2.1. Sampling and Preconcentration of TGM
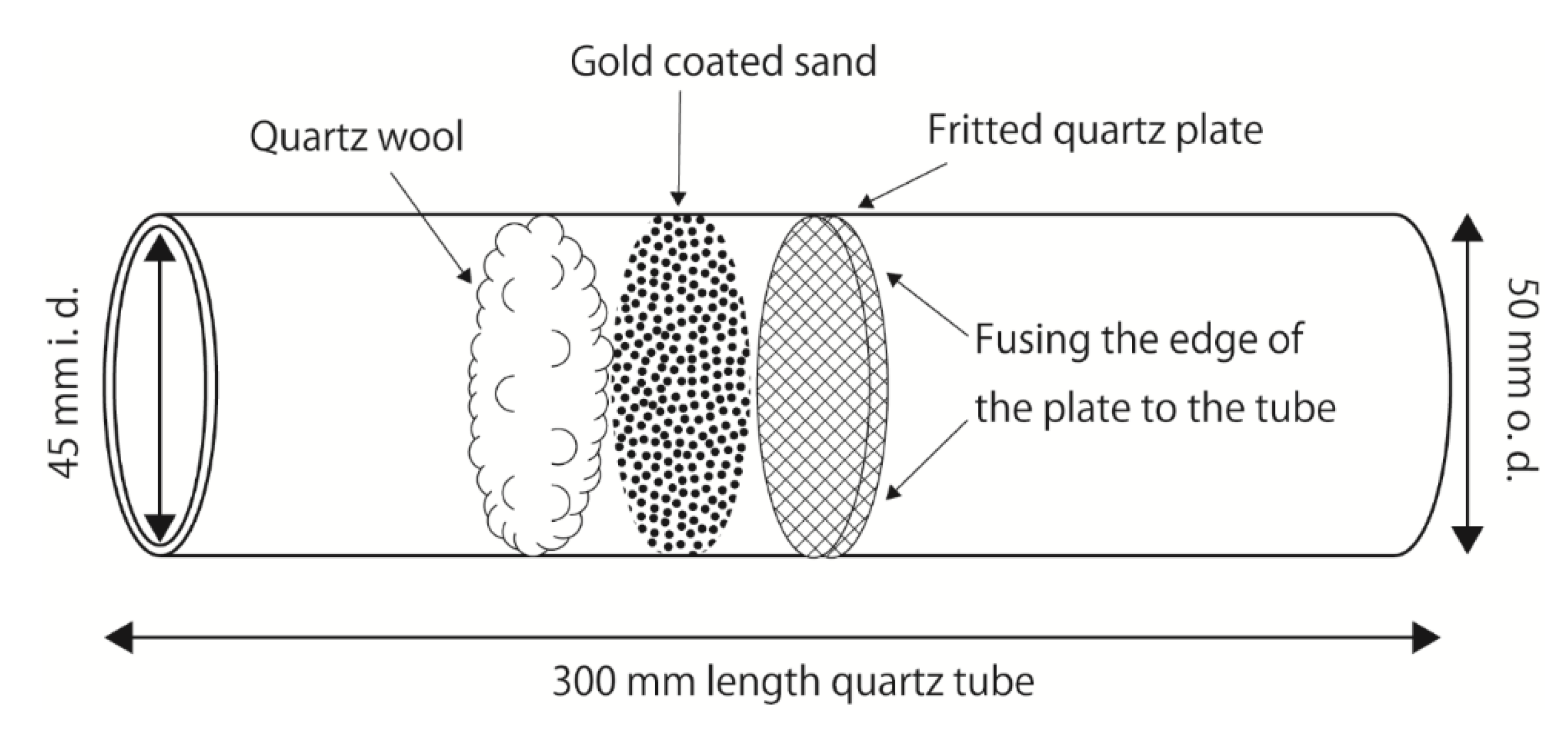
TGM was collected using a big gold-coated sand trap (BAuT) (
Figure 1). A BAuT is a custom-made 50 (o.d.) × 45 (i.d.) × 300 mm (length) quartz tube with a 45 (o.d.) × 5 mm (thickness) fritted quartz plate (COSMOS VID, Fukuoka, Japan). Approximately 10.5 g gold-coated sand, which traps TGM at room temperature and releases TGM as GEM at high temperatures and is reused more than 50,000 times (Nippon Instruments Corporation, Osaka, Japan) was layered on the fritted plate, and approximately 0.1 g quartz wool was then stuffed on the top of the sand. The BAuT was used vertically during sampling because the trapping material settled, but was not secured onto the fritted plate. Compared to a commercially available conventional gold-coated sand trap (a 4 (i.d.) × 160 mm quartz tube with 0.08 g gold-coated silica sand, capable of capturing 70 μg of TGM; Nippon Instruments Corporation), the mouth of the BAuT is approximately 11 times wider, and the amount of trapping material is approximately 131 times greater, allowing a larger air flow with the same collection efficiency and capturing capacity of 9.2 mg of TGM. To meet the same linear velocities as those of the conventional trap under the typical flow rates of 0.5 and 1.0 L min
−1, the sampling flow rates used for the BAuT were 63.3 and 126.6 L min
−1, respectively. Techniques reported in the literature use sampling flow rates of 2–20 L min
−1 with a sampling duration of 12 h–20 days (using a carbon trap impregnated with iodine or chlorine [
10,
16]), 14.4 L min
−1 with 2–3 days (1.8 L min
−1 × 8 conventional gold traps [
5,
8]), and 16 L min
−1 with 3 days (1 L min
−1 × 16 conventional gold traps [
9]). Assuming identical collection efficiencies, the BAuT sampling is 3–9-fold faster than that of the other methods. An additional advantage of the BAuT is the ease of preconditioning, its long lifetime, and the use of a single trap that makes the postsampling procedure simple.
