Zolmitriptan Intranasal Spanlastics for Enhanced Migraine Treatment; Formulation Parameters Optimized via Quality by Design Approach
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Zolmitriptan-Loaded Spanlastic (ZLS) Formulations
2.3. Statistic Optimization of Spanlastic Formulations
The Design of the Experiment
2.4. Characterization of Zolmitriptan-Loaded Spanlastic (ZLS) Formulations
2.4.1. Entrapment Efficiency Percentage (EE%) Determination
2.4.2. Particle Size Analysis
2.5. Design Space Optimization and Model Validation
2.6. Optimized Formula Characterization
2.6.1. Entrapment Efficiency Percentage Determination
2.6.2. Determination of Particle Size, Polydispersity Index (PDI) and Zeta Potential (ZP)
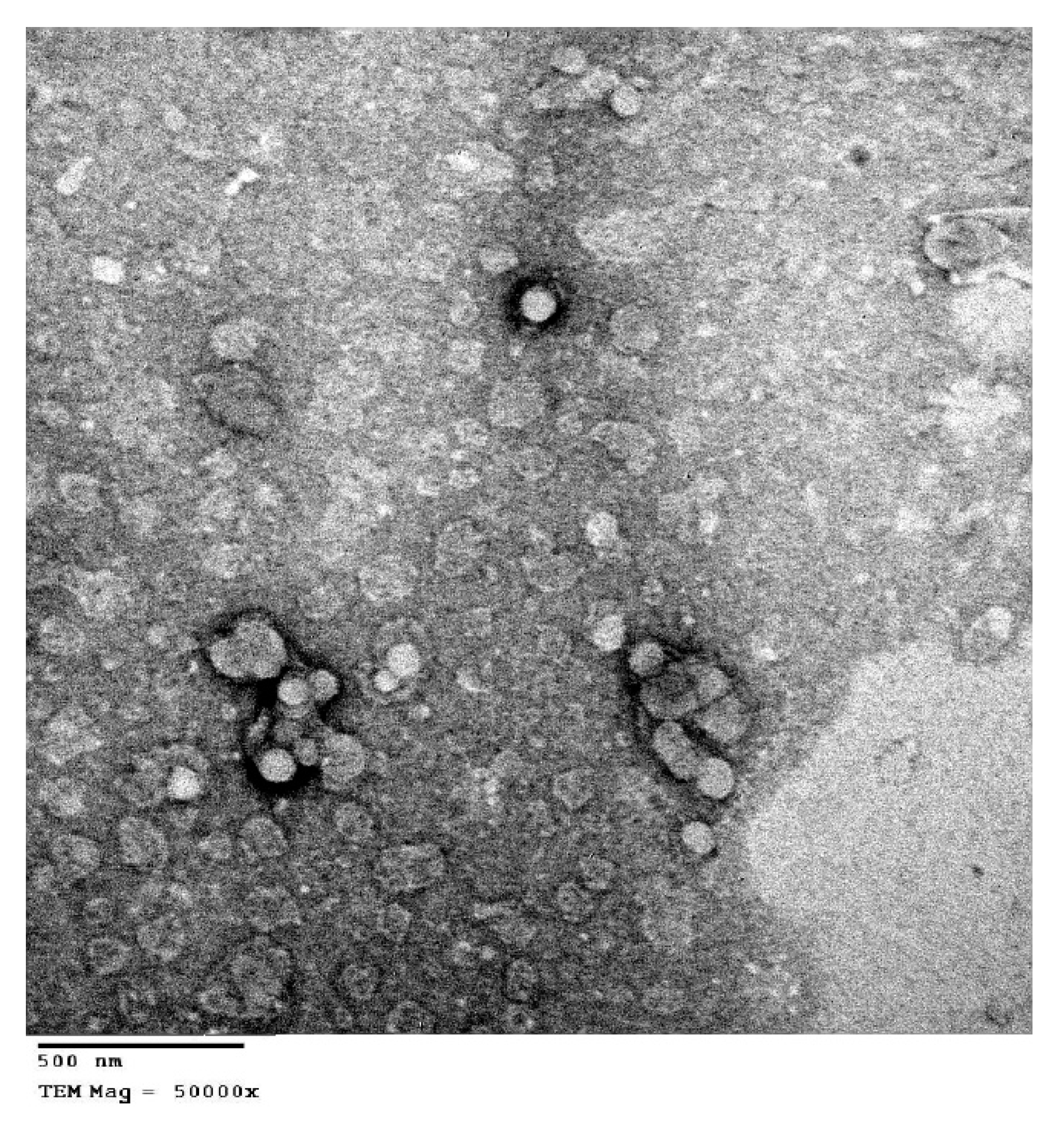
2.6.3. Transmission Electron Microscope (TEM) Imaging
2.7. Preparation of Zolmitriptan-Loaded Spanlastic (ZLS) Nasal Gel
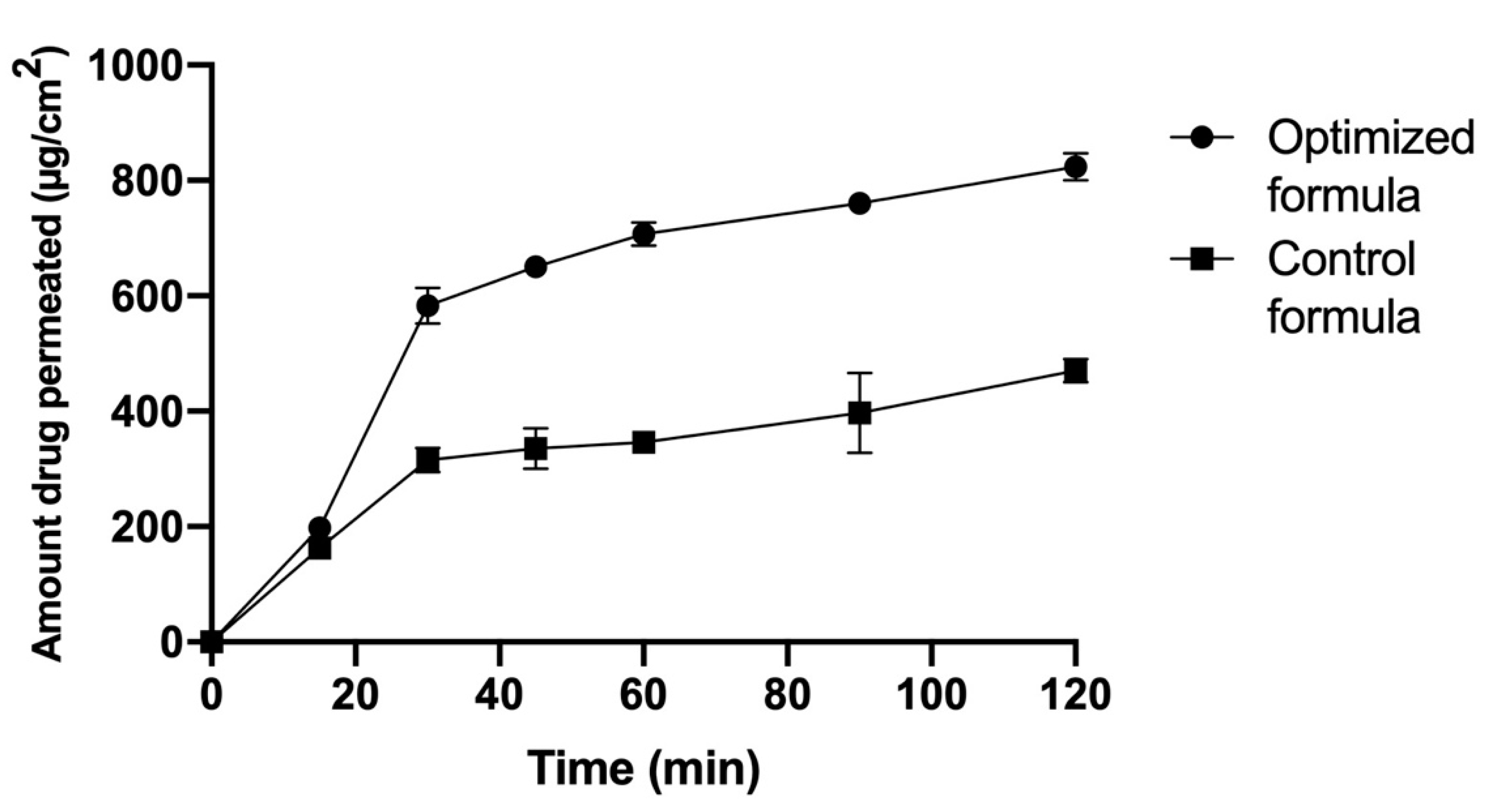
2.8. Ex Vivo Permeation
3. Results and Discussion
3.1. Influence of Formulation Factors on CQAs
3.1.1. Influence of Formulation Parameters on Entrapment Efficiency Percentage
3.1.2. Influence of Formulation Parameters on Particle Size
3.2. Optimized Formula Characterization
3.2.1. Entrapment Efficiency, Particle Size, Zeta Potential and Polydispersity Index (PDI) of the Optimized Formula
3.2.2. Transmission Electron Microscope (TEM) Imaging
3.2.3. Ex Vivo Permeation
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- De Vries, T.; Villalón, C.M.; MaassenVanDenBrink, A. Pharmacological treatment of migraine: CGRP and 5-HT beyond the triptans. Pharmacol. Ther. 2020, 211, 107528. [Google Scholar] [CrossRef]
- Mahmoud, A.A.; Salah, S. Fast relief from migraine attacks using fast-disintegrating sublingual zolmitriptan tablets. Drug Dev. Ind. Pharm. 2012, 38, 762–769. [Google Scholar] [CrossRef]
- Malhotra, R. Understanding migraine: Potential role of neurogenic inflammation. Ann. Indian Acad. Neurol. 2016, 19, 175–182. [Google Scholar] [CrossRef]
- Poply, K.; Bahra, A.; Mehta, V. Migraine. BJA Educ. 2016, 16, 357–361. [Google Scholar] [CrossRef] [Green Version]
