Evaluation of an Algorithm for Testis-Sparing Surgery in Boys with Testicular Tumors: A Retrospective Cohort Study
Abstract
:1. Introduction
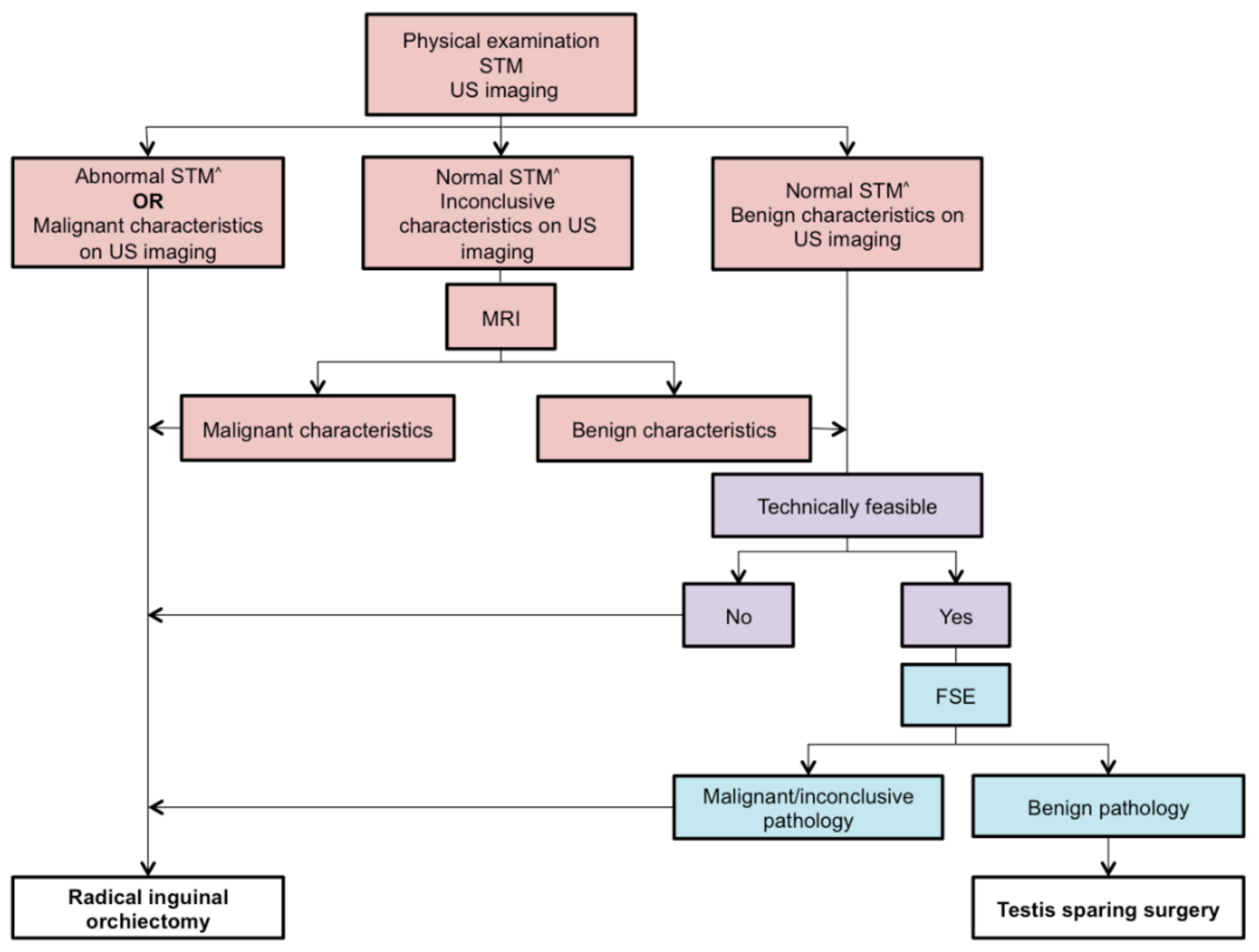
2. Materials and Methods
3. Results
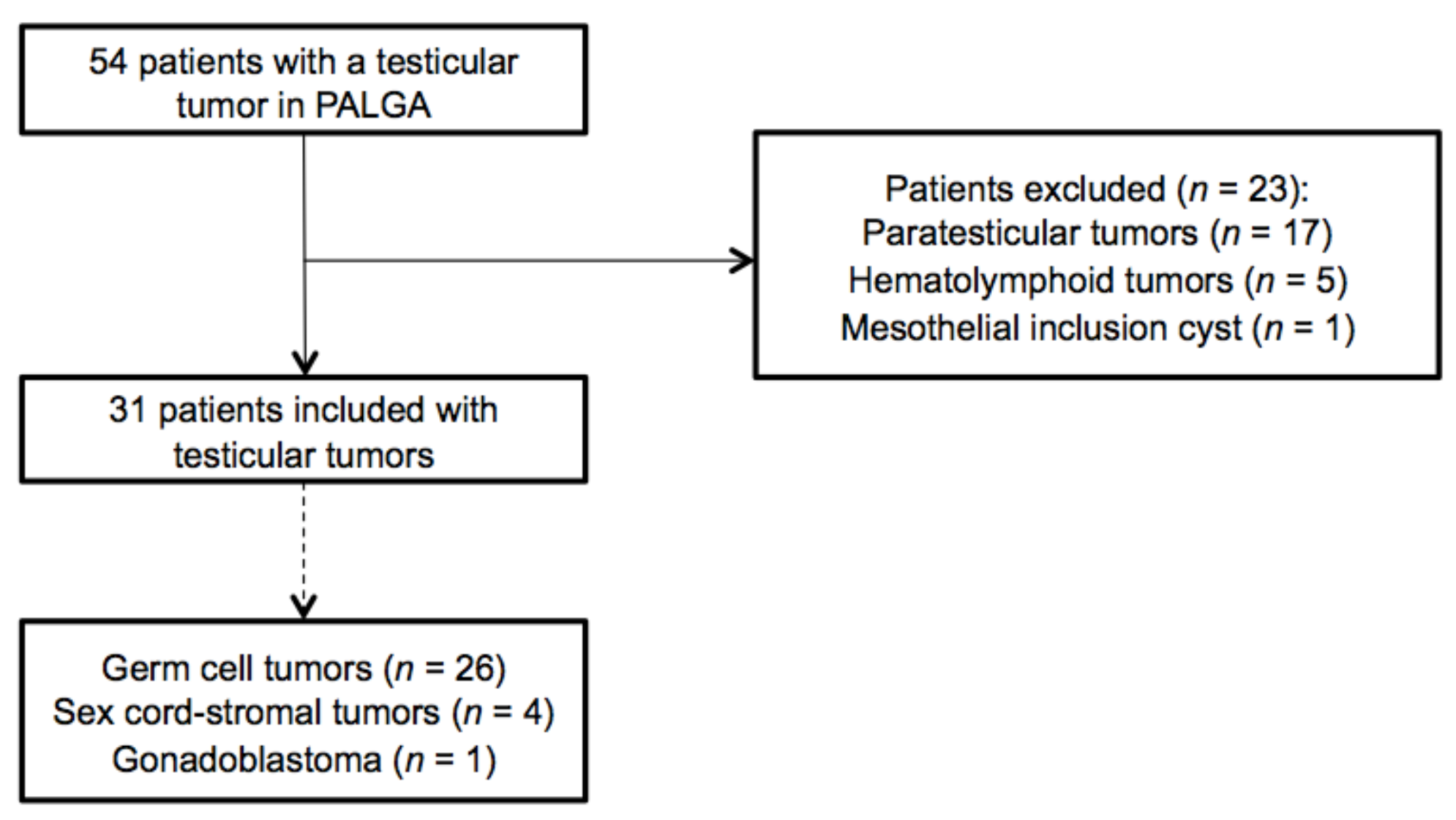
3.1. Search and Selection
3.2. Clinical Characteristics
3.3. Diagnostic Findings, Treatment, and Outcome
3.4. Performance, Considerations, and Outcome of TSS
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Shanmugalingam, T.; Soultati, A.; Chowdhury, S.; Rudman, S.; van Hemelrijck, M. Global incidence and outcome of testic-ular cancer. Clin. Epidemiol. 2013, 5, 417–427. [Google Scholar] [PubMed] [Green Version]
- Purdue, M.P.; Devesa, S.S.; Sigurdson, A.J.; McGlynn, K.A. International patterns and trends in testis cancer incidence. Int. J. Cancer 2005, 115, 822–827. [Google Scholar] [CrossRef] [PubMed]
- Cheng, L.; Albers, P.; Berney, D.M.; Feldman, D.R.; Daugaard, G.; Gilligan, T.; Looijenga, L.H. Testicular cancer. Nat. Rev. Dis. Primers 2018, 4, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Maizlin, I.I.; Dellinger, M.; Gow, K.W.; Goldin, A.B.; Goldfarb, M.; Nuchtern, J.G.; Langer, M.; Vasudevan, S.A.; Doski, J.J.; Raval, M.V.; et al. Testicular tumors in prepubescent patients. J. Pediatr. Surg. 2018, 53, 1748–1752. [Google Scholar] [CrossRef] [PubMed]
- Karmazyn, B.; Weatherly, D.; Lehnert, S.J.; Cain, M.; Fan, R.; Jennings, S.G.; Ouyang, F.; Kaefer, M. Characteristics of testicular tumors in prepubertal children (age 5–12 years). J. Pediatr. Urol. 2018, 14, 259.e1–259.e6. [Google Scholar] [CrossRef] [Green Version]
- Ahmed, H.U.; Arya, M.; Muneer, A.; Mushtaq, I.; Sebire, N.J. Testicular and paratesticular tumours in the prepubertal pop-ulation. Lancet Oncol. 2010, 11, 476–483. [Google Scholar] [CrossRef]
- Hung, G.-Y.; Horng, J.-L.; Yen, H.-J.; Lee, C.-Y. Pre-pubertal and adolescent germ cell neoplasms in Taiwan: Time trends and geographic variation. Andrology 2015, 3, 895–901. [Google Scholar] [CrossRef] [Green Version]
- Looijenga, L.H.; Van der Kwast, T.H.; Grignon, D.; Egevad, L.; Kristiansen, G.; Kao, C.S.; Idrees, M.T. Report From the International Society of Urological Pathology (ISUP) Consultation Conference on Mo-lecular Pathology of Urogenital Cancers. Am. J. Surg. Pathol. 2020, 44, e66–e79. [Google Scholar]
