Novel Azoles as Antiparasitic Remedies against Brain-Eating Amoebae
Abstract
:1. Introduction
2. Results
2.1. Characterization of Azoles
2.1.1. 2-[3-Bromo-4,5-bis(methoxy)phenyl]-1H-benzimidazole (A1)
2.1.2. 2-(5-Methylfuran-2-yl)-1H-benzimidazole (A2)
2.1.3. (E)-N-(2″-Chlorophenyl)-2-(6,6-dimethyl-1-phenyl-1,5,6,7-tetrahydro-4H-indazol-4-ylidene)hydrazine-1-carbothioamide (A3) [27]
2.1.4. (E)-2-(6,6-Dimethyl-1-phenyl-1,5,6,7-tetrahydro-4H-indazol-4-ylidene)hydrazine-1-carbothioamide (A4) [27]
2.1.5. 5-(3’Chlorobenzyl)-1H-tetrazole (A5) [28]
2.1.6. 5-Phenyl-1H-tetrazole (A6) [28]
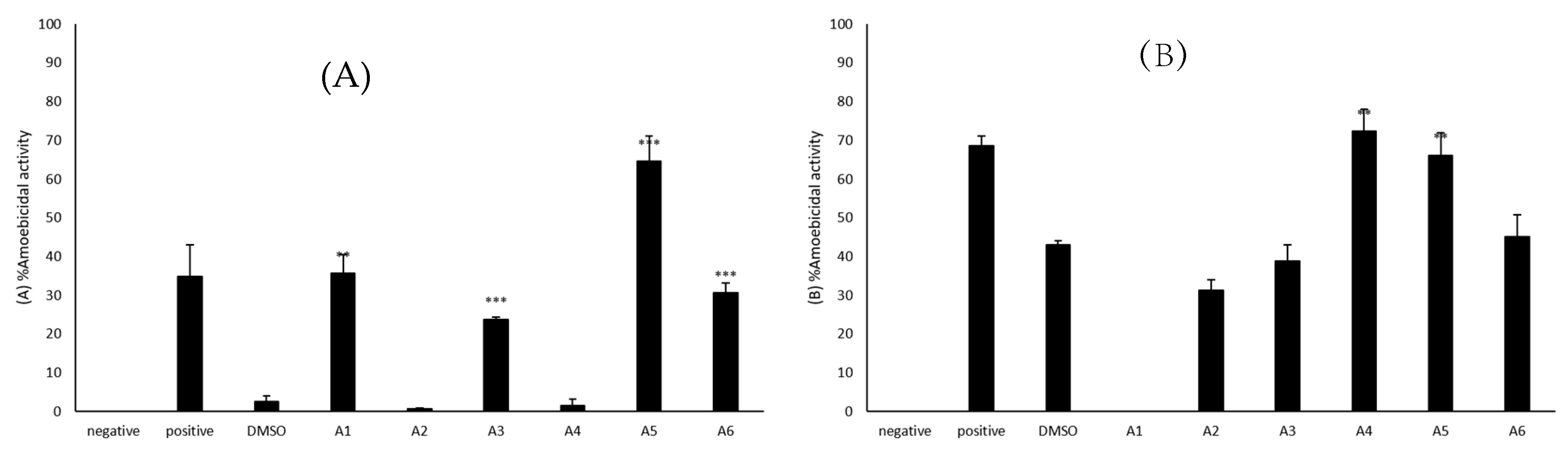
2.2. A1, A3, A5 and A6 Showed Significant Amoebicidal Activity against B. mandrillaris
2.3. A4 and A5 Showed Significant Amoebicidal Activity against N. fowleri
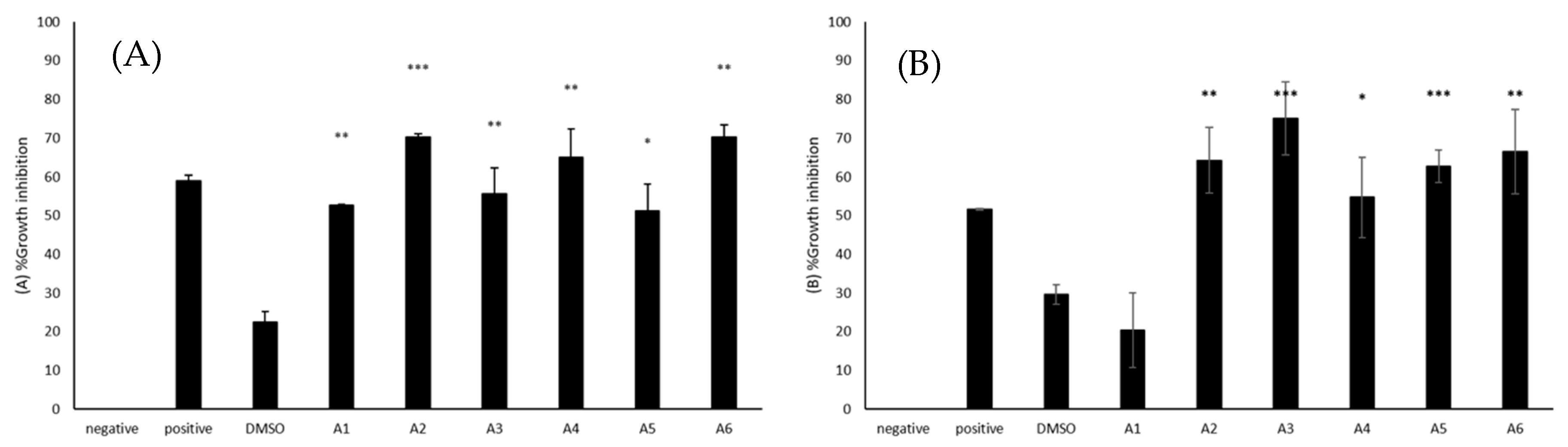
2.4. All Azoles Showed Significant Amoebistatic Activity against B. mandrillaris
2.5. Most Azoles Showed Significant Amoebistatic Activity against N. fowleri
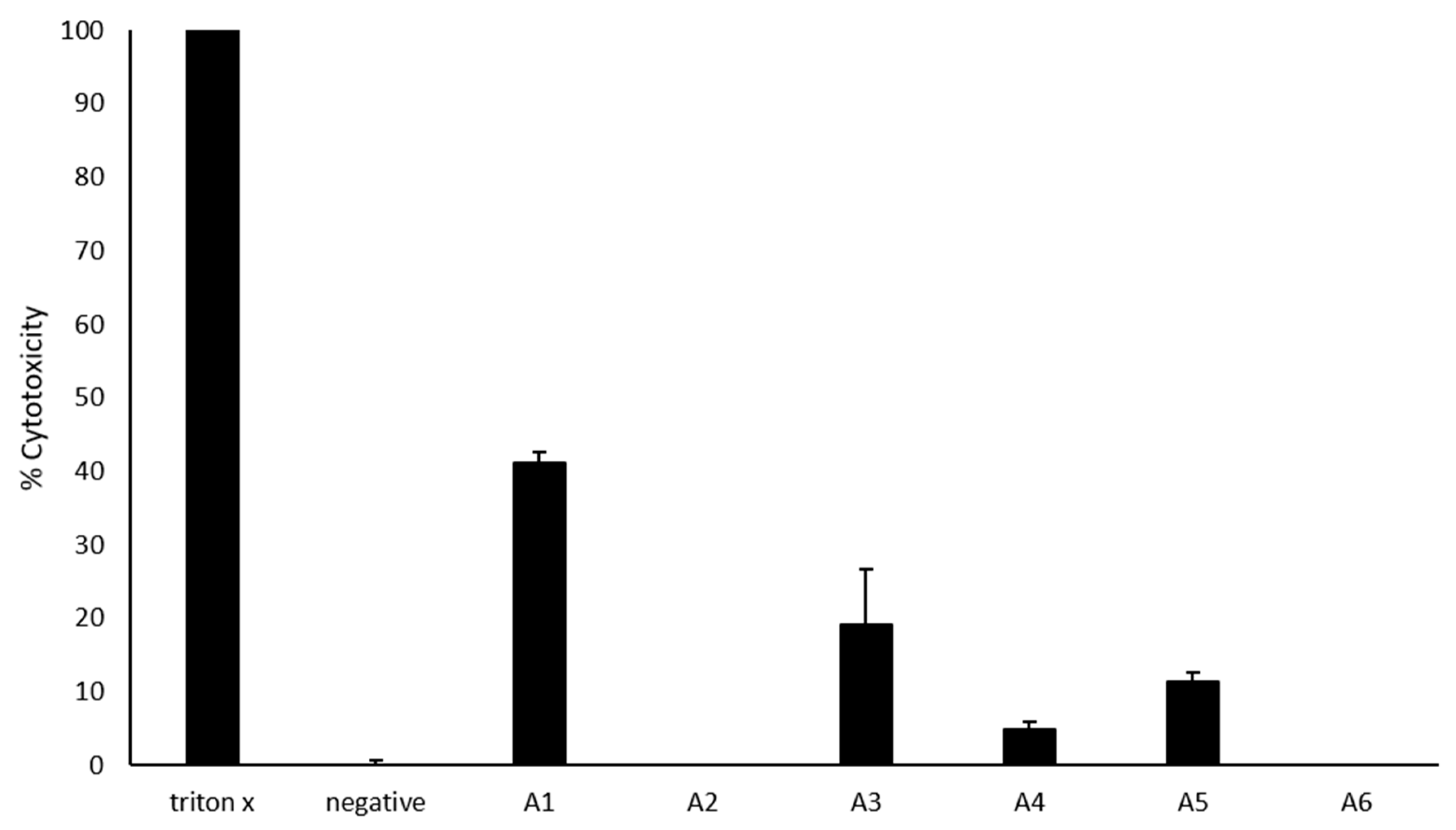
2.6. Most Azole Compounds Did Not Exhibit High Cytotoxicity
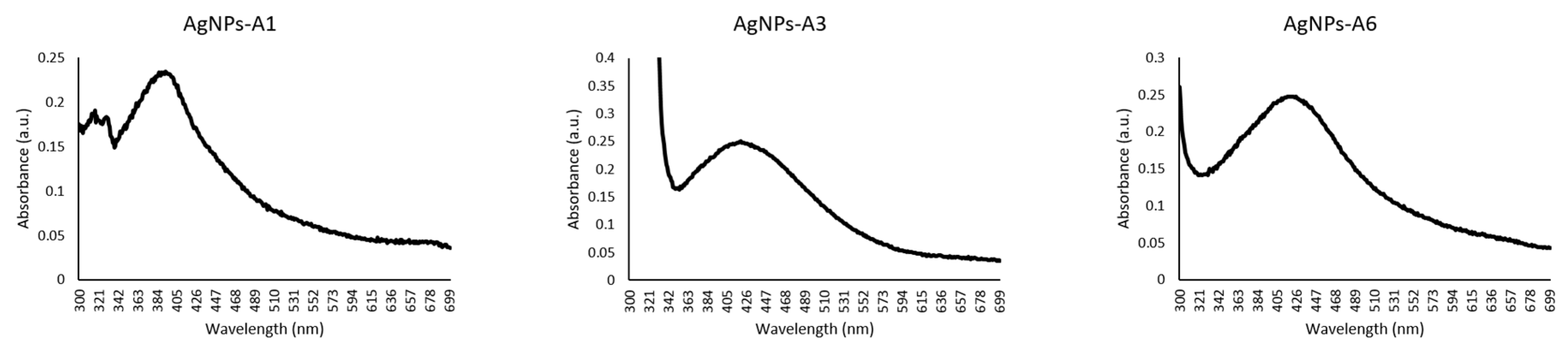
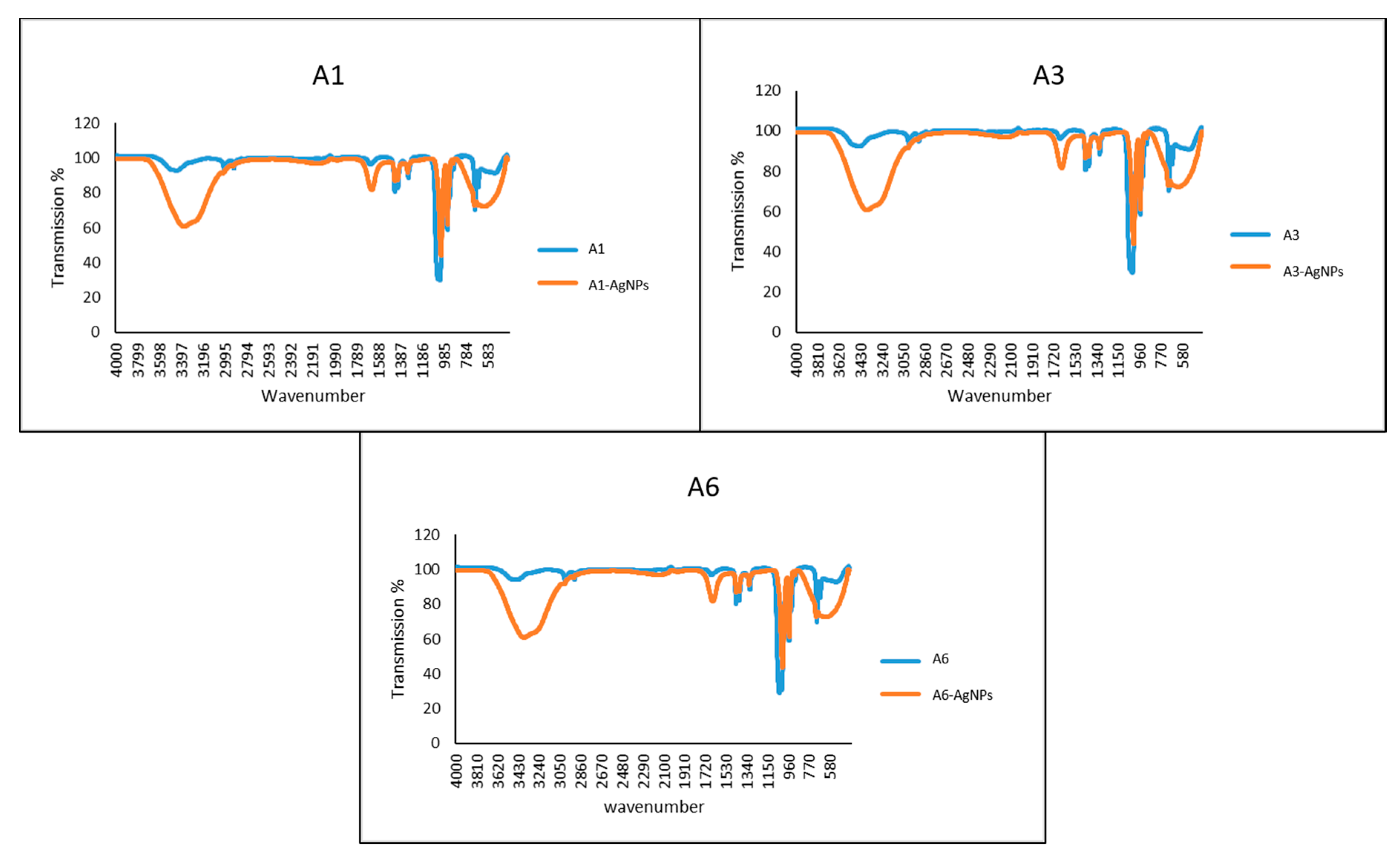
2.7. Characterization of Azoles-AgNPs
2.8. Conjugation with Silver Nanoparticles Significantly Improved Amoebicidal Effects of A4 against B. mandrillaris
2.9. Conjugation with Silver Nanoparticles Significantly Improved Amoebicidal Effects of A1 against N. fowleri
3. Discussion
4. Methods
4.1. Human Cervical Adenocarcinoma Cells (Hela) Cultivation
4.2. Cultivation of Amoebae
4.3. Amoebicidal Assays
4.4. Amoebistatic Assays
