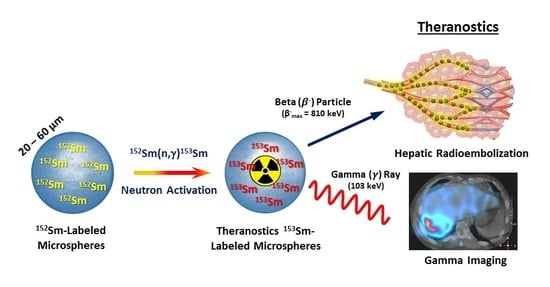
Preparation and In Vitro Evaluation of Neutron-Activated, Theranostic Samarium-153-Labeled Microspheres for Transarterial Radioembolization of Hepatocellular Carcinoma and Liver Metastasis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Samarium-152 Labeled Microspheres
2.2. Preparation of Samarium-152 Carbonate Labeled Microspheres
2.3. Neutron Activation of 152Sm- and 152SmC-Labeled Microspheres
2.4. Gamma Spectrometry
2.5. Physicochemical Characterization of Sm- and SmC-Labeled Microspheres
2.5.1. Field Emission Scanning Electron Microscopy and Energy Dispersive X-ray
2.5.2. Particle Size Analyzer
2.5.3. Fourier Transform Infrared (FTIR) Spectroscopy
2.5.4. Density Measurements
2.5.5. Viscosity Measurements
2.6. In Vitro Stability Test of the 153Sm- and 153SmC-Labeled Microspheres
2.7. Statistical Analysis
3. Results
3.1. Determination of Optimum Neutron Activation Protocol for 152Sm- and 152Smc-Labeled Microspheres
3.2. Gamma Spectrometry of 153Sm- and 153Smc-Labeled Microspheres
3.3. Effects of Neutron Activation on the Physicochemical Characteristics of 152Sm- and 152Smc-Labeled Microspheres
3.3.1. Field Emission Scanning Electron Microscopy and Energy Dispersive X-ray
3.3.2. Particle Size Analyzer
3.3.3. FTIR Spectroscopy
3.3.4. Density Measurement
3.3.5. Viscosity Measurement
3.4. In Vitro Stability Test of the 153Sm- and 153Smc-Labeled Microspheres
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- De Baere, T.; Arai, Y.; Lencioni, R.; Geschwind, J.-F.; Rilling, W.; Salem, R.; Matsui, O.; Soulen, M.C. Treatment of Liver Tumors with Lipiodol TACE: Technical Recommendations from Experts Opinion. Cardiovasc. Intervent. Radiol. 2016, 39, 334–343. [Google Scholar] [CrossRef] [PubMed]
- Bruix, J.; Sherman, M. Management of hepatocellular carcinoma. Hepatology 2005, 42, 1208–1236. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M.; Burroughs, A.; Bruix, J. Hepatocellular carcinoma. Lancet 2003, 362, 1907–1917. [Google Scholar] [CrossRef]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- El-Serag, H.B. Hepatocellular carcinoma. N. Engl. J. Med. 2011, 365, 1118–1127. [Google Scholar] [CrossRef] [PubMed]
- Riihimäki, M.; Thomsen, H.; Hemminki, A.; Sundquist, K.; Hemminki, K. Comparison of survival of patients with metastases from known versus unknown primaries: survival in metastatic cancer. BMC Cancer 2013, 13, 36. [Google Scholar] [CrossRef] [PubMed]
- Jemal, A.; Siegel, R.; Ward, E.; Hao, Y.; Xu, J.; Thun, M.J. Cancer statistics, 2009. CA Cancer J. Clin. 2009, 59, 225–249. [Google Scholar] [CrossRef] [PubMed]
- Tsoulfas, G.; Pramateftakis, M.G.; Kanellos, I. Surgical treatment of hepatic metastases from colorectal cancer. World. J. Gastrointest. Oncol. 2011, 3, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Bruix, J.; Reig, M.; Sherman, M. Evidence-Based Diagnosis, Staging, and Treatment of Patients With Hepatocellular Carcinoma. Gastroenterology 2016, 150, 835–853. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, S.; Vimalnath, K.; Dash, A. Preparation and preliminary in vivo evaluation of 166Ho-labeled microspheres for possible use in radioembolic therapy of liver cancer. J. Labelled Compd. Radiopharmaceut. 2018, 61, 509–514. [Google Scholar] [CrossRef] [PubMed]
