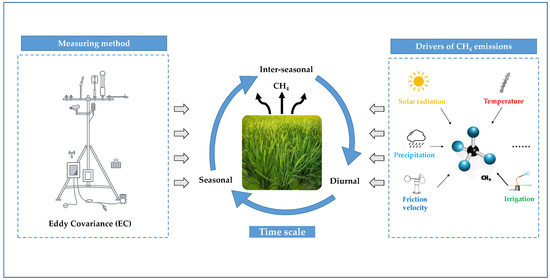
Variations and Drivers of Methane Fluxes from Double-Cropping Paddy Fields in Southern China at Diurnal, Seasonal and Inter-Seasonal Timescales
Abstract
:1. Introduction
2. Materials and Methods
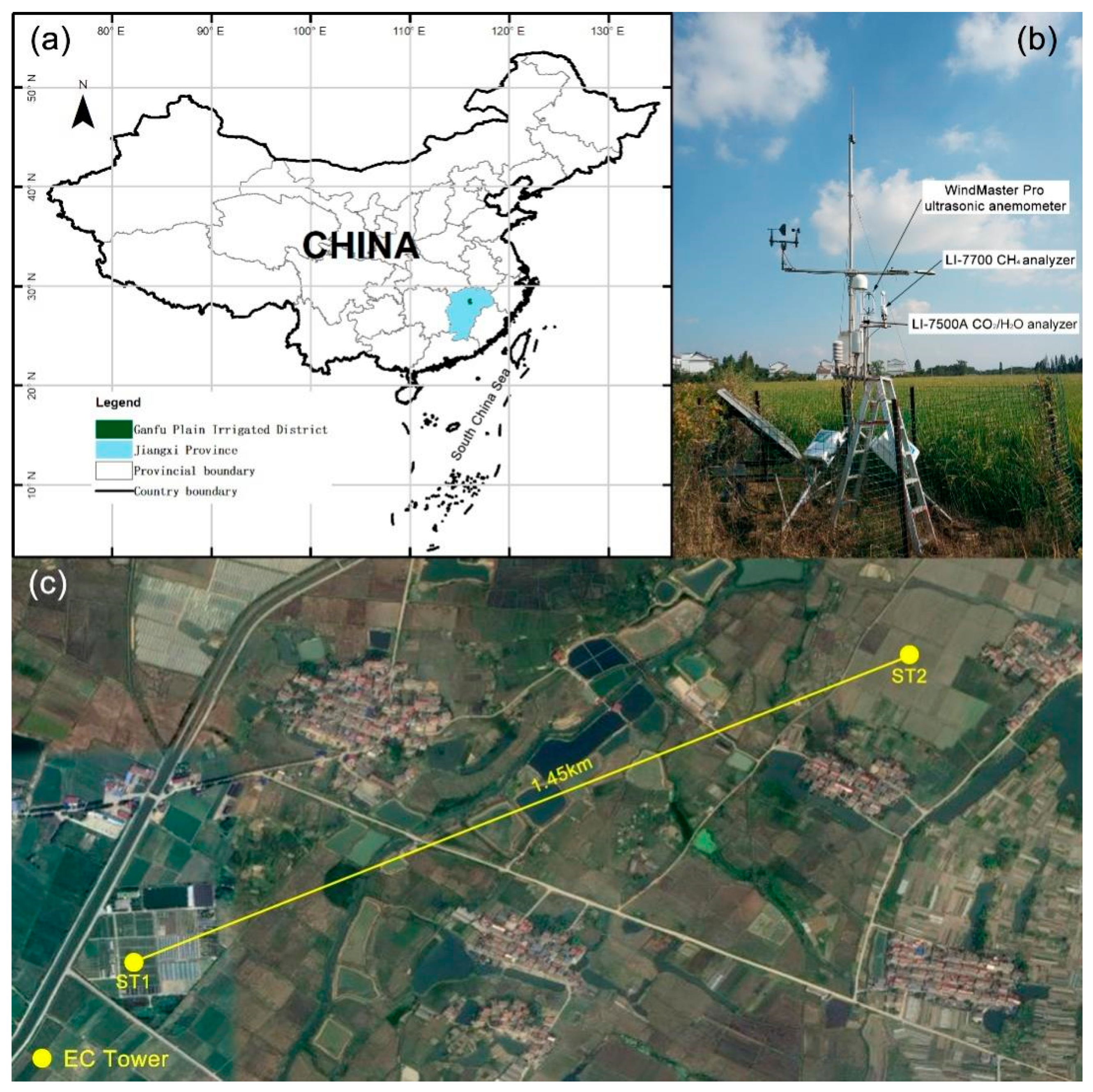
2.1. Study Site and Crop Management
2.2. Eddy Covariance and Meteorological Measurements
2.3. Data Processing
2.4. Data Analysis
2.4.1. Spectral Analysis at the Diurnal Scale
2.4.2. Semiempirical Multiplicative Model Construction at the Seasonal Scale
3. Results and Discussion
3.1. Diurnal Variations in CH4 Fluxes and Driving Factors
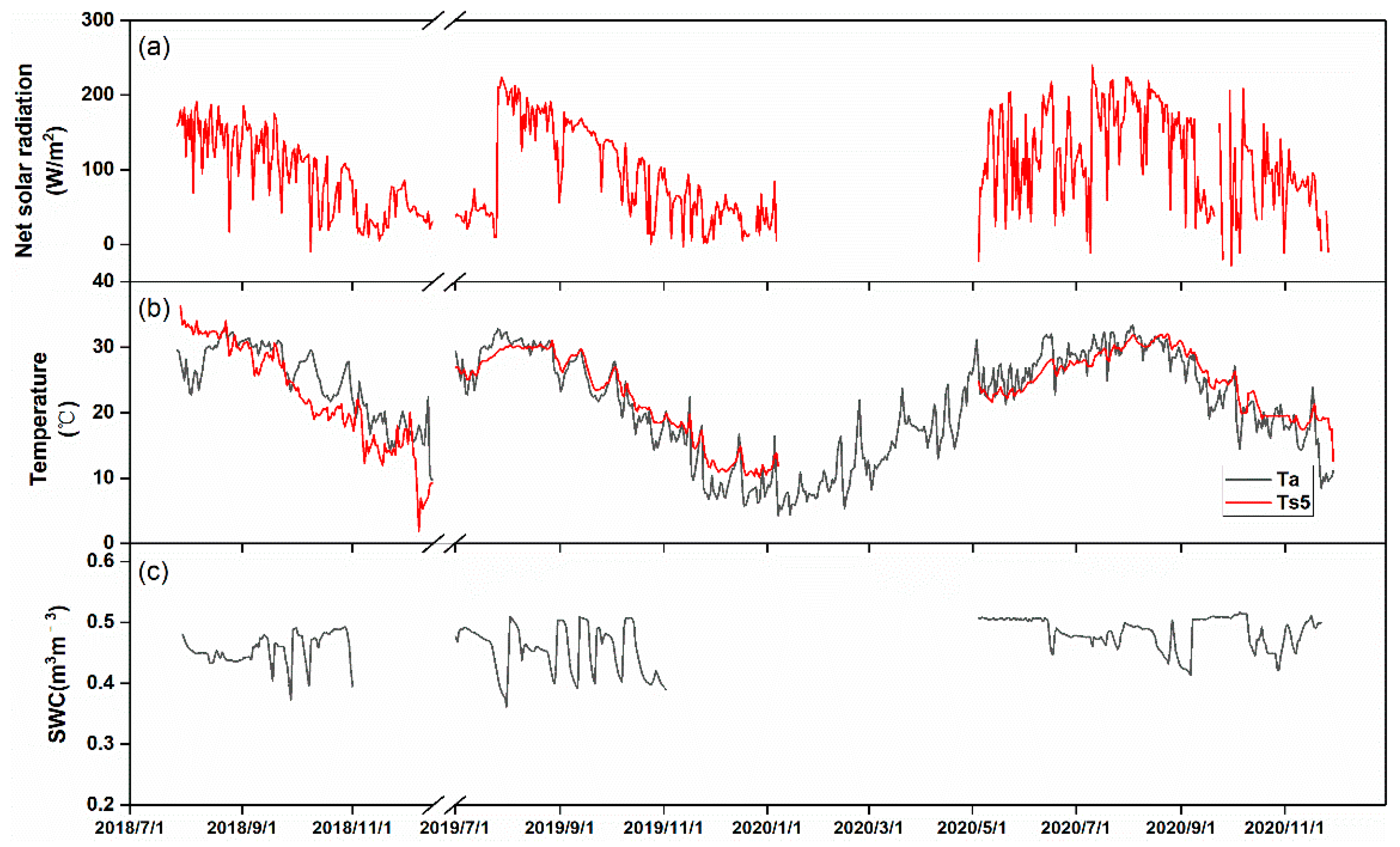
3.2. Seasonal Variations in Biophysical Factors and CH4 Fluxes
3.3. Driving Factors of Seasonal CH4 Fluxes
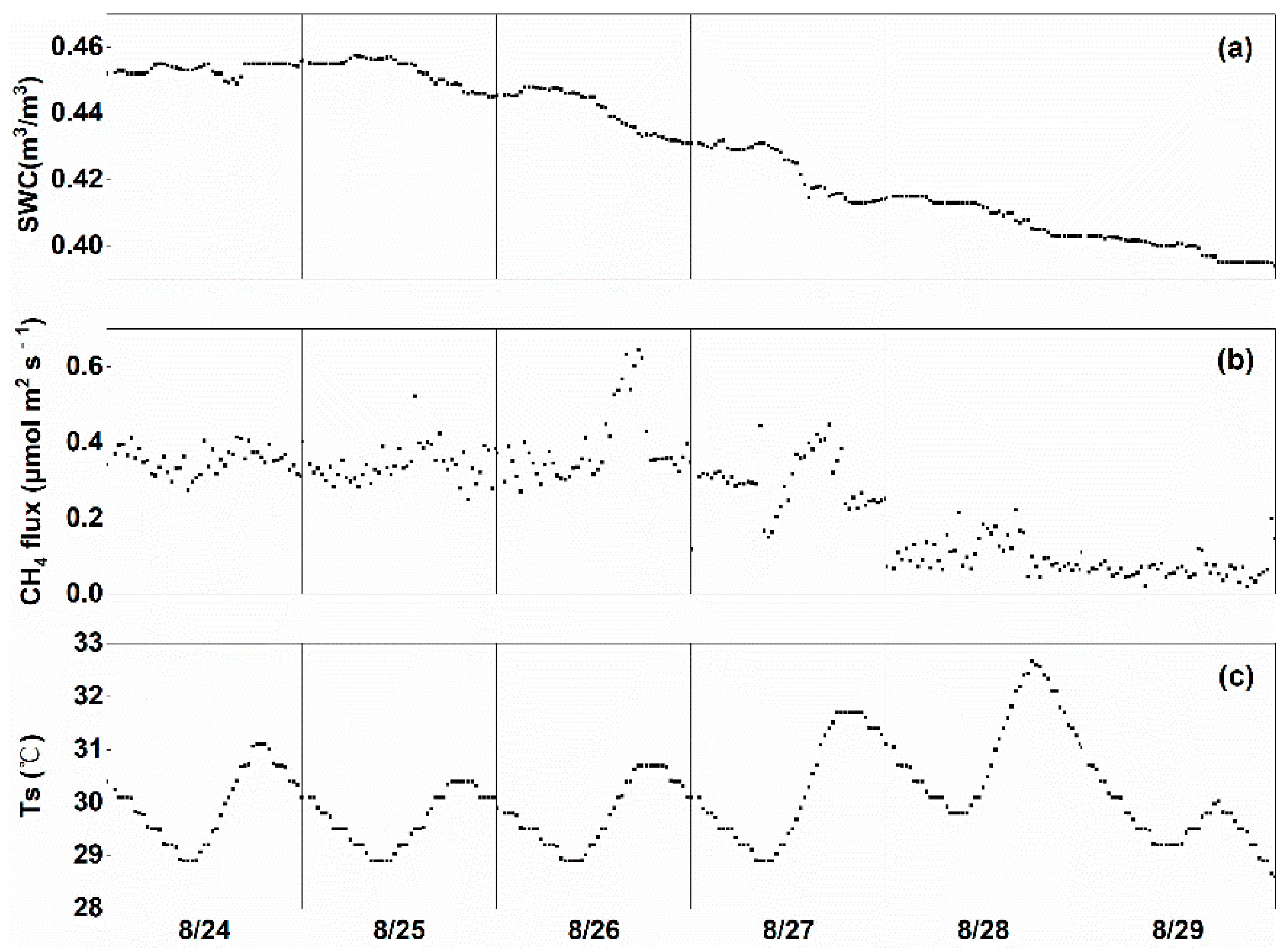
3.4. Effect of Drainage on CH4 Fluxes
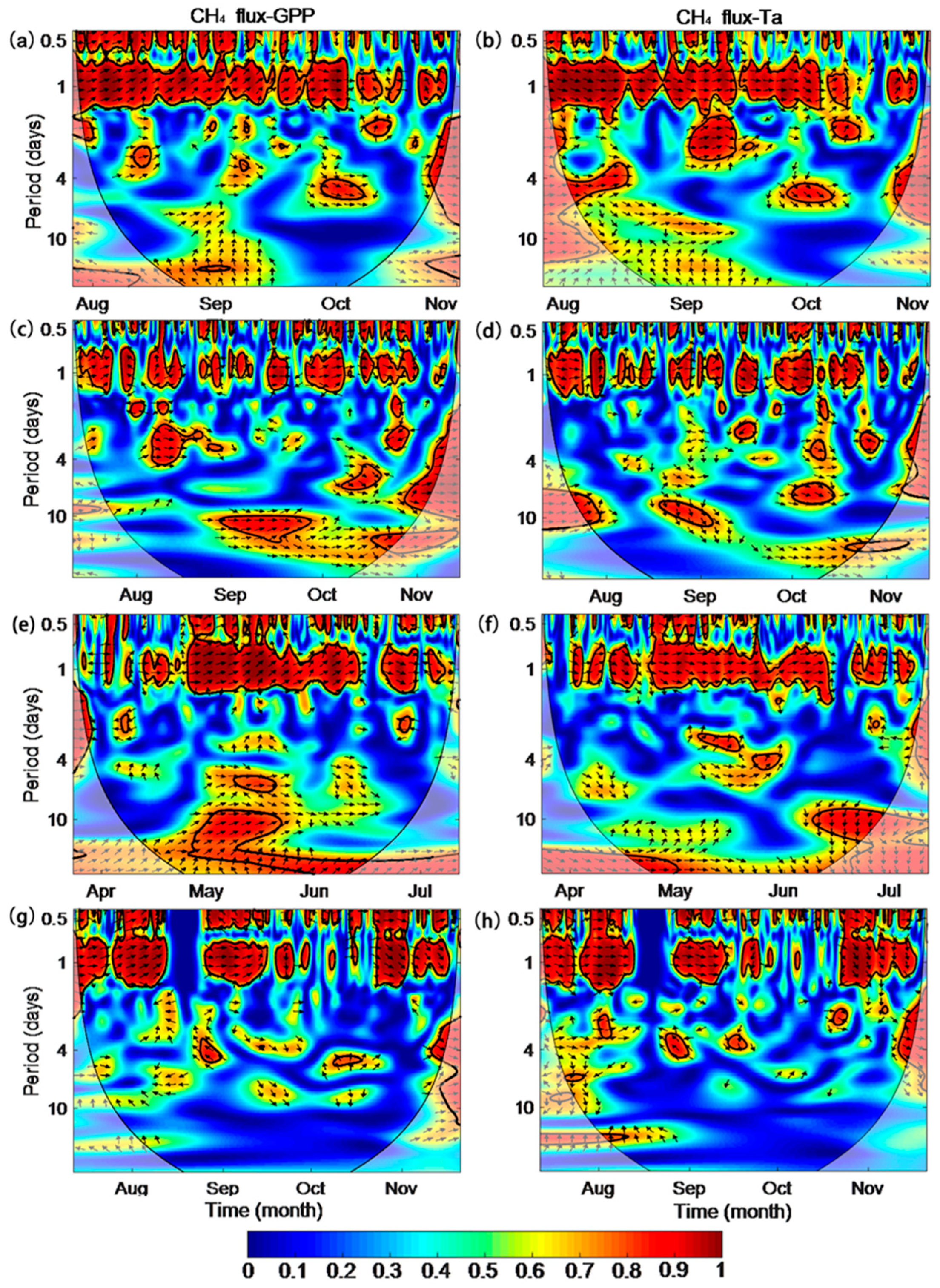
3.5. Inter-Seasonal Variations in CH4 Fluxes and Driving Factors
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hwang, Y.; Ryu, Y. Comprehensive assessments of carbon dynamics in an intermittently-irrigated rice paddy. Agric. For. Meteorol. 2020, 285–286, 107933. [Google Scholar] [CrossRef]
- Jia, Q.; Yu, W. Methane emissions from typical paddy fields in Liaohe plain and Sanjiang plain, northeast China. Environ. Res. Commun. 2019, 1, 011006. [Google Scholar]
- Stocker, T.F.; Qin, D. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. In Climate Change 2013: The Physical Science Basis; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013. [Google Scholar]
- Saunois, M.; Stavert, A.R.; Poulter, B.; Bousquet, P.; Zhuang, Q. The Global Methane Budget 2000–2017. Earth Sys. Sci. Data 2020, 12, 1561–1623. [Google Scholar] [CrossRef]
- FAO. Statistical Yearbook 2013: World Food and Agriculture; Food and Agriculture Organization of the United Nations: Rome, Italy, 2013; p. 289. [Google Scholar]
- Wu, X.; Wang, W. Combined effects of straw and water management on CH4 emissions from rice fields. J. Environ. Manag. 2018, 231, 1257–1262. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Yuan, Y. Effects of different fertilizers on methane emissions and methanogenic community structures in paddy rhizosphere soil. Sci. Total Environ. 2018, 627, 770–781. [Google Scholar] [CrossRef]
- Ma, Y.; Liu, D.L.; Schwenke, G.; Yang, B. The global warming potential of straw-return can be reduced by application of straw-decomposing microbial inoculants and biochar in rice-wheat production systems. Environ. Pollut. 2019, 252, 835–845. [Google Scholar] [CrossRef] [PubMed]