4.5. Cytotoxicity Assays
4.6. Synthesis of Azoles
4.6.1. Overall Technique for the Synthesis of Benzimidazoles (A1 and A2)
4.6.2. Overall Technique for the Synthesis of (E)-2-(6,6-dimethyl-1-phenyl-1,5,6,7-tetrahydro-4H-indazol-4-ylidene)hydrazine-1-carbothioamide (A4)
4.6.3. Overall Technique for the Synthesis of Tetrazoles (A5 and A6)
4.7. Nanoparticle Conjugation
4.8. Characterization of Nanoparticle Conjugation
4.8.1. Spectrometric Analysis
4.8.2. Dynamic Light Scattering
4.8.3. Fourier-Transform Infrared Spectroscopy
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Schuster, F.L.; Visvesvara, G.S. Opportunistic amoebae: Challenges in prophylaxis and treatment. Drug Resist. Updates 2004, 7, 41–51. [Google Scholar] [CrossRef]
- Visvesvara, G.S. Infections with free-living amebae. In Handbook of Clinical Neurology; Elsevier: Amsterdam, The Netherlands, 2013; Volume 114, pp. 153–168. [Google Scholar]
- La Hoz, R.M.; Morris, M.I. Infectious Diseases Community of Practice of the American Society of Transplantation. Tissue and Blood Protozoa including Toxoplasmosis, Chagas disease, Leishmaniasis, Babesia, Acanthamoeba, Balamuthia, & Naegleria in Solid Organ Transplant Recipients-Guidelines from the Infectious Diseases Community of Practice of the American Society of Transplantation. Clin. Transplant. 2019, 33, e13546. [Google Scholar]
- Matin, A.; Siddiqui, R.; Jayasekera, S.; Khan, N.A. Increasing importance of Balamuthia mandrillaris. Clin. Microbiol. Rev. 2008, 21, 435–448. [Google Scholar] [CrossRef] [Green Version]
