Journal Description
Photochem
Photochem
is an international, peer-reviewed, open access journal on photochemistry published quarterly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, EBSCO, and other databases.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 23.4 days after submission; acceptance to publication is undertaken in 7.9 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: APC discount vouchers, optional signed peer review, and reviewer names published annually in the journal.
- Photochem is a companion journal of Molecules.
Latest Articles
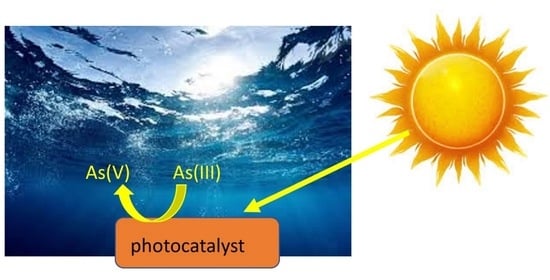
A Review of Visible Light Responsive Photocatalysts for Arsenic Remediation in Water
Photochem 2024, 4(2), 198-218; https://0-doi-org.brum.beds.ac.uk/10.3390/photochem4020012 - 17 Apr 2024
Abstract
This review summarizes the progress over the last fifteen years in visible light reactive photocatalysts for environmental arsenic remediation. The design and performance of several materials including (1) doped and surface functionalized TiO2, (2) binary composites combining TiO2 with another
[...] Read more.
This review summarizes the progress over the last fifteen years in visible light reactive photocatalysts for environmental arsenic remediation. The design and performance of several materials including (1) doped and surface functionalized TiO2, (2) binary composites combining TiO2 with another semiconductor that absorbs visible light radiation or a metal (Pt), (3) ternary composites incorporating TiO2, a conductive polymer that can retard electron-hole recombination and an excellent adsorbent material for the removal of As(V), (4) tungsten, zinc, and bismuth oxides, (5) g-C3N4 based catalysts, and (6) M@AgCl core–shell structures. These results show that long reaction time remains a major challenge in achieving high As(III) oxidation.
Full article
(This article belongs to the Special Issue Exclusive Papers of the Editorial Board Members of the Journal Photochem)
►
Show Figures
Open AccessArticle
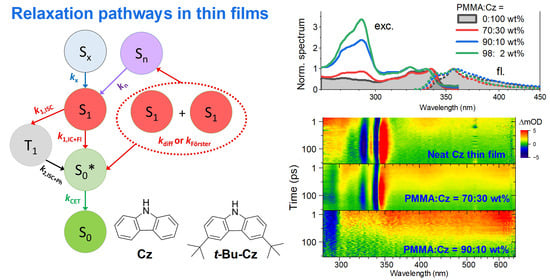
Excited-State Dynamics of Carbazole and tert-Butyl-Carbazole in Thin Films
by
Konstantin Moritz Knötig, Domenic Gust, Kawon Oum and Thomas Lenzer
Photochem 2024, 4(2), 179-197; https://0-doi-org.brum.beds.ac.uk/10.3390/photochem4020011 - 09 Apr 2024
Abstract
Thin films of carbazole (Cz) derivatives are frequently used in organic electronics, such as organic light-emitting diodes (OLEDs). Because of the proximity of the Cz units, the excited-state relaxation in such films is complicated, as intermolecular pathways, such as singlet–singlet annihilation (SSA), kinetically
[...] Read more.
Thin films of carbazole (Cz) derivatives are frequently used in organic electronics, such as organic light-emitting diodes (OLEDs). Because of the proximity of the Cz units, the excited-state relaxation in such films is complicated, as intermolecular pathways, such as singlet–singlet annihilation (SSA), kinetically compete with the emission. Here, we provide an investigation of two benchmark systems employing neat carbazole and 3,6-di-tert-butylcarbazole (t-Bu-Cz) films and also their thin film blends with poly(methyl methacrylate) (PMMA). These are investigated by a combination of atomic force microscopy (AFM), femtosecond and nanosecond transient absorption spectroscopy (fs-TA and ns-TA) and time-resolved fluorescence. Excitonic J-aggregate-type features are observed in the steady-state absorption and emission spectra of the neat films. The S1 state shows a broad excited-state absorption (ESA) spanning the entire UV–Vis–NIR range. At high S1 exciton number densities of about 4 × 1018 cm−3, bimolecular diffusive S1–S1 annihilation is found to be the dominant SSA process in the neat films with a rate constant in the range of 1–2 × 10−8 cm3 s−1. SSA produces highly vibrationally excited molecules in the electronic ground state (S0*), which cool down slowly by heat transfer to the quartz substrate. The results provide relevant photophysical insight for a better microscopic understanding of carbazole relaxation in thin-film environments.
Full article
(This article belongs to the Special Issue Feature Papers in Photochemistry II)
►▼
Show Figures
Graphical abstract
Open AccessArticle
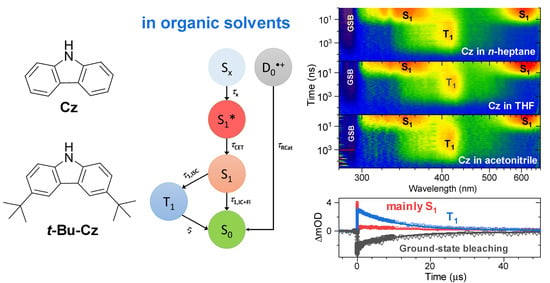
Excited-State Dynamics of Carbazole and tert-Butyl-Carbazole in Organic Solvents
by
Konstantin Moritz Knötig, Domenic Gust, Thomas Lenzer and Kawon Oum
Photochem 2024, 4(2), 163-178; https://0-doi-org.brum.beds.ac.uk/10.3390/photochem4020010 - 30 Mar 2024
Cited by 1
Abstract
Carbazole-based molecular units are ubiquitous in organic optoelectronic materials; however, the excited-state relaxation of these compounds is still underexplored. Here, we provide a detailed investigation of carbazole (Cz) and 3,6-di-tert-butylcarbazole (t-Bu-Cz) in organic solvents using femtosecond and nanosecond UV–Vis–NIR
[...] Read more.
