Ureteral Stent and Percutaneous Nephrostomy in Managing Malignant Ureteric Obstruction of Gastrointestinal Origin: A 10 Years’ Experience
Abstract
:1. Introduction
- Outcomes data regarding renal function, hospitalization, complications, stent failure, and overall survival (OS) following insertion of US and NT
- Differences between patients treated with NT and those treated with US
- The rate and the characteristics of patients who received further oncological treatment after stent/nephrostomy insertion
- Potential risk factors associated with worse outcomes.
2. Results
3. Discussion
4. Materials and Methods
4.1. Surgical Technique
4.2. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Turo, R.; Horsu, S.; Broome, J.; Das, S.; Gulur, D.M.; Pettersson, B.; Doyle, G.; Awsare, N. Complications of Percutaneous Nephrostomy in a District General Hospital. Turk. J. Urol. 2018, 44, 478–483. [Google Scholar] [CrossRef] [PubMed]
- Erlich, T.; Shoshany, O.; Golan, S.; Livne, P.; Lifshitz, D.; Rosenzweig, B.; Eisner, A.; Kleinman, N.; Mor, Y.; Ramon, J.; et al. Mp30-09 Ureteric Stent Versus Percutaneous Nephrostomy for Acute Ureteral Obstruction-Clinical Outcome and Quality of Life: A Bi-Center Prospective Study. J. Urol. 2015, 193, 350. [Google Scholar] [CrossRef]
- Ferrara, M.; Kann, B.R. Urological Injuries during Colorectal Surgery. Clin. Colon Rectal Surg. 2019, 32, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Jeong, I.G.; Han, K.S.; Joung, J.Y.; Seo, H.K.; Chung, J. The Outcome with Ureteric Stents for Managing Non-Urological Malignant Ureteric Obstruction. BJU Int. 2007, 100, 1288–1291. [Google Scholar] [CrossRef]
- Wong, L.M.; Cleeve, L.K.; Milner, A.D.; Pitman, A.G. Malignant Ureteral Obstruction: Outcomes After Intervention. Have Things Changed? J. Urol. 2007, 178, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Folkard, S.S.; Banerjee, S.; Menzies-Wilson, R.; Reason, J.; Psallidas, E.; Clissold, E.; Al-Mushatat, A.; Chaudhri, S.; Green, J.S.A. Percutaneous Nephrostomy in Obstructing Pelvic Malignancy Does Not Facilitate Further Oncological Treatment. Int. Urol. Nephrol. 2020, 52, 1625–1628. [Google Scholar] [CrossRef]
- Borda, A.P.; Charney-Sonnek, F.; Fonteyne, V.; Papaioannou, E.G. European Association of Urology Guidelines on Pain Management & Palliative Care. 2014. Available online: https://uroweb.org/wp-content/uploads/25-Pain-Management_LR.pdf (accessed on 23 October 2020).
- Prentice, J.; Amer, T.; Tasleem, A.; Aboumarzouk, O. Malignant Ureteric Obstruction Decompression: How Much Gain for How Much Pain? A Narrative Review. J. R. Soc. Med. 2018, 111, 125–135. [Google Scholar] [CrossRef] [Green Version]
- Haas, C.R.; Shah, O.; Hyams, E.S. Temporal Trends and Practice Patterns for Inpatient Management of Malignant Extrinsic Ureteral Obstruction in the United States. J. Endourol. 2020, 34, 828–835. [Google Scholar] [CrossRef]
- O’Connor, E.M.; Nason, G.J.; Kiely, E.A. Urological Management of Extramural Malignant Ureteric Obstruction: A Survey of Irish Urologists. Curr. Urol. 2017, 11, 21–25. [Google Scholar] [CrossRef]
