Minimally Invasive Surgery for Posterior Spinal Instrumentation and Fusion in Adolescent Idiopathic Scoliosis: Current Status and Future Application
Abstract
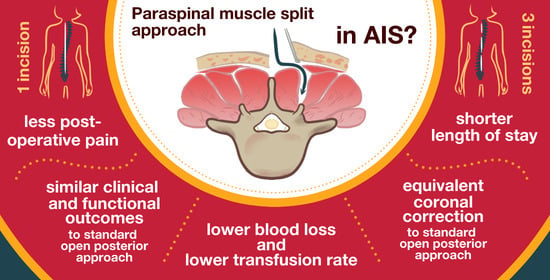
:1. Introduction
2. Surgical Technique
3. Deformity Correction and Fusion
3.1. Coronal Correction
3.2. Sagittal Correction
3.3. Fusion Rate
4. Perioperative Morbidity
4.1. Estimated Blood Loss and Allogeneic Transfusion Rate
4.2. Operative Time
4.3. Postoperative Pain and Average Opioid Consumption
4.4. Hospital Length of Stay (LOS)
4.5. Intraoperative, Perioperative, and Long-Term Complications
5. Clinical and Functional Outcomes
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Asher, M.A.; Burton, D.C. Adolescent Idiopathic Scoliosis: Natural History and Long Term Treatment Effects. Scoliosis 2006, 1, 2. [Google Scholar] [CrossRef]
- Willner, S.; Udén, A. A Prospective Prevalence Study of Scoliosis in Southern Sweden. Acta Orthop. Scand. 1982, 53, 233–237. [Google Scholar] [CrossRef]
- Rogala, E.J.; Drummond, D.S.; Gurr, J. Scoliosis: Incidence and Natural History. A Prospective Epidemiological Study. J. Bone Jt. Surg. Am. 1978, 60, 173–176. [Google Scholar] [CrossRef]
- Luk, K.D.K.; Lee, C.F.; Cheung, K.M.C.; Cheng, J.C.Y.; Ng, B.K.W.; Lam, T.P.; Mak, K.H.; Yip, P.S.F.; Fong, D.Y.T. Clinical Effectiveness of School Screening for Adolescent Idiopathic Scoliosis: A Large Population-Based Retrospective Cohort Study. Spine 2010, 35, 1607–1614. [Google Scholar] [CrossRef]
- Pesenti, S.; Jouve, J.-L.; Morin, C.; Wolff, S.; Sales De Gauzy, J.; Chalopin, A.; Ibnoulkhatib, A.; Polirsztok, E.; Walter, A.; Schuller, S.; et al. Evolution of Adolescent Idiopathic Scoliosis: Results of a Multicenter Study at 20 Years’ Follow-Up. Orthop. Traumatol. Surg. Res. 2015, 101, 619–622. [Google Scholar] [CrossRef]
- Cheng, J.C.; Castelein, R.M.; Chu, W.C.; Danielsson, A.J.; Dobbs, M.B.; Grivas, T.B.; Gurnett, C.A.; Luk, K.D.; Moreau, A.; Newton, P.O.; et al. Adolescent Idiopathic Scoliosis. Nat. Rev. Dis. Primers 2015, 1, 1–21. [Google Scholar] [CrossRef]
- Lonner, B.S.; Ren, Y.; Yaszay, B.; Cahill, P.J.; Shah, S.A.; Betz, R.R.; Samdani, A.F.; Shufflebarger, H.L.; Newton, P.O. Evolution of Surgery for Adolescent Idiopathic Scoliosis Over 20 Years: Have Outcomes Improved? Spine 2018, 43, 402–410. [Google Scholar] [CrossRef]
- Block, A.M.; Tamburini, L.M.; Zeng, F.; Mancini, M.R.; Jackson, C.A.; Antonacci, C.L.; Karsmarski, O.P.; Stelzer, J.W.; Wellington, I.J.; Lee, M.C. Surgical Treatment of Pediatric Scoliosis: Historical Origins and Review of Current Techniques. Bioengineering 2022, 9, 600. [Google Scholar] [CrossRef]
- Suk, S.-I.; Kim, W.-J.; Lee, S.-M.; Kim, J.-H.; Chung, E.-R. Thoracic Pedicle Screw Fixation in Spinal Deformities: Are They Really Safe? Spine 2001, 26, 2049–2057. [Google Scholar] [CrossRef]
- Suk, S.I.; Lee, C.K.; Kim, W.J.; Chung, Y.J.; Park, Y.B. Segmental Pedicle Screw Fixation in the Treatment of Thoracic Idiopathic Scoliosis. Spine 1995, 20, 1399–1405. [Google Scholar] [CrossRef]