Carbazole-based molecular units are ubiquitous in organic optoelectronic materials; however, the excited-state relaxation of these compounds is still underexplored. Here, we provide a detailed investigation of carbazole (Cz) and 3,6-di-tert-butylcarbazole (t-Bu-Cz) in organic solvents using femtosecond and nanosecond UV–Vis–NIR transient absorption spectroscopy, as well as time-resolved fluorescence experiments upon photoexcitation in the deep-UV range. The initially prepared Sx singlet state has a (sub-)picosecond lifetime and decays to the S1 state by internal conversion (IC). The S1 state exhibits absorption peaks at 350, 600 and 1100 nm and has a lifetime of 13–15 ns, which is weakly dependent on the solvent. Energy transfer from vibrationally hot S1 molecules (S1*) to the surrounding solvent molecules takes place with a time constant of 8–20 ps. The T1 triplet state is populated by intersystem crossing (ISC) from S1 with a typical quantum yield of 51–56% and shows a lifetime which is typically in the few microseconds regime. The S1 and T1 states of both carbazole compounds in solution are strongly quenched by O2. Two-photon excitation leads to the formation of a small amount of the respective radical cation. The influence of the tert-butyl substituents on the photophysics is relatively weak and mainly reflects itself in a small increase in the Stokes shift. The results provide important photophysical information for the interpretation of carbazole relaxation in more complex environments.
Full article
(This article belongs to the Special Issue Feature Papers in Photochemistry II)
►▼
Show Figures
Graphical abstract
Open AccessArticle
Charge-Selective Photocatalytic Degradation of Organic Dyes Driven by Naturally Occurring Halloysite Nanotubes
by
Ashim Pramanik, Martina Maria Calvino, Luisa Sciortino, Pooria Pasbakhsh, Giuseppe Cavallaro, Giuseppe Lazzara, Fabrizio Messina and Alice Sciortino
Photochem 2024, 4(2), 151-162; https://0-doi-org.brum.beds.ac.uk/10.3390/photochem4020009 - 28 Mar 2024
Abstract
►▼
Show Figures
This study explores the use of Halloysite NanoTubes (HNTs) as photocatalysts capable of decomposing organic dyes under exposure to visible or ultraviolet light. Through a systematic series of photocatalytic experiments, we unveil that the photodegradation of Rhodamine B, used as a model cationic
[...] Read more.
This study explores the use of Halloysite NanoTubes (HNTs) as photocatalysts capable of decomposing organic dyes under exposure to visible or ultraviolet light. Through a systematic series of photocatalytic experiments, we unveil that the photodegradation of Rhodamine B, used as a model cationic dye, is significantly accelerated in the presence of HNTs. We observe that the extent of RhB photocatalytic degradation in 100 min in the presence of the HNTs is ~four times higher compared to that of bare RhB. Moreover, under optimized conditions, the as-extracted photodegradation rate of RhB (~0.0022 min−1) is comparable to that of the previously reported work on the photodegradation of RhB in the presence of tubular nanostructures. A parallel effect is observed for anionic Coumarin photodegradation, albeit less efficiently. Our analysis attributes this discrepancy to the distinct charge states of the two dyes, influencing their attachment sites on HNTs. Cationic Rhodamine B molecules preferentially attach to the outer surface of HNTs, while anionic Coumarin molecules tend to attach to the inner surface. By leveraging the unique properties of HNTs, a family of naturally occurring nanotube structures, this research offers valuable insights for optimizing photocatalytic systems in the pursuit of effective and eco-friendly solutions for environmental remediation.
Full article
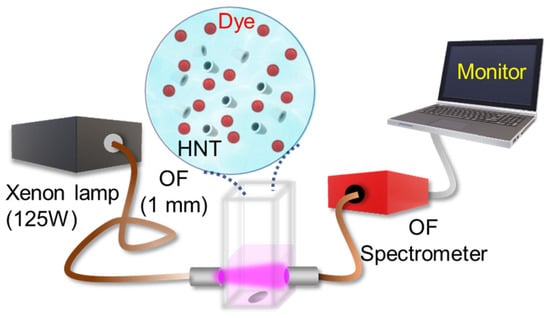
Figure 1
Open AccessArticle
Torsional Disorder in Tetraphenyl [3]-Cumulenes: Insight into Excited State Quenching
by
David Bain, Julia Chang, Yihuan Lai, Thomas Khazanov, Phillip J. Milner and Andrew J. Musser
Photochem 2024, 4(1), 138-150; https://0-doi-org.brum.beds.ac.uk/10.3390/photochem4010008 - 09 Feb 2024
Abstract
►▼
Show Figures
Cumulenes are linear molecules consisting of consecutive double bonds linking chains of sp-hybridized carbon atoms. They have primarily been of interest for potential use as molecular wires or in other nanoscale electronic devices, but more recently, other applications such as catalysis or even
[...] Read more.
Cumulenes are linear molecules consisting of consecutive double bonds linking chains of sp-hybridized carbon atoms. They have primarily been of interest for potential use as molecular wires or in other nanoscale electronic devices, but more recently, other applications such as catalysis or even light harvesting through singlet fission have been speculated. Despite the recent theoretical and experimental interest, the photoexcitation of cumulenes typically results in quenching on the picosecond timescale, and the exact quenching mechanism for even the simplest of [3]-cumulenes lacks a clear explanation. In this report, we perform transient absorption spectroscopy on a set of model [3]-cumulene derivatives in a wide range of environmental conditions to demonstrate that the planarization of phenyl groups ultimately quenches the excited state. By restricting this intramolecular motion, we increase the excited state lifetime by a few nanoseconds, strongly enhancing photoluminescence and demonstrating an approach to stabilize them for photochemical applications.