- Subramaniam, S.; Kelley, R.K.; Venook, A.P. A review of hepatocellular carcinoma (HCC) staging systems. Chin. Clin. Oncol. 2013, 2. [Google Scholar]
- Khajornjiraphan, N.; Thu, N.A.; Chow, P.K.H. Yttrium-90 microspheres: a review of its emerging clinical indications. Liver Cancer 2015, 4, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Kallini, J.R.; Gabr, A.; Salem, R.; Lewandowski, R.J. Transarterial radioembolization with yttrium-90 for the treatment of hepatocellular carcinoma. Adv. Ther. 2016, 33, 699–714. [Google Scholar] [CrossRef] [PubMed]
- Salem, R.; Thurston, K.G. Radioembolization with Yttrium-90 Microspheres: A State-of-the-Art Brachytherapy Treatment for Primary and Secondary Liver Malignancies: Part 3: Comprehensive Literature Review and Future Direction. J. Vasc. Interv. Radiol. 2006, 17, 1571–1593. [Google Scholar] [CrossRef] [PubMed]
- Kao, Y.-H.; Steinberg, J.D.; Tay, Y.-S.; Lim, G.K.; Yan, J.; Townsend, D.W.; Takano, A.; Burgmans, M.C.; Irani, F.G.; Teo, T.K.; et al. Post-radioembolization yttrium-90 PET/CT-part 1: diagnostic reporting. EJNMMI Res. 2013, 3, 56. [Google Scholar] [CrossRef] [PubMed]
- Pasciak, A.S.; Bourgeois, A.C.; McKinney, J.M.; Chang, T.T.; Osborne, D.R.; Acuff, S.N.; Bradley, Y.C. Radioembolization and the Dynamic Role of (90)Y PET/CT. Front. Oncol. 2014, 4. [Google Scholar] [CrossRef] [PubMed]
- Lopci, E.; Chiti, A.; Castellani, M.R.; Pepe, G.; Antunovic, L.; Fanti, S.; Bombardieri, E. Matched pairs dosimetry: 124I/131I metaiodobenzylguanidine and 124I/131I and 86Y/90Y antibodies. Eur. J. Nucl. Med. Mol. Imaging 2011, 38, 28. [Google Scholar] [CrossRef] [PubMed]
- Nayak, T.K.; Brechbiel, M.W. 86Y based PET radiopharmaceuticals: radiochemistry and biological applications. Med. Chem. 2011, 7, 380–388. [Google Scholar] [CrossRef] [PubMed]
- Walrand, S.; Flux, G.D.; Konijnenberg, M.W.; Valkema, R.; Krenning, E.P.; Lhommel, R.; Pauwels, S.; Jamar, F. Dosimetry of yttrium-labelled radiopharmaceuticals for internal therapy: 86Y or 90Y imaging? Eur. J. Nucl. Med. Mol. I. 2011, 38, 57–68. [Google Scholar] [CrossRef] [PubMed]
- Riaz, A.; Awais, R.; Salem, R. Side Effects of Yttrium-90 Radioembolization. Front. Oncol. 2014, 4, 198. [Google Scholar] [CrossRef] [PubMed]
- Koch, W.; Tatsch, K. Nuclear Medicine Procedures for Treatment Evaluation. In Liver Radioembolization with 90Y Microspheres, Bilbao, J.I., Reiser, M.F., Eds.; Springer Berlin Heidelberg: Berlin, Heidelberg, 2008; pp. 75–91. [Google Scholar] [CrossRef]
- Gupta, T.; Virmani, S.; Neidt, T.M.; Szolc-Kowalska, B.; Sato, K.T.; Ryu, R.K.; Lewandowski, R.J.; Gates, V.L.; Woloschak, G.E.; Salem, R.; et al. MR Tracking of Iron-labeled Glass Radioembolization Microspheres during Transcatheter Delivery to Rabbit VX2 Liver Tumors: Feasibility Study. Radiology 2008, 249, 845–854. [Google Scholar] [CrossRef] [PubMed]
- Qaim Syed, M. Therapeutic radionuclides and nuclear data. Radiochim. Acta. 2001, 89, 297–304. [Google Scholar]