- Miyata, A.; Leuning, R. Carbon dioxide and methane fluxes from an intermittently flooded paddy field. Agric. For. Meteorol. 2000, 102, 287–303. [Google Scholar] [CrossRef]
- Weller, S.; Kraus, D. Diurnal patterns of methane emissions from paddy rice fields in the Philippines. J. Plant Nutr. Soil Sci. 2015, 178, 755–767. [Google Scholar] [CrossRef]
- Long, K.D.; Flanagan, L.B. Diurnal and seasonal variation in methane emissions in a northern Canadian peatland measured by eddy covariance. Glob. Chang. Biol. 2009, 16, 2420–2435. [Google Scholar] [CrossRef]
- Kim, Y.; Johnson, M.S. Gap-filling approaches for eddy covariance methane fluxes: A comparison of three machine learning algorithms and a traditional method with principal component analysis. Glob. Chang. Biol. 2020, 26, 1499–1518. [Google Scholar] [CrossRef] [PubMed]
- Hollinger, D.Y.; Kelliher, F.M. Initial assessment of multi-scale measures of CO2 and H2O flux in the Siberian taiga. J. Biogeogr. 1995, 22, 425–431. [Google Scholar] [CrossRef]
- Moncrieff, J.B.; Massheder, J.M. A system to measure surface fluxes of momentum, sensible heat, water vapour and carbon dioxide. J. Hydrol. 1997, 188–189, 589–611. [Google Scholar] [CrossRef]
- Jiang, S.Z.; Liang, C. Water use efficiency and its drivers in four typical agroecosystems based on flux tower measurements. Agric. For. Meteorol. 2020, 295, 108200. [Google Scholar] [CrossRef]
- Wagle, P.; Gowda, P.H. Dynamics of CO2 and H2O fluxes in Johnson grass in the U.S. Southern Great Plains. Sci. Total Environ. 2020, 739, 140077. [Google Scholar] [CrossRef]
- Alberto, M.C.R.; Hirano, T. Influence of climate variability on seasonal and interannual variations of ecosystem CO2 exchange in flooded and non-flooded rice fields in the Philippines. Field Crops Res. 2012, 134, 80–94. [Google Scholar] [CrossRef]
- Schmidt, M.; Reichenau, T.G. The carbon budget of a winter wheat field: An eddy covariance analysis of seasonal and inter-annual variability. Agric. For. Meteorol. 2012, 165, 114–126. [Google Scholar] [CrossRef]
- Richardson, A.D.; Hollinger, D.Y. A multi-site analysis of random error in tower-based measurements of carbon and energy fluxes. Agric. For. Meteorol. 2006, 136, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Leuning, R.; Gorsel, E.V. Reflections on the surface energy imbalance problem. Agric. For. Meteorol. 2012, 156, 65–74. [Google Scholar] [CrossRef]