- Spierings, E.L.; Brandes, J.L.; Kudrow, D.B.; Weintraub, J.; Schmidt, P.C.; Kellerman, D.J.; Tepper, S.J. Randomized, double-blind, placebo-controlled, parallel-group, multi-center study of the safety and efficacy of ADAM zolmitriptan for the acute treatment of migraine. Cephalalgia 2018, 38, 215–224. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hargreaves, R.; Olesen, J. Calcitonin Gene-Related Peptide Modulators—The History and Renaissance of a New Migraine Drug Class. Headache J. Head Face Pain 2019, 59, 951–970. [Google Scholar] [CrossRef] [PubMed]
- Tepper, S.J.; Rapoport, A.M.; Sheftell, F.D. Mechanisms of Action of the 5-HT1B/1D Receptor Agonists. Arch. Neurol. 2002, 59, 1084–1088. [Google Scholar] [CrossRef] [PubMed]
- Prajapati, S.T.; Patel, M.V.; Patel, C.N. Preparation and evaluation of sublingual tablets of zolmitriptan. Int. J. Pharm. Investig. 2014, 4, 27–31. [Google Scholar] [CrossRef] [Green Version]
- Saxena, P.R.; Tfelt-Hansen, P. Success and failure of triptans. J. Headache Pain 2001, 2, 3–11. [Google Scholar] [CrossRef] [Green Version]
- Mostafa, D.A.E.; Khalifa, M.K.A.; Gad, S.S. Zolmitriptan brain targeting via intranasal route using solid lipid nanoparticles for migraine therapy: Formulation, characterization, in-vitro and in-vivo assessment. Int. J. Appl. Pharm. 2020, 10, 86–93. [Google Scholar] [CrossRef] [Green Version]
- Aurora, S.K.; Papapetropoulos, S.; Kori, S.H.; Kedar, A.; Abell, T.L. Gastric stasis in migraineurs: Etiology, characteristics, and clinical and therapeutic implications. Cephalalgia 2013, 33, 408–415. [Google Scholar] [CrossRef] [Green Version]
- Meairs, S.; Alonso, A. Ultrasound, microbubbles and the blood-brain barrier. Prog. Biophys. Mol. Biol. 2007, 93, 354–362. [Google Scholar] [CrossRef]
- Pailla, S.R.; Talluri, S.; Rangaraj, N.; Ramavath, R.; Challa, V.S.; Doijad, N.; Sampathi, S. Intranasal Zotepine Nanosuspension: Intended for improved brain distribution in rats. DARU J. Pharm. Sci. 2019, 27, 541–556. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Liu, L. Modern methods for delivery of drugs across the blood-brain barrier. Adv. Drug Deliv. Rev. 2012, 64, 640–665. [Google Scholar] [CrossRef] [PubMed]