- Spiller, C.M.; Bowles, J. Germ cell neoplasia in situ: The precursor cell for invasive germ cell tumors of the testis. Int. J. Biochem. Cell Biol. 2017, 86, 22–25. [Google Scholar] [CrossRef] [Green Version]
- Moch, H.; Cubilla, A.L.; Humphrey, P.A.; Reuter, V.E.; Ulbright, T.M. The 2016 WHO Classification of Tumours of the Urinary System and Male Genital Organs—Part A: Renal, Penile, and Testicular Tumours. Eur. Urol. 2016, 70, 93–105. [Google Scholar] [CrossRef]
- Pohl, H.G.; Shukla, A.R.; Metcalf, P.D.; Cilento, B.G.; Retik, A.B.; Bagli, D.J.; Huff, D.S.; Gil Rushton, H. Prepubertal Testis Tumors: Actual Prevalence Rate of Histological Types. J. Urol. 2004, 172, 2370–2372. [Google Scholar] [CrossRef] [PubMed]
- Calaminus, G.; Schneider, D.T.; von Schweinitz, D.; Jürgens, H.; Infed, N.; Schönberger, S.; Olson, T.A.; Albers, P.; Vokuhl, C.; Stein, R.; et al. Age-Dependent Presentation and Clinical Course of 1465 Patients Aged 0 to Less than 18 Years with Ovarian or Testicular Germ Cell Tumors; Data of the MAKEI 96 Protocol Revisited in the Light of Prenatal Germ Cell Biology. Cancers 2020, 12, 611. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patel, H.D.; Gupta, M.; Cheaib, J.G.; Sharma, R.; Zhang, A.; Bass, E.B.; Pierorazio, P. Testis-sparing surgery and scrotal violation for testicular masses suspicious for malignancy: A systematic review and meta-analysis. Urol. Oncol. Semin. Orig. Investig. 2020, 38, 344–353. [Google Scholar] [CrossRef] [PubMed]
- Woo, L.L.; Ross, J.H. The role of testis-sparing surgery in children and adolescents with testicular tumors. Urol. Oncol. Semin. Orig. Investig. 2016, 34, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Djaladat, H. Organ-sparing surgery for testicular tumours. Curr. Opin. Urol. 2015, 25, 116–120. [Google Scholar] [CrossRef] [PubMed]
- Zuniga, A.; Lawrentschuk, N.; Jewett, M.A. Organ-sparing approaches for testiclar masses. Nat. Rev. Urol. 2010, 7, 454–464. [Google Scholar] [CrossRef]
- Ferreira, U.; Netto, N.R.; Esteves, S.C.; Rivero, M.A.; Schirren, C. Comparative Study of the Fertility Potential of Men with only one Testis. Scand. J. Urol. Nephrol. 1991, 25, 255–259. [Google Scholar] [CrossRef]