- Liu, B.; Cui, Y.; Luo, Y.; Shi, Y.; Liu, M.; Liu, F. Energy partitioning and evapotranspiration over a rotated paddy field in Southern China. Agric. For. Meteorol. 2019, 276-277, 107626. [Google Scholar] [CrossRef]
- Dengel, S.; Levy, P.E. Methane emissions from sheep pasture, measured with an open-path eddy covariance system. Glob. Chang. Biol. 2011, 17, 3524–3533. [Google Scholar] [CrossRef]
- Morin, T.H.; Bohrer, G. The seasonal and diurnal dynamics of methane flux at a created urban wetland. Ecol. Eng. 2014, 72, 74–83. [Google Scholar] [CrossRef]
- Podgrajsek, E.; Sahlée, E. Diurnal cycle of lake methane flux. J. Geophys. Res. 2014, 119, 236–248. [Google Scholar] [CrossRef]
- Koebsch, F.; Jurasinski, G. Controls for multi-scale temporal variation in ecosystem methane exchange during the growing season of a permanently inundated fen. Agric. For. Meteorol. 2015, 204, 94–105. [Google Scholar] [CrossRef]
- Fortuniak, K.; Pawlak, W.O. Methane and carbon dioxide fluxes of a temperate mire in Central Europe. Agric. For. Meteorol. 2017, 232, 306–318. [Google Scholar] [CrossRef]
- Yagi, K.; Tsuruta, H. Effect of water management on methane emission from a Japanese rice paddy field: Automated methane monitoring. Glob. Biogeochem. Cycles 1996, 10, 255–267. [Google Scholar] [CrossRef]
- Acosta, M.; Dušek, J. Autumnal fluxes of CH4 and CO2 from Mediterranean reed wetland based on eddy covariance and chamber methods. Catena 2019, 183, 104191. [Google Scholar] [CrossRef]
- Suyker, A.E.; Verma, S.B. Methane flux in a boreal fen: Season-long measurement by eddy correlation. J. Geophys. Res. 1996, 101, 28637–28647. [Google Scholar] [CrossRef]
- Tokida, T.; Miyazaki, T. Ebullition of methane from peat with falling atmospheric pressure. Geophys. Res. Lett. 2005, 32, 13823. [Google Scholar] [CrossRef]
- Kim, Y.; Talucder, M.S.A. Interannual variations in methane emission from an irrigated rice paddy caused by rainfalls during the aeration period. Agric. Ecosyst. Environ. 2016, 223, 67–75. [Google Scholar] [CrossRef]
- Hatala, J.A.; Detto, M. Gross ecosystem photosynthesis causes a diurnal pattern in methane emission from rice. Geophys. Res. Lett. 2012, 39, 06409. [Google Scholar] [CrossRef] [Green Version]
- Bridgham, S.D.; Cadillo-Quiroz, H. Methane emissions from wetlands: Biogeochemical, microbial, and modeling perspectives from local to global scales. Glob. Chang. Biol. 2013, 19, 1325–1346. [Google Scholar] [CrossRef] [PubMed]