TGM was sampled by the BAuT using a diaphragm pump (N860FTE, KNF, Freiburg, Germany or DOA-P501-DB, GAST MFG Corp., Benton Harbor, MI, USA) through a flow meter (RK230, KOFLOC, Kyoto, Japan). PTFE connectors for the BAuT were custom-made (COSMOS VID, Fukuoka, Japan), and half an inch or 3/8 of an inch (o.d.) PFA tubing (Tombo 9003-PFA, NICHIAS Corporation, Tokyo, Japan), PFA connectors (Swagelok, Solon, OH, USA), and/or PTFE connectors (Flowell 30 series, Flowell Corporation, Yokohama, Japan) were also used.
TGM trapped in the BAuT was preconcentrated to a conventional gold trap prior to the plastic bag extraction. The BAuT was heated to 873 K for 1 h under a 0.5 L min−1 zero air flow, which was supplied by compressed room air (super oil free BEBICON 0.4LE-8SB, Hitachi Industrial Equipment Systems Co., Ltd., Tokyo, Japan) through a dryer stuffed with silica gel (Kanto Chemical Co., Inc., Tokyo, Japan) and a mercury trap stuffed with activated charcoal (Hokuetsu MA-HG, Ajinomoto Fine Techno Co., Inc., Yokohama, Japan). The background TGM concentration in the zero air was 0.03 pg L−1 or less. The flow rate was controlled using a mass flow controller (SEC-E40 and PE-D20, HORIBA, Ltd., Kyoto, Japan). The BAuT was heated by a handmade heater consisting of a nickel-chrome wire (1.0 mm (o.d.), Sunko Corporation, Kyoto, Japan), quartz wool (GL Sciences, Tokyo, Japan), insulating glass tape (Komeri Corporation, Ltd., Niigata, Japan), variable transformer (RAS-10, Tokyo Rikosha Co., Ltd., Saitama, Japan), and temperature controller (TR-KN, AS ONE Corporation, Osaka, Japan). After preconcentration, the BAuT was baked at 853 K in a muffle furnace (FUL230BL, Toyo Seisakusho Kaisha, Ltd., Chiba, Japan) for several days to remove organic and mercury contaminants. Prior to sampling, the background mercury in the BAuT was preconcentrated to a conventional trap under 873 K for 1 h with a 0.5 L min−1 zero air flow. Then, it was quantitatively analyzed by a cold vapor atomic florescent spectrometer (CV-AFS) (Nippon Instruments Corporation) which was calibrated with saturated mercury vapor at a specific temperature (MB-1, Nippon Instruments Corporation). The observed background mercury was less than 70 pg.