- Radford, A.; Peycelon, M.; Haid, B.; Powis, M.; Lakshminarayanan, B. Testicular-sparing surgery in the pediatric popula-tion: Multicenter review of practice with review of the literature. Curr. Opin. Urol. 2019, 29, 481–486. [Google Scholar] [CrossRef]
- Kooij, C.D.; Hulsker, C.C.; Kranendonk, M.E.; Zsiros, J.; Littooij, A.S.; Looijenga, L.H.; Klijn, A.J.; Mavinkurve-Groothuis, A.M. Testis Sparing Surgery in Pediatric Testicular Tumors. Cancers 2020, 12, 2867. [Google Scholar] [CrossRef]
- Overbeek, L.; Van Kemenade, F.; Meijer, G. Het Pathologisch-Anatomisch Landelijk Geautomatiseerd Archief. Tijdschr. Voor Gezondh. 2012, 90, 276–279. [Google Scholar] [CrossRef]
- Kusler, K.A.; Poynter, J.N. International testicular cancer incidence rates in children, adolescents and young adults. Cancer Epidemiol. 2018, 56, 106–111. [Google Scholar] [CrossRef] [PubMed]
- Nistal, M.; Paniagua, R.; González-Peramato, P.; Reyes-Múgica, M. Perspectives in pediatric pathology, chapter 25. Testic-ular and paratesticular tumors in the pediatric age group. Pediatr. Dev. Pathol. 2016, 19, 471–492. [Google Scholar] [CrossRef] [PubMed]
- Chung, J.M.; Lee, S.D. Overview of Pediatric Testicular Tumors in Korea. Korean J. Urol. 2014, 55, 789–796. [Google Scholar] [CrossRef] [PubMed]
- Stoop, H.; Kirkels, W.; Dohle, G.R.; Gillis, A.J.M.; Bakker, M.A.D.; Biermann, K.; Oosterhuis, J.W.; Looijenga, L.H. Diagnosis of testicular carcinoma in situ‘(intratubular and microinvasive)’ seminoma and embryonal carcinoma using direct enzymatic alkaline phosphatase reactivity on frozen histological sections. Histopathology 2011, 58, 440–446. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barlow, L.J.; Badalato, G.M.; McKiernan, J.M. Serum tumor markers in the evaluation of male germ cell tumors. Nat. Rev. Urol. 2010, 7, 610–617. [Google Scholar] [CrossRef]
- Mittal, P.K.; Abdalla, A.S.; Chatterjee, A.; Baumgarten, D.A.; Harri, P.A.; Patel, J.; Moreno, C.C.; Gabriel, H.A.; Miller, F.H. Spectrum of Extratesticular and Testicular Pathologic Conditions at Scrotal MR Imaging. Radiographics 2018, 38, 806–830. [Google Scholar] [CrossRef] [Green Version]