- Mungroo, M.R.; Anwar, A.; Khan, N.A.; Siddiqui, R. Brain-eating amoebae infection: Challenges and opportunities in chemotherapy. Mini Rev. Med. Chem. 2019, 19, 980–987. [Google Scholar] [CrossRef] [PubMed]
- Shaharyar, M.; Mazumder, A. Benzimidazoles: A biologically active compounds. Arab. J. Chem. 2017, 10, S157–S173. [Google Scholar]
- Kopańska, K.; Najda, A.; Żebrowska, J.; Chomicz, L.; Piekarczyk, J.; Myjak, P.; Bretner, M. Synthesis and activity of 1H-benzimidazole and 1H-benzotriazole derivatives as inhibitors of Acanthamoeba castellanii. Bioorg. Med. Chem. 2004, 12, 2617–2624. [Google Scholar] [CrossRef] [PubMed]
- Torres-Gómez, H.; Hernández-Núñez, E.; León-Rivera, I.; Guerrero-Alvarez, J.; Cedillo-Rivera, R.; Moo-Puc, R.; Argotte-Ramos, R.; del Carmen Rodríguez-Gutiérrez, M.; Chan-Bacab, M.J.; Navarrete-Vázquez, G. Design, synthesis and in vitro antiprotozoal activity of benzimidazole-pentamidine hybrids. Bioorg. Med. Chem. Lett. 2008, 18, 3147–3151. [Google Scholar] [CrossRef] [PubMed]
- Mavrova, A.T.; Vuchev, D.; Anichina, K.; Vassilev, N. Synthesis, antitrichinnellosis and antiprotozoal activity of some novel thieno-[2,3-d]-pyrimidin-4(3H)-ones containing benzimidazole ring. Eur. J. Med. Chem. 2010, 45, 5856–5861. [Google Scholar] [CrossRef]
- Valdez-Padilla, D.; Rodríguez-Morales, S.; Hernández-Campos, A.; Hernández-Luis, F.; Yépez-Mulia, L.; Tapia-Contreras, A.; Castillo, R. Synthesis and antiprotozoal activity of novel 1-methylbenzimidazole derivatives. Bioorg. Med. Chem. 2009, 17, 1724–1730. [Google Scholar] [CrossRef]