- Lonner, B.S.; Auerbach, J.D.; Boachie-Adjei, O.; Shah, S.A.; Hosogane, N.; Newton, P.O. Treatment of Thoracic Scoliosis: Are Monoaxial Thoracic Pedicle Screws the Best Form of Fixation for Correction? Spine 2009, 34, 845–851. [Google Scholar] [CrossRef]
- Lonner, B.S.; Auerbach, J.D.; Estreicher, M.B.; Kean, K.E. Thoracic Pedicle Screw Instrumentation: The Learning Curve and Evolution in Technique in the Treatment of Adolescent Idiopathic Scoliosis. Spine 2009, 34, 2158–2164. [Google Scholar] [CrossRef]
- Sarwahi, V.; Wollowick, A.L.; Sugarman, E.P.; Horn, J.J.; Gambassi, M.; Amaral, T.D. Minimally Invasive Scoliosis Surgery: An Innovative Technique in Patients with Adolescent Idiopathic Scoliosis. Scoliosis 2011, 6, 16. [Google Scholar] [CrossRef]
- Anand, N.; Baron, E.M.; Thaiyananthan, G.; Khalsa, K.; Goldstein, T.B. Minimally Invasive Multilevel Percutaneous Correction and Fusion for Adult Lumbar Degenerative Scoliosis: A Technique and Feasibility Study. J. Spinal Disord. Tech. 2008, 21, 459–467. [Google Scholar] [CrossRef]
- Dakwar, E.; Cardona, R.F.; Smith, D.A.; Uribe, J.S. Early Outcomes and Safety of the Minimally Invasive, Lateral Retroperitoneal Transpsoas Approach for Adult Degenerative Scoliosis. Neurosurg. Focus. 2010, 28, E8. [Google Scholar] [CrossRef]
- Brodano, G.B.; Martikos, K.; Vommaro, F.; Greggi, T.; Boriani, S. Less Invasive Surgery in Idiopathic Scoliosis: A Case Report. Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 24–28. [Google Scholar]
- De Bodman, C.; Ansorge, A.; Tabard-Fougère, A.; Amirghasemi, N.; Dayer, R. Clinical and Radiological Outcomes of Minimally-Invasive Surgery for Adolescent Idiopathic Scoliosis at a Minimum Two Years’ Follow-Up. Bone Jt. J. 2020, 102-B, 506–512. [Google Scholar] [CrossRef]
- De Bodman, C.; Miyanji, F.; Borner, B.; Zambelli, P.-Y.; Racloz, G.; Dayer, R. Minimally Invasive Surgery for Adolescent Idiopathic Scoliosis: Correction of Deformity and Peri-Operative Morbidity in 70 Consecutive Patients. Bone Jt. J. 2017, 99-B, 1651–1657. [Google Scholar] [CrossRef]
- Yang, J.H.; Chang, D.-G.; Suh, S.W.; Damani, N.; Lee, H.-N.; Lim, J.; Mun, F. Safety and Effectiveness of Minimally Invasive Scoliosis Surgery for Adolescent Idiopathic Scoliosis: A Retrospective Case Series of 84 Patients. Eur. Spine J. 2020, 29, 761–769. [Google Scholar] [CrossRef]
- Park, S.C.; Son, S.W.; Yang, J.H.; Chang, D.-G.; Suh, S.W.; Nam, Y.; Kim, H.J. Novel Surgical Technique for Adolescent Idiopathic Scoliosis: Minimally Invasive Scoliosis Surgery. JCM 2022, 11, 5847. [Google Scholar] [CrossRef]
- Samdani, A.F.; Asghar, J.; Miyanji, F.; Haw, J.; Haddix, K. Minimally Invasive Treatment of Pediatric Spinal Deformity. Semin. Spine Surg. 2011, 23, 72–75. [Google Scholar] [CrossRef]
- Amer Samdani, F.M. Minimally Invasive Surgery for AIS: An Early Prospective Comparison with Standard Open Posterior Surgery. J. Spine 2013, 5, 1. [Google Scholar] [CrossRef]
- Miyanji, F.; Desai, S. Minimally Invasive Surgical Options for Adolescent Idiopathic Scoliosis. Semin. Spine Surg. 2015, 27, 39–44. [Google Scholar] [CrossRef]