- Pearce, M.; Salotti, J.A.; Little, M.P.; McHugh, K.; Lee, C.; Kim, K.P.; Howe, N.L.; Ronckers, C.M.; Rajaraman, P.; Craft, A.W.S.; et al. Radiation exposure from CT scans in childhood and subsequent risk of leukaemia and brain tumours: A retrospective cohort study. Lancet 2012, 380, 499–505. [Google Scholar] [CrossRef] [Green Version]
- Hisamatsu, E.; Takagi, S.; Nakagawa, Y.; Sugita, Y.; Yoshino, K.; Ueoka, K.; Tanikaze, S. Prepubertal testicular tumors: A 20-year experience with 40 cases. Int. J. Urol. 2010, 17, 956–959. [Google Scholar] [CrossRef]
- Friend, J.; Barker, A.; Khosa, J.; Samnakay, N. Benign scrotal masses in children–some new lessons learned. J. Pediatr. Surg. 2016, 51, 1737–1742. [Google Scholar] [CrossRef]
- Matei, D.V.; Vartolomei, M.D.; Renne, G.; Tringali, V.M.L.; Russo, A.; Bianchi, R.; Cozzi, G.; Bottero, D.; Musi, G.; Mazzarol, G.; et al. Reliability of Frozen Section Examination in a Large Cohort of Testicular Masses: What Did We Learn? Clin. Genitourin. Cancer 2017, 15, e689–e696. [Google Scholar] [CrossRef]
| Patient Characteristics | |
|---|---|
| Number of Patients | 31 |
| Median age in years (range) | 5.5 (0–16.9) |
| Presenting Symptoms | |
| Enlarged testis | 19 (61.3%) |
| Scrotal pain | 6 (19.4%) |
| Palpable mass | 5 (16.1%) |
| Cryptorchidism | 1 (3.2%) |
| Side (left/right) | 13/18 |
| Imaging | |
| US | 13 |
| US + CT | 12 |
| US + MRI | 2 |
| US + CT + MRI | 3 |
| None | 1 |
| Median follow-up in months (range) | 34 (5–94) |
| Histopathological Diagnosis | Stage * | Median Age in Years (Range) | Pubertal Status ^ | Findings on Imaging (US/CT/MRI) | n (%) with Elevated AFP # | n (%) with Elevated bHCG # | Surgical Approach | FSE | Adjuvant Treatment | Outcome |
|---|---|---|---|---|---|---|---|---|---|---|
| GCTs | ||||||||||
| Benign | ||||||||||
| MT | I (4) | 5.3 (2.8–5.8) | Pre (4) | Suspected of benign pathology (3), inconclusive on US (1) | 0 (0%) | 0 (0%) | TSS (3), RIO (1) | Yes (2), no (2) | None (4) | NED (4) |
| MT + IT | I (2) | 5.5 | Pre (2) | Suspected of malignancy (1), inconclusive on US (1) | 2 (100%) | 0 (0%) | RIO (2) | No (2) | None (1), ND (1) | NED (1), ND (1) |
| EpC | I (1) | 11.7 | Pre (1) | Inconclusive on US (1) | 0 (0%) | 0 (0%) | RIO (1) | No (1) | None (1) | ND (1) |