- Navarrete-Vázquez, G.; Cedillo, R.; Hernández-Campos, A.; Yépez, L.; Hernández-Luis, F.; Valdez, J.; Morales, R.; Cortés, R.; Hernández, M.; Castillo, R. Synthesis and antiparasitic activity of 2-(trifluoromethyl) benzimidazole derivatives. Bioorg. Med. Chem. Lett. 2001, 11, 187–190. [Google Scholar] [CrossRef]
- Navarrete-Vázquez, G.; de Monserrat Rojano-Vilchis, M.; Yépez-Mulia, L.; Meléndez, V.; Gerena, L.; Hernández-Campos, A.; Castillo, R.; Hernández-Luis, F. Synthesis and antiprotozoal activity of some 2-(trifluoromethyl)-1H-benzimidazole bioisosteres. Eur. J. Med. Chem. 2006, 41, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Ismail, M.A.; Brun, R.; Wenzler, T.; Tanious, F.A.; Wilson, W.D.; Boykin, D.W. Dicationic biphenyl benzimidazole derivatives as antiprotozoal agents. Bioorg. Med. Chem. 2004, 12, 5405–5413. [Google Scholar] [CrossRef] [PubMed]
- Gaikwad, D.D.; Chapolikar, A.D.; Devkate, C.G.; Warad, K.D.; Tayade, A.P.; Pawar, R.P.; Domb, A.J. Synthesis of indazole motifs and their medicinal importance: An overview. Eur. J. Med. Chem. 2015, 90, 707–731. [Google Scholar] [CrossRef] [PubMed]
- Cerecetto, H.; Gerpe, A.; González, M.; Aran, V.J.; de Ocariz, C.O. Pharmacological properties of indazole derivatives: Recent developments. Mini Rev. Med. Chem. 2005, 5, 869–878. [Google Scholar] [CrossRef] [PubMed]
- Vyas, D.H.; Tala, S.D.; Akbari, J.D.; Dhaduk, M.F.; Joshi, H.S. Synthesis, antimicrobial and antitubercular activity of some cyclohexenone and indazole derivatives. Indian J. Chem. Sect. B 2009, 48, 1405–1410. [Google Scholar]