- Eary, J.F.; Collins, C.; Stabin, M.; Vernon, C.; Petersdorf, S.; Baker, M.; Hartnett, S.; Ferency, S.; Addison, S.J.; Appelbaum, F.; et al. Samarium-153-EDTMP biodistribution and dosimetry estimation. J. Nucl. Med. 1993, 34, 1031–1036. [Google Scholar] [PubMed]
- Forughi, S.; Hamidi, S.; Khalafi, H.; Sheibani, S.; Shahidi, A. Production of medical radioisotope 153Sm in the Tehran Research Reactor (TRR) through theoretical calculations and practical tests. Ann. Nucl. Energy 2013, 57, 16–21. [Google Scholar] [CrossRef]
- Ramamoorthy, N.; Saraswathy, P.; Das, M.K.; Mehra, K.S.; Ananthakrishnan, M. Production logistics and radionuclidic purity aspects of 153Sm for radionuclide therapy. Nucl. Med. Commun. 2002, 23, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Yeong, C.-H.; Abdullah, B.J.J.; Ng, K.-H.; Chung, L.-Y.; Goh, K.-L.; Sarji, S.A.; Perkins, A.C. Neutron-activated 153Sm-ion-exchange resin as a tracer for gastrointestinal scintigraphy. Nucl. Med. Commun. 2011, 32, 1256–1260. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yeong, C.-H.; Abdullah, B.J.J.; Ng, K.-H.; Chung, L.-Y.; Goh, K.-L.; Sarji, S.A.; Perkins, A.C. Production and first use of 153SmCl3-ion exchange resin capsule formulation for assessing gastrointestinal motility. Appl. Radiat. Isot. 2012, 70, 450–455. [Google Scholar] [CrossRef] [PubMed]
- Yeong, C.-H.; Abdullah, B.J.J.; Ng, K.-H.; Chung, L.-Y.; Goh, K.-L.; Perkins, A.C. Fusion of gamma scintigraphic and magnetic resonance images improves the anatomical delineation of radiotracer for the assessment of gastrointestinal transit. Nucl. Med. Commun. 2013, 34, 645–651. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hashikin, N.A.A.; Yeong, C.-H.; Abdullah, B.J.J.; Ng, K.-H.; Chung, L.-Y.; Dahalan, R.; Perkins, A.C. Neutron Activated Samarium-153 Microparticles for Transarterial Radioembolization of Liver Tumour with Post-Procedure Imaging Capabilities. PLoS ONE 2015, 10, e0138106. [Google Scholar] [CrossRef] [PubMed]
- Hashikin, N.A.A.; Yeong, C.H.; Abdullah, B.J.J.; Ng, K.H.; Chung, L.Y.; Dahalan, R.; Perkins, A.C. Samarium-153 Labelled Microparticles For Targeted Radionuclide Therapy Of Liver Tumor. In Proceedings of the World Congress on Medical Physics and Biomedical Engineering, Toronto, ON, Canada, 7–12 June 2015; Springer: Cham, Switzerland, 2009; pp. 471–474. [Google Scholar]
- Hashikin, N.A.A.; Yeong, C.H.; Guatelli, S.; Abdullah, B.J.J.; Ng, K.H.; Malaroda, A.; Rosenfeld, A.B.; Perkins, A.C. Organ doses from hepatic radioembolization with90Y,153Sm,166Ho and177Lu: A Monte Carlo simulation study using Geant4. J. Phys. Conf. Ser. 2016, 694, 012059. [Google Scholar] [CrossRef] [Green Version]
- Kim, D.-H.; Chen, J.; Omary, R.A.; Larson, A.C. MRI Visible Drug Eluting Magnetic Microspheres for Transcatheter Intra-Arterial Delivery to Liver Tumors. Theranostics 2015, 5, 477–488. [Google Scholar] [CrossRef] [PubMed]
- Hubicki, Z.; Kołodyńska, D. Selective removal of heavy metal ions from waters and waste waters using ion exchange methods. In Ion Exchange Technologies; Springer Netherlands: Dordrecht, Netherlands, 2012; pp. 193–240. [Google Scholar]
- Elder, D.P. Pharmaceutical Applications of Ion-Exchange Resins. J. Chem. Educ. 2005, 82, 575. [Google Scholar] [CrossRef]
