Lactobacillus reuteri DSM 17938 Protects against Gastric Damage Induced by Ethanol Administration in Mice: Role of TRPV1/Substance P Axis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Animals
2.3. Experimental Design on the Ethanol-Induced Gastric Mucosal Damage
2.4. Evaluation of the Role TRPV1 and SP in Mediating the Protective Effects of DSM on Ethanol-Induced Gastric Mucosal Damage
2.5. Histopathological Analysis of Gastric Damage
2.6. TRPV1 Immunohistochemistry in Gastric Tissue
2.7. Immunohistochemical Analysis
2.8. Enzyme-Linked Immunosorbent Assay (ELISA) for SP
2.9. Malondialdehyde Levels
2.10. Nitrate/Nitrite Levels in Gastric Tissue
2.11. Reduced Glutathione Assay
2.12. Superoxide Dismutase Assay
2.13. Gastric Wall Mucus
2.14. Gastric Acid Secretion
2.15. Statistic Analysis
3. Results
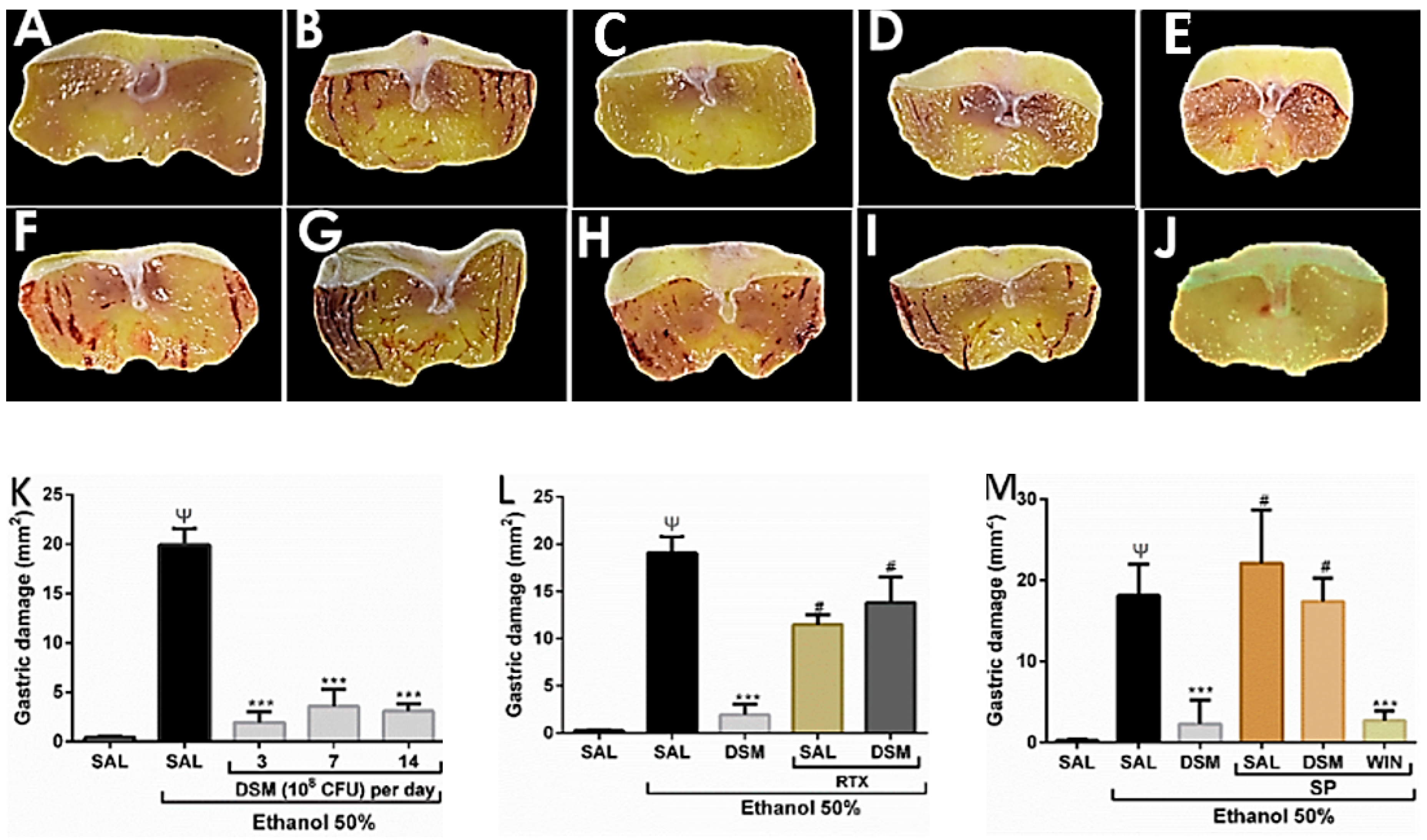
3.1. Effect of DSM on Ethanol-Induced Gastric Damag
3.2. Evaluation of the Possible role TRPV1 and NK1 Receptor in the Protective Effects of DSM on Ethanol-Induced Gastric Injury
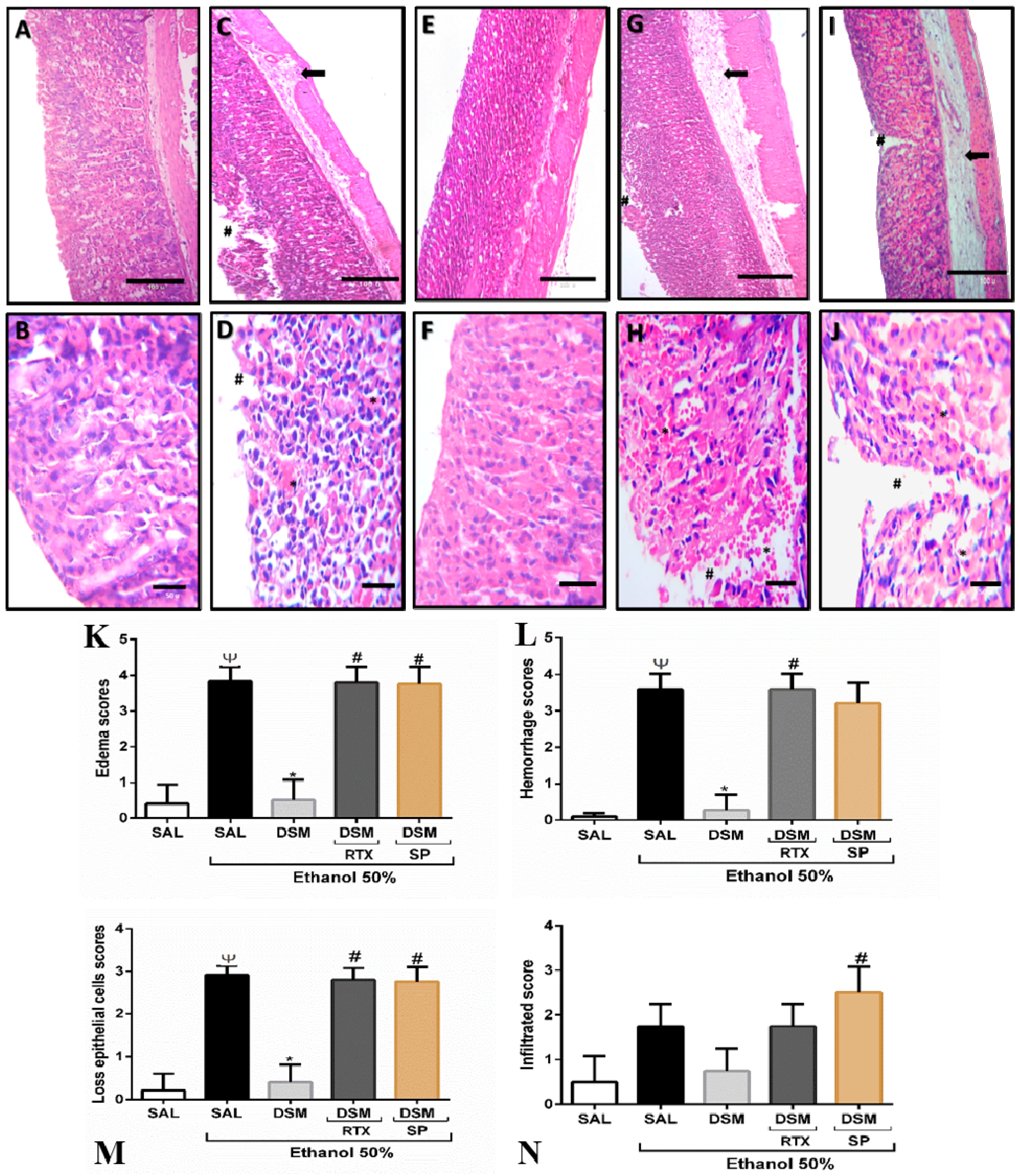
3.3. Histopathological Parameters of DSM on Ethanol-Induced Gastric Damage
3.4. Immunohistochemistry Detection of the TRPV1 Receptor
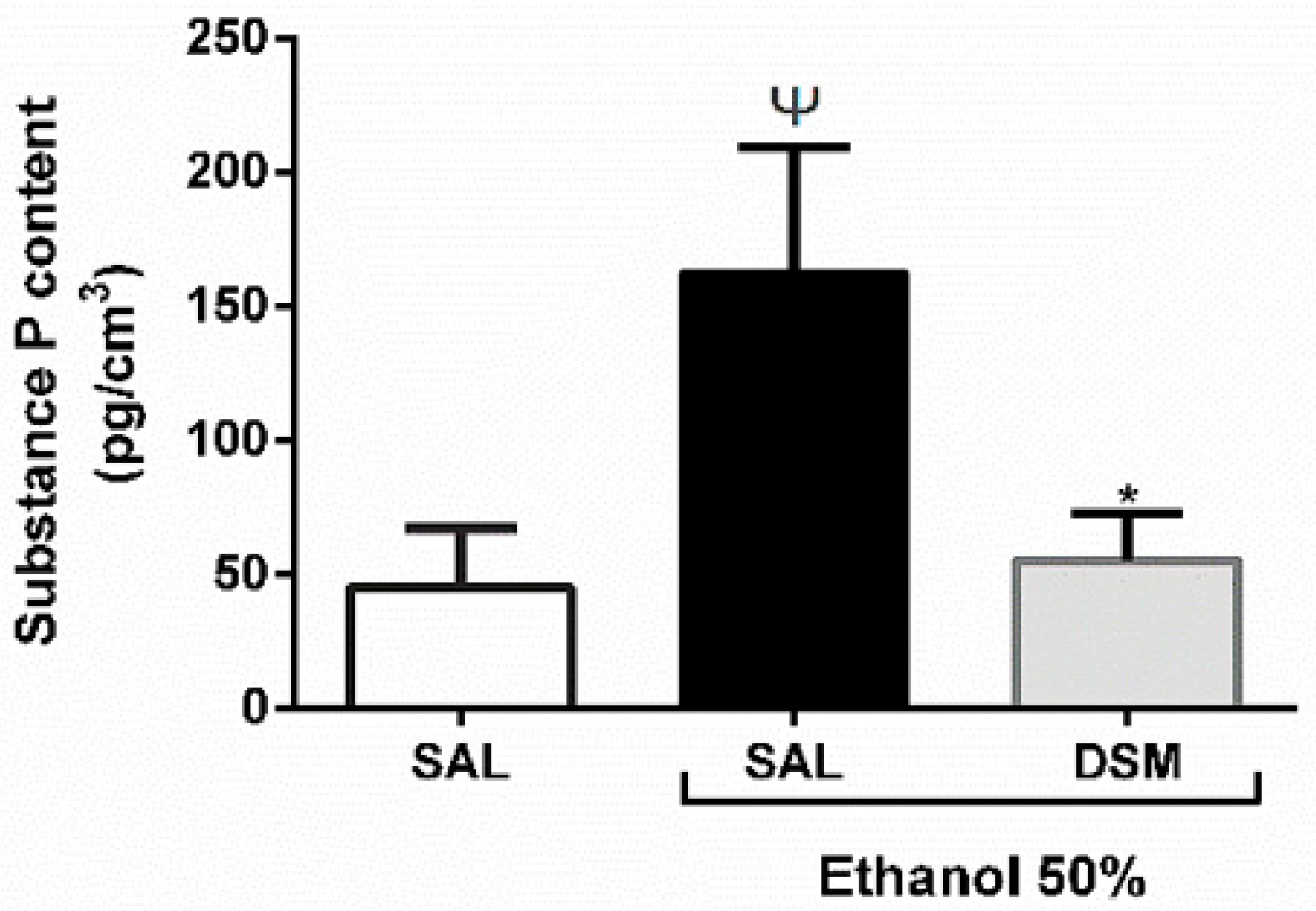
3.5. SP Levels
3.6. Effect of DSM on Biomarkers of Oxidative Stress and Antioxidant Parameters
3.7. Gastric Wall Mucus and Acid Secretion
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Global Status Report on Alcohol and Health 2018; World Health Organization: Geneva, Switzerland, 2018; pp. 38–84. ISBN 978-92-4-156563-9. [Google Scholar]
- Li, G.; Zhu, L.; Cao, Z.; Wang, J.; Zhou, F.; Wang, X.; Li, X.; Nie, G. A New Participant in the Pathogenesis of Alcoholic Gastritis: Pyroptosis. Cell Physiol. Biochem. 2018, 49, 406–418. [Google Scholar] [CrossRef] [PubMed]
- Rubbens, J.; Brouwers, J.; Wolfs, K.; Adams, E.; Tack, J.; Augustijns, P. Ethanol concentrations in the human gastrointestinal tract after intake of alcoholic beverages. Eur. J. Pharm. Sci. 2016, 86, 91–95. [Google Scholar] [CrossRef] [PubMed]
- Abdul-Rahman, A.K.; Card, T.R.; Grainge, M.J.; Fleming, K.M. All-cause and cause-specific mortality rates of patients treated for alcohol use disorders: A meta-analysis. Subst. Abuse 2018. [CrossRef] [PubMed]
