Cocculus hirsutus (L.) W.Theob. (Menispermaceae): A Review on Traditional Uses, Phytochemistry and Pharmacological Activities
Abstract
:1. Introduction
2. Traditional Uses
3. Chemical Constituents
4. Pharmacological Activities
4.1. Anti-Microbial Activity
4.2. Anti-Malarial and Insecticidal Activity
4.3. Anti-Cancer Activity
4.4. Immunomodulatory Activity
4.5. Anti-Diabetic Acitivty
4.6. Anti-Oxidant Activity
4.7. Hepatoprotective Activity
4.8. Diruetic and Nephroprotective Activity
4.9. Other Activities
5. Toxicological Studies
6. Patents
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- De Wet, H.; Struwig, M.; Van Wyk, B.E. Taxonomic notes on the genus Cocculus (Menispermaceae) in southern Africa. S. Afr. J. Bot. 2015, 96, 99–104. [Google Scholar] [CrossRef] [Green Version]
- World Flora Online (WFO). Cocculus hirsutus (L.) W.Theob; Published on the Internet. Available online: http://www.worldfloraonline.org/taxon/wfo-0000613546 (accessed on 3 November 2020).
- Panda, B.R.; Mohanta, S.R.; Mishra, U.S.; Kar, S.; Panda, B.K.; Chakraborty, P. Antibacterial activity of the leaves of Cocculus hirsutus. Indian Drugs 2007, 44, 108–116. [Google Scholar]
- Shrestha, K.K.; Bhattarai, S.; Bhandari, P. Gymnosperms and Angiosperms: Cycadaceae—Betulaceae. In Handbook of Flowering Plants of Nepal; Scientific Publishers: Jodhpur, India, 2018; Volume 1. [Google Scholar]
- Ahmad, V.U.; Iqbal, S. Jamtinine, an alkaloid from Cocculus hirsutus. Phytochemistry 1993, 33, 735–736. [Google Scholar] [CrossRef]
- Fahmy, A.G.; El-Bakry, A.A. Phytogeographical significance of Wadi Wateer (Sinai), Egypt, with special reference to Cocculus hirsutus (L.) W.Theob. Plant Syst. Evol. 2011, 297, 299–303. [Google Scholar] [CrossRef]
- Global Biodiversity Information Facility Secretariat (GBIF). Cocculus hirsutus (L.) Diels. Available online: https://www.gbif.org/species/7930800 (accessed on 3 November 2020).
- India Biodiversity Portal. Cocculus hirsutus (L.) Diels. Available online: https://indiabiodiversity.org/species/show/229234 (accessed on 3 February 2020).
- Chadha, Y. The Wealth of India; CSIR: New delhi, India, 1950. [Google Scholar]
- Ganapaty, S.; Vijay, K. Hypoglycemic activity of aerial parts of Cocculus hirsutus in alloxan indued diabetes. Indian J. Nat. Prod. 2006, 22, 17–20. [Google Scholar]
- Nayak, S.; Singhai, A. Antiinflammatory and analgesic activity of roots of Cocculus hirsutus. Indian J. Nat. Prod. 1993, 9, 12–14. [Google Scholar]
- Clinical Trials Registry-India. Clinical Trials Registry-India. Available online: http://ctri.nic.in/Clinicaltrials/login.php (accessed on 3 November 2020).
- Rama Rao, N.; Henry, A.N. The Ethnobotany of Eastern Ghats in Andhra Pradesh, India; Botanical Survey of India: Kolkata, India, 1996.