2.2. Plastic Bag Extraction of TGM
Preconcentrated TGM was extracted with a 5 mL trapping solution in a plastic bag. Plastic bags were chosen for this purpose because, compared to a hard container, they are light and easy to evacuate and fill with the gas and have a large inner surface area, which is needed to increase the contact area of the trapping solution with the sample gas. The preconcentrated TGM in the conventional trap was flushed into a 2 L Tedlar (polyvinyl fluoride) bag through the attached PTFE stopcock (AS ONE Corporation, Osaka, Japan) for 4 min under 973 K and a 0.5 L min−1 zero air flow using another handmade heater. To enable efficient transfer, flushing was started as soon as the temperature reached 573 K. Prior to the transfer, 5 mL inversed aqua regia, a 2:1 (v/v) mixture of nitric and hydrochloric acids (PMA grade, Kanto Chemical Co., Inc., Tokyo, Japan) in ultrapure water (Milli-Q Direct 3, Merck KGaA, Darmstadt, Germany), was pipetted into the bag. The plastic bag was shaken for 3 min, then left for 3‒24 h. The bag was shaken for 1 min three more times during the rest of the period. The bag was also weighed at the start and at the end of the extraction using an electronic balancer (ML4002T, Mettler-Toledo GmbH, Greifensee, Switzerland) to check for leaks during the extraction.
2.3. CV–MC–ICP–MS Measurements
The mercury isotope ratio is expressed using the following delta notation:
where x and the bracketed isotope ratios with sample and reference stand for the mass of the mercury isotope and the mercury isotope ratios of the mass x to the mass 198 for the sample and reference materials, respectively. The standard reference material 3133 (or SRM 3133; NIST, Gaithersburg, MD, USA) was used for the reference material.
The following is a brief description of the stable mercury isotope measurement using a CV (HGX-200, Teledyne CETAC Technologies, Omaha, NE, USA), followed by an MC–ICP–MS (Neptune Plus, Thermo Fisher Scientific GmbH, Bremen, Germany). A sample solution and a 5% (w/w) SnCl2 in 10% (v/v) hydrochloric acid solution, which instantaneously reduces Hg2+ in a solution to GEM, were introduced into the CV by a peristaltic pump (Perimax, Spetec GmbH, Erding, Germany) under a flow rate of 0.58 mL min−1. Thallium standard reference material (SRM 997, NIST, Gaithersburg, MD, USA), of which the isotope ratio of 203Tl to 205Tl is known, was also introduced into the CV in order to correct artificial mass-dependent isotope fractionations as they occurred on the surfaces of the mercury and thallium at the ICP. This injection was made by introducing the dried thallium chloride aerosols generated from the hydrochloric acid/thallium (SRM 997, NIST, Gaithersburg, MD, USA) solution using an aerosol generator (Aridus II, Teledyne CETAC Technologies, Gaithersburg, NE, USA), which was operated with a 100 μL min−1 PFA nebulizer (SP820A, Teledyne CETAC Technologies) under 383 K for the spray chamber and a desolvator, with 0.40 mL min–1 and 2.9 L min–1 argon flow for the nebulizer and desolvator, respectively. Argon carrier gas for the CV (known as the “counter flow” for a straight gas–liquid separator) flowed into the CV from one end at 0.56 mL min–1 and flowed out from the other end so that all gases and aerosols inside the CV, including the sample GEM and the generated thallium aerosols, were carried into the MC-ICP-MS system. The MC-ICP-MS was tuned on a daily basis using a 10 ng g–1 solution of SRM 3133 (NIST, Gaithersburg, MD, USA) prepared in 5% inversed aqua regia (2:1 nitric acid/hydrochloric acid) prior to the measurements. A sample was preanalyzed by the CV-MC-ICP-MS to determine the concentration. When the concentration was higher than 10 ng g–1, the sample was diluted to 10 ng g–1—a concentration equivalent to the reference solution. When the concentration was below 10 ng g–1, the concentration of the reference solution was diluted to an identical concentration of the sample. The typical sensitivity of the instruments under the conditions above was 0.19 V min∙g ng−1 mL−1 for 202Hg. This value is low compared to other reported values (e.g., 1 V min∙g ng−1 mL−1). The variation in the sensitivity was very likely caused by the use of different instruments (i.e., CV-MC-ICP-MS).