- Sommansson, A.; Saudi, W.S.W.; Nylander, O.; Sjöblom, M. The Ethanol-Induced Stimulation of Rat Duodenal Mucosal Bicarbonate Secretion In Vivo Is Critically Dependent on Luminal Cl−. PLoS ONE 2014, 9, e102654. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.S.; He, S.Z.; Xu, H.Z.; Zhan, X.J.; Yang, X.N.; Xiao, H.M.; Shi, H.X.; Ren, J.L. Oxidative stress disturbs energy metabolism of mitochondria in ethanol-induced gastric mucosa injury. World J. Gastroenterol. 2008, 14, 5857–5867. [Google Scholar] [CrossRef] [PubMed]
- Lucetti, L.T.; Silva, R.O.; Santana, A.P.M.; de Melo Tavares, B.; Vale, M.L.; Soares, P.M.G.; Lima Júnior, F.J.B.; Magalhães, P.J.C.; Cunha, F.Q.; Ribeiro, R.A.; et al. Nitric oxide and hydrogen sulfide interact when modulating gastric physiological functions in rodents. Dig. Dis. Sci. 2017, 62, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Tarnawski, A.S.; Ahluwalia, A.; Jones, M.K. Angiogenesis in gastric mucosa: An important component of gastric erosion and ulcer healing and its impairment in aging. J. Gastroenterol. Hepatol. 2014, 29, 112–123. [Google Scholar] [CrossRef] [Green Version]
- Gazzieri, D.; Trevisani, M.; Springer, J.; Harrison, S.; Cottrell, G.S.; Andre, E.; Nicoletti, P.; Massi, D.; Zecchi, S.; Nosi, D.; et al. Substance P released by TRPV1-expressing neurons produces reactive oxygen species that mediate ethanol-induced gastric injury. Free Radic. Biol. Med. 2007, 43, 581–589. [Google Scholar] [CrossRef]
- Hayashi, H.; Nishiyama, K.; Majima, M.; Katori, M.; Saigenji, K. Role of endogenous substance P in ethanol-induced mucosal damage in the rat stomach. J. Gastroenterol. 1996, 31, 314–322. [Google Scholar] [CrossRef]
- Karmeli, F.; Eliakim, R.; Okon, E.; Rachmilewitz, D. Gastric Mucosal Damage by Ethanol Is Mediated by Substance P and Prevented by Ketotifen, a Mast Cell Stabilizer. Gastroenterology 1991, 100, 1206–1216. [Google Scholar] [CrossRef]
- Getzoff, E.D.; Tainer, J.A.; Weiner, P.K.; Kollman, P.A.; Richardson, P.J.S.; Richardson, D.C. Electrostatic recognition between superoxide and copper, zinc superoxide dismutase. Nature 1983, 306, 287–290. [Google Scholar] [CrossRef] [PubMed]
- Kwiecien, S.; Jasnos, K.; Magierowski, M.; Sliwowski, Z.; Pajdo, R.; Brzozowski, B.; Mach, T.; Wojcik, D.; Brzozowski, T. Lipid peroxidation, reactive oxygen species and antioxidative factors in the pathogenesis of gastric mucosal lesions and mechanism of protection against oxidative stress-induced gastric injury. J. Physiol. Pharmacol. 2014, 65, 613–622. [Google Scholar] [PubMed]