Full article

Figure 1
Open AccessArticle
Evaluation of MAA Analogues as Potential Candidates to Increase Photostability in Sunscreen Formulations
by
Jacobo Soilán, Leonardo López-Cóndor, Beatriz Peñín, José Aguilera, María Victoria de Gálvez, Diego Sampedro and Raúl Losantos
Photochem 2024, 4(1), 128-137; https://0-doi-org.brum.beds.ac.uk/10.3390/photochem4010007 - 06 Feb 2024
Abstract
Avobenzone is one of the most widely used sunscreens in skin care formulations, but suffers from some drawbacks, including photo instability. To mitigate this critical issue, the use of octocrylene as a stabilizer is a common approach in these products. However, octocrylene has
[...] Read more.
Avobenzone is one of the most widely used sunscreens in skin care formulations, but suffers from some drawbacks, including photo instability. To mitigate this critical issue, the use of octocrylene as a stabilizer is a common approach in these products. However, octocrylene has been recently demonstrated to show potential phototoxicity. The aim of this work is to analyze the performance of a series of mycosporine-like amino acid (MAA)-inspired compounds to act as avobenzone stabilizers as an alternative to octocrylene. Different avobenzone/MAA analogue combinations included in galenic formulations were followed under increasing doses of solar-simulated UV radiation. Some of the synthetic MAA analogues analyzed were able to increase by up to two times the UV dose required for 50% of avobenzone photobleaching. We propose some of these MAA analogues as new candidates to act as avobenzone-stabilizing compounds in addition to their UV absorbance and antioxidant properties, together with a facile synthesis.
Full article
(This article belongs to the Special Issue Feature Papers in Photochemistry II)
►▼
Show Figures
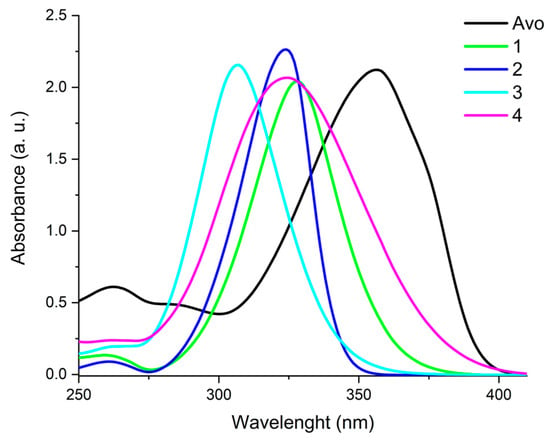
Figure 1
Open AccessArticle
Physicochemical Properties of Tungsten Trioxide Photoanodes Fabricated by Wet Coating of Soluble, Particulate, and Mixed Precursors
by
Valli Kamala Laxmi Ramya Chittoory, Marketa Filipsika, Radim Bartoš, Marcela Králová and Petr Dzik
Photochem 2024, 4(1), 111-127; https://0-doi-org.brum.beds.ac.uk/10.3390/photochem4010006 - 01 Feb 2024
Abstract
Advanced oxidation processes are emerging technologies for the decomposition of organic pollutants in various types of water by harnessing solar energy. The purpose of this study is to examine the physicochemical characteristics of tungsten(VI) oxide (WO3) photoanodes, with the aim of
[...] Read more.
Advanced oxidation processes are emerging technologies for the decomposition of organic pollutants in various types of water by harnessing solar energy. The purpose of this study is to examine the physicochemical characteristics of tungsten(VI) oxide (WO3) photoanodes, with the aim of enhancing oxidation processes in the treatment of water. The fabrication of WO3 coatings on conductive fluorine-doped tin oxide (FTO) substrates was achieved through a wet coating process that utilized three different liquid formulations: a dispersion of finely milled WO3 particles, a fully soluble WO3 precursor (acetylated peroxo tungstic acid), and a combination of both (applying a brick-and-mortar strategy). Upon subjecting the WO3 coatings to firing at a temperature of 450 °C, it was observed that their properties exhibited marked variations. The fabricated photoanodes are examined using a range of analytical techniques, including profilometry, thermo-gravimetric analysis (TGA), X-ray diffraction (XRD), and voltammetry. The experimental data suggest that the layers generated through the combination of particulate ink and soluble precursor (referred to as the brick-and-mortar building approach) display advantageous physicochemical properties, rendering them suitable for use as photoanodes in photoelectrochemical cells.
Full article
(This article belongs to the Topic New Materials and Advanced Applications in Photocatalysis)
►▼
Show Figures
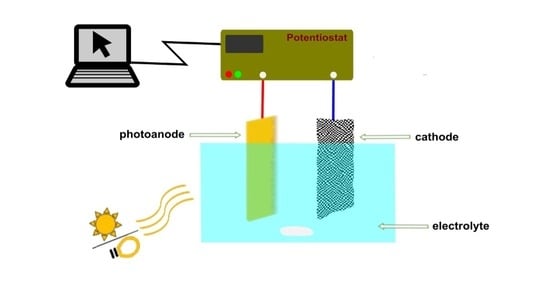
Graphical abstract
Open AccessArticle
Inverse Problems in Pump–Probe Spectroscopy
by
Denis S. Tikhonov, Diksha Garg and Melanie Schnell
Photochem 2024, 4(1), 57-110; https://0-doi-org.brum.beds.ac.uk/10.3390/photochem4010005 - 31 Jan 2024
Abstract
Ultrafast pump–probe spectroscopic studies allow for deep insights into the mechanisms and timescales of photophysical and photochemical processes. Extracting valuable information from these studies, such as reactive intermediates’ lifetimes and coherent oscillation frequencies, is an example of the inverse problems of chemical kinetics.