- Sarwahi, V.; Horn, J.J.; Kulkarni, P.M.; Wollowick, A.L.; Lo, Y.; Gambassi, M.; Amaral, T.D. Minimally Invasive Surgery in Patients With Adolescent Idiopathic Scoliosis: Is It Better than the Standard Approach? A 2-Year Follow-up Study. Clin. Spine Surg. 2016, 29, 331–340. [Google Scholar] [CrossRef]
- Urbanski, W.; Zaluski, R.; Kokaveshi, A.; Aldobasic, S.; Miekisiak, G.; Morasiewicz, P. Minimal invasive posterior correction of Lenke 5C idiopathic scoliosis: Comparative analysis of minimal invasive vs. open surgery. Arch. Orthop. Trauma. Surg. 2019, 139, 1203–1208. [Google Scholar] [CrossRef]
- Yang, J.H.; Kim, H.J.; Chang, D.-G.; Suh, S.W. Comparative Analysis of Radiologic and Clinical Outcomes Between Conventional Open and Minimally Invasive Scoliosis Surgery for Adolescent Idiopathic Scoliosis. World Neurosurg. 2021, 151, e234–e240. [Google Scholar] [CrossRef]
- Si, G.; Li, T.; Wang, Y.; Liu, X.; Li, C.; Yu, M. Minimally Invasive Surgery versus Standard Posterior Approach for Lenke Type 1–4 Adolescent Idiopathic Scoliosis: A Multicenter, Retrospective Study. Eur. Spine J. 2021, 30, 706–713. [Google Scholar] [CrossRef]
- Sarwahi, V.; Galina, J.M.; Hasan, S.; Atlas, A.; Ansorge, A.; De Bodman, C.; Lo, Y.; Amaral, T.D.; Dayer, R. Minimally Invasive Versus Standard Surgery in Idiopathic Scoliosis Patients: A Comparative Study. Spine 2021, 46, 1326–1335. [Google Scholar] [CrossRef]
- Alhammoud, A.; Alborno, Y.; Baco, A.M.; Othman, Y.A.; Ogura, Y.; Steinhaus, M.; Sheha, E.D.; Qureshi, S.A. Minimally Invasive Scoliosis Surgery Is a Feasible Option for Management of Idiopathic Scoliosis and Has Equivalent Outcomes to Open Surgery: A Meta-Analysis. Glob. Spine J. 2022, 12, 483–492. [Google Scholar] [CrossRef]
- Yang, H.; Jia, X.; Hai, Y. Posterior Minimally Invasive Scoliosis Surgery versus the Standard Posterior Approach for the Management of Adolescent Idiopathic Scoliosis: An Updated Meta-Analysis. J. Orthop. Surg. Res. 2022, 17, 58. [Google Scholar] [CrossRef]
- Wiltse, L.L.; Bateman, J.G.; Hutchinson, R.H.; Nelson, W.E. The Paraspinal Sacrospinalis-Splitting Approach to the Lumbar Spine. JBJS 1968, 50, 919. [Google Scholar] [CrossRef]
- Wiltse, L.L.; Spencer, C.W. New Uses and Refinements of the Paraspinal Approach to the Lumbar Spine. Spine 1988, 13, 696–706. [Google Scholar] [CrossRef]
- Yang, J.H. Fusion Rates Based on Type of Bone Graft Substitute Using Minimally Invasive Scoliosis Surgery for Adolescent Idiopathic Scoliosis. BMC Musculoskelet. Disord. 2023, 24, 30. [Google Scholar] [CrossRef]
- Li, G.; Lv, G.; Passias, P.; Kozanek, M.; Metkar, U.S.; Liu, Z.; Wood, K.B.; Rehak, L.; Deng, Y. Complications Associated with Thoracic Pedicle Screws in Spinal Deformity. Eur. Spine J. 2010, 19, 1576–1584. [Google Scholar] [CrossRef]
- Upendra, B.N.; Meena, D.; Chowdhury, B.; Ahmad, A.; Jayaswal, A. Outcome-Based Classification for Assessment of Thoracic Pedicular Screw Placement. Spine 2008, 33, 384–390. [Google Scholar] [CrossRef]
- Welch, N.; Mota, F.; Birch, C.; Hutchinson, L.; Hedequist, D. Robotics Coupled With Navigation for Pediatric Spine Surgery: Initial Intraoperative Experience With 162 Cases. J. Pediatr. Orthop. 2023, 43, e337–e342. [Google Scholar] [CrossRef]