- Song, C.Q.; Liu, W. Characteristics and drivers of methane fluxes from a rice paddy based on the flux measurement. Adv. Earth Sci. 2019, 34, 1141–1151, (In Chinese with English Abstract). [Google Scholar]
- Cicerone, R.J.; Shetter, J.D. Seasonal variation of methane flux from a California rice paddy. J. Geophys. Res. 1983, 88, 11022–11024. [Google Scholar] [CrossRef] [Green Version]
- Wassmann, R.; Aulakh, M.S. The role of rice plants in regulating mechanisms of methane missions. Biol. Fertil. Soils 2000, 31, 20–29. [Google Scholar] [CrossRef]
- Cai, Z.C.; Tsuruta, H. Methane emission from rice fields in China: Measurements and influencing factors. J. Geophys. Res. 2000, 105, 17231–17242. [Google Scholar] [CrossRef] [Green Version]
- Conrad, R. Control of microbial methane production in wetland rice fields. Nutr. Cycl. Agroecosyst. 2002, 64, 59–69. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, X. Methane emissions from a rice agroecosystem in South China: Effects of water regime, straw incorporation and nitrogen fertilizer. Nutr. Cycl. Agroecosyst. 2012, 93, 103–112. [Google Scholar] [CrossRef]
- Liao, B.; Wu, X. Effects of mild alternate wetting and drying irrigation and mid-season drainage on CH4 and N2O emissions in rice cultivation. Sci. Total Environ. 2020, 698, 134212. [Google Scholar] [CrossRef] [PubMed]
- Iwata, H.; Mano, M. Exploring sub-daily to seasonal variations in methane exchange in a single-crop rice paddy in central Japan. Atmos. Environ. 2018, 179, 156–165. [Google Scholar] [CrossRef]
- Moore, T.R.; Dalva, M. The influence of temperature and water table position on carbon dioxide and methane emissions from laboratory columns of peatland soils. J. Soil Sci. 2010, 44, 651–664. [Google Scholar] [CrossRef]
- Knox, S.H.; Sturtevant, C. Agricultural peatland restoration: Effects of land-use change on greenhouse gas (CO2 and CH4) fluxes in the Sacramento-San Joaquin Delta. Glob. Chang. Biol. 2014, 21, 750–765. [Google Scholar] [CrossRef] [PubMed]
- Knox, S.H.; Matthes, J.H. Biophysical controls on interannual variability in ecosystem-scale CO2 and CH4 exchange in a California rice paddy. J. Geophys. Res. 2016, 121, 978–1001. [Google Scholar] [CrossRef]
