Effect of Vitamin D Deficiency on COVID-19 Status: A Systematic Review
Abstract
:1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Eligibility Criteria
2.3. Data Extraction and Study Quality Assessment
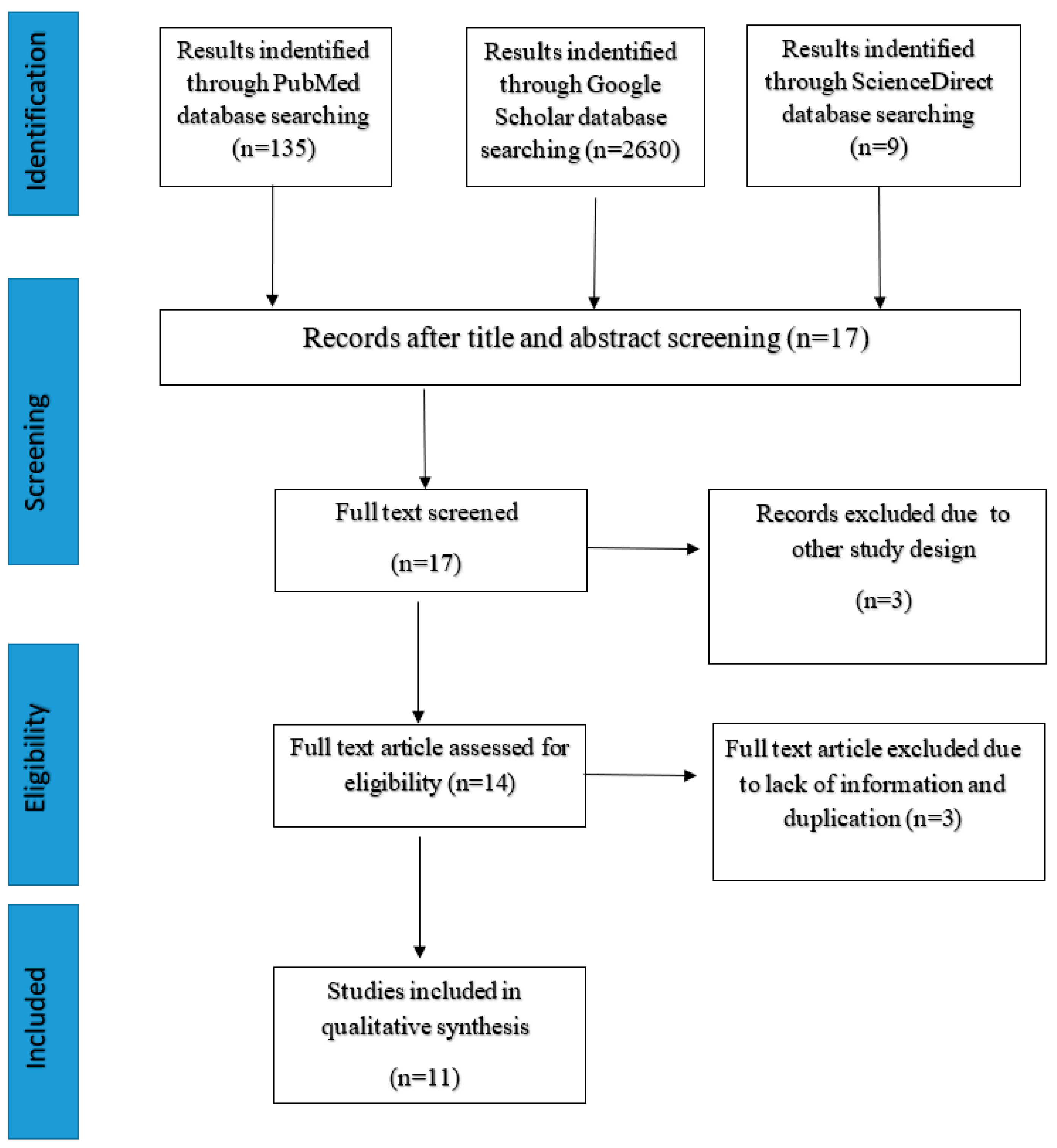
3. Results
3.1. Search Results
3.2. Study Characteristics and Main Findings
3.3. Findings from All the Studies
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AOR | adjusted odds Ratio |
| HR | Hazard ratio |
References
- World Health Organisation. WHO Coronavirus Disease (COVID-19) Dashboard. 2020. Available online: https://covid19.who.int/ (accessed on 11 November 2020).