- Sonvico, F.; Clementino, A.; Buttini, F.; Colombo, G.; Pescina, S.; Guterres, S.S.; Pohlmann, A.R.; Nicoli, S. Surface-Modified Nanocarriers for Nose-to-Brain Delivery: From Bioadhesion to Targeting. Pharmaceutics 2018, 10, 34. [Google Scholar] [CrossRef] [Green Version]
- Abdelmonem, R.; El Nabarawi, M.; Attia, A. Development of novel bioadhesive granisetron hydrochloride spanlastic gel and insert for brain targeting and study their effects on rats. Drug Deliv. 2018, 25, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Li, B.; Zhang, Y.; Chen, T.; Chen, C.; Jiang, W.; Wang, Q.; Chen, T. Intranasal delivery of paeoniflorin nanocrystals for brain targeting. Asian J. Pharm. Sci. 2020, 15, 326–335. [Google Scholar] [CrossRef]
- Nowak, M.; Brown, T.D.; Graham, A.; Helgeson, M.E.; Mitragotri, S. Size, shape, and flexibility influence nanoparticle transport across brain endothelium under flow. Bioeng. Transl. Med. 2020, 5, e10153. [Google Scholar] [CrossRef] [Green Version]
- Abdelrahman, F.E.; Elsayed, I.; Gad, M.K.; Elshafeey, A.H.; Mohamed, M.I. Response surface optimization, ex vivo and in vivo investigation of nasal spanlastics for bioavailability enhancement and brain targeting of risperidone. Int. J. Pharm. 2017, 530, 1–11. [Google Scholar] [CrossRef]
- El-Nabarawy, N.A.; Teaima, M.H.; Helal, D.A. Assessment of spanlastic vesicles of zolmitriptan for treating migraine in rats. Drug Des. Dev. Ther. 2019, 13, 3929. [Google Scholar] [CrossRef] [Green Version]
- Kakkar, S.; Kaur, I.P. Spanlastics—A novel nanovesicular carrier system for ocular delivery. Int. J. Pharm. 2011, 413, 202–210. [Google Scholar] [CrossRef]
- Yassin, G.E.; Amer, R.I.; Fayez, A.M. Carbamazepine loaded vesicular structures for enhanced brain targeting via intranasal route: Optimization, in vitro evaluation, and in vivo study. Int. J. Appl. Pharm. 2019, 11, 264–274. [Google Scholar] [CrossRef]