- Hyams, E.S.; Shah, O. Malignant Extrinsic Ureteral Obstruction: A Survey of Urologists and Medical Oncologists Regarding Treatment Patterns and Preferences. Urology 2008, 72, 51–56. [Google Scholar] [CrossRef]
- Tanaka, T.; Yanase, M.; Takatsuka, K. Clinical Course in Patients with Percutaneous Nephrostomy for Hydronephrosis Associated with Advanced Cancer. Acta Urol. Jpn. 2004, 50, 457–462. [Google Scholar]
- Kamiyama, Y.; Matsuura, S.; Kato, M.; Abe, Y.; Takyu, S.; Yoshikawa, K.; Arai, Y. Stent Failure in the Management of Malignant Extrinsic Ureteral Obstruction: Risk Factors. Int. J. Urol. 2011, 18, 379–382. [Google Scholar] [CrossRef] [PubMed]
- Ohtaka, M.; Kawahara, T.; Takamoto, D.; Mochizuki, T.; Hattori, Y.; Teranishi, J.I.; Makiyama, K.; Miyoshi, Y.; Yumura, Y.; Yao, M.; et al. Gastrointestinal Cancer and Bilateral Hydronephrosis Resulted in a High Risk of Ureteral Stent Failure. BMC Urol. 2018, 18, 35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chung, S.Y.; Stein, R.J.; Landsittel, D.; Davies, B.J.; Cuellar, D.C.; Hrebinko, R.L.; Tarin, T.; Averch, T.D. 15-Year Experience with the Management of Extrinsic Ureteral Obstruction with Indwelling Ureteral Stents. J. Urol. 2004, 172, 592–595. Available online: http://www.jurology.com/doi/10.1097/01.ju.0000130510.28768.f5 (accessed on 23 October 2020). [CrossRef] [PubMed]
- Guachetá-Bomba, P.L.; Echeverría-García, F.; García-Perdomo, H.A. Predictors for Failure of Endoscopic Ureteric Stenting in Patients with Malignant Ureteric Obstruction: Systematic Review and Meta-Analysis. BJU Int. 2020. [Google Scholar] [CrossRef]
- Jalbani, M.H.; Deenari, R.A.; Dholia, K.R.; Oad, A.K.; Arbani, I.A. Role of Percutaneous Nephrostomy (PCN) in Malignant Ureteral Obstruction. J. Pak. Med. Assoc. 2010, 60, 280–283. [Google Scholar]
- Pavlovic, K.; Lange, D.; Chew, B.H. Stents for Malignant Ureteral Obstruction. Asian J. Urol. 2016, 3, 142–149. [Google Scholar] [CrossRef] [Green Version]
- Little, B.; Ho, K.J.; Gawley, S.; Young, M. Use of Nephrostomy Tubes in Ureteric Obstruction from Incurable Malignancy. Int. J. Clin. Pract. 2003, 57, 180–181. Available online: http://0-www-ncbi-nlm-nih-gov.brum.beds.ac.uk/pubmed/12723719 (accessed on 23 October 2020).
- Donat, S.M.; Russo, P. Ureteral Decompression in Advanced Nonurologic Malignancies. Ann. Surg. Oncol. 1996, 3, 393–399. Available online: http://0-link-springer-com.brum.beds.ac.uk/10.1007/BF02305670 (accessed on 23 November 2020). [CrossRef]
- Wilson, J.R.; Urwin, G.H.; Stower, M.J. The Role of Percutaneous Nephrostomy in Malignant Ureteric Obstruction. Ann. R. Coll. Surg. Engl. 2005, 87, 21–24. [Google Scholar] [CrossRef] [Green Version]
- WHO. Classificazione Delle Malattie, dei Traumatismi, Degli Interventi Chirurgici e Delle Procedure Diagnostiche e Terapeutiche-ICD 9-CM. Ist Poligr e Zecca dello Stato—Libr dello Stato. 2007; pp. 1–1116. Available online: http://www.salute.gov.it/imgs/C_17_pubblicazioni_2251_allegato.pdf (accessed on 24 October 2020).