- Farrell, C.P.; Morgan, M.; Rudolph, D.S.; Hwang, A.; Albert, N.E.; Valenzano, M.C.; Wang, X.; Mercogliano, G.; Mullin, J.M. Proton Pump Inhibitors Interfere With Zinc Absorption and Zinc Body Stores. Gastroenterol. Res. 2011, 4, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef] [PubMed]
- Azmi, L.; Gupta, S.S.; Shukla, I.; Kant, P.; Upreti, D.K.; Rao, C.V. Gastro protective effects of usnea longissima metabolites on probiotic Lactobacillus casei. Int. J. Pharmacogn. 2016, 3, 140–148. [Google Scholar] [CrossRef]
- Liu, C.F.; Hu, C.L.; Chiang, S.S.; Tseng, K.C.; Yu, R.C.; Pan, T.M. Beneficial Preventive Effects of Gastric Mucosal Lesion for Soy—Skim Milk Fermented by Lactic Acid Bacteria. J. Agric. Food Chem. 2009, 57, 4433–4438. [Google Scholar] [CrossRef]
- Senol, A.; Isler, M.; Karahan, A.G.; Kilic, G.B.; Kuleasan, H.; Kaya, S.; Keskin, M.; Goren, I.; Saritas, U.; Aridogan, B.C.; et al. Preventive effect of probiotics and α-tocopherol on ethanol-induced gastric mucosal injury in rats. J. Med. Food 2011, 14, 173–179. [Google Scholar] [CrossRef]
- Suo, H.; Zhao, X.; Qian, Y.; Sun, P.; Zhu, K.; Li, J.; Sun, B. Lactobacillus fermentum Suo attenuates HCl/Ethanol induced gastric injury in mice through its antioxidant effects. Nutrients 2016, 8, 155. [Google Scholar] [CrossRef]
- Wagnerberger, S.; Spruss, A.; Kanuri, G.; Stahl, C.; Schröder, M.; Vetter, W.; Bischoff, S.C.; Bergheim, I. Lactobacillus casei Shirota protects from fructose-induced liver steatosis: A mouse model. J. Nutr. Biochem. 2013, 24, 531–538. [Google Scholar] [CrossRef]
- Rosander, A.; Connolly, E.; Roos, S. Removal of antibiotic resistance gene-carrying plasmids from Lactobacillus reuteri ATCC 55730 and characterization of the resulting daughter strain, L. reuteri DSM 17938. Appl. Environ. Microbial. 2008, 74, 6032–6040. [Google Scholar] [CrossRef]
- Casas, I.A.; Dobrogosz, W.J. Validation of the Probiotic Concept: Lactobacillus reuteri Confers Broad-spectrum Protection against Disease in Humans and Animals. Microb. Ecol. Health Dis. 2000, 12, 247–285. [Google Scholar] [CrossRef]
- Fatheree, N.Y.; Liu, Y.; Taylor, C.M.; Hoang, T.K.; Cai, C.; Rahbar, M.H.; Hessabi, M.; Ferris, M.; McMurtry, V.; Wong, C.; et al. Lactobacillus reuteri for infants with colic: A double-blind, placebo-controlled, randomized clinical trial. J. Pediatr. 2017, 191, 170–178. [Google Scholar] [CrossRef] [PubMed]