- Namjoshi, S.; Dabbaghi, M.; Roberts, M.S.; Grice, J.E.; Mohammed, Y. Quality by Design: Development of the Quality Target Product Profile (QTPP) for Semisolid Topical Products. Pharmaceutics 2020, 12, 287. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shirsat, A.E.; Chitlange, S.S. Application of quality by design approach to optimize process and formulation parameters of rizatriptan loaded chitosan nanoparticles. J. Adv. Pharm. Technol. Res. 2015, 6, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Barakat, N.S. Optimization of physical characterization, skin permeation of naproxen from glycofurol-based topical gel. Asian J. Pharm. 2014, 4. [Google Scholar] [CrossRef]
- Sharma, A.; Pahwa, S.; Bhati, S.; Kudeshia, P. Spanlastics: A modern approach for nanovesicular drug delivery system. Int. J. Pharm. Sci. Res. 2020, 11, 1057–1065. [Google Scholar] [CrossRef]
- Ranade, S.; Thiagarajan, P. Selection of a design for response surface. IOP Conf. Ser. Mater. Sci. Eng. 2017, 263, 022043. [Google Scholar] [CrossRef] [Green Version]
- Elsenosy, F.M.; Abdelbary, G.A.; Elshafeey, A.H.; Elsayed, I.; Fares, A.R. Brain Targeting of Duloxetine HCL via Intranasal Delivery of Loaded Cubosomal Gel: In Vitro Characterization, ex vivo Permeation, and in vivo Biodistribution Studies. Int. J. Nanomed. 2020, 15, 9517–9537. [Google Scholar] [CrossRef]
- Yoshioka, T.; Sternberg, B.; Florence, A.T. Preparation and properties of vesicles (niosomes) of sorbitan monoesters (Span 20, 40, 60 and 80) and a sorbitan triester (Span 85). Int. J. Pharm. 1994, 105, 1–6. [Google Scholar] [CrossRef]
- Al-Mahallawi, A.M.; Abdelbary, A.A.; Aburahma, M.H. Investigating the potential of employing bilosomes as a novel vesicular carrier for transdermal delivery of tenoxicam. Int. J. Pharm. 2015, 485, 329–340. [Google Scholar] [CrossRef]
- Fahmy, A.M.; El-Setouhy, D.A.; Habib, B.A.; Tayel, S.A. Enhancement of Transdermal Delivery of Haloperidol via Spanlastic Dispersions: Entrapment Efficiency vs. Particle Size. AAPS PharmSciTech 2019, 20, 95. [Google Scholar] [CrossRef]
- Shilo, M.; Sharon, A.; Baranes, K.; Motiei, M.; Lellouche, J.-P.M.; Popovtzer, R. The effect of nanoparticle size on the probability to cross the blood-brain barrier: An in-vitro endothelial cell model. J. Nanobiotechnol. 2015, 13, 19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mazyed, E.; Helal, D.; Elkhoudary, M.; Elhameed, A.A.; Yasser, M. Formulation and Optimization of Nanospanlastics for Improving the Bioavailability of Green Tea Epigallocatechin Gallate. Pharmaceuticals 2021, 14, 68. [Google Scholar] [CrossRef] [PubMed]