- Croghan, S.M.; Zaborowski, A.; Mohan, H.M.; Mulvin, D.; McGuire, B.B.; Murphy, M.; Galvin, D.J.; Lennon, G.; Quinlan, D.; Winter, D.C. The Sentinel Stent? A Systematic Review of the Role of Prophylactic Ureteric Stenting Prior to Colorectal Resections. Int. J. Colorectal Dis. 2019, 1161–1178. [Google Scholar] [CrossRef] [PubMed]
- Oken, M.M.; Creech, R.H.; Tormey, D.C.; Horton, J.; Davis, T.E.; McFadden, E.T.; Carbone, P.P. Toxicity and Response Criteria of the Eastern Cooperative Oncology Group. Am. J. Clin. Oncol. 1982, 5, 649–655. Available online: http://0-www-ncbi-nlm-nih-gov.brum.beds.ac.uk/pubmed/7165009 (accessed on 24 November 2020). [CrossRef] [PubMed]
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.L.; Castro, A.F.; Feldman, H.I.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Greene, T.; et al. A New Equation to Estimate Glomerular Filtration Rate. Ann. Intern. Med. 2009, 150, 604–612. Available online: http://0-www-ncbi-nlm-nih-gov.brum.beds.ac.uk/pubmed/19414839 (accessed on 24 November 2020). [CrossRef] [PubMed]
| Parameter | Overall (n = 51) |
|---|---|
| Age at surgery (years) | 70 (58–76) |
| Male n | 20 (39.2%) |
| Female n | 31 (60.8%) |
| ECOG # Performance status n (%) | |
| 0–1 | 37 (72.5%) |
| 2–3 | 14 (27.5%) |
| Type of malignancy n (%) | |
| Rectal | 14 (27.5%) |
| Colon | 28 (54.9%) |
| Gastric | 5 (9.8%) |
| Pancreatic | 3 (5.9%) |
| Appendicular | 1 (1.9%) |
| Location of obstruction | |
| Upper | 3 (5.9%) |
| Middle | 10 (19.6%) |
| Lower | 38 (74.5%) |
| Side | |
| Right | 14 (27.4%) |
| Left | 16 (31.4%) |
| Bilateral | 21 (41.2%) |
| Cause of obstruction | |
| Unknown | 7 (13.7%) |
| Mass | 21 (41.2%) |
| Lymphadenopathy | 4 (7.8%) |
| Ureteral infiltration | 9 (17.7%) |
| Carcinomatosis | 10 (19.6%) |
| Hydronephrosis degree | |
| Mild | 7 (13.7%) |
| Moderate | 23 (45.1%) |