- Li, X.; Chu, S.; Feher, V.A.; Khalili, M.; Nie, Z.; Margosiak, S.; Nikulin, V.; Levin, J.; Sprankle, K.G.; Tedder, M.E.; et al. Structure-based design, synthesis, and antimicrobial activity of indazole-derived SAH/MTA nucleosidase inhibitors. J. Med. Chem. 2003, 46, 5663–5673. [Google Scholar] [CrossRef]
- Kaushik, N.; Kumar, N.; Kumar, A.; Singh, U.K. Tetrazoles: Synthesis and Biological Activity. Immunol. Endocr. Metab. Agents Med. Chem. Former. Curr. Med. Chem. Immunol. Endocr. Metab. Agents 2018, 18, 3–21. [Google Scholar] [CrossRef]
- Upadhayaya, R.S.; Jain, S.; Sinha, N.; Kishore, N.; Chandra, R.; Arora, S.K. Synthesis of novel substituted tetrazoles having antifungal activity. Eur. J. Med. Chem. 2004, 39, 579–592. [Google Scholar] [CrossRef]
- Matysiak, J.; Niewiadomy, A.; Krajewska-Kułak, E.; Mącik-Niewiadomy, G. Synthesis of some 1-(2,4-dihydroxythiobenzoyl) imidazoles,-imidazolines and-tetrazoles and their potent activity against Candida species. Il Farm. 2003, 58, 455–461. [Google Scholar] [CrossRef]
- Mohite, P.B.; Bhaskar, V.H. Synthesis and antifungal activity of 3-aryl-1-(5-phenyl-1H-tetrazol-1-yl)-prop-2-en-1-one. Orbital Electron. J. Chem. 2011, 2, 311–315. [Google Scholar]
- Wittenberger, S.J. Recent developments in tetrazole chemistry. A review. Org. Prep. Proced. Int. 1994, 26, 499–531. [Google Scholar] [CrossRef]
- Mohite, P.B.; Bhaskar, V.H. Potential pharmacological activities of tetrazoles in the new millennium. Int. J. PharmTech Res. 2011, 3, 1557–1566. [Google Scholar]
- Rajendran, K.; Anwar, A.; Khan, N.A.; Siddiqui, R. Brain-Eating Amoebae: Silver Nanoparticle Conjugation Enhanced Efficacy of Anti-Amoebic Drugs against Naegleria fowleri. ACS Chem. Neurosci. 2017, 8, 2626–2630. [Google Scholar] [CrossRef]