[...] Read more.
Ultrafast pump–probe spectroscopic studies allow for deep insights into the mechanisms and timescales of photophysical and photochemical processes. Extracting valuable information from these studies, such as reactive intermediates’ lifetimes and coherent oscillation frequencies, is an example of the inverse problems of chemical kinetics. This article describes a consistent approach for solving this inverse problem that avoids the common obstacles of simple least-squares fitting that can lead to unreliable results. The presented approach is based on the regularized Markov Chain Monte-Carlo sampling for the strongly nonlinear parameters, allowing for a straightforward solution of the ill-posed nonlinear inverse problem. The software to implement the described fitting routine is introduced and the numerical examples of its application are given. We will also touch on critical experimental parameters, such as the temporal overlap of pulses and cross-correlation time and their connection to the minimal reachable time resolution.
Full article
(This article belongs to the Special Issue Feature Papers in Photochemistry II)
►▼
Show Figures

Figure 1
Open AccessArticle
The Rhodamine–Perylene Compact Electron Donor–Acceptor Dyad: Spin-Orbit Charge-Transfer Intersystem Crossing and the Energy Balance of the Triplet Excited States
by
Muhammad Imran, Dongyi Liu, Kaiyue Ye, Xue Zhang and Jianzhang Zhao
Photochem 2024, 4(1), 40-56; https://0-doi-org.brum.beds.ac.uk/10.3390/photochem4010004 - 29 Jan 2024
Abstract
We prepared a rhodamine (RB)–perylene (Pery) compact electron donor/acceptor dyad (RB–Pery) to study the spin-orbit charge-transfer intersystem crossing (SOCT–ISC). The UV–vis absorption spectrum indicates a negligible electronic interaction between the donor and acceptor at ground state. However, the fluorescence of both
[...] Read more.
We prepared a rhodamine (RB)–perylene (Pery) compact electron donor/acceptor dyad (RB–Pery) to study the spin-orbit charge-transfer intersystem crossing (SOCT–ISC). The UV–vis absorption spectrum indicates a negligible electronic interaction between the donor and acceptor at ground state. However, the fluorescence of both the RB and Pery units are quenched in the dyad, which is attributed to the photoinduced electron transfer, supported by the electrochemical studies. Nanosecond transient absorption (ns-TA) spectra show delocalized triplet states, i.e., there is an excited-state equilibrium between Pery and the RB triplet states. The triplet state lifetime was determined as 109.8 μs. With intermolecular triplet–triplet energy transfer, monitored using ns-TA spectra, the triplet-state energy balance between RB and Pery in RB–Pery was confirmed. The proposed cascade photophysical processes of the dyad are 1RB*-Pery→RB–Pery+•→[3RB*-Pery↔RB-3Pery*]. Moreover, long-lived rhodamine radical cation (in milliseconds) was detected in both deaerated/aerated non-polar or low-polarity solvents (i.e., p-xylene, toluene). The potential energy curve of the dyad against the variation in the dihedral angle between the two units indicates large torsional freedom (53°~128°) in RB–Pery, which leads to inefficient SOCT–ISC; consequently, low singlet-oxygen quantum yields (ΦΔ = 2~8%) were observed.
Full article
(This article belongs to the Special Issue Exclusive Papers of the Editorial Board Members of the Journal Photochem)
►▼
Show Figures
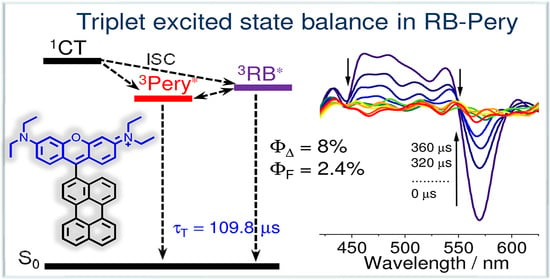
Graphical abstract
Open AccessArticle
Interactions of CdSe Nanocrystals with Cationic Proteins Extracted from Moringa oleifera Seeds
by
Likius Shipwiisho Daniel, Salatiel Kapofi, Martha Kandawa-Schulz and Habauka Majority Kwaambwa
Photochem 2024, 4(1), 24-39; https://0-doi-org.brum.beds.ac.uk/10.3390/photochem4010003 - 15 Jan 2024
Abstract
►▼
Show Figures
Even with significant developments in nanoscience, relatively little is known about the interactions of nanocrystal semiconducting materials with bio-macromolecules. To investigate the interfacial phenomena of cadmium selenide quantum dot (CdSe QD) nanocrystals with proteins extracted from Moringa oleifera seeds, different concentrations of cadmium
[...] Read more.
Even with significant developments in nanoscience, relatively little is known about the interactions of nanocrystal semiconducting materials with bio-macromolecules. To investigate the interfacial phenomena of cadmium selenide quantum dot (CdSe QD) nanocrystals with proteins extracted from Moringa oleifera seeds, different concentrations of cadmium selenide quantum dots–Moringa oleifera seed protein (CdSe–MSP) complexes were prepared. Respective CdSe QDs with hexagonal phase and crystalline size in the range of 4–7 nm were synthesized and labelled with the purified mesoporous MSP having a surface area of 8.4 m2/g. The interaction mechanism between CdSe QDs and MSP was studied using UV–Vis absorption, fluorescence emission and Fourier Transform Infrared spectroscopies. The UV–Vis absorption spectra showed absorption bands of CdSe–MSP complexes at 546.5 nm. The fluorescence intensity of CdSe QDs was found to decrease with increasing concentration of MSP. The thermodynamic potentials

Figure 1
Open AccessArticle
Survey of T1 and T2 Energies of Intramolecular Singlet Fission Chromophores
by
Guoying Yao, Zhenyu Yang and Tao Zeng
Photochem 2024, 4(1), 14-23; https://0-doi-org.brum.beds.ac.uk/10.3390/photochem4010002 - 10 Jan 2024
Abstract
►▼
Show Figures
Singlet fission is a desired process in photovoltaics since it enhances photoelectric conversion efficiency. Intramolecular singlet fission is of special interest as the fission efficiency can be improved through tuning configurations between chromophore units that are covalently connected. However, intramolecular singlet fission chromophores
[...] Read more.