| Severe | 21 (41.2%) |
| Bladder invasion | |
| Yes | 7 (13.7%) |
| Parameter | Overall (n = 51) |
|---|---|
| Type of urinary diversion | |
| Ureteral stent | 27 (53%) |
| Nephrostomy tube | 24 (47%) |
| Hospital stay (days) | 9 (3–17) |
| Stent failure n | 8 (15.7%) |
| Time to stent failure (days) | 30 (23.5–128.5) |
| Complications n (%) | |
| No | 44 (86.3%) |
| Yes | 7 (13.7%) |
| Type of complication n (%) (n = 7) | |
| Fever | 2 (28.6%) |
| Pyelonephritis | 1 (14.2%) |
| Encrustation | 2 (28.6%) |
| Sepsis | 2 (28.6%) |
| Deaths n (%) | 43 (84.3%) |
| Overall survival (months) | 10.5 (4–17.2) |
| Parameter | US Group (n = 27) | NT Group (n = 24) | p |
|---|---|---|---|
| Age at surgery (years) | 70 (60–77) | 68 (57–73.7) | 0.44 |
| Gender | 0.8 | ||
| Female | 16 (59.2%) | 15 (62.5%) | |
| Male | 11 (40.8%) | 9 (37.5%) | |
| ECOG Performance status n (%) | 0.8 | ||
| 0–1 | 20 (74%) | 17 (70.8%) | |
| 2–3 | 7 (26%) | 7 (29.2%) | |
| Type of malignancy n (%) * | 0.8 | ||
| Upper GI tract | 5 (18.5%) | 4 (16.7%) | |
| Lower GI tract | 22 (81.5%) | 20 (83.3%) | |
| Location of obstruction | 0.4 | ||
| Upper | 1 (3.7%) | 2 (8.3%) | |
| Middle | 7 (25.9%) | 3 (12.5%) | |
| Lower | 19 (70.4%) | 19 (79.2%) | |
| Laterality | 0.5 | ||
| Monolateral | 17 (63%) | 13 (54.2%) | |
| Bilateral | 10 (37%) | 11 (45.8%) | |
| Cause of obstruction | 0.8 | ||
| Unknown | 3 (11.1%) | 4 (16.7%) | |
| Mass | 11 (40.7%) | 10 (41.7%) | |
| Lymphadenopathy | 2 (7.4%) | 2 (8.3%) | |
| Ureteral infiltration | 4 (14.9%) | 5 (20.8%) | |
| Carcinomatosis | 7 (25.9%) | 3 (12.5%) | |
| Hydronephrosis degree | 0.3 | ||
| Mild | 2 (7.4%) | 5 (20.8%) | |
| Moderate | 14 (51.9%) | 9 (37.5%) | |
| Severe | 11 (40.7%) | 10 (41.7%) | |
| Bladder invasion | 0.03 | ||
| Yes | 1 (3.7%) | 6 (25%) | |
| No | 26 (96.3%) | 18 (75%) | |