- Anwar, A.; Masri, A.; Rao, K.; Rajendran, K.; Khan, N.A.; Shah, M.R.; Siddiqui, R. Antimicrobial activities of green synthesized gums-stabilized nanoparticles loaded with flavonoids. Sci. Rep. 2019, 9, 3122. [Google Scholar] [CrossRef] [PubMed]
- Padzik, M.; Hendiger, E.B.; Chomicz, L.; Grodzik, M.; Szmidt, M.; Grobelny, J.; Lorenzo-Morales, J. Tannic acid-modified silver nanoparticles as a novel therapeutic agent against Acanthamoeba. Parasitol. Res. 2018, 117, 3519–3525. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rafique, R.; Khan, K.M.; Chigurupati, S.; Wadood, A.; Rehman, A.U.; Karunanidhi, A.; Hameed, S.; Taha, M.; al-Rashida, M. Synthesis of new indazole based dual inhibitors of α-glucosidase and α-amylase enzymes, their in vitro, in silico and kinetics studies. Bioorg. Chem. 2020, 94, 103195. [Google Scholar] [CrossRef]
- Fatima, I.; Zafar, H.; Khan, K.M.; Saad, S.M.; Javaid, S.; Perveen, S.; Choudhary, M.I. Synthesis, molecular docking and xanthine oxidase inhibitory activity of 5-aryl-1H-tetrazoles. Bioorg. Chem. 2018, 79, 201–211. [Google Scholar] [CrossRef]
- Gerpe, A.; Aguirre, G.; Boiani, L.; Cerecetto, H.; González, M.; Olea-Azar, C.; Rigol, C.; Maya, J.D.; Morello, A.; Piro, O.E.; et al. Indazole N-oxide derivatives as antiprotozoal agents: Synthesis, biological evaluation and mechanism of action studies. Bioorg. Med. Chem. 2006, 14, 3467–3480. [Google Scholar] [CrossRef] [Green Version]
- Blanco, E.; Shen, H.; Ferrari, M. Principles of nanoparticle design for overcoming biological barriers to drug delivery. Nat. Biotechnol. 2015, 33, 941. [Google Scholar] [CrossRef]