Singlet fission is a desired process in photovoltaics since it enhances photoelectric conversion efficiency. Intramolecular singlet fission is of special interest as the fission efficiency can be improved through tuning configurations between chromophore units that are covalently connected. However, intramolecular singlet fission chromophores feature a large tetraradical character, and may tend to dissatisfy the
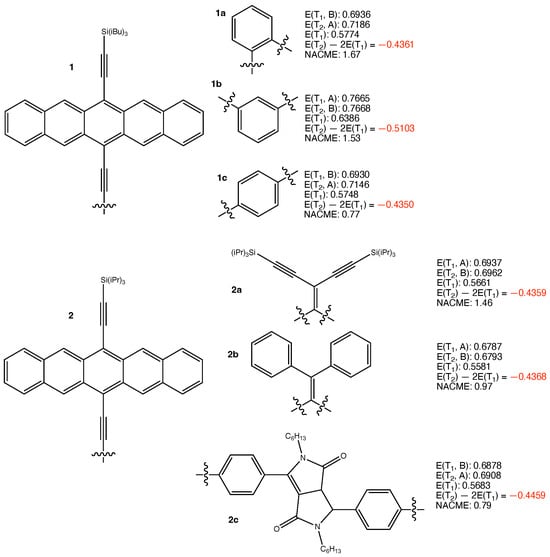
Figure 1
Open AccessArticle
Twisted 8-Acyl-1-dialkyl-amino-naphthalenes Emit from a Planar Intramolecular Charge Transfer Excited State
by
Christopher Abelt and Kirsten Sweigart
Photochem 2024, 4(1), 1-13; https://0-doi-org.brum.beds.ac.uk/10.3390/photochem4010001 - 04 Jan 2024
Abstract
►▼
Show Figures
Fluorescence from dialkylamino donor–acyl acceptor substituted 1,8-naphthalene derivatives can occur either from a planar (PICT) or a twisted (TICT) intramolecular charge transfer excited state. The photophysical properties of 8-acetyl-1-(dimethyl-amino)naphthalene (3) and 8-pivaloyl-1-(dimethyl-amino)naphthalene (4) are compared with 1-methyl-2,3-dihydronaphtho[1,8-bc]azepin-4(1H)-one (5
[...] Read more.
Fluorescence from dialkylamino donor–acyl acceptor substituted 1,8-naphthalene derivatives can occur either from a planar (PICT) or a twisted (TICT) intramolecular charge transfer excited state. The photophysical properties of 8-acetyl-1-(dimethyl-amino)naphthalene (3) and 8-pivaloyl-1-(dimethyl-amino)naphthalene (4) are compared with 1-methyl-2,3-dihydronaphtho[1,8-bc]azepin-4(1H)-one (5). In 3 and 4, both the carbonyl and amino groups are forced to twist out of the plane of the naphthalene ring. In 5, these groups are nearly coplanar with the naphthalene. Neither 3 nor 4 fluoresce as strongly as 5, but all three show similar degrees of solvato-chromism and all are strongly quenched by alcohol solvents. Nitrile 6, 8-cyano-1-(dimethyl-amino)naphthalene, does not show the same degree of solvato-chromism as 3–5, nor is it as affected by alcohols. Calculations corroborate the experimental results, indicating that 3–5 emit from a PICT excited state.
Full article
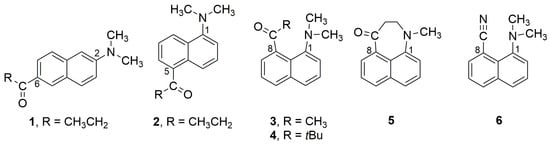
Figure 1
Open AccessReview
Recent Advances in Visible Light Photoinitiating Systems Based on Flavonoids
by
Frédéric Dumur
Photochem 2023, 3(4), 495-529; https://0-doi-org.brum.beds.ac.uk/10.3390/photochem3040030 - 12 Dec 2023
Abstract
►▼
Show Figures
The design of biosourced and/or bioinspired photoinitiators is an active research field as it offers a unique opportunity to develop photoinitiating systems exhibiting better biocompatibility as well as reduced toxicity. In this field, flavonoids can be found in numerous fruits and vegetables so
[...] Read more.
The design of biosourced and/or bioinspired photoinitiators is an active research field as it offers a unique opportunity to develop photoinitiating systems exhibiting better biocompatibility as well as reduced toxicity. In this field, flavonoids can be found in numerous fruits and vegetables so these structures can be of interest for developing, in the future, polymerization processes, offering a reduced environmental impact but also better biocompatibility of the polymers. In this review, the different flavonoids reported to date as photoinitiators of polymerization are presented. Over the years, different modifications of the flavonoid scaffold have been examined including the grafting of well-known chromophores, the preparation of Type II photoinitiators or the introduction of photocleavable groups enabling the generation of Type I photoinitiators. Different families of flavonoids have also been investigated, enabling to design of high-performance photoinitiating systems.
Full article

Graphical abstract
Open AccessArticle
Photoprotective Steering of Previtamin D3 Photochemistry by Phenylalanine in Solution
by
Gordon A. Ochsner, Jaren S. Meikle and Jacob C. Dean
Photochem 2023, 3(4), 477-494; https://0-doi-org.brum.beds.ac.uk/10.3390/photochem3040029 - 28 Nov 2023
Abstract
►▼
Show Figures
Provitamin D3 is the biological precursor to naturally formed vitamin D3 in humans, and its conversion is initiated via photoexcitation by near ultraviolet light. Following an initial photolysis, the primary intermediate, known as previtamin D3, is prone to light-induced
[...] Read more.