- Runkle, B.R.; Suvoscarev, K.; Reba, M.L.; Reavis, C.W.; Smith, S.F.; Chiu, Y.L.; Fong, B. Methane Emission Reductions from the Alternate Wetting and Drying of Rice Fields Detected Using the Eddy Covariance Method. Environ. Sci. Technol. 2019, 53, 671–681. [Google Scholar] [CrossRef] [PubMed]
- Meijide, A.; Manca, G. Seasonal trends and environmental controls of methane emissions in a rice paddy field in Northern Italy. Biogeosciences 2011, 8, 3809–3821. [Google Scholar] [CrossRef] [Green Version]
- Meijide, A.; Gruening, C. Water management reduces greenhouse gas emissions in a Mediterranean rice paddy field. Agric. Ecosyst. Environ. 2017, 238, 168–178. [Google Scholar] [CrossRef]
- Centeno, C.A.R.; Alberto, M.C.R. Assessing diel variation of CH4 flux from rice paddies through temperature patterns. Atmos. Environ. 2017, 167, 23–39. [Google Scholar] [CrossRef]
- Ge, H.X.; Zhang, H.S. The characteristics of methane flux from an irrigated rice farm in East China measured using the eddy covariance method. Agric. For. Meteorol. 2018, 249, 228–238. [Google Scholar] [CrossRef]
- Dai, S.; Ju, W. Variations and drivers of methane fluxes from a rice-wheat rotation agroecosystem in eastern China at seasonal and diurnal scales. Sci. Total Environ. 2019, 690, 973–990. [Google Scholar] [CrossRef]
- Berg, M.; Hengsdijk, H. The impact of increasing farm size and mechanization on rural income and rice production in Zhejiang province, China. Agric. Syst. 2007, 94, 841–850. [Google Scholar] [CrossRef]
- Chu, G.; Chen, T. Morphological and physiological traits of roots and their relationships with water productivity in water-saving and drought-resistant rice. Field Crops Res. 2014, 162, 108–119. [Google Scholar] [CrossRef]
- Carrijo, D.R.; Lundy, M.E. Rice yields and water use under alternate wetting and drying irrigation: A meta-analysis. Field Crops Res. 2017, 203, 173–180. [Google Scholar] [CrossRef]
- Chen, X.Y.; Wu, F.; Chen, H.X.; Wang, C.G. Analysis of the characteristics and causes of severe summer and autumn droughts in Jiangxi in 2019. Meteorol. Disaster Reduct. Res. 2020, 43, 170–179, (In Chinese with English Abstract). [Google Scholar]
- LI-COR Biosciences. EddyPro 5.0: Help and User’s Guide; LI-COR Biosciences: Lincoln, NE, USA, 2013. [Google Scholar]
- Webb, E.K.; Pearman, G.I. Correction of Flux Measurements for Density Effects Due to Heat and Water Vapor Transfer. Q. J. R. Meteorolog. Soc. 1980, 106, 100. [Google Scholar] [CrossRef]