For a sample or reference run, the operation software for the MC-ICP-MS was programmed to preinject a sample solution for 3 min for flushing, to measure the isotope ratios in 50 cycles and one block, and then to average out the measurement results. A 10 ng g−1 standard solution of SRM 3133 in 5% inversed aqua regia was measured before and after the sample run (i.e., the bracketing method), and the average of those reference isotope ratios was used as the reference isotope ratio in the calculation of Equation (1).
To check the performance of the instrument, the dependence of the measurement accuracy on the signal intensity (or sample concentration) was evaluated using SRM 8610 (NIST, Gaithersburg, MD, USA) and SRM 3133 in different concentrations dissolved in 5% inversed aqua regia. We called this the linearity of the isotope measurements. In this evaluation, the concentrations of SRM 8610 were identical to those of SRM 3133, the reference solution. The measurement results show that the offsets of the isotope ratios from the reference values (offset = measured isotope ratio − reference isotope ratio) or the measurement accuracy for δ
199Hg, δ
200Hg, δ
201Hg, δ
202Hg, and δ
204Hg were less than ± 0.1‰ when the concentrations were between 2.5 and 10 ng g
−1, and their uncertainties indicate that the differences are insignificant (
Figure 2). Depending on the mass, the offset became slightly larger as the concentration decreased. For samples with a mercury concentration of 1.3 or 0.8 ng g
−1, the offset was as large as 0.18‰ ± 0.09‰ or −0.33‰ ± 0.12‰, respectively. The offsets were insignificant as 2σ confidence intervals of the uncertainties were considered for the 1.3 ng g
−1 solution and 3σ intervals for the 0.8 ng g
−1 solution. We conclude that the high accuracy and precision can be maintained when the sample concentrations are higher than 2.5 ng g
−1, and that the precision decreases when the concentration falls below 2.5 ng g
−1.
2.4. Methodology Test
We performed tests for the sampling and plastic bag extraction in laboratory and field studies. The BAuT performance was evaluated by the collection efficiency: TGM was sampled using two BAuTs connected in series (double BAuT sampling), and the collection efficiency was then determined by comparing the mass of TGM in the backup BAuT with that in the primary BAuT. During this evaluation, the concentration of the preconcentrated