Provitamin D3 is the biological precursor to naturally formed vitamin D3 in humans, and its conversion is initiated via photoexcitation by near ultraviolet light. Following an initial photolysis, the primary intermediate, known as previtamin D3, is prone to light-induced isomerization or recyclization, which creates byproducts that limit the desired final thermal conversion to vitamin D3. The branching of the photochemical reaction is highly wavelength-dependent, whereby excitation toward the blue edge of the provitamin D3 absorption spectrum tends to terminate the reaction with the most undesired tachysterol byproduct and the lowest previtamin D3 concentration. In this work, the influence of introducing the natural amino acid phenyalanine as an excitation energy donor to the photochemical reaction is investigated. We find that the incorporation of phenylalanine into provitamin D3 solution results in greater intermediate concentrations and prolonged lifetimes of the desired previtamin D3 while simultaneously reducing the final concentration of tachysterol when exposing the solution to wavelengths at the blue edge of the provitamin D3 spectrum. The results, coupled with quantum chemical analysis, suggest that phenylalanine indeed helps to funnel energy from shorter wavelengths more effectively into the provitamin D3 precursor, while simultaneously screening those wavelengths from direct excitation, which otherwise leads to enhanced concentrations of tachysterol byproduct at the expense of previtamin D3.
Full article
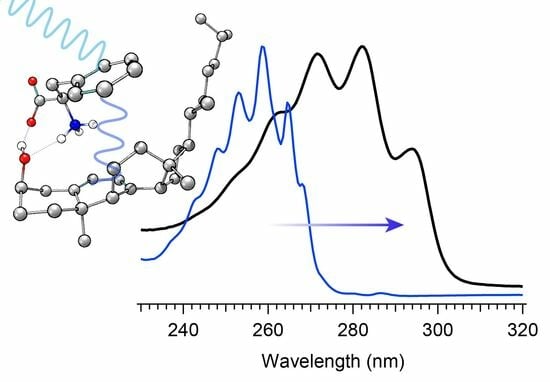
Graphical abstract
Open AccessArticle
Investigation of the Self-Cleaning Property of Photocatalytic Coatings at a Laboratory Scale
by
Julie Hot, Kevin Castelló Lux and Erick Ringot
Photochem 2023, 3(4), 461-476; https://0-doi-org.brum.beds.ac.uk/10.3390/photochem3040028 - 25 Nov 2023
Abstract
Self-cleaning products are commercially available to protect surfaces against soiling and avoid the high consumption of energy and chemical detergents necessary for cleaning. They are based on semiconductor oxides, mostly titanium dioxide (TiO2), which induce photocatalytic oxidation activity and superhydrophilicity. Therefore,
[...] Read more.
Self-cleaning products are commercially available to protect surfaces against soiling and avoid the high consumption of energy and chemical detergents necessary for cleaning. They are based on semiconductor oxides, mostly titanium dioxide (TiO2), which induce photocatalytic oxidation activity and superhydrophilicity. Therefore, we present an experimental procedure at a lab scale to assess the self-cleaning ability of various photocatalytic coatings (five TiO2-based commercial products and one lab-grade zinc oxide (ZnO) product) applied to mortar surfaces. The samples were artificially stained with three types of soiling: Congo red dye, diesel soot, and motor oil. They were exposed to the environmental cycle of UV illumination and water flow for two weeks and the changes in stain colors were first assessed with visual inspection. Then, spectrophotometry measurements were conducted before and after the self-cleaning experiment to calculate the color differences for each stain in the CIELab color space data. In addition, the coatings were characterized via X-ray diffraction analyses and water contact angle measurements. Results highlighted color changes for each stain and higher wettability (induced by OH radicals) of the coated surfaces, which favored surface washing and thus stain removal. Light also had a positive effect on the attenuation of the stains, particularly for the Congo red dye.
Full article
(This article belongs to the Topic New Materials and Advanced Applications in Photocatalysis)
►▼
Show Figures

Figure 1
Open AccessArticle
Photolytic Degradation of the Insecticide Clothianidin in Hydrochar Aquatic Suspensions and Extracts
by
Artemis Pappa, Feidias Bairamis and Ioannis Konstantinou
Photochem 2023, 3(4), 442-460; https://0-doi-org.brum.beds.ac.uk/10.3390/photochem3040027 - 07 Nov 2023
Abstract
In this study, the aqueous photolytic degradation of the neonicotinoid pesticide clothianidin was studied in suspensions and aqueous extracts of hydrochar produced from olive kernels. A slight and nonsignificant decrease in the photodegradation rate of clothianidin in aqueous extracts of hydrochar (HCw) with
[...] Read more.
In this study, the aqueous photolytic degradation of the neonicotinoid pesticide clothianidin was studied in suspensions and aqueous extracts of hydrochar produced from olive kernels. A slight and nonsignificant decrease in the photodegradation rate of clothianidin in aqueous extracts of hydrochar (HCw) with an initial concentration of hydrochar ranged from 50 to 400 mg L−1 (rate constants ranged between k = 0.0034 and 0.0039 min−1) was observed in comparison to the respective rate in the bi-distilled water (k = 0.0040 min−1). On the contrary, in the presence of hydrochar suspensions (HCp), a significant decrease was observed for 50 mg L−1 hydrochar particle concentration (k = 0.020 min−1), while for higher concentrations (100 to 400 mg L−1), rate constants increased but with nonsignificant differences compared with the kinetics followed in the absence of them. Generally, the photodegradation rate of clothianidin, in the presence of HCw and HCp, is reduced compared to the photodegradation rate in bi-distilled aqueous solutions, except in the case of the aqueous suspension with an HCp concentration of 200 mg L−1. The transformation products (TPs) of clothianidin formed in the photolytic degradation processes were identified using ultrahigh-performance liquid chromatography coupled with accurate high-resolution mass spectrometry technique (UHPLC-LTQ-ORBITRAP). The formation profiles of TPs varied according to the matrix showing different degrees of participation of direct and indirect (photosensitized) phototransformation pathways. Photolytic degradation of clothianidin takes place mainly through denitration, hydroxylation and dechlorination pathways. Finally, the toxicity of the identified TPs was studied using the Vibrio fischeri bioassay. Toxicity was slightly reduced after 300 min of irradiation while maximum value was observed after 180–240 min of irradiation showing the formation of more toxic TPs along the photochemical degradation.