- Berlin, C.; Quante, M.; Thomsen, B.; Koeszegvary, M.; Platz, U.; Ivanits, D.; Halm, H. Intraoperative Radiation Exposure to Patients in Idiopathic Scoliosis Surgery with Freehand Insertion Technique of Pedicle Screws and Comparison to Navigation Techniques. Eur. Spine J. 2020, 29, 2036–2045. [Google Scholar] [CrossRef]
- Dabaghi Richerand, A.; Christodoulou, E.; Li, Y.; Caird, M.S.; Jong, N.; Farley, F.A. Comparison of Effective Dose of Radiation During Pedicle Screw Placement Using Intraoperative Computed Tomography Navigation Versus Fluoroscopy in Children With Spinal Deformities. J. Pediatr. Orthop. 2016, 36, 530–533. [Google Scholar] [CrossRef]
- Su, A.W.; McIntosh, A.L.; Schueler, B.A.; Milbrandt, T.A.; Winkler, J.A.; Stans, A.A.; Larson, A.N. How Does Patient Radiation Exposure Compare With Low-Dose O-Arm Versus Fluoroscopy for Pedicle Screw Placement in Idiopathic Scoliosis? J. Pediatr. Orthop. 2017, 37, 171–177. [Google Scholar] [CrossRef]
- Syundyukov, A.R.; Nikolaev, N.S.; Vissarionov, S.V.; Kornyakov, P.N.; Bhandarkar, K.S.; Emelianov, V.U. Less Correction with Minimally Invasive Surgery for Adolescent Idiopathic Scoliosis Compared to Open Surgical Correction. J. Child. Orthop. 2023, 17, 141–147. [Google Scholar] [CrossRef]
- Sarwahi, V.; Visahan, K.; Hasan, S.; Patil, A.; Grunfeld, M.; Atlas, A.; Galina, J.; Ansorge, A.; Lo, Y.; Amaral, T.D.; et al. SLIM: Single Long-Incision Minimally Invasive Surgery. Spine, 2023; epub ahead of print. [Google Scholar] [CrossRef]
- Newton, P.O.; Yaszay, B.; Upasani, V.V.; Pawelek, J.B.; Bastrom, T.P.; Lenke, L.G.; Lowe, T.; Crawford, A.; Betz, R.; Lonner, B.; et al. Preservation of Thoracic Kyphosis Is Critical to Maintain Lumbar Lordosis in the Surgical Treatment of Adolescent Idiopathic Scoliosis. Spine 2010, 35, 1365–1370. [Google Scholar] [CrossRef]
- Bridwell, K.H. Surgical Treatment of Idiopathic Adolescent Scoliosis. Spine 1999, 24, 2607–2616. [Google Scholar] [CrossRef]
- Majdouline, Y.; Aubin, C.-E.; Robitaille, M.; Sarwark, J.F.; Labelle, H. Scoliosis Correction Objectives in Adolescent Idiopathic Scoliosis. J. Pediatr. Orthop. 2007, 27, 775–781. [Google Scholar] [CrossRef]
- Sudo, H.; Abe, Y.; Kokabu, T.; Ito, M.; Abumi, K.; Ito, Y.M.; Iwasaki, N. Correlation Analysis between Change in Thoracic Kyphosis and Multilevel Facetectomy and Screw Density in Main Thoracic Adolescent Idiopathic Scoliosis Surgery. Spine J. 2016, 16, 1049–1054. [Google Scholar] [CrossRef]
- Bridwell, K.H.; O’Brien, M.F.; Lenke, L.G.; Baldus, C.; Blanke, K. Posterior Spinal Fusion Supplemented with Only Allograft Bone in Paralytic Scoliosis. Does It Work? Spine 1994, 19, 2658–2666. [Google Scholar] [CrossRef]
- Yang, J.H.; Kim, H.J.; Chang, D.-G.; Nam, Y.; Suh, S.W. Learning Curve for Minimally Invasive Scoliosis Surgery in Adolescent Idiopathic Scoliosis. World Neurosurg. 2023, 175, e201–e207. [Google Scholar] [CrossRef]