| Time between malignancy diagnosis and urinary diversion (months) | 18 (7–36) | 1 (0–12) | 0.005 |
| Previous chemotherapy | 0.7 | ||
| Yes | 17 (63%) | 14 (58.3%) | |
| No | 10 (37%) | 10 (41.7%) | |
| Previous radiotherapy | |||
| Yes | 6 (22.2%) | 9 (37.5%) | 0.2 |
| No | 21 (77.8%) | 15 (62.5%) | |
| Preoperative creatinine (mg/dL) | 1.5 (1–3.7) | 1.4 (0.9–2) | 0.6 |
| Preoperative eGFR (mL/min/1.73 m2) | 37 (13.2–71.5) | 41.5 (24.2–71.5) | 0.5 |
| Hospital stay (days) | 5 (2–13) | 12 (4.2–21.7) | 0.04 |
| Postoperative creatinine (mg/dL) | 1 (0.6–1.5) | 0.9 (0.7–1.2) | 0.8 |
| Postoperative eGFR (mL/min/1.73 m2) | 68 (40–92.5) | 72 (45.2–88.2) | 0.6 |
| Complications | |||
| Yes | 6 (22.2%) | 1 (4.2%) | 0.06 |
| No | 21 (77.8%) | 23 (95.8%) | |
| Postoperative oncological treatment | 0.1 | ||
| Yes | 16 (59.3%) | 9 (37.5%) | |
| No | 11 (40.7%) | 15 (62.5%) | |
| Overall survival (months) | 11 (6.7–19) | 8 (3–16.5) | 0.2 |
| Parameter | Without Therapy (n = 26) | With Therapy (n = 25) | p |
|---|---|---|---|
| Age at surgery (years) | 65.5 (58–74.5) | 71 (57–76) | 0.8 |
| Gender | 0.06 | ||
| Female | 19 (73.1%) | 12 (48%) | |
| Male | 7 (26.9%) | 13 (52%) | |
| ECOG Performance status n (%) | 0.015 | ||
| 0–1 | 15 (57.7%) | 22 (88%) | |
| 2–3 | 11 (42.3%) | 3 (12%) | |
| Type of malignancy n (%) * | 0.3 | ||
| Upper GI tract | 6 (23.1%) | 3 (12%) | |
| Lower GI tract | 20 (76.9%) | 22 (88%) | |
| Location of obstruction | 0.7 | ||
| Upper | 1 (3.8%) | 2 (8%) | |
| Middle | 6 (23.1%) | 4 (16%) | |
| Lower | 19 (73.1%) | 19 (76%) | |
| Laterality | 0.5 | ||
| Monolateral | 14 (53.8%) | 16 (64%) | |
| Bilateral | 12 (46.2%) | 9 (36%) | |
| Cause of obstruction | 0.2 | ||
| Unknown | 4 (15.4%) | 3 (12%) | |
| Mass | 8 (30.8%) | 13 (52%) | |
| Lymphadenopathy | 1 (3.8%) | 3 (12%) | |
| Ureteral infiltration | 5 (19.2%) | 4 (16%) | |
| Carcinomatosis | 8 (30.8%) | 2 (8%) | |