- Szymański, H.; Szajewska, H. Efficacy of Lactobacillus Reuteri DSM 17938 for the Treatment of Acute Gastroenteritis in Children: Protocol of a Randomized Controlled Trial. JMIR Res. Protoc. 2017, 6, e164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Coccorullo, P.; Strisciuglio, C.; Martinelli, M.; Miele, E.; Greco, L.; Staiano, A. Lactobacillus reuteri (DSM 17938) in infants with functional chronic constipation: A double-blind, randomized, placebo-controlled study. J. Pediatr. 2010, 157, 598–602. [Google Scholar] [CrossRef]
- Francavilla, R.; Lionetti, E.; Castellaneta, S.; Ciruzzi, F.; Indrio, F.; Masciale, A.; Fontana, C.; La Rosa, M.M.; Cavallo, L.; Francavilla, A. Randomised clinical trial: Lactobacillus reuteri DSM 17938 vs. placebo in children with acute diarrhea—A double-blind study. Aliment. Pharmacol. Ther. 2012, 36, 363–369. [Google Scholar] [CrossRef]
- Haileselassie, Y.; Navis, M.; Vu, N.; Qazi, K.R.; Rethi, B.; Sverremark-Ekström, E. Postbiotic modulation of retinoic acid imprinted mucosal-like dendritic cells by probiotic Lactobacillus reuteri 17938 in vitro. Front. Immunol. 2016, 7, 1–11. [Google Scholar] [CrossRef]
- Perez-burgos, A.; Wang, L.; Neufeld, K.A.M.; Mao, Y.K.; Ahmadzai, M.; Janssen, L.J.; Stanisz, A.M.; Bienenstock, J.; Kunze, W.A. The TRPV1 channel in rodents is a major target for antinociceptive effect of the probiotic Lactobacillus reuteri DSM 17938. J. Physiol. 2015, 593, 3943–3957. [Google Scholar] [CrossRef]
- Medeiros, J.V.R.; Bezerra, V.H.; Gomes, A.S.; Barbosa, A.L.R.; Lima-Júnior, R.C.P.; Soares, P.M.G.; Brito, G.A.C.; Ribeiro, R.A.; Cunha, F.Q.; Souza, M.H.L.P. Hydrogen sulfide prevents ethanol-induced gastric damage in mice: Role of ATP-sensitive potassium channels and capsaicin-sensitive primary afferent neurons. J. Pharmacol. Exp. Ther. 2009, 330, 764–770. [Google Scholar] [CrossRef]
- Tramontana, M.; Renzi, D.; Panerai, C.; Surrenti, C.; Nappi, F.; Abelli, L.; Evangelista, S. Capsaicin-like effect of resiniferatoxin in the rat stomach. Neuropeptides 1994, 26, 29–32. [Google Scholar] [CrossRef]
- Laine, L.; Weinstein, W.M. Histology of alcoholic hemorrhagic ‘‘gastritis’’: A prospective evaluation. Gastroenterology 1988, 94, 1254–1262. [Google Scholar] [CrossRef]
- Buckinx, R.; Nassauw, L.V.; Avula, L.R.; Alpaerts, K.; Adriaensen, D.; Timmermans, J.P. Transient receptor potential vanilloid type 1 channel (TRPV1) immunolocalization in the murine enteric nervous system is affected by the targeted C-terminal epitope of the applied antibody. J. Histochem. Cytoch. 2013, 61, 421–432. [Google Scholar] [CrossRef] [PubMed]