- Looi, M.K. Covid-19: Is a second wave hitting Europe? BMJ 2020, 371, m4113. [Google Scholar] [CrossRef]
- Aleta, A.; Martin-Corral, D.; y Piontti, A.P.; Ajelli, M.; Litvinova, M.; Chinazzi, M.; Dean, N.E.; Halloran, M.E.; Longini, I.M., Jr.; Merler, S.; et al. Modeling the impact of social distancing, testing, contact tracing and household quarantine on second-wave scenarios of the COVID-19 epidemic. medRxiv 2020. [Google Scholar] [CrossRef]
- Leung, K.; Wu, J.T.; Liu, D.; Leung, G.M. First-wave COVID-19 transmissibility and severity in China outside Hubei after control measures, and second-wave scenario planning: A modelling impact assessment. Lancet 2020, 395, 1382–1393. [Google Scholar] [CrossRef]
- Laird, E.; Kenny, R.A. Vitamin D Deficiency in Ireland–Implications for COVID-19. Results from the Irish Longitudinal Study on Ageing (TILDA). 2020. Available online: https://tilda.tcd.ie/publications/reports/pdf/Report_Covid19VitaminD.pdf (accessed on 21 November 2020).
- Richardson, D.P.; Lovegrove, J.A. Nutritional status of micronutrients as a possible and modifiable risk factor for COVID-19: A UK perspective. Br. J. Nutr. 2021, 125, 678–684. [Google Scholar] [CrossRef]
- Ebadi, M.; Montano-Loza, A.J. Perspective: Improving vitamin D status in the management of COVID-19. Eur. J. Clin. Nutr. 2020, 74, 856–859. [Google Scholar] [CrossRef] [PubMed]
- Grant, W.B.; Lahore, H.; McDonnell, S.L.; Baggerly, C.A.; French, C.B.; Aliano, J.L.; Bhattoa, H.P. Evidence that vitamin D supplementation could reduce risk of influenza and COVID-19 infections and deaths. Nutrients 2020, 12, 988. [Google Scholar] [CrossRef] [Green Version]
- McCartney, D.M.; Byrne, D.G. Optimisation of vitamin D status for enhanced Immuno-protection against Covid-19. Ir. Med. J. 2020, 113, 58. [Google Scholar]
- Di Rosa, M.; Malaguarnera, M.; Nicoletti, F.; Malaguarnera, L. Vitamin D3: A helpful immuno-modulator. Immunology 2020, 134, 123–139. [Google Scholar] [CrossRef] [PubMed]
- Zhong, J.; Tang, J.; Ye, C.; Dong, L. The immunology of COVID-19: Is immune modulation an option for treatment? Lancet Rheumatol. 2020, 2, e428–e436. [Google Scholar] [CrossRef]
- Panarese, A.; Shahini, E. Covid-19, and vitamin D. Aliment. Pharmacol. Ther. 2020, 51, 993. [Google Scholar] [CrossRef]
- Grasselli, G.; Tonetti, T.; Protti, A.; Langer, T.; Girardis, M.; Bellani, G.; Laffey, J.; Carrafiello, G.; Carsana, L.; Rizzuto, C.; et al. Pathophysiology of COVID-19-associated acute respiratory distress syndrome: A multicentre prospective observational study. Lancet Respir. Med. 2020, 8, 1201–1208. [Google Scholar] [CrossRef]
- Ginde, A.A.; Mansbach, J.M.; Camargo, C.A. Association between serum 25-hydroxyvitamin D level and upper respiratory tract infection in the Third National Health and Nutrition Examination Survey. Arch. Intern. Med. 2009, 169, 384–390. [Google Scholar] [CrossRef] [Green Version]
- Jolliffe, D.A.; Griffiths, C.J.; Martineau, A.R. Vitamin D in the prevention of acute respiratory infection: Systematic review of clinical studies. J. Steroid Biochem. Mol. Biol. 2020, 136, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Bergman, P.; Norlin, A.C.; Hansen, S.; Rekha, R.S.; Agerberth, B.; Björkhem-Bergman, L.; Ekström, L.; Lindh, J.D.; Andersson, J. Vitamin D3 supplementation in patients with frequent respiratory tract infections: A randomised and double-blind intervention study. BMJ Open 2012, 2, e001663. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martineau, A.R.; Forouhi, N.G. Vitamin D for COVID-19: A case to answer? Lancet Diabetes Endocrinol. 2020, 8, 735–736. [Google Scholar] [CrossRef]
- Ali, N. Role of vitamin D in preventing of COVID-19 infection, progression and severity. J. Infect. Public Health 2020, 13, 1373–1380. [Google Scholar] [CrossRef]