| DC | I (2) | 10.9 | Pre (1), Pub (1) | Suspected of benign pathology (2) | 0 (0%) | 0 (0%) | TSS (1), RIO (1) | No (2) | None (2) | NED (2) |
| Malignant | ||||||||||
| YST | I (10) | 1.4 (0.3–2.3) | Pre (10) | Suspected of malignancy (3), inconclusive on US (2), inconclusive on US + CT (4), inconclusive on US + MRI (1) | 10 (100%) | 0 (0%) | RIO (10) | Yes (2), no (8) | None (3), BEP (1), 2PE (6) | NED (9), ND (1) |
| MGCT | II (1) | 0.4 | Pre (1) | Suspected of malignancy (1) | 1 (100%) | 0 (0%) | SO (1) | No (1) | 4PEI + RLND (1) | NED (1) |
| I (4) | 15.4 (14.5–16.4) | Pub (4) | Suspected of malignancy (2), inconclusive on US + CT (2) | 3 (75%) | 3 (75%) | RIO (4) | No (4) | 2PE (2), 2PEI (1), 1PE + 2PEI (1) | NED (4) | |
| IIIB (2) | 16.7 | Pub (2) | Suspected of malignancy (2) | 2 (100%) | 2 (100%) | RIO (2) | No (2) | 4PEI (1), 2PEI + JEP (1) | NED (2) | |
| SCSTs | ||||||||||
| SCT | I (2) | 4.9 | Pre (2) | Inconclusive on US (1), inconclusive on US + MRI (1) | 0 (0%) | 0 (0%) | RIO (1), SO (1) | No (2) | None (2) | NED (2) |
| JGCT | I (2) | 0.2 | Pre (2) | Inconclusive on US (2) | 1 (50%) | 1 (50%) | RIO (2) | Yes (1), no (1) | None (2) | NED (2) |
| GCT/SCST | ||||||||||
| Gonadoblastoma | I (1) | 0 | Pre (1) | ND | ND | ND | RIO (1) | No (1) | None (1) | NED (1) |
| Patient Number | Age in Years | AFP Level | bHCG Level | Size on US * | Findings on US | Considerations against TSS | Surgical Approach | FSE | Final Pathology | Testicular Atrophy | Postoperative Complications |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 13.8 | 1.9 | <2.0 | 1.3 × 1.8 × 1.3 | Suspected of benign pathology | None | TSS | No | Dermoid cyst | No | None |
| 2 | 6 | 1.0 | <2.0 | ND | Suspected of benign pathology | None | TSS | Yes | Mature teratoma | No | None |
| 3 | 2.2 | 2.5 | <2.0 | ND | Suspected of benign pathology | None | TSS | No | Mature teratoma | No | Abscess |
| 4 | 4.9 | 2.0 | <2.0 | 0.7 × 0.8 × 0.8 | Suspected of benign pathology | None | TSS | Yes | Mature teratoma | No | None |
| 5 | 5.8 | <1.0 | <2.0 | 2.0 × 1.3 × 2.0 | Inconclusive | Technically unfeasible, according to attending physician | RIO | No | Mature teratoma | NA | None |