- Fazal Ur, R.; Khan, S.N. Therapeutic Applications of Ion Exchange Resins. In Ion Exchange Technology II: Applications; Inamuddin, D., Luqman, M., Eds.; Springer Netherlands: Dordrecht, Netherlands, 2012; pp. 149–168. [Google Scholar] [CrossRef]
- Fazal Ur, R.; Khan, S.N. Application of Ion Exchange Resins in Kidney Dialysis. In Ion Exchange Technology II: Applications; Inamuddin, D., Luqman, M., Eds.; Springer Netherlands: Dordrecht Netherlands, 2012; pp. 169–182. [Google Scholar] [CrossRef]
- Guo, X.; Chang, R.K.; Hussain, M.A. Ion-exchange resins as drug delivery carriers. J. Pharm. Sci. 2009, 98, 3886–3902. [Google Scholar] [CrossRef] [PubMed]
- Schubiger, P.A.; Beer, H.F.; Geiger, L.; Rösler, H.; Zimmermann, A.; Triller, J.; Mettler, D.; Schilt, W. 90Y-resin particles—Animal experiments on pigs with regard to the introduction of superselective embolization therapy. Int. J. Radiat. Appl. Instrum. Part B 1991, 18, 305–311. [Google Scholar] [CrossRef]
- Turner, J.; Claringbold, P.; Klemp, P.; Cameron, P.; Martindale, A.; Glancy, R.; Norman, P.; Hetherington, E.; Najdovski, L.; Lambrecht, R. 166Ho-microsphere liver radiotherapy: A preclinical SPECT dosimetry study in the pig. Nucl. Med. Commun. 1994, 15, 545–553. [Google Scholar] [CrossRef] [PubMed]
- Dormandy, J.A. Clinical significance of blood viscosity. Ann. R. Coll. Surg. Engl. 1970, 47, 211–228. [Google Scholar] [PubMed]
- Mumper, R.J.; Ryo, U.Y.; Jay, M. Neutron-activated holmium-166-poly (l-lactic acid) microspheres: a potential agent for the internal radiation therapy of hepatic tumors. J. Nucl. Med. 1991, 32, 2139–2143. [Google Scholar] [PubMed]
- Poorbaygi, H.; Reza Aghamiri, S.M.; Sheibani, S.; Kamali-asl, A.; Mohagheghpoor, E. Production of glass microspheres comprising 90Y and 177Lu for treating of hepatic tumors with SPECT imaging capabilities. Appl. Radiat. Isot. 2011, 69, 1407–1414. [Google Scholar] [CrossRef] [PubMed]
- Häfeli, U.O.; Casillas, S.; Dietz, D.W.; Pauer, G.J.; Rybicki, L.A.; Conzone, S.D.; Day, D.E. Hepatic tumor radioembolization in a rat model using radioactive rhenium (186Re/188Re) glass microspheres. Int. J. Radiat. Oncol. Biol. Phys. 1999, 44, 189–199. [Google Scholar] [CrossRef]
- Gray, B. Polymer based radionuclide containing particulate material. U.S. Patent Application 10/173,496, 9 January 2003. [Google Scholar]
- Arranja, A.G.; Hennink, W.E.; Denkova, A.G.; Hendrikx, R.W.A.; Nijsen, J.F.W. Radioactive holmium phosphate microspheres for cancer treatment. Int. J. Pharm. 2018, 548, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Nijsen, J.F.W.; Zonnenberg, B.A.; Woittiez, J.R.W.; Rook, D.W.; Swildens-van Woudenberg, I.A.; van Rijk, P.P.; van het Schip, A.D. Holmium-166 poly lactic acid microspheres applicable for intra-arterial radionuclide therapy of hepatic malignancies: effects of preparation and neutron activation techniques. Eur. J. Nucl. Med. 1999, 26, 699–704. [Google Scholar] [CrossRef] [PubMed]
- International Atomic Energy Agency. Research Reactor Database (RRDB). International Atomic Energy Agency. Available online: https://nucleus.iaea.org/RRDB/RR/ReactorSearch.aspx?rf=1 (accessed on 1 August 2019).