- Ilie, P.C.; Stefanescu, S.; Smith, L. The role of vitamin D in the prevention of coronavirus disease 2019 infection and mortality. Aging Clin. Exp. Res. 2020, 32, 1195–1198. [Google Scholar] [CrossRef]
- Walrand, S. Autumn COVID-19 surge dates in Europe correlated to latitudes, not to temperature-humidity, pointing to vitamin D as contributing factor. Sci. Rep. 2021, 11, 1981. [Google Scholar] [CrossRef] [PubMed]
- Klingberg, E.; Oleröd, G.; Konar, J.; Petzold, M.; Hammarsten, O. Seasonal variations in serum 25-hydroxy vitamin D levels in a Swedish cohort. Endocrine 2015, 49, 800–808. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abrishami, A.; Dalili, N.; Torbati, P.M.; Asgari, R.; Arab-Ahmadi, M.; Behnam, B.; Sanei-Taheri, M. Possible association of vitamin D status with lung involvement and outcome in patients with COVID-19: A retrospective study. Eur. J. Nutr. 2020, 60, 2249–2257. [Google Scholar] [CrossRef]
- Radujkovic, A.; Hippchen, T.; Tiwari-Heckler, S.; Dreher, S.; Boxberger, M.; Merle, U. Vitamin D deficiency and outcome of COVID-19 patients. Nutrients 2020, 12, 2757. [Google Scholar] [CrossRef] [PubMed]
- Maghbooli, Z.; Sahraian, M.A.; Ebrahimi, M.; Pazoki, M.; Kafan, S.; Tabriz, H.M.; Hadadi, A.; Montazeri, M.; Nasiri, M.; Shirvani, A.; et al. Vitamin D sufficiency, a serum 25-hydroxyvitamin D at least 30 ng/mL reduced risk for adverse clinical outcomes in patients with COVID-19 infection. PLoS ONE 2020, 15, e0239799. [Google Scholar] [CrossRef]
- Macaya, F.; Espejo Paeres, C.; Valls, A.; Fernández-Ortiz, A.; Del Castillo, J.G.; Martín-Sánchez, F.J.; Runkle, I.; Rubio Herrera, M.Á. Interaction between age and vitamin D deficiency in severe COVID-19 infection. Nutr. Hosp. 2020, 37, 1039–1042. [Google Scholar] [CrossRef] [PubMed]
- Ye, K.; Tang, F.; Liao, X.; Shaw, B.A.; Deng, M.; Huang, G.; Qin, Z.; Peng, X.; Xiao, H.; Chen, C.; et al. Does Serum Vitamin D Level Affect COVID-19 Infection and Its Severity?-A Case-Control Study. J. Am. Coll. Nutr. 2020, 1–8. [Google Scholar] [CrossRef]
- Merzon, E.; Tworowski, D.; Gorohovski, A.; Vinker, S.; Golan Cohen, A.; Green, I.; Frenkel-Morgenstern, M. Low plasma 25(OH) vitamin D level is associated with increased risk of COVID-19 infection: An Israeli population-based study. FEBS J. 2020, 287, 3693–3702. [Google Scholar] [CrossRef]
- Meltzer, D.O.; Best, T.J.; Zhang, H.; Vokes, T.; Arora, V.; Solway, J. Association of Vitamin D Status and Other Clinical Characteristics With COVID-19 Test Results. JAMA Netw. Open 2020, 3, e2019722. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, H.W.; Niles, J.K.; Kroll, M.H.; Bi, C.; Holick, M.F. SARS-CoV-2 positivity rates associated with circulating 25-hydroxyvitamin D levels. PLoS ONE 2020, 15, e0239252. [Google Scholar] [CrossRef]
- D’avolio, A.; Avataneo, V.; Manca, A.; Cusato, J.; De Nicolò, A.; Lucchini, R.; Keller, F.; Cantù, M. 25-hydroxyvitamin D concentrations are lower in patients with positive PCR for SARS-CoV-2. Nutrients 2020, 12, 1359. [Google Scholar] [CrossRef]
- Baktash, V.; Hosack, T.; Patel, N.; Shah, S.; Kandiah, P.; Van den Abbeele, K.; Mandal, A.K.J.; Missouris, C.G. Vitamin D status and outcomes for hospitalised older patients with COVID-19. Postgrad. Med. J. 2020, 2, 1–6. [Google Scholar] [CrossRef]
- Hastie, C.E.; Mackay, D.F.; Ho, F.; Celis-Morales, C.A.; Katikireddi, S.V.; Niedzwiedz, C.L.; Jani, B.D.; Welsh, P.; Mair, F.S.; Gray, S.R.; et al. Vitamin D concentrations and COVID-19 infection in UK Biobank. Diabetes Metab. Syndr. Clin. Res. Rev. 2020, 14, 561–565. [Google Scholar] [CrossRef]
| Author | Study Design | Country | Sample Size | Mean/Median Age/Age Interval (Years) | Vitamin D Categories | Outcome Assessed |
|---|---|---|---|---|---|---|
| Radujkovic et al., 2020 | Prospective cohort | Germany | 185 | 60 | Deficiency: <12 ng/mL | Death |