- Fratini, G.; Ibrom, A. Relative humidity effects on water vapour fluxes measured with closed-path eddy-covariance systems with short sampling lines. Agric. For. Meteorol. 2012, 165, 53–63. [Google Scholar] [CrossRef]
- Falge, E.; Baldocchi, D. Gap filling strategies for defensible annual sums of net ecosystem exchange. Agric. For. Meteorol. 2001, 107, 43–69. [Google Scholar] [CrossRef] [Green Version]
- Taylor, J.L.A. On the Temperature Dependence of Soil Respiration. Funct. Ecol. 1994, 8, 315–323. [Google Scholar]
- Minoda, T.; Kimura, M. Photosynthates as dominant source of CH4 and CO2 in soil water and CH4 emitted to the atmosphere from paddy fields. J. Geophys. Res. 1996, 101, 21091–21097. [Google Scholar] [CrossRef]
- Miyata, A.; Iwata, T. Seasonal variation of carbon dioxide and methane fluxes at single cropping paddy fields in central and western Japan. Phyton 2005, 45, 89–97. [Google Scholar]
- Gogoi, N.; Baruah, K.K. Methane emission characteristics and its relations with plant and soil parameters under irrigated rice ecosystem of northeast India. Chemosphere 2005, 59, 1677–1684. [Google Scholar] [CrossRef]
- Neue, H.U.; Wassmann, R. Factors and processes controlling methane emissions from rice fields. Nutr. Cycl. Agroecosyst. 1997, 49, 111–117. [Google Scholar] [CrossRef]
- Wang, M.; Dai, A. CH4 emission from Chinese rice paddy field. Acta Meteorol. Sin. 1990, 4, 265–275. [Google Scholar]
- Marik, T.; Fischer, H. Seasonal variations in stable carbon and hydrogen isotope ratios in methane from rice fields. Glob. Biogeochem. Cycles 2002, 16, 41-1–41-11. [Google Scholar] [CrossRef]
- Alberto, M.C.R.; Wassmann, R. Measuring methane flux from irrigated rice fields by eddy covariance method using open-path gas analyzer. Field Crops Res. 2014, 160, 12–21. [Google Scholar] [CrossRef]
- Simpson, I.J.; Thurtell, G.W. Tunable diode laser measurements of methane fluxes from an irrigated rice paddy field in the Philippines. J. Geophys. Res. 1995, 100, 7283–7290. [Google Scholar] [CrossRef]
- Xu, X.; Kuzyakov, Y. Root-derived respiration and non-structural carbon of rice seedlings. Eur. J. Soil Biol. 2008, 44, 22–29. [Google Scholar] [CrossRef]
- Holzapfel-Pschorn, A.; Seiler, W. Methane emission during a cultivation period from an Italian rice paddy. J. Geophys Res. 1986, 91, 11803–11814. [Google Scholar] [CrossRef]
- Inubushi, K.; Wada, H. Methane emission from paddy fields. Presented at the 14th International Congress of Soil Science, Kyoto, Japan, 12–18 August 1990. [Google Scholar]