- Helps, S.C.; Thornton, E.; Kleinig, T.J.; Manavis, J.; Vink, R. Automatic nonsubjective estimation of antigen content visualized by immunohistochemistry using color deconvolution. Appl. Immunohistochem. Mol. Morphol. 2012, 20, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Ruifrok, A.C.; Johnston, D.A. Quantification of Histochemical Staining by Color Deconvolution. Anal. Quant. Cytol. Histol. 2001, 23, 291–299. [Google Scholar] [PubMed]
- Ma, J.; Altomare, A.; de la Monte, S.; Tong, M.; Rieder, F.; Fiocchi, C.; Behar, J.; Shindou, H.; Biancani, P.; Harnett, K.M. HCl-induced inflammatory mediators in esophageal mucosa increase migration and production of H2O2 by peripheral blood leukocytes. Am. J. Physiol. Gastrointest. Liver Physiol. 2010, 299, 791–798. [Google Scholar] [CrossRef] [PubMed]
- Uchiyama, M.; Mihara, M. Determination of malonaldehyde precursor in tissues by thiobarbituric acid test. Anal. Biochem. 1978, 86, 271–278. [Google Scholar] [CrossRef]
- Green, L.C.; Wagner, D.A.; Glogowski, J.; Skipper, P.L.; Wishnok, J.S.; Tannenbaum, S.R. Analysis of nitrate, nitrite and (15N) nitrate in biological fluids. Anal. Biochem. 1982, 126, 131–138. [Google Scholar] [CrossRef]
- Sedlak, J.; Lindsay, R.H. Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman’s reagent. Anal. Biochem. 1968, 25, 192–205. [Google Scholar] [CrossRef]
- Das, K.; Samanta, L.; Chainy, G.B.N. A modified spectrophotometric assay of superoxide dismutase using nitrite formation by superoxide radicals. Indian J. Biochem. Biophys. 2000, 37, 201–204. [Google Scholar]
- Corne, S.J.; Morrissey, S.M.; Woods, R.J. Proceedings: A method for the quantitative estimation of gastric barrier mucus. J. Physiol. 1974, 242, 116–117. [Google Scholar]
- Shay, M.; Kamarov, S.A.; Fels, D.; Meranze, D.; Gruenstein, H.; Siplet, H. A simple method for the uniform production of gastric ulceration in the rats. Gastroenterology 1945, 5, 43–61. [Google Scholar]
- Gamallat, Y.; Meyiah, A.; Kuugbee, E.D.; Hago, A.M.; Chiwala, G.; Awadasseid, A.; Bamba, D.; Zhang, X.; Shang, X.; Luo, F.; et al. Lactobacillus rhamnosus induced epithelial cell apoptosis, ameliorates inflammation and prevents colon cancer development in an animal model. Biomed. Pharmacother. 2016, 83, 536–541. [Google Scholar] [CrossRef] [PubMed]