- Hariharan, A.R.; Shah, S.A.; Petfield, J.; Baldwin, M.; Yaszay, B.; Newton, P.O.; Lenke, L.G.; Lonner, B.S.; Miyanji, F.; Sponseller, P.D.; et al. Complications Following Surgical Treatment of Adolescent Idiopathic Scoliosis: A 10-Year Prospective Follow-up Study. Spine Deform. 2022, 10, 1097–1105. [Google Scholar] [CrossRef]


| Authors | Year | Study Design | No. of Cases | MISS (%) | OM (%) | p-Value |
|---|---|---|---|---|---|---|
| Miyanji et al. [22] | 2013 | Pros. comp. | 32 | 63 | 68 | n/a a |
| Miyanji et al. [23] | 2015 | Retro. comp. | 46 | 58 | 68 | 0.001 |
| Sarwahi et al. [24] | 2016 | Retro. comp. | 22 | 79 | 85 | 0.503 |
| Urbanski et al. [25] | 2019 | Retro. comp. | 8 | 68 b | 78 | 0.072 |
| Yang et al. [26] | 2021 | Retro. comp. | 49 | 65 | 70 | 0.017 |
| Si et al. [27] | 2021 | Retro. comp. | 112 | 65 | 64 | 0.862 |
| Sarwahi et al. [28] | 2021 | Retro. comp. | 485 | 69 | 68 | 0.46 |
| Syundyukov et al. [40] | 2023 | Retro. comp. | 82 | 78 | 88 | <0.001 |
| Sarwahi et al. [41] | 2023 | Retro. comp. | 532 | 69 or 62 c | 68 | 0.49 |
| Yang et al. [30] | 2022 | Meta-analysis | 713 | n/a d | 0.518 | |
| Authors | Year | Study Design | No. of Cases | MISS (mL) | OM (mL) | p-Value |
|---|---|---|---|---|---|---|
| Miyanji et al. [22] | 2013 | Pros. comp. | 32 | 277 | 388 | n/a a |
| Miyanji et al. [23] | 2015 | Retro. comp. | 46 | 261.5 | 471.1 | 0.000 |
| Sarwahi et al. [24] | 2016 | Retro. comp. | 22 | 600 | 800 | 0.051 |
| Urbanski et al. [25] | 2019 | Retro. comp. | 8 | 138.75 b | 450 | 0.016 |
| Yang et al. [26] | 2021 | Retro. comp. | 49 | 1279 | 2503 | <0.001 |
| Si et al. [27] | 2021 | Retro. comp. | 112 | 502 | 808 | <0.001 |
| Sarwahi et al. [28] | 2021 | Retro. comp. | 485 | 300 | 500 | <0.001 |
| Alhammoud et al. [29] | 2022 | Meta-analysis | 107 | 271.1 | 527 | 0.019 |
| Syundyukov et al. [40] | 2023 | Retro. comp. | 82 | 208.7 | 564.3 | <0.001 |
| Sarwahi et al. [41] | 2023 | Retro. comp. | 532 | 302 vs. 325 c | 500 | 0.005 |
| Yang et al. [30] | 2023 | Meta-analysis | 767 | n/a d | <0.001 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bazin, L.; Ansorge, A.; Vendeuvre, T.; Cochard, B.; Tabard-Fougère, A.; Vazquez, O.; De Marco, G.; Sarwahi, V.; Dayer, R. Minimally Invasive Surgery for Posterior Spinal Instrumentation and Fusion in Adolescent Idiopathic Scoliosis: Current Status and Future Application. Children 2023, 10, 1882. https://0-doi-org.brum.beds.ac.uk/10.3390/children10121882
Bazin L, Ansorge A, Vendeuvre T, Cochard B, Tabard-Fougère A, Vazquez O, De Marco G, Sarwahi V, Dayer R. Minimally Invasive Surgery for Posterior Spinal Instrumentation and Fusion in Adolescent Idiopathic Scoliosis: Current Status and Future Application. Children. 2023; 10(12):1882. https://0-doi-org.brum.beds.ac.uk/10.3390/children10121882
Chicago/Turabian StyleBazin, Ludmilla, Alexandre Ansorge, Tanguy Vendeuvre, Blaise Cochard, Anne Tabard-Fougère, Oscar Vazquez, Giacomo De Marco, Vishal Sarwahi, and Romain Dayer. 2023. "Minimally Invasive Surgery for Posterior Spinal Instrumentation and Fusion in Adolescent Idiopathic Scoliosis: Current Status and Future Application" Children 10, no. 12: 1882. https://0-doi-org.brum.beds.ac.uk/10.3390/children10121882