- Wassmann, R.; Neue, H.U. Temporal patterns of methane emissions from wetland rice fields treated by different modes of N application. J. Geophys. Res. 1994, 991, 16457–16462. [Google Scholar] [CrossRef]
- Bhattacharyya, P.; Neogi, S. Tropical low land rice ecosystem is a net carbon sink. Agric. Ecosyst. Environ. 2014, 189, 127–135. [Google Scholar] [CrossRef]
- Lindau, C.W.; Bollich, P.K. Effect of urea fertilizer and environmental factors on CH4 emissions from a Louisiana, USA rice field. Plant Soil 1991, 136, 195–203. [Google Scholar] [CrossRef]



| Growth Stage | Field Management | Fertilizer Application | |||
|---|---|---|---|---|---|
| 2018–late rice–ST1 (WT a: AWD) | |||||
| Vegetative stage | 27 July–25 August | Field ponding | 20 July | Basal fertilizer | 26 July |
| Reproductive stage | 26 August–6 October | Transplanting | 26 July | Tillering fertilizer | 2 August |
| Ripening stage | 7 October–31 October | Mid-season drainage | 21 August–25 August | Panicle fertilizer | 28 August |
| Harvest | 1 November | ||||
| 2019–late rice–ST2 (WT a: local farms’ traditional irrigation method) | |||||
| Vegetative stage | 12 July–30 August | Field ponding | 6 July | Basal fertilizer | 11 July |
| Reproductive stage | 31 August–2 October | Seeding | 10 July | Tillering fertilizer | 20 August |
| Ripening stage | 3 October–14 November | Mid-season drainage | 26 August–30 August | ||
| Fallow season | 15 November–10 March | Harvest | 15 November | ||
| 2020–early rice–ST2 (WT a: local farms’ traditional irrigation method) | |||||
| Vegetative stage | 24 March–13 May | Field ponding | 20 March | Basal fertilizer | 24 March |
| Reproductive stage | 14 May–16 Jun | Seeding | 23 March | Tillering fertilizer | 1 May |
| Ripening stage | 17 June–11 July | Mid-season drainage | 8 May–13 May | ||
| Harvest | 12 July | ||||
| 2020–late rice–ST2 (WT a: local farms’ traditional irrigation method) | |||||
| Vegetative stage | 12 July–7 September | Field ponding | 6 July | Basal fertilizer | 12 July |
| Reproductive stage | 8 September–10 October | Seeding | 10 July | Tillering fertilizer | 19 August |
| Ripening stage | 11 October–20 November | Mid-season drainage | 28 August–7 September | ||
| Harvest | 21 November | ||||
| Model-1 | Model-2 | ||||||
|---|---|---|---|---|---|---|---|
| P1 b | P2 b | P3 b | |||||
| Variable | R2 | Variable | R2 | Variable | R2 | Variable | R2 |
| 2018 late rice | |||||||