- Jain, S.K. Glimpses of Indian Ethnobotany; Jain, S.K., Ed.; Oxford and IBH Publishing Co.: New Delhi, India, 1981; pp. 13–36. [Google Scholar]
- Bedi, S.J. Ethnobotany of the Ratan Mahal Hills, Gujarat, India. Econ. Bot. 1978, 32, 278–284. [Google Scholar] [CrossRef]
- Saxena, H.O.; Brahman, M.; Datta, P.K. Ethnobotanical Studies in Orissa. In Glimpses of Indian Ethnobotany; Jain, S.K., Ed.; Oxford and IBH Publishing Co.: New Delhi, India, 1981; pp. 232–244. [Google Scholar]
- Chaudhuri, R.H.N.; Pal, D.C.; Tarafdar, C.R. Less known uses of some plants from the tribal areas of Orissa. Bull. Bot. Surv. India 1975, 17, 132–136. [Google Scholar]
- Shah, G.L.; Yadav, S.S.; Badri, N. Medicinal plants from Dahanu forest division in Maharashtra state. J. Econ. Taxon. Bot. 1983, 4, 141–151. [Google Scholar]
- Joshi, P. An ethnobotanical study of Bhils-A preliminary survey. J. Econ. Taxon. Bot. 1982, 3, 257–266. [Google Scholar]
- Hemadri, K.; Rao, S.S. Leucorrhoea and menorrhagia: Tribal medicines. Anc. Sci. Life 1983, 3, 40–41. [Google Scholar] [PubMed]
- Goel, A.K.; Sahoo, A.K.; Mudgal, V. A contribution to ethnobotany of Santal Pargana, Bihar. Bot. Surv. India 1984, 31, 22–26. [Google Scholar]
- Ahmad, V.U.; Iqbal, S. Haiderine, a new isoquinoline alkaloid from Cocculus hirsutus. Nat. Prod. Lett. 1993, 2, 105–109. [Google Scholar] [CrossRef]
- Audichya, K.C.; Billore, K.V.; Joseph, T.H.; Chaturvedi, D.D. Role of indigenous folk remedies for certain acute illnesses in primary health care. Nagarjun 1983, 26, 199–201. [Google Scholar]
- Gairola, S.; Sharma, J.; Gaur, R.D.; Siddiqi, T.O.; Painuli, R.M. Plants used for treatment of dysentery and diarrhoea by the Bhoxa community of district Dehradun, Uttarakhand, India. J. Ethnopharmacol. 2013, 150, 989–1006. [Google Scholar] [CrossRef]
- Singh, U.; Wadhwani, A.M.; Johri, B.M. Dictionary of Economic Plants of India; Indian Council of Agricultural Research (ICAR): New delhi, India, 1965. [Google Scholar]
- Agrawal, V.S. Economic Plants of India; Bishen Singh Mahendra Pal Singh and Kailash Prakashan: Calcutta, India, 1986. [Google Scholar]
- Sudhakar, S.; Rolla, R.S. Medicinal plants of Upper East Godavari District (Andhra Pradesh) and need for establishment of medicinal farm. J. Econ. Taxon. Bot. 1985, 7, 399–406. [Google Scholar]
- Shah, G.L.; Manon, A.R.; Gopal, V. An account of Ethnobotany of Saurasthra in Gujarat state (India). J. Econ. Taxon. Bot. 1981, 2, 173–182. [Google Scholar]
- Shah, G.L. Some economically important plants of Salsette island near Bombay. J. Econ. Taxon. Bot. 1984, 5, 753–765. [Google Scholar]
- Malhotra, S.K.; Moorthy, S. Some useful and medicinal plants of Chandanpur district (maharasthra State). J. Econ. Taxon. Bot. 1973, 15, 13–21. [Google Scholar]