| 6 | 0.5 | 18 # | <2.0 | 1.7 × 1.9 × 2.4 | Inconclusive | Elevated AFP | RIO | No | Mature teratoma with immature components | NA | None |
| 7 | 0.4 | 45.9 # | <2.0 | 3.6 × 2.6 × 2.2 | Suspected of malignancy | Elevated AFP + suspected of malignancy on US | RIO | No | Mature teratoma with immature components | NA | ND |
| 8 | 11.7 | <2.0 | <2.0 | 1.1 ^ | Inconclusive | Technically unfeasible, according to attending physician | RIO | No | Epidermoid cyst | NA | ND |
| 9 | 8.1 | 1.2 | <2.0 | 2 × 1.2 × 3 | Suspected of benign pathology | Non-functioning testis, according to attending physician | RIO | No | Dermoid cyst | NA | None |
| 10 | 9.8 | 2.0 | <0.2 | 1 × 0.9 × 0.7 | Inconclusive | MRI could not rule out malignancy | SO | No | Sertoli cell tumor | NA | None |
| 11 | 0 | 11,000 | <2.0 | 2.8 × 2.0 × 2.2 | Inconclusive | None | RIO | No | Sertoli cell tumor | NA | None |
| 12 | 0.3 | 230 # | <2.0 | 1.1 × 1.7 × 1.8 | Inconclusive | Elevated AFP | RIO | Yes | Juvenile granulosa cell tumor | NA | None |
| 13 | 0 | 15 721 | 84.0 | 1.1 × 1.4 × 1.5 | Inconclusive | Elevated bHCG | RIO | No | Juvenile granulosa cell tumor | NA | None |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kooij, C.D.; Hulsker, C.C.C.; Kranendonk, M.E.G.; Zsiros, J.; Littooij, A.S.; Looijenga, L.H.J.; Klijn, A.J.; Mavinkurve-Groothuis, A.M.C. Evaluation of an Algorithm for Testis-Sparing Surgery in Boys with Testicular Tumors: A Retrospective Cohort Study. Surgeries 2021, 2, 9-19. https://0-doi-org.brum.beds.ac.uk/10.3390/surgeries2010002
Kooij CD, Hulsker CCC, Kranendonk MEG, Zsiros J, Littooij AS, Looijenga LHJ, Klijn AJ, Mavinkurve-Groothuis AMC. Evaluation of an Algorithm for Testis-Sparing Surgery in Boys with Testicular Tumors: A Retrospective Cohort Study. Surgeries. 2021; 2(1):9-19. https://0-doi-org.brum.beds.ac.uk/10.3390/surgeries2010002
Chicago/Turabian StyleKooij, Cezanne D., Caroline C.C. Hulsker, Mariëtte E.G. Kranendonk, József Zsiros, Annemieke S. Littooij, Leendert H.J. Looijenga, Aart J. Klijn, and Annelies M.C. Mavinkurve-Groothuis. 2021. "Evaluation of an Algorithm for Testis-Sparing Surgery in Boys with Testicular Tumors: A Retrospective Cohort Study" Surgeries 2, no. 1: 9-19. https://0-doi-org.brum.beds.ac.uk/10.3390/surgeries2010002