- Singh, R.; Lillard, J.W., Jr. Nanoparticle-based targeted drug delivery. Exp. Mol. Pathol. 2009, 86, 215–223. [Google Scholar] [CrossRef] [Green Version]
- Siddiqui, R.; Jeyamogan, S.; Ali, S.M.; Abbas, F.; Sagathevan, K.A.; Khan, N.A. Crocodiles and alligators: Antiamoebic and antitumor compounds of crocodiles. Exp. Parasitol. 2017, 183, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Mariappan, M.; Alagarsamy, R.; Panneerselvam, A.; Veerappan, A.; Rajendran, S.; Arunachalam, J. Synthesis, solvatochromism, photochemistry, DNA binding, photocleavage, cytotoxicity and molecular docking studies of a ruthenium (II) complex bearing photoactive subunit. J. Photochem. Photobiol. A Chem. 2018, 356, 617–626. [Google Scholar] [CrossRef]
- Kulsoom, H.; Baig, A.M.; Siddiqui, R.; Khan, N.A. Combined drug therapy in the management of granulomatous amoebic encephalitis due to Acanthamoeba spp., and Balamuthia mandrillaris. Exp. Parasitol. 2014, 145, S115–S120. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.A. Pathogenicity, Morphology, and Differentiation of Acanthamoeba. Curr. Microbiol. 2001, 43, 391–395. [Google Scholar] [CrossRef] [PubMed]
- Adegboye, A.A.; Khan, K.M.; Salar, U.; Aboaba, S.A.; Chigurupati, S.; Fatima, I.; Taha, M.; Wadood, A.; Mohammad, J.I.; Khan, H.; et al. 2-Aryl benzimidazoles: Synthesis, In vitro α-amylase inhibitory activity, and molecular docking study. Eur. J. Med. Chem. 2018, 150, 248–260. [Google Scholar] [CrossRef]
- Anwar, A.; Shah, M.R.; Muhammad, S.P.; Afridi, S.; Ali, K. Thio-pyridinium capped silver nanoparticle based supramolecular recognition of Cu (I) in real samples and T-lymphocytes. New J. Chem. 2016, 40, 6480–6486. [Google Scholar] [CrossRef]