Full article
(This article belongs to the Special Issue Exclusive Papers of the Editorial Board Members of the Journal Photochem)
►▼
Show Figures
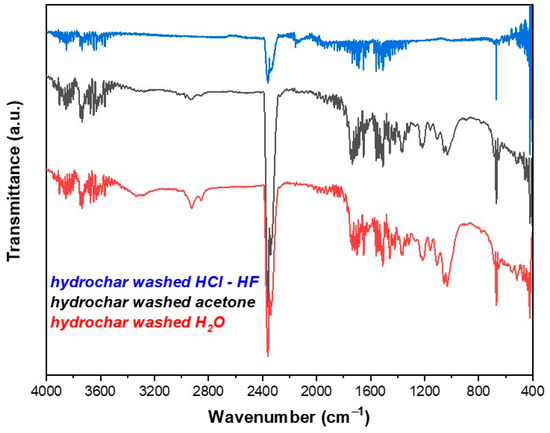
Figure 1
Open AccessArticle
Metal Ion-Induced Formation of Metallogels by Visible-Light-Responsive Phenylalanine-Functionalized Arylazopyrazole Ligands
by
Mikayla Browning, Alexandra Jefferson, Jazz Geter and Kesete Ghebreyessus
Photochem 2023, 3(4), 427-441; https://0-doi-org.brum.beds.ac.uk/10.3390/photochem3040026 - 24 Oct 2023
Cited by 1
Abstract
►▼
Show Figures
A visible-light-responsive arylazopyrazole-functionalized phenylalanine (4-MeS-AAP-NF) derived ligand was designed and synthesized, and it was found to form metallogels with reversible photo-responsive properties in mixed methanol/water (MeOH/H2O) solvents. The gelation behavior of the 4-MeS-AAP-NF ligand in the presence of different divalent metal
[...] Read more.
A visible-light-responsive arylazopyrazole-functionalized phenylalanine (4-MeS-AAP-NF) derived ligand was designed and synthesized, and it was found to form metallogels with reversible photo-responsive properties in mixed methanol/water (MeOH/H2O) solvents. The gelation behavior of the 4-MeS-AAP-NF ligand in the presence of different divalent metal ions in mixed methanol/water (MeOH/H2O) solvents at pH~11.60 was studied. It was found that the 4-MeS-AAP-NF ligand alone could not self-assemble to form any gels. However, in the presence of divalent metal ions, it readily formed the assembled metallogels in an alkaline aqueous/methanol solution with various morphologies. The results suggest that the gelation process was triggered by divalent metal ions. The presence of the AAP moiety in the gel matrix rendered the metallogel assemblies photo-responsive, and the reversible gel-to-sol phase transition was studied by UV-vis spectroscopy. The gels showed a slow, reversible visible-light-induced gel-to-sol phase transition under blue (λ = 405 nm) and then sol-to-gel transition by green light (λ = 530 nm) irradiation, resulting in the re-formation of the original gel state. The morphology and viscoelastic properties of the yellow–orange opaque metallogels were characterized by scanning electron microscopy (SEM) and rheological measurement, respectively.
Full article

Figure 1
Open AccessArticle
Photochemistry of β-γ-Unsaturated Spirolactones
by
Werner Fudickar, Melanie Metz, Tobias Krüger-Braunert, Alexandra Kelling, Eric Sperlich, Pablo Wessig and Torsten Linker
Photochem 2023, 3(4), 408-426; https://0-doi-org.brum.beds.ac.uk/10.3390/photochem3040025 - 17 Oct 2023
Abstract
►▼
Show Figures
β-γ-unsaturated spirolactones are easily available by Birch reduction. We describe their photochemistry in the presence of or without carbonyl compounds. The spirolactones show a distinct absorption band at 230 nm, which is not present in other cyclohexadienes. We explain this behavior by an
[...] Read more.
β-γ-unsaturated spirolactones are easily available by Birch reduction. We describe their photochemistry in the presence of or without carbonyl compounds. The spirolactones show a distinct absorption band at 230 nm, which is not present in other cyclohexadienes. We explain this behavior by an interaction of the double bonds with the carbonyl group through space, further proven by TDDFT calculations. This allows their direct excitation with UV-C light. Interestingly, we obtain only products of an oxa-di-π-methane rearrangement, hitherto unknown for lactones. This speaks for a reaction pathway starting from singlet states, confirmed by calculated relative energies of biradical intermediates. Although polymerization is the main side reaction, we were able to isolate tricyclic lactones in moderate yields in a pure form. In the presence of benzaldehyde or benzophenone, excitation with UV-B light was possible, leading to H-atom abstraction in the allylic position and formation of alcohols. With an electron-rich double bond, the Paternó–Büchi products were isolated as well. The different diastereomers were separated by column chromatography or HPLC. Their relative configurations were determined using NOESY measurements or X-ray structure analysis. Overall, β-γ-unsaturated spirolactones show a remarkably different photochemistry compared to other cyclohexadienes, affording new products in only a few steps.