- Štofilová, J.; Langerholc, T.; Botta, C.; Treven, P.; Gradišnik, L.; Salaj, R.; Šoltésová, A.; Bertková, I.; Hertelyová, Z.; Bomba, A. Cytokine production in vitro and in rat model of colitis in response to Lactobacillus plantarum LS/07. Biomed. Pharmacother. 2017, 94, 1176–1185. [Google Scholar] [CrossRef] [PubMed]
- Walsham, A.D.S.; Mackenzie, D.A.; Cook, V.; Wemyss-holden, S.; Hews, C.L.; Juge, N.; Schüller, S. Lactobacillus reuteri Inhibition of Enteropathogenic Escherichia coli Adherence to Human Intestinal Epithelium. Front. Microbiol. 2016, 7, 244. [Google Scholar] [CrossRef] [PubMed]
- Dore, M.P.; Cuccu, M.; Pes, G.M.; Manca, A.; Graham, D.Y. Lactobacillus reuteri in the treatment of Helicobacter pylori infection. Intern. Emerg. Med. 2014, 9, 649–654. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Tran, D.Q.; Fatheree, N.Y.; Rhoads, J.M. Lactobacillus reuteri DSM 17938 differentially modulates effector memory T cells and Foxp3+ regulatory T cells in a mouse model of necrotizing enterocolitis. Am. J. Physiol. Gastrointest. Liver Physiol. 2014, 307, 177–186. [Google Scholar] [CrossRef]
- Liu, Y.; Fatheree, N.Y.; Mangalat, N.; Rhoads, J.M. Lactobacillus reuteri strains reduce incidence and severity of experimental necrotizing enterocolitis via modulation of TLR4 and NF-κB signaling in the intestine. Am. J. Physiol. Gastrointest. Liver Physiol. 2012, 302, 608–617. [Google Scholar] [CrossRef]
- Lam, E.K.Y.; Tai, E.K.K.; Koo, M.W.L.; Wong, H.P.S.; Wu, W.K.K.; Yu, L.; So, W.H.L.; Woo, P.C.Y.; Cho, C.H. Enhancement of gastric mucosal integrity by Lactobacillus rhamnosus GG. Life Sci. 2007, 80, 2128–2136. [Google Scholar] [CrossRef]
- Guslandi, M. Effects of ethanol on the gastric mucosa. Dig. Dis. 1987, 5, 21–32. [Google Scholar] [CrossRef]
- Choi, Y.J.; Kim, N.; Lee, J.Y.; Nam, R.H.; Seo, J.H.; Lee, S.; Kim, H.J.; Choi, Y.J.; Lee, H.S.; Lee, D.H. Gastroprotective Effects of PMK-S005 against Ethanol-Induced Acute Gastric Damage in Rats. Gut Liver 2016, 10, 348–355. [Google Scholar] [CrossRef]
- Batran, R.A.; Al-bayaty, F.; Al-obaidi, M.M.J.; Abdualkader, A.M.; Hadi, H.A.; Ali, H.M.; Abdulla, M.A. In Vivo Antioxidant and Antiulcer Activity of Parkia speciosa Ethanolic Leaf Extract against Ethanol-Induced Gastric Ulcer in Rats. PLoS ONE 2013, 8, e64751. [Google Scholar] [CrossRef]
- Zhao, X.; Suo, H.Y.; Qian, Y.; Li, G.J.; Liu, Z.H.; Li, J. Therapeutic effects of Lactobacillus casei Qian treatment in activated carbon-induced constipated mice. Mol. Med. Rep. 2015, 12, 3191–3199. [Google Scholar] [CrossRef] [PubMed]