| Base Bead | Hydroxylated Methacrylic (HW)-65 |
|---|---|
| Functional group | Sulfopropyl (S) strong cation exchange groups |
| Pore size (mean) | 100 nm |
| Particle size (mean) | 35 µm |
| Ligand type | strong cation |
| Ligand pKa | 1.2 |
| Dynamic binding capacity (DBC) | >150 g/L |
| Pressure rating | 0.3 MPa |
| Shipping buffer | 20% ethanol |
| pH stability | 3–13 |
| Shelf life (estimated) | 10 years |
| Parameters | Pneumatic Transfer System (PTS) | Rotary Rack (RR) |
|---|---|---|
| Thermal Neutron Flux, θth (n·cm−2·s−1) | 5.0 × 1012 | 2.0 × 1012 |
| Irradiation Time | Maximum 5 min | Maximum 6 h |
| Irradiation Location | Near to the Core | Peripheral to the Core |
| Sample Delivery | Automatic | Manual |
| Physicochemical Properties | 153Sm-labeled Microspheres | 153SmC-labeled Microspheres |
|---|---|---|
| Mean size (µm) | 35.70 ± 0.15 | 35.63 ± 0.16 |
| Density (g·cm−3) | 1.3681 ± 0.0009 | 1.3689 ± 0.0005 |
| Viscosity of 2.5% (w/v) microspheres suspension at 37 °C (g·cm−1·s−1) | 0.0116 ± 0.00003 | 0.0125 ± 0.0003 |
| Particle concentration (number of particles per g) | 30,677,684 | 30,858,851 |
| Specific activity (GBq·g−1) | 2.53 ± 0.08 | 2.40 ± 0.13 |
| Activity per microsphere (Bq) | 82.47 ± 2.60 | 77.77 ± 4.2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wong, Y.H.; Tan, H.Y.; Kasbollah, A.; Abdullah, B.J.J.; Yeong, C.H. Preparation and In Vitro Evaluation of Neutron-Activated, Theranostic Samarium-153-Labeled Microspheres for Transarterial Radioembolization of Hepatocellular Carcinoma and Liver Metastasis. Pharmaceutics 2019, 11, 596. https://0-doi-org.brum.beds.ac.uk/10.3390/pharmaceutics11110596
Wong YH, Tan HY, Kasbollah A, Abdullah BJJ, Yeong CH. Preparation and In Vitro Evaluation of Neutron-Activated, Theranostic Samarium-153-Labeled Microspheres for Transarterial Radioembolization of Hepatocellular Carcinoma and Liver Metastasis. Pharmaceutics. 2019; 11(11):596. https://0-doi-org.brum.beds.ac.uk/10.3390/pharmaceutics11110596
Chicago/Turabian StyleWong, Yin How, Hun Yee Tan, Azahari Kasbollah, Basri Johan Jeet Abdullah, and Chai Hong Yeong. 2019. "Preparation and In Vitro Evaluation of Neutron-Activated, Theranostic Samarium-153-Labeled Microspheres for Transarterial Radioembolization of Hepatocellular Carcinoma and Liver Metastasis" Pharmaceutics 11, no. 11: 596. https://0-doi-org.brum.beds.ac.uk/10.3390/pharmaceutics11110596