- Mehanna, M.M.; Motawaa, A.M.; Samaha, M.W. Nanovesicular carrier-mediated transdermal delivery of tadalafil: I-Formulation and physicsochemical characterization. Drug Dev. Ind. Pharm. 2015, 41, 714–721. [Google Scholar] [CrossRef] [PubMed]
- Kamboj, S.; Saini, V.; Bala, S. Formulation and Characterization of Drug Loaded Nonionic Surfactant Vesicles (Niosomes) for Oral Bioavailability Enhancement. Sci. World J. 2014, 2014, 959741. [Google Scholar] [CrossRef] [Green Version]
- Wang, N.; Hsu, C.; Zhu, L.; Tseng, S.; Hsu, J.-P. Influence of metal oxide nanoparticles concentration on their zeta potential. J. Colloid Interface Sci. 2013, 407, 22–28. [Google Scholar] [CrossRef]
- Junyaprasert, V.B.; Teeranachaideekul, V.; Supaperm, T. Effect of Charged and Non-ionic Membrane Additives on Physicochemical Properties and Stability of Niosomes. AAPS PharmSciTech 2008, 9, 851–859. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, Y.; Ran, F.; Cui, Y.; Liu, C.; Zhao, Q.; Gao, Y.; Wang, D.; Wang, S. A comparison between sphere and rod nanoparticles regarding their in vivo biological behavior and pharmacokinetics. Sci. Rep. 2017, 7, 4131. [Google Scholar] [CrossRef]
- Vyas, T.K.; Shahiwala, A.; Marathe, S.; Misra, A. Intranasal Drug Delivery for Brain Targeting. Curr. Drug Deliv. 2005, 2, 165–175. [Google Scholar] [CrossRef]
- Kreuter, J.; Shamenkov, D.; Petrov, V.; Ramge, P.; Cychutek, K.; Koch-Brandt, C.; Alyautdin, R. Apolipoprotein-mediated Transport of Nanoparticle-bound Drugs across the Blood-Brain Barrier. J. Drug Target. 2002, 10, 317–325. [Google Scholar] [CrossRef]
- El Zaafarany, G.M.; Awad, G.A.; Holayel, S.M.; Mortada, N.D. Role of edge activators and surface charge in developing ultradeformable vesicles with enhanced skin delivery. Int. J. Pharm. 2010, 397, 164–172. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, R.; Sandhu, S.K.; Sharma, I.; Kaur, I.P. Development and Evaluation of Curcumin-loaded Elastic Vesicles as an Effective Topical Anti-inflammatory Formulation. AAPS PharmSciTech 2015, 16, 364–374. [Google Scholar] [CrossRef] [PubMed]