| Class of Compound | Code | Structure | IUPAC Name |
|---|---|---|---|
| Benzimidazole | A1 |  | 2-[3-Bromo-4,5-bis(methoxy)phenyl]-1H-benzimidazole |
| Benzimidazole | A2 |  | 2-(5-Methylfuran-2-yl)-1H-benz]imidazole |
| Indazole | A3 |  | (E)-N-(2-Chlorophenyl)-2-(6,6-dimethyl-1-phenyl-1,5,6,7-tetrahydro-4H-indazol-4-4ylidene)hydrazine-1-carbothioamide |
| Indazole | A4 |  | (E)-2-(6,6-Dimethyl-1-phenyl-1,5,6,7-tetrahydro-4H-indazol-4-4ylidene)hydrazine-1-carbothioamide |
| Tetrazole | A5 |  | 5-(3-Chlorobenzyl)-1H-tetrazole |
| Tetrazole | A6 |  | 5-Phenyl-1H-tetrazole |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anwar, A.; Mungroo, M.R.; Khan, S.; Fatima, I.; Rafique, R.; Kanwal; Khan, K.M.; Siddiqui, R.; Khan, N.A. Novel Azoles as Antiparasitic Remedies against Brain-Eating Amoebae. Antibiotics 2020, 9, 188. https://0-doi-org.brum.beds.ac.uk/10.3390/antibiotics9040188
Anwar A, Mungroo MR, Khan S, Fatima I, Rafique R, Kanwal, Khan KM, Siddiqui R, Khan NA. Novel Azoles as Antiparasitic Remedies against Brain-Eating Amoebae. Antibiotics. 2020; 9(4):188. https://0-doi-org.brum.beds.ac.uk/10.3390/antibiotics9040188
Chicago/Turabian StyleAnwar, Ayaz, Mohammad Ridwane Mungroo, Simal Khan, Itrat Fatima, Rafaila Rafique, Kanwal, Khalid Mohammed Khan, Ruqaiyyah Siddiqui, and Naveed Ahmed Khan. 2020. "Novel Azoles as Antiparasitic Remedies against Brain-Eating Amoebae" Antibiotics 9, no. 4: 188. https://0-doi-org.brum.beds.ac.uk/10.3390/antibiotics9040188