| Meltzer et al., 2020 | Retrospective cohort | USA | 489 | 49.2 | Deficiency: <20 ng/mL | Infection |
| Kaufman et al., 2020 | Cross-sectional | USA | 188,028 | 54 | Deficiency: <20 ng/mL | Infection |
| Macaya F et al., 2020 | Retrospective cohort | Spain | 80 | 50–84 | Deficiency: <20 ng/mL | Severity |
| Hastie et al., 2020 | Cross-sectional | UK | 1474 | 38–58 | Deficiency: <10 ng/mL | Infection |
| Abrishami et al., 2020 | Retrospective cohort | Iran | 73 | 55.18 | Deficiency: <25 ng/mL | Death |
| Merzon et al., 2020 | Cross-sectional | Israel | 7807 | 46.17 | Deficiency: <30 ng/ml | Infection |
| Baktash et al., 2020 | Prospective cohort study | UK | 105 | 81.29 | Deficiency: ≤12 ng/mL | Infection |
| Maghbooli et al., 2020 | Cross-sectional | Iran | 235 | 58.72 | Deficiency: <30 ng/mL | Severity |
| Ye et al., 2020 | Case-control study | China | 142 | 43 | Deficiency: <20 ng/mL | Severity |
| D’avolio et al., 2020 | Retrospective cohort | Switzerland | 107 | 73 | Not categorized | Infection |
| Author | Main Findings |
|---|---|
| Radujkovic et al., 2020 | Vitamin D deficiency was associated with higher risk of death (HR = 14.73, p < 0.05) |
| Meltzer et al., 2020 | Patients with likely deficient vitamin D status at the time of COVID-19 testing had an increased relative risk of testing positive for COVID-19 (relative risk, 1.77; 95%CI, 1.12–2.81; p < 0.05) compared with patients with likely sufficient status at the time of COVID-19 testing, for an estimated mean rate in the deficient group of 21.6% vs. 12.2% in the sufficient group |
| Kaufman et al., 2020 | The SARS-CoV-2 positivity rate was lower in the 27,870 patients with “adequate” 25(OH)D values (30–34 ng/mL) (8.1%), than in the 39,190 patients with “deficiency” (<20 ng/mL) (12.5%) (difference 35%; p < 0.05). Similarly, the SARS-CoV-2 positivity rate was lower in the 12,321 patients with 25(OH)D values >55 ng/mL (5.9%) than in patients with adequate values (difference 27%; p < 0.05) |
| Macaya F et al., 2020 | After adjusting for age, gender, obesity, and severe CKD, the odds ratio for vitamin D-deficient people to have severe COVID-19 was 3.2 (95% CI: 0.9–11.4), p < 0.05 compared to vitamin D-sufficient people |
| Hastie et al., 2020 | Vitamin D deficiency has significant effect on COVID-19 infection in absence of confounders which is deficient people are more likely to be positive [OR = 1.37, p < 0.05]. |
| Abrishami et al., 2020 |
|
| Merzon et al., 2020 |
|
| Baktash et al., 2020 | Vitamin D levels in the COVID-19-positive group were overall significantly lower compared with that in the COVID-19-negative group (27.00 nmol/L vs. 52.00 nmol/L) (p < 0.05) |
| Maghbooli et al., 2020 |
|
| Ye et al., 2020 |
|
| D’avolio et al., 2020 | Observed statistically significant (p < 0.05) lower 25(OH)D levels (11.1 ng/mL) in patients positive for the SARS-CoV-2 PCR compared with the negative patients (24.6 ng/mL) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Das, P.; Samad, N.; Ahinkorah, B.O.; Hagan, J.E., Jr.; Peprah, P.; Mohammed, A.; Seidu, A.-A. Effect of Vitamin D Deficiency on COVID-19 Status: A Systematic Review. COVID 2021, 1, 97-104. https://0-doi-org.brum.beds.ac.uk/10.3390/covid1010008
Das P, Samad N, Ahinkorah BO, Hagan JE Jr., Peprah P, Mohammed A, Seidu A-A. Effect of Vitamin D Deficiency on COVID-19 Status: A Systematic Review. COVID. 2021; 1(1):97-104. https://0-doi-org.brum.beds.ac.uk/10.3390/covid1010008
Chicago/Turabian StyleDas, Pranta, Nandeeta Samad, Bright Opoku Ahinkorah, John Elvis Hagan, Jr., Prince Peprah, Aliu Mohammed, and Abdul-Aziz Seidu. 2021. "Effect of Vitamin D Deficiency on COVID-19 Status: A Systematic Review" COVID 1, no. 1: 97-104. https://0-doi-org.brum.beds.ac.uk/10.3390/covid1010008