- Singh, V.; Pandey, R.P. Medicinal plantlore of the tribals of eastern Rajasthan. J. Econ. Taxon. Bot. 1980, 1, 137–147. [Google Scholar]
- Rajan, S.; Sethuraman, M.; Mukherjee, P.K. Ethnobiology of the Nilgiri Hills, India. Phyther. Res. 2002, 16, 98–116. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, V.J.; Ray, A.B.; Dasgupta, B. Alkaloids of Cocculus hirsutus DC. Indian J. Chem. Sect. B Org. Chem. Incl. Med. Chem. 1976, 14, 62–63. [Google Scholar]
- Jagannadha Rao, K.V.; Row, L.R.M. Chemical examination of Cocculus hirsutus DC. J. Sci. Ind. Res. 1961, 20b, 125–126. [Google Scholar]
- Naik, R.M.; Merchant, J.R. Chemical investigation of Cocculus hirsutus. Curr. Sci. 1956, 25, 324–325. [Google Scholar]
- Merchant, J.R.; Naik, R.M.; Hirwe, S.N. Chemical investigation of Cocculus hirsutus. J. Indian Chem. Soc. 1962, 39, 411–416. [Google Scholar]
- Patil, V.; Angadi, S.; Devdhe, S.; Wakte, P. Recent Progress in Simultaneous Estimation of Rutin, Quercetin and Liquiritin in Cocculus Hirsutus by HPTLC. Res. J. Pharmacogn. 2015, 2, 49–55. [Google Scholar]
- Ahmad, V.U.; Mohammad, F.V.; Rasheed, T. Hirsudiol a triterpenoid from Cocculus hirsutus. Phytochemistry 1987, 26, 793–794. [Google Scholar] [CrossRef]
- Iyer, S.V.; Shankul, K.; Parikh, P.M. Isolation of phytoconstituents from the aerial parts of Cocculus hirsutus Linn. J.Pharm.Res. 2011, 4, 1946–1947. [Google Scholar]
- Meena, M.K.; Singh, N.; Patni, V. Determination of bioactive components of the leaves of Cocculus hirsutus (L.) Diels using GC-MS analysis. Int. J. Pharm. Pharm. Sci. 2014, 6, 327–329. [Google Scholar]
- Thavamani, B.S.; Mathew, M.; Dhanabal, S.P. Gas Chromatography—Mass Spectroscopy (GC-MS) analysis of various extracts of Cocculus hirsutus. Biosci. Biotechnol. Res. Asia 2013, 10, 925–928. [Google Scholar] [CrossRef]
- Ahmad, V.U.; Atta-ur-Rahman; Rasheed, T.; Habib-ur-Rehman. Jamtine-N-oxide—A new isoquinoline alkaloid from Cocculus hirsutus. Heterocycles 1987, 26, 1251–1255. [Google Scholar]
- Rasheed, T.; Khan, M.N.I.; Zhadi, S.S.A.; Durrani, S. Hirsutine: A new alkaloid from Cocculus hirsutus. J. Nat. Prod. 1991, 54, 582–585. [Google Scholar] [CrossRef]
- Ahmad, V.U.; Iqbal, S. Cohirsitinine, a new isoquinoline alkaloid from Cocculus hirsutus. J. Nat. Prod. 1992, 55, 237–240. [Google Scholar] [CrossRef]
- Ahmad, V.U.; Atta-ur-Rahman; Rasheed, T.; Habib-ur-Rehman; Khan, A.Q. Cohirsine—A novel isoquinolone alkaloid from Cocculus hirsutus. Tetrahedron 1987, 43, 5865–5872. [Google Scholar] [CrossRef]
- Ahmad, V.U.; Rasheed, T.; Iqbal, S. Cohirsinine, an alkaloid from Cocculus hirsutus. Phytochemistry 1991, 30, 1350–1351. [Google Scholar] [CrossRef]