- Ioele, G.; Tavano, L.; De Luca, M.; Ragno, G.; Picci, N.; Muzzalupo, R. Photostability and ex-vivo permeation studies on diclofenac in topical niosomal formulations. Int. J. Pharm. 2015, 494, 490–497. [Google Scholar] [CrossRef] [PubMed]

| Formulation Parameters (Factors) | Levels | ||
|---|---|---|---|
| −1 | 0 | 1 | |
| X1: Span 60 concentration | 65 mg | 75 mg | 85mg |
| X2: Tween 80 concentration | 15 mg | 25 mg | 35 mg |
| Response (CQAs) | constraints | ||
| Y1: Entrapment efficiency | maximize | ||
| Y2: Particle size | minimize | ||
| Formulation Parameters | CQAs (Responses) | |||
|---|---|---|---|---|
| Run | X1: Span 60 Concentration | X2: Tween 80 Concentration | Y1: Entrapment Efficiency (%) | Y2: Particle Size (nm) |
| 1 | 0 | −1.41421 | 131.2 | 53.42 |
| 2 | 0 | 0 | 96.12 | 48.32 |
| 3 | −1 | 1 | 40.75 | 20.7 |
| 4 | 0 | 1.41421 | 90.17 | 40.21 |
| 5 | 0 | 0 | 96.12 | 48.32 |
| 6 | 0 | 0 | 96.12 | 48.32 |
| 7 | −1 | −1 | 21.07 | 55.21 |
| 8 | 0 | 0 | 96.12 | 48.32 |
| 9 | 1.41421 | 0 | 172.3 | 61.92 |
| 10 | −1.41421 | 0 | 60.73 | 23.22 |
| 11 | 1 | −1 | 175.24 | 63.47 |
| 12 | 0 | 0 | 96.12 | 48.32 |
| 13 | 1 | 1 | 168.4 | 57.98 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saleh, A.; Khalifa, M.; Shawky, S.; Bani-Ali, A.; Eassa, H. Zolmitriptan Intranasal Spanlastics for Enhanced Migraine Treatment; Formulation Parameters Optimized via Quality by Design Approach. Sci. Pharm. 2021, 89, 24. https://0-doi-org.brum.beds.ac.uk/10.3390/scipharm89020024
Saleh A, Khalifa M, Shawky S, Bani-Ali A, Eassa H. Zolmitriptan Intranasal Spanlastics for Enhanced Migraine Treatment; Formulation Parameters Optimized via Quality by Design Approach. Scientia Pharmaceutica. 2021; 89(2):24. https://0-doi-org.brum.beds.ac.uk/10.3390/scipharm89020024
Chicago/Turabian StyleSaleh, Asmaa, Maha Khalifa, Seham Shawky, Amjaad Bani-Ali, and Heba Eassa. 2021. "Zolmitriptan Intranasal Spanlastics for Enhanced Migraine Treatment; Formulation Parameters Optimized via Quality by Design Approach" Scientia Pharmaceutica 89, no. 2: 24. https://0-doi-org.brum.beds.ac.uk/10.3390/scipharm89020024