- Suo, H.; Zhao, X.; Qian, Y.; Li, G.; Liu, Z.; Xie, J.; Li, J. Therapeutic Effect of Activated Carbon-Induced Constipation Mice with Lactobacillus fermentum Suo on Treatment. Int. J. Mol. Sci. 2014, 15, 21875–21895. [Google Scholar] [CrossRef] [PubMed]
- Souza, L.K.M.; Nicolau, L.A.D.; Sousa, N.A.; Araújo, T.S.L.; Sousa, F.B.M.; Costa, D.S.; Souza, F.M.; Pacífico, D.M.; Martins, C.S.; Silva, R.O.; et al. Diminazene aceturate, an angiotensin-converting enzyme II activator, prevents gastric mucosal damage in mice: Role of the angiotensin-(1-7)/Mas receptor axis. Biochem. Pharmacol. 2016, 112, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.S.; Deitrich, R.A. Ethanol metabolism and effects: Nitric oxide and its interaction. Curr. Clin. Pharmacol. 2007, 2, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Gago, B.; Nyström, T.; Cavaleiro, C.; Rocha, B.S.; Barbosa, R.M.; Laranjinha, J.; Lundberg, J.O. The potent vasodilator ethyl nitrite is formed upon reaction of nitrite and ethanol under gastric conditions. Free Radic. Biol. Med. 2008, 45, 404–412. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Halliwell, B.; Aeschbach, R.; Loliger, J.; Aruoma, O.I. The characterization of antioxidants. Food Chem. Toxicol. 1995, 33, 601–617. [Google Scholar] [CrossRef]
- Bhattacharyya, A.; Chattopadhyay, R.; Mitra, S.; Crowe, S.E. Oxidative stress: An essential factor in the pathogenesis of gastrointestinal mucosal diseases. Physiol. Rev. 2014, 94, 329–354. [Google Scholar] [CrossRef] [PubMed]
- Loguercio, C.; Taranto, D.; Beneduce, F.; Blanco, C.V.; Vincentiis, A.; Nardi, G.; Romano, M. Glutathione prevents ethanol induced gastric mucosal damage and depletion of sulfhydryl compounds in humans. Gut 1983, 34, 161–165. [Google Scholar] [CrossRef]
- Bompart, G.J.; Prévot, D.S.; Bascands, J.L. Rapid automated analysis of glutathione reductase, peroxidase, and S-transferase activity: Application to cisplatin-induced toxicity. Clin. Biochem. 1990, 23, 501–504. [Google Scholar] [CrossRef]
- Carvalho, N.S.; Silva, M.M.; Silva, R.O.; Nicolau, L.A.D.; Sousa, F.B.M.; Damasceno, S.R.B.; Silva, D.A.; Barbosa, A.L.R.; Leite, J.R.S.A.; Medeiros, J.V.R. Gastroprotective Properties of Cashew Gum, a Complex Heteropolysaccharide of Anacardium occidentale, in Naproxen-Induced Gastrointestinal Damage in Rats. Drug Dev. Res. 2015, 76, 143–151. [Google Scholar] [CrossRef]