- Yadav, S.B.; Tripathi, V. A minor phenolic alkaloid from Cocculus hirsutus Diels. Indian J. Chem.-Sect. B Org. Med. Chem. 2005, 44, 212–214. [Google Scholar] [CrossRef]
- El-Shabrawy, A.O.; Schiff, P.L., Jr.; Slatkin, D.J.; Das Gupta, B.; Ray, A.B.; Tripathi, V.J. Cocsuline-N-2-Oxide, a New Alkaloid from Cocculus hirsutus DC. Heterocycles 1984, 22, 993. [Google Scholar]
- Jethva, K.; Bhatt, D.; Zaveri, M. Antimycobacterial screening of selected medicinal plants against Mycobacterium tuberculosis H37Rv using agar dilution method and the microplate resazurin assay. Int. J. Mycobacteriology 2020, 9, 150–155. [Google Scholar]
- Gupta, V.K.; Kaushik, A.; Chauhan, D.S.; Ahirwar, R.K.; Sharma, S.; Bisht, D. Anti-mycobacterial activity of some medicinal plants used traditionally by tribes from Madhya Pradesh, India for treating tuberculosis related symptoms. J. Ethnopharmacol. 2018, 227, 113–120. [Google Scholar] [CrossRef]
- Devi, M.D.; Kaviyarasu, S.; Arunsagararaj, I.; Ranjithkumar, J. Optimization of Conditions for Flavonoid Extraction from the Leaves of Cocculus hirsutus (L.) And its Antibacterial Activity. Int. J. Microbiol. Res. 2019, 10, 42–47. [Google Scholar]
- Nayak, S.; Singhai, A.K. Antimicrobial activity of the roots of Cocculus hirsutus. Anc. Sci. Life 2003, 22, 101–105. [Google Scholar] [PubMed]
- Devi, K.B.; Pavankumar, P.; Bhadraiah, B. Antifungal activity of plant extracts against post-harvest fungal pathogen. Int. J. Curr. Microbiol. Appl. Sci. 2017, 6, 669–679. [Google Scholar] [CrossRef]
- Brahmam, P.; Sunita, K. Phytochemical investigation and in vitro antimalarial activity of Acalypha indica (L.) and Cocculus hirsutus (L.) from Prakasam district, Andhra Pradesh, India. Biomed. Pharmacol. J. 2018, 11, 2123–2134. [Google Scholar] [CrossRef]
- Elango, G.; Rahuman, A.A.; Kamaraj, C.; Bagavan, A.; Zahir, A.A. Efficacy of medicinal plant extracts against malarial vector, Anopheles subpictus Grassi. Parasitol. Res. 2011, 108, 1437–1445. [Google Scholar] [CrossRef] [PubMed]
- Elango, G.; Rahuman, A.A.; Bagavan, A.; Kamaraj, C.; Zahir, A.A.; Venkatesan, C. Laboratory study on larvicidal activity of indigenous plant extracts against Anopheles subpictus and Culex tritaeniorhynchus. Parasitol. Res. 2009, 104, 1381–1388. [Google Scholar] [CrossRef] [PubMed]
- De Wet, H.; Fouche, G.; Van Heerden, F.R. In vitro cytotoxicity of crude alkaloidal extracts of South African Menispermaceae against three cancer cell lines. Afr. J. Biotechnol. 2009, 8, 3332–3335. [Google Scholar]
- Thavamani, B.S.; Mathew, M.; Dhanabal, S.P. In vitro cytotoxic activity of menispermaceae plants against HeLa cell line. Anc. Sci. Life 2013, 33, 81. [Google Scholar]
- Thavamani, B.S.; Mathew, M.; Palaniswamy, D.S. Anticancer activity of Cocculus hirsutus against Dalton’s lymphoma ascites (DLA) cells in mice. Pharm. Biol. 2014, 52, 867–872. [Google Scholar] [CrossRef]
