 (±3.58) | 33.53 (±6.63) | 25.33 (± 7.88) |
| E | 3.20 (±0.00) | 1.50 (±0.00) | 37.00 * (±0.00) | 44.50 * (±0.00) | 15.00 (±0.00) |
| Undetermined | 7.14 (±1.41) | 2.50 (±0.25) | 18.80 (±2.69) | 21.32 (±4.45) | 18.00 (±4.06) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Janahi, E.M.; Ilyas, Z.; Al-Othman, S.; Darwish, A.; Sanad, S.J.; Almusaifer, B.; Al-Mannai, M.; Golbahar, J.; Perna, S. Hepatitis B Virus Genotypes in the Kingdom of Bahrain: Prevalence, Gender Distribution and Impact on Hepatic Biomarkers. Medicina 2019, 55, 622. https://0-doi-org.brum.beds.ac.uk/10.3390/medicina55100622
Janahi EM, Ilyas Z, Al-Othman S, Darwish A, Sanad SJ, Almusaifer B, Al-Mannai M, Golbahar J, Perna S. Hepatitis B Virus Genotypes in the Kingdom of Bahrain: Prevalence, Gender Distribution and Impact on Hepatic Biomarkers. Medicina. 2019; 55(10):622. https://0-doi-org.brum.beds.ac.uk/10.3390/medicina55100622
Chicago/Turabian StyleJanahi, Essam M., Zahra Ilyas, Sara Al-Othman, Abdulla Darwish, Sanad J. Sanad, Budoor Almusaifer, Mariam Al-Mannai, Jamal Golbahar, and Simone Perna. 2019. "Hepatitis B Virus Genotypes in the Kingdom of Bahrain: Prevalence, Gender Distribution and Impact on Hepatic Biomarkers" Medicina 55, no. 10: 622. https://0-doi-org.brum.beds.ac.uk/10.3390/medicina55100622