| Hydronephrosis degree | 0.6 | ||
| Mild | 4 (15.4%) | 3 (12%) | |
| Moderate | 13 (50%) | 10 (40%) | |
| Severe | 9 (34.6%) | 12 (48%) | |
| Bladder invasion | 0.2 | ||
| Yes | 5 (19.2%) | 2 (8%) | |
| No | 21 (80.8%) | 23 (92%) | |
| Time between malignancy diagnosis and urinary diversion (months) | 3.5 (0–12.7) | 18.5 (1.25–37.5) | 0.007 |
| Previous chemotherapy | 0.3 | ||
| Yes | 14 (53.8%) | 17 (68%) | |
| No | 12 (46.2%) | 8 (32%) | |
| Previous radiotherapy | 0.8 | ||
| Yes | 8 (30.8%) | 7 (28%) | |
| No | 18 (69.2%) | 18 (72%) | |
| Preoperative creatinine (mg/dL) | 1.9 (1.1–3.7) | 1.4 (0.8–1.8) | 0.07 |
| Preoperative eGFR (mL/min/1.73 m2) | 28 (12.5–60) | 43 (33.5–73.5) | 0.07 |
| Type of urinary diversion | 0.1 | ||
| US | 11 (42.3%) | 16 (64%) | |
| NT | 15 (57.7%) | 9 (36%) | |
| Stent failure | 0.5 | ||
| Yes | 5 (19.2%) | 3 (12%) | |
| No | 21 (80.8%) | 22 (88%) | |
| Hospital stay (days) | 13 (4.7–20.2) | 4 (2–11.5) | 0.06 |
| Postoperative creatinine (mg/dL) | 1 (0.7–1.4) | 0.9 (0.6–1.4) | 0.4 |
| Postoperative eGFR (mL/min/1.73 m2) | 68 (40–86.5) | 75 (41.7–95) | 0.3 |
| Complications | |||
| Yes | 3 (11.5%) | 4 (16%) | 0.6 |
| no | 23 (88.5%) | 21 (84%) | |
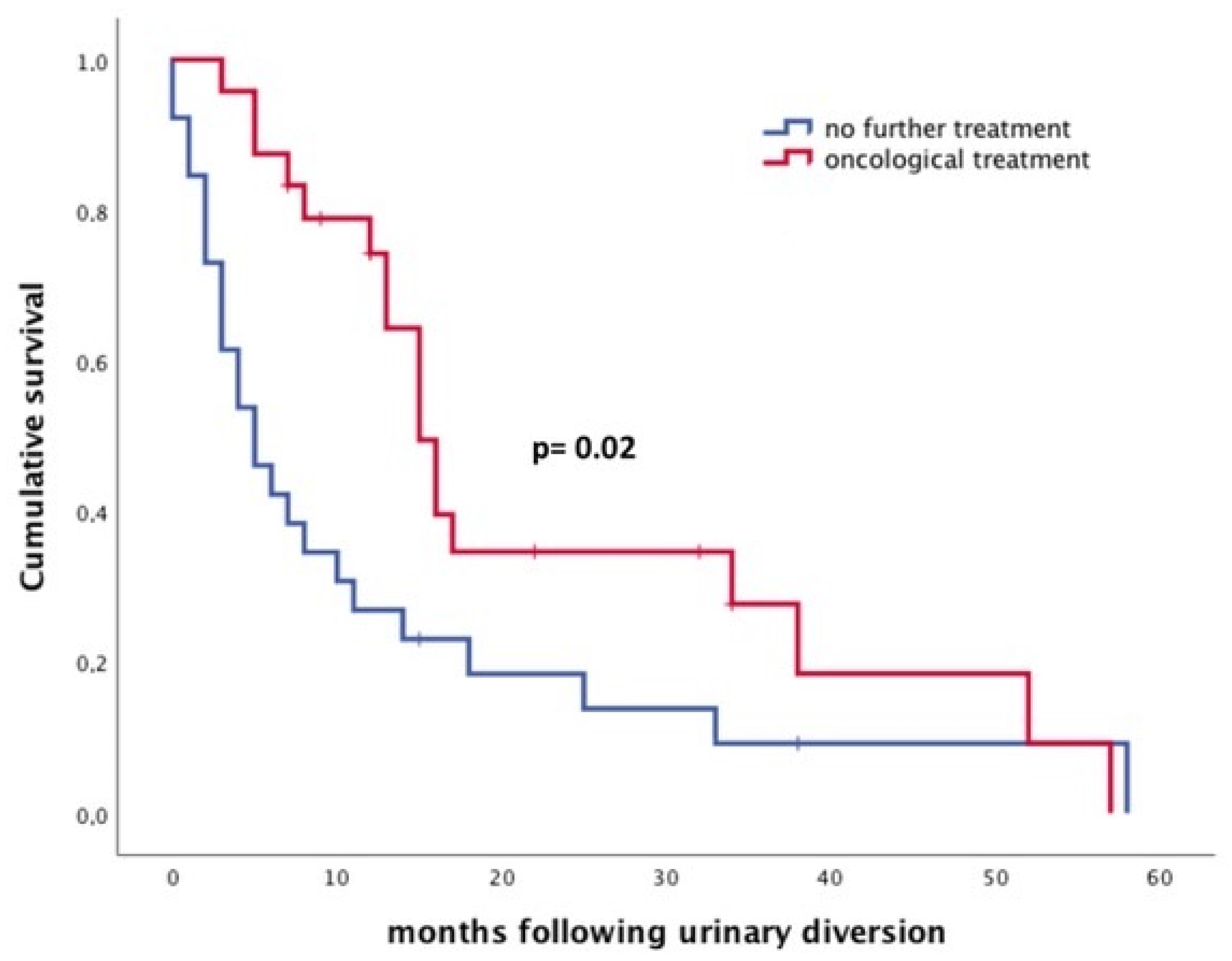
| Overall survival (months) | 5 (2–14.2) | 15 (8.2–29.5) | 0.003 |
| Parameter | P Value | 95% CI |
|---|---|---|
| Age at surgery (years) | 0.96 | 0.95–1.05 |
| Gender | 0.07 | 0.89–0.96 |
| ECOG Performance status | 0.02 | 0.04–0.78 |
| Type of malignancy * Upper GI tract Lower GI tract | 0.3 | 0.48–9.98 |
| Location of obstruction | 0.9 | 0.37–2.49 |
| Laterality | 0.4 | 0.21–2.01 |
| Cause of obstruction | 0.1 | 0.47–1.07 |
| Hydronephrosis degree | 0.4 | 0.63–3.22 |
| Bladder invasion | 0.2 | 0.06–2.08 |
| Time between malignancy diagnosis and urinary diversion | 0.037 | 1.002–1.07 |