- Mallik, A.; Nayak, S. Study the Immunomodulatory Effects of Combined Extracts of Sesbania grandiflora Flowers and Cocculus hirsutus Leaves on the Circulating Antibody Response. Am. J. Phytomed. Clin. Ther. 2015, 3, 199–208. [Google Scholar]
- Rastogi, B.; Tiwari, U.; Dubey, A.; Bawara, B.; Chauhan, N.S.; Saraf, D.K. Immunostimulant activity of Cocculus hirsutus on immunosuppressed rat. Pharmacologyonline 2008, 3, 38–57. [Google Scholar]
- Badole, S.; Patel, N.; Bodhankar, S.; Jain, B.; Bhardwaj, S. Antihyperglycemic activity of aqueous extract of leaves of Cocculus hirsutus (L.) Diels in alloxan-induced diabetic mice. Indian J. Pharmacol. 2006, 38, 49–53. [Google Scholar]
- Sangameswaran, B.; Jayakar, B. Anti-diabetic and spermatogenic activity of Cocculus hirsutus (L) diels. Afr. J. Biotechnol. 2007, 6, 1212–1216. [Google Scholar]
- Srikanta, B.M.; Dharmesh, S.M. Anti-Helicobacter pylori, proton pump inhibitory and antioxidant properties of selected dietary/medicinal plants. Int. J. Phytomed. 2012, 4, 573–581. [Google Scholar]
- Rakkimuthu, R.; Kavithakrishna, R.; Suganyadevi, P.; Aravinthan, K.M. Quantitative phytochemical analysis and their antioxidant activity of Cocculus hirsutus (l.) Diels fruit. Int. J. Phytomedicine 2012, 4, 447–455. [Google Scholar]
- Thakare, S.P.; Deore, H.V.; Patil, S.D.; Yende, S.R.; Upadhyay, U.M. Evaluation of hepatoprotective effect of Cocculus hirsutus (L) diels on ethanol induced hepatic damage in albino wistar rats. Pharmacologyonline 2010, 3, 810–816. [Google Scholar]
- Ganapaty, S.; Dash, G.K.; Subburaju, T.; Suresh, P. Diuretic, laxative and toxicity studies of Cocculus hirsutus aerial parts. Fitoterapia 2002, 73, 28–31. [Google Scholar] [CrossRef]
- Badole, S.L.; Bodhankar, S.L.; Patel, N.M.; Bhardwaj, S. Acute and chronic diuretic effect of ethanolic extract of leaves of Cocculus hirsutus (L.) Diles in normal rats. J. Pharm. Pharmacol. 2009, 61, 387–393. [Google Scholar] [CrossRef]
- Gadapuram, T.K.; Murthy, J.S.N.; Rajannagari, R.R.; Kandati, V.; Choda, P.K.; Shukla, R. Nephroprotective activity of cocculus hirsutus leaf extract in 5/6 nephrectomized rat model. J. Basic Clin. Physiol. Pharmacol. 2013, 24, 299–306. [Google Scholar] [CrossRef]
- Ranjan, P.B.; Mohanta, S.R.; Sii, S.; Das, G.K. Preliminary Phytochemical Analysis and Wound Healing Activity from the Leaves of Cocculus hirsutus Diels. Int. J. Pharm. Sci. Nanotechnol. 2009, 2, 675–678. [Google Scholar] [CrossRef]
- Elango, G.; Rahuman, A.A. Evaluation of medicinal plant extracts against ticks and fluke. Parasitol. Res. 2011, 108, 513–519. [Google Scholar] [CrossRef]