- Ermund, A.; Schütte, A.; Johansson, M.E.V.; Gustafsson, J.K.; Hansson, G.C. Studies of mucus in mouse stomach, small intestine, and colon. I. Gastrointestinal mucus layers have different properties depending on location as well as over the Peyer’s patches. Am. J. Physiol. Gastrointest. Liver Physiol. 2013, 305, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Hersey, S.J.; Sachs, G. Gastric acid secretion. Physiol. Rev. 1995, 75, 155–189. [Google Scholar] [CrossRef] [PubMed]
- Hung, C.R.; Neu, S.L. Acid-induced gastric damage in rats is aggravated by starvation and prevented by several nutrients. J. Nutr. 1997, 127, 630–636. [Google Scholar] [CrossRef] [PubMed]
- Yokotani, K.; Fujiwara, M. Effects of substance P on cholinergically stimulated gastric acid secretion and mucosal blood flow in rats. J. Pharmacol. Exp. Ther. 1985, 232, 826–830. [Google Scholar] [PubMed]


| Treatment | Volume (mL) | pH (Units) | [H+] (mEq/mL/4 h) | Mucus Levels (ug/g Tissue) |
|---|---|---|---|---|
| Saline | 0.028 ± 0.02 | 5.6 ± 0.2 | 0.14 ± 0.02 | 137.20 ± 16.58 |
| Ethanol | 0.350 ± 0.08 Ψ | 3.5 ± 0.2 Ψ | 3.70 ± 0.83 Ψ | 58.34 ± 8.95 Ψ |
| Omeprazole | 0.046 ± 0.02 *** | 5.2 ± 0.5 * | 0.20 ± 0.19 * | - |
| DSM | 0.055 ± 0.02 ** | 4.6 ± 0.5 * | 0.12 ± 0.05 * | 103.20 ± 6.11 ** |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oliveira, A.P.; Souza, L.K.M.; Araújo, T.S.L.; Araújo, S.d.; Nogueira, K.M.; Sousa, F.B.M.; Silva, R.O.; Pacífico, D.M.; Martins, C.S.; Brito, G.A.d.C.; et al. Lactobacillus reuteri DSM 17938 Protects against Gastric Damage Induced by Ethanol Administration in Mice: Role of TRPV1/Substance P Axis. Nutrients 2019, 11, 208. https://0-doi-org.brum.beds.ac.uk/10.3390/nu11010208
Oliveira AP, Souza LKM, Araújo TSL, Araújo Sd, Nogueira KM, Sousa FBM, Silva RO, Pacífico DM, Martins CS, Brito GAdC, et al. Lactobacillus reuteri DSM 17938 Protects against Gastric Damage Induced by Ethanol Administration in Mice: Role of TRPV1/Substance P Axis. Nutrients. 2019; 11(1):208. https://0-doi-org.brum.beds.ac.uk/10.3390/nu11010208
Chicago/Turabian StyleOliveira, Ana P., Luan K. M. Souza, Thiago S. L. Araújo, Simone de Araújo, Kerolayne M. Nogueira, Francisca Beatriz M. Sousa, Renan O. Silva, Dvison M. Pacífico, Conceição S. Martins, Gerly Anne de C. Brito, and et al. 2019. "Lactobacillus reuteri DSM 17938 Protects against Gastric Damage Induced by Ethanol Administration in Mice: Role of TRPV1/Substance P Axis" Nutrients 11, no. 1: 208. https://0-doi-org.brum.beds.ac.uk/10.3390/nu11010208