| Previous chemotherapy | 0.3 | 0.58–5.68 |
| Previous radiotherapy | 0.8 | 0.26–2.92 |
| Preoperative creatinine | 0.4 | 0.75–1.12 |
| Preoperative eGFR | 0.07 | 0.99–1.04 |
| Type of urinary diversion | 0.12 | 0.13–1.27 |
| Hospital stay (days) | 0.13 | 0.89–1.01 |
| Postoperative creatinine | 0.5 | 0.22–2.07 |
| Postoperative eGFR | 0.3 | 0.99–1.03 |
| Complications | 0.6 | 0.29–7.30 |
| Parameter | p Value | Hazard Ratio | 95% CI |
|---|---|---|---|
| ECOG Performance status | 0.000 | 4.5 | 2.2–9.1 |
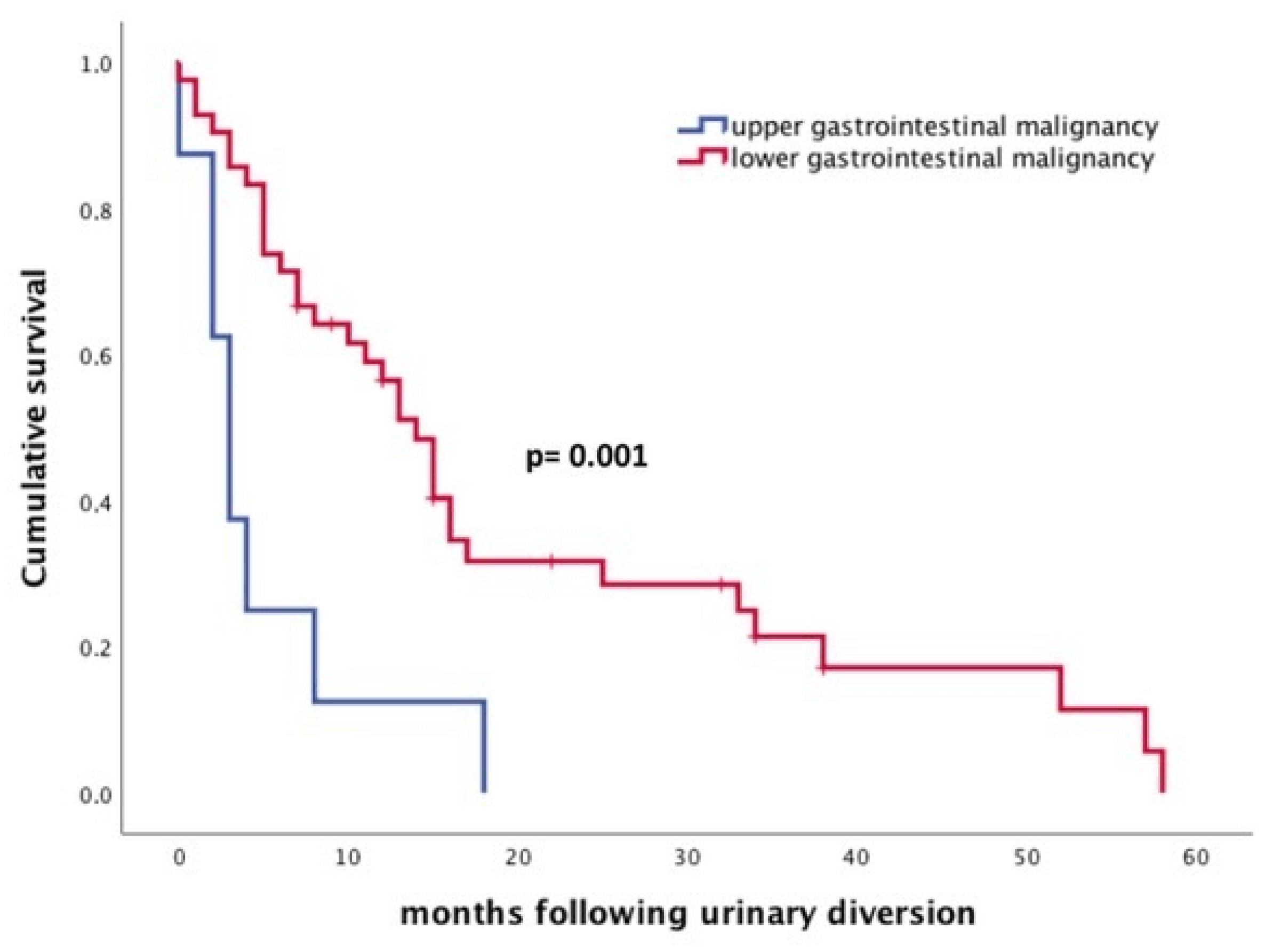
| Type of malignancy (upper/lower GI tract) | 0.003 | 3.4 | 1.5–7.5 |
| Postoperative oncological treatment | 0.026 | 0.49 | 0.26–0.92 |
| Time between diagnosis and urinary diversion (</> 8 months) | 0.048 | 0.51 | 0.27–0.99 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Lorenzis, E.; Lievore, E.; Turetti, M.; Gallioli, A.; Galassi, B.; Boeri, L.; Montanari, E. Ureteral Stent and Percutaneous Nephrostomy in Managing Malignant Ureteric Obstruction of Gastrointestinal Origin: A 10 Years’ Experience. Gastrointest. Disord. 2020, 2, 456-468. https://0-doi-org.brum.beds.ac.uk/10.3390/gidisord2040041
De Lorenzis E, Lievore E, Turetti M, Gallioli A, Galassi B, Boeri L, Montanari E. Ureteral Stent and Percutaneous Nephrostomy in Managing Malignant Ureteric Obstruction of Gastrointestinal Origin: A 10 Years’ Experience. Gastrointestinal Disorders. 2020; 2(4):456-468. https://0-doi-org.brum.beds.ac.uk/10.3390/gidisord2040041
Chicago/Turabian StyleDe Lorenzis, Elisa, Elena Lievore, Matteo Turetti, Andrea Gallioli, Barbara Galassi, Luca Boeri, and Emanuele Montanari. 2020. "Ureteral Stent and Percutaneous Nephrostomy in Managing Malignant Ureteric Obstruction of Gastrointestinal Origin: A 10 Years’ Experience" Gastrointestinal Disorders 2, no. 4: 456-468. https://0-doi-org.brum.beds.ac.uk/10.3390/gidisord2040041