Full article
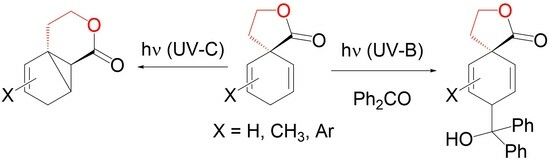
Graphical abstract
Open AccessArticle
Photocatalytic Degradation of Methylene Blue by Magnetic Opal/Fe3O4 Colloidal Crystals under Visible Light Irradiation
by
Abraham Jorge Carmona-Carmona, Enrique Sánchez Mora, Jesús Ivan Peña Flores, César Márquez-Beltrán, María Dolores Castañeda-Antonio, Marlén Alexis González-Reyna, María Concepción Barrera, Khashayar Misaghian, Jesús Eduardo Lugo and Miller Toledo-Solano
Photochem 2023, 3(4), 390-407; https://0-doi-org.brum.beds.ac.uk/10.3390/photochem3040024 - 08 Oct 2023
Cited by 1
Abstract
►▼
Show Figures
In this study, opal–magnetite photocatalysts based on SiO2 artificial opal crystals infiltrated with different concentrations of Fe3O4 nanoparticles (NPs) were synthesized using a combination of lateral infiltration and co-assembly methods. By adjusting the concentration of Fe3O4
[...] Read more.
In this study, opal–magnetite photocatalysts based on SiO2 artificial opal crystals infiltrated with different concentrations of Fe3O4 nanoparticles (NPs) were synthesized using a combination of lateral infiltration and co-assembly methods. By adjusting the concentration of Fe3O4 NPs in the SiO2 opal crystal, the energy band gap (Eg) was tuned to enable efficient degradation of methylene blue (MB) under visible light (410 nm and 575 nm). The photocatalytic process involved two stages: MB adsorption on the surface due to charge differences in the composite film and subsequent degradation through oxidative radicals on the catalyst’s surface. The developed material exhibited potential for applications in water remediation.
Full article
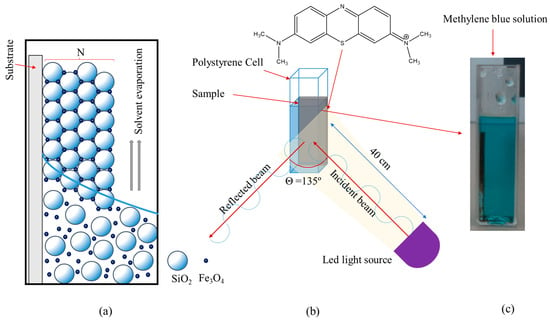
Figure 1
Open AccessCommunication
5-Chlorocoumaranone-Conjugates as Chemiluminescent Protecting Groups (CLPG) and Precursors to Fluorescent Protecting Groups (FPG)
by
Tim Lippold, Axel G. Griesbeck, Robert Herzhoff, Mathias S. Wickleder, Laura Straub and Niko T. Flosbach
Photochem 2023, 3(3), 373-389; https://0-doi-org.brum.beds.ac.uk/10.3390/photochem3030023 - 07 Sep 2023
Abstract
►▼
Show Figures
The introduction and cleavage of protecting groups and the subsequent release of protected molecules is an important tool in synthetic organic chemistry. When polyfunctionalized substrates are involved, the reactivity of similar functional groups must be differentiated and selectively switched on and off. A
[...] Read more.
The introduction and cleavage of protecting groups and the subsequent release of protected molecules is an important tool in synthetic organic chemistry. When polyfunctionalized substrates are involved, the reactivity of similar functional groups must be differentiated and selectively switched on and off. A very useful switching agent is visible or UV-light in photoremovable protecting groups (PRPG), allowing the PG release upon interaction with electromagnetic radiation. The reverse principle is the release of a protected molecule, which is accompanied by emission of light, i.e., chemiluminescent protecting groups (CLPG). This principle is proposed and investigated for phenylalanine (using ureido carboxylic acid 2 and its methyl ester derivative 3) and the carbamate derivatives of paracetamol 4 and L-menthol 7, protected as the corresponding urea-/carbamate-coumaranones 5A, 5E, 6 and 8, respectively. While the carbamate derivative 6 released the protected substrate with a short and strong emission of blue light when treated with a base under atmospheric oxygen, 8 had to be treated additionally with potassium hydroxide in boiling ethanol to completely cleave the PG. Both urea-coumaranone derivatives 5A/5E had a flash-like CL without release of the protected amino acid and, thus, were converted into a fluorescent protecting group (FPG).
Full article
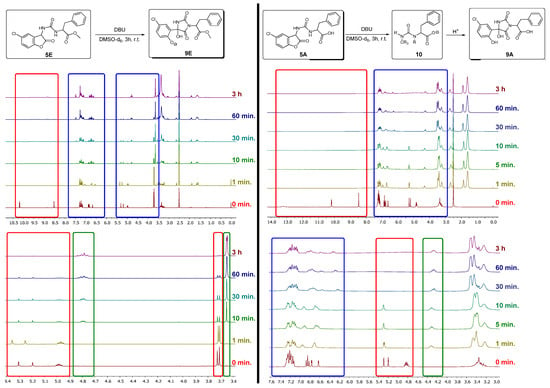
Figure 1
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics

Conferences
Special Issues
Special Issue in
Photochem
Advanced Topics in Photodynamic Therapy (PDT) and Photo Diagnosis (PD)
Guest Editor: Juliana Ferreira-StrixinoDeadline: 31 May 2024
Special Issue in
Photochem
Fluorescing Dyes and Fluorescing Probes
Guest Editor: Anna Cleta CroceDeadline: 31 October 2024
Special Issue in
Photochem
Exclusive Papers of the Editorial Board Members of the Journal Photochem
Guest Editor: Dirk M. GuldiDeadline: 31 December 2024