TGM in the backup BAuT was directly measured by CV-AFS, and that in the primary BAuT was measured by CV-MC-ICP-MS. The extraction tests were performed by introducing 103 ng GEM prepared from SRM 8610 into the Tedlar bag. The dependence of the recovery yields on different inversed aqua regia concentrations (20%–40%) and the extraction time (3–24 h) was evaluated. The concentrations were chosen based on the preliminary tests, which showed that the use of 5% inversed aqua regia required 7 days to yield 90% GEM and that concentrations higher than 40% resulted in lower signal intensity (see
subsection S1 in the Supplementary Materials).
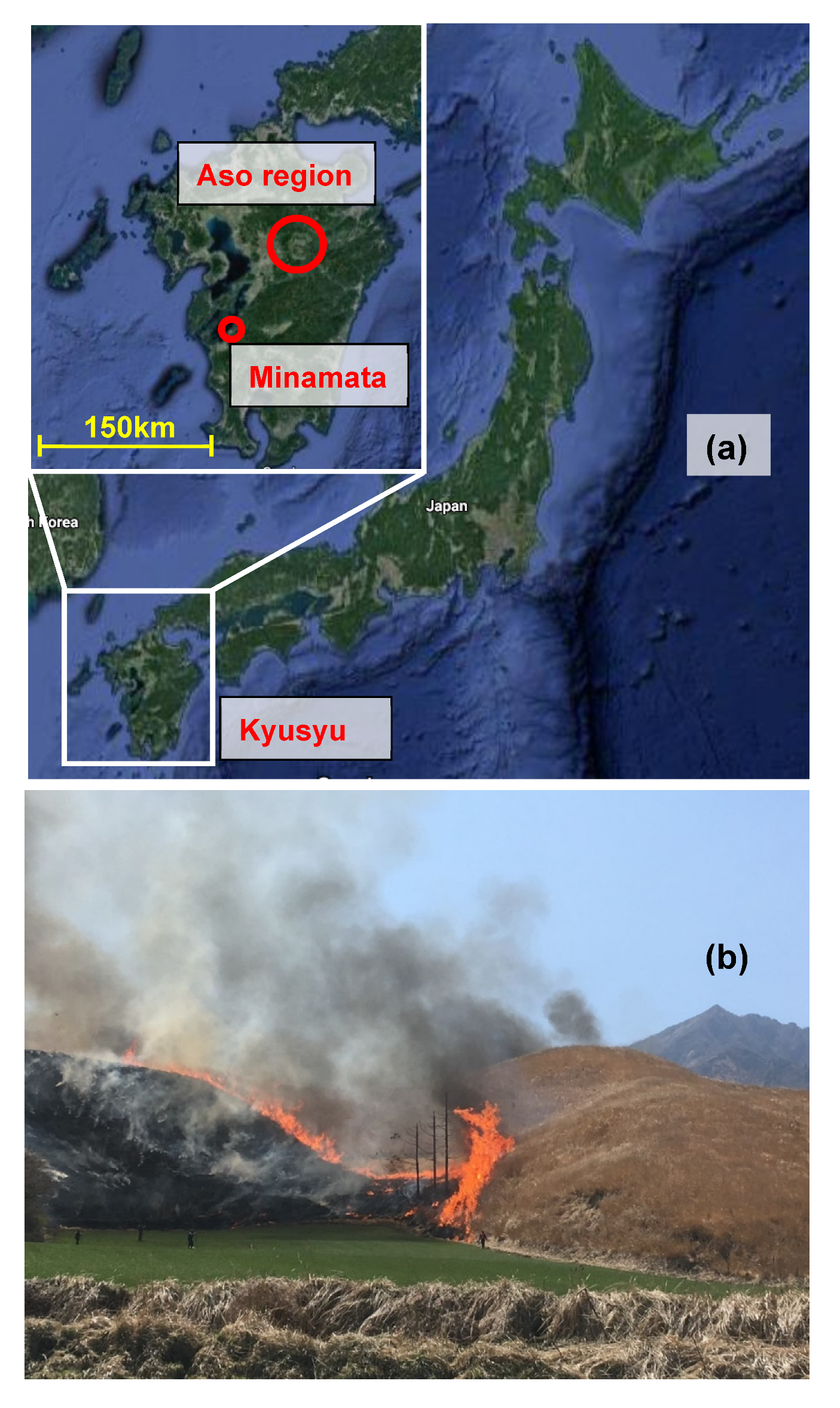
The overall methodology was evaluated in source, air quality, and laboratory studies. TGM from the biomass burning was sampled on 24 March and 6 April 2019 during the Aso open field burning, also referred to as Noyaki (
Figure 3). The air was drawn through a 47 mm (o.d.) Teflon-coated glass fiber filter (Pallflex Emfab, Pall Corporation, Port Washington, NY, USA) placed on an open-face filter holder (NL-O-01, NILU, Kjeller, Norway) to remove the soot. The electric power for the sampling pump was supplied from an uninterruptible power supply (SURTA1500XLJ, APC, West Kingston, RI, USA). The background concentration of TGM in the regional air was also collected on 31 March and 23 May 2019. Moreover, one outdoor and two indoor air samplings were also conducted at the institute the author affiliates. The BAuT was also tested for sampling GEM in the gas mixture used in the laboratory experiment. The experimental conditions were similar to those reported in [
17,
18]. Briefly, approximately 200 ng of GEM prepared from SRM 8610, oxidant precursors, and dried zero air were introduced into a 1.5 m
3 photochemical reactor. Then, 366 nm UV lamps (UV lamp 4, 8 W, CAMAG, Muttenz, Switzerland) were switched on and off to initiate and stop the reactions with OH radicals, one of the major sinking pathways of atmospheric GEM. After the reaction, GEM in the gas mixture was sampled using the BAuT through a KCl-coated eight-channel annular denuder (URG-2000-30CF, URG Corporation, Chapel Hill, NC, USA), which removes GOM [
19]. The sampling conditions are presented in
Supplementary Table S1, together with the weather conditions. The preconcentration and extraction procedures were the same as those referred to previously.