| Ts5 | 0.921 | Ts5 | 0.690 | Ts5 | 0.789 | ||
| SWC | 0.941 | GPP | 0.735 | SWC | 0.839 | ||
| U* | 0.942 | RH | 0.737 | GPP | 0.850 | ||
| GPP | 0.942 | SWC | 0.739 | RH | 0.850 | ||
| RH | 0.942 | U* | 0.739 | U* | 0.850 | ||
| 2019 late rice | |||||||
| Ts5 | 0.793 | SWC | 0.809 | GPP | 0.856 | Ts5 | 0.903 |
| GPP | 0.836 | RH | 0.848 | RH | 0.872 | SWC | 0.906 |
| SWC | 0.847 | Ts5 | 0.878 | U* | 0.881 | GPP | 0.909 |
| RH | 0.854 | U* | 0.882 | Ts5 | 0.881 | U* | 0.911 |
| U* | 0.856 | GPP | 0.882 | SWC | 0.881 | RH | 0.911 |
| 2020 early rice | |||||||
| SWC | 0.163 | GPP | 0.108 | SWC | 0.838 | ||
| Ts5 | 0.584 | SWC | 0.534 | GPP | 0.860 | ||
| RH | 0.702 | RH | 0.541 | RH | 0.875 | ||
| GPP | 0.702 | Ts5 | 0.555 | Ts5 | 0.876 | ||
| U* | 0.702 | U* | 0.555 | U* | 0.876 | ||
| 2020 late rice | |||||||
| Ts5 | 0.826 | U* | 0.142 | RH | 0.458 | Ts5 | 0.791 |
| RH | 0.852 | RH | 0.292 | GPP | 0.578 | GPP | 0.814 |
| GPP | 0.874 | SWC | 0.328 | U* | 0.647 | SWC | 0.825 |
| SWC | 0.881 | Ts5 | 0.381 | SWC | 0.728 | U* | 0.827 |
| U* | 0.881 | GPP | 0.449 | Ts5 | 0.731 | RH | 0.827 |
| Rice Season | CH4 (g CH4 m−2) | GPP (g CO2 m−2) | Mean Ta (°C) | F(CH4/GPP) % | ||
|---|---|---|---|---|---|---|
| Whole Season | Before Mid-Season Drainage | Whole Season | Before Mid-Season Drainage | |||
| 2018 late rice | 21.63 | 13.70 | 889.53 | 242.26 | 25.83 | 2.43 |
| 2019 late rice | 19.13 | 16.24 | 1245.95 | 471.40 | 26.46 | 1.54 |
| 2020 early rice | 14.52 | 6.78 | 746.57 | 134.71 | 23.11 | 1.94 |
| 2020 late rice | 23.95 | 21.14 | 1081.13 | 460.60 | 24.74 | 2.22 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, L.; Liu, B.; Cui, Y.; Shi, Y. Variations and Drivers of Methane Fluxes from Double-Cropping Paddy Fields in Southern China at Diurnal, Seasonal and Inter-Seasonal Timescales. Water 2021, 13, 2171. https://0-doi-org.brum.beds.ac.uk/10.3390/w13162171
Ma L, Liu B, Cui Y, Shi Y. Variations and Drivers of Methane Fluxes from Double-Cropping Paddy Fields in Southern China at Diurnal, Seasonal and Inter-Seasonal Timescales. Water. 2021; 13(16):2171. https://0-doi-org.brum.beds.ac.uk/10.3390/w13162171
Chicago/Turabian StyleMa, Linhua, Bo Liu, Yuanlai Cui, and Yuanzhi Shi. 2021. "Variations and Drivers of Methane Fluxes from Double-Cropping Paddy Fields in Southern China at Diurnal, Seasonal and Inter-Seasonal Timescales" Water 13, no. 16: 2171. https://0-doi-org.brum.beds.ac.uk/10.3390/w13162171