- Nayyar, K.; Arora, U.; Palla, S.; Saravan, A.; Prasad, M.; Madan, S.; Sood, R.; Khanna, N.; Potdar, A.; Shukla, R.; et al. Use of Cocculus hirsutus Extract for Treating Dengue. Indian Patent Application IN 201,821,046,412, 2020. (Filing date 9 December 2019). [Google Scholar]
- Nayyar, K.; Arora, U.; Palla, S.; Prasad, M.; Saravan, A.; Madan, S.; Sood, R.; Khanna, N.; Potdar, A.; Shukla, R.; et al. Use of Cocculus hirsutus Extract for Treating Dengue. PCT International Application WO 2,020,115,723, 2020. [Google Scholar]
- Dhirajal, J.K.; Narendrakumar, Z.M.; Shrikant, D.S. Herbal Formula Containing Cocculus Hirsutus Extract for the Treatment and Management of Tuberculosis. Indian Patent Application IN 201,821,050,047, 2019. [Google Scholar]
- Mina, M. Herbal Omposition for Therapeutic Managemnt of Respiratory Tract Diseases. Indian Patent Application IN 201,611,023,226, 2018. [Google Scholar]


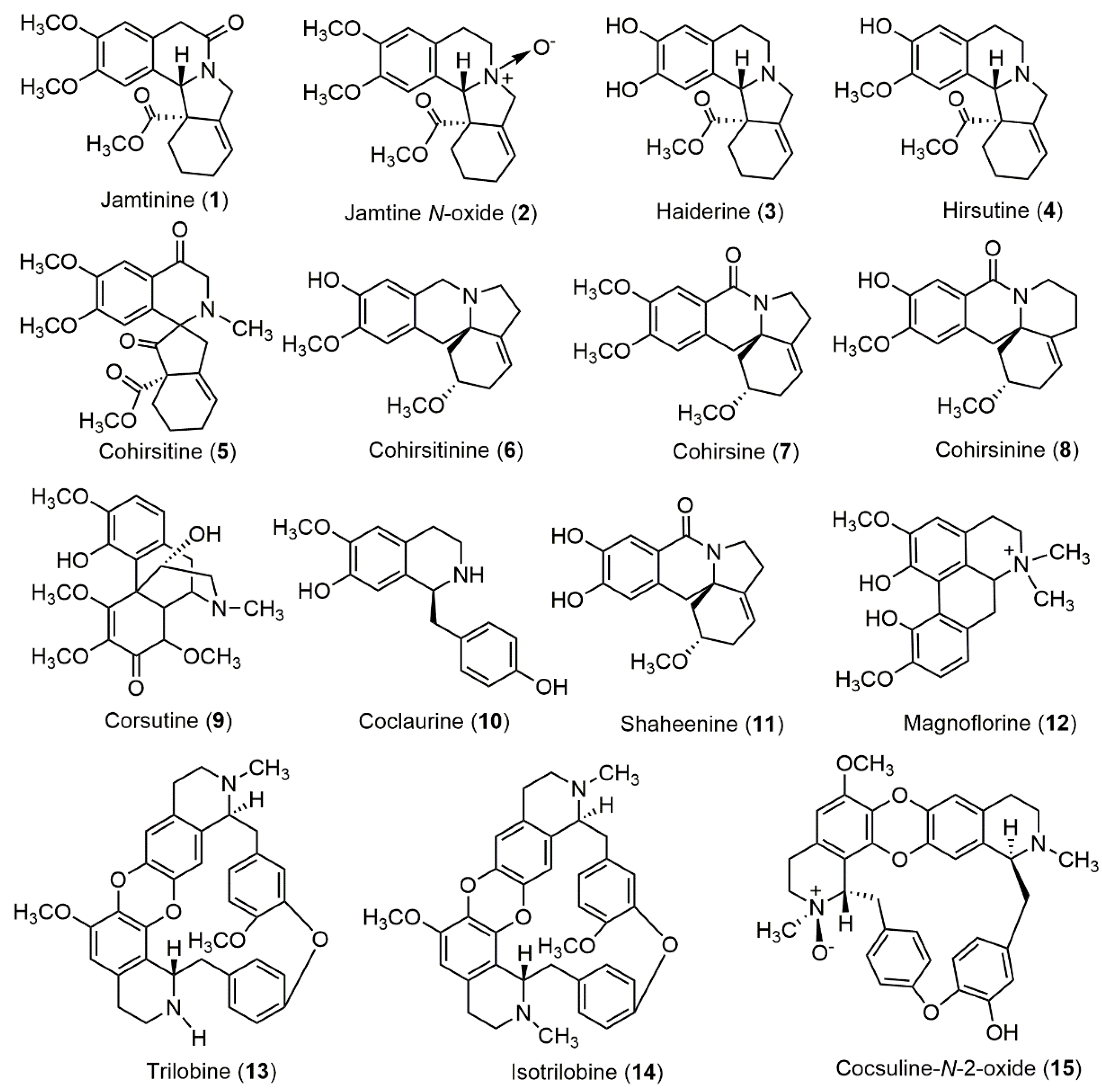
| S.N. | Compound Name | Plant Part/Extract | Reference |
|---|---|---|---|
| 1 | Jamtinine | Whole plant/ethanol extract | [5,22] |
| 2 | Jamtine N-oxide | Stems and roots | [22,42] |
| 3 | Haiderine | Whole plant/ethanol extract | [22] |
| 4 | Hirsutine | Whole plant/ethanol extract | [22,43] |
| 5 | Cohirsitine | Whole plant/ethanol extract | [22] |
| 6 | Cohirsitinine | Whole plant/ethanol extract | [22,44] |
| 7 | Cohirsine | Whole plant/ethanol extract | [22,45] |
| 8 | Cohirsinine | Whole plant/ethanol extract | [22,46] |
| 9 | Corsutine | Stems and roots/ethanol extract | [47] |
| 10 | Coclaurine | Stems and roots, whole plant/ethanol extract | [22,33,34] |
| 11 | Shaheenine | Stems and roots | [22,43] |
| 12 | Magnoflorine | Stems and roots | [22,33] |
| 13 | Trilobine | Stems and roots | [22,33,34] |
| 14 | Isotrilobine | Stems and roots | [22,33] |
| 15 | Cocsuline-N-2-oxide | Whole plant/ ethanol extract | [48] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Logesh, R.; Das, N.; Adhikari-Devkota, A.; Devkota, H.P. Cocculus hirsutus (L.) W.Theob. (Menispermaceae): A Review on Traditional Uses, Phytochemistry and Pharmacological Activities. Medicines 2020, 7, 69. https://0-doi-org.brum.beds.ac.uk/10.3390/medicines7110069
Logesh R, Das N, Adhikari-Devkota A, Devkota HP. Cocculus hirsutus (L.) W.Theob. (Menispermaceae): A Review on Traditional Uses, Phytochemistry and Pharmacological Activities. Medicines. 2020; 7(11):69. https://0-doi-org.brum.beds.ac.uk/10.3390/medicines7110069
Chicago/Turabian StyleLogesh, Rajan, Niranjan Das, Anjana Adhikari-Devkota, and Hari Prasad Devkota. 2020. "Cocculus hirsutus (L.) W.Theob. (Menispermaceae): A Review on Traditional Uses, Phytochemistry and Pharmacological Activities" Medicines 7, no. 11: 69. https://0-doi-org.brum.beds.ac.uk/10.3390/medicines7110069






