Behavioral and Physiological Plasticity Provides Insights into Molecular Based Adaptation Mechanism to Strain Shift in Spodoptera frugiperda
Abstract
:1. Introduction
2. Materials and Methods
2.1. Insect Collection and Rearing
2.2. Rearing and Host Plant Selection
2.3. Larval Feeding Choice in Y-Tube Olfactometers
2.4. Larval Feeding Choice Bioassays on Whole Plants in Cages
2.5. Female Adult Host Preference Experiment in Y-Tube Olfactometer
2.6. Female Oviposition Choice Experiments of Fall Armyworm in Cages
2.7. Larval Performance Experiments
2.8. Sample’s Preparation, RNA Extraction, Library Preparation and Sequencing
2.9. RNA Quantification and Qualification RNA Integrity
2.10. Library Preparation for Transcriptome Sequencing
2.11. Reads Mapping to the Reference Genome
2.12. Novel Transcripts Prediction
2.13. Quantification of Gene Expression Level
2.14. Differential Expression Analysis
2.15. GO and KEGG Enrichment Analysis of Differentially Expressed Genes
2.16. Weighted Correlation Network Analysis
2.17. Validation of Transcriptome Data Using RTq-PCR and Data Analysis
2.18. Statistical Analysis
3. Results
3.1. Feeding Choice of Fall Armyworm on Preferred and Alternate Host Plants in Y-Tube Olfactometer
3.2. Feeding Choice of Fall Armyworm on Preferred and Alternate Host Plants in Plastic Cages
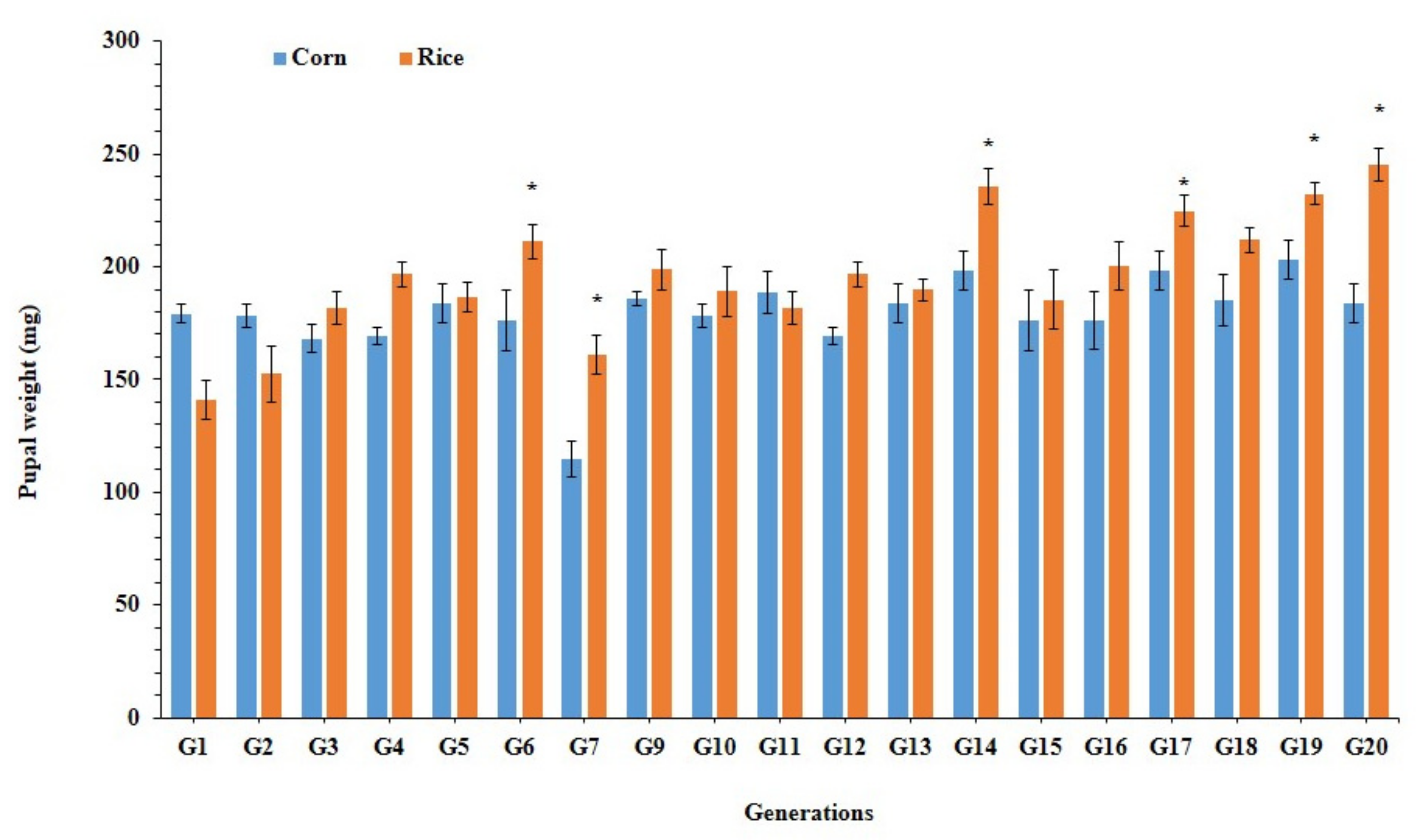
3.3. Larval Performance of Fall Armyworm on Preferred and Alternate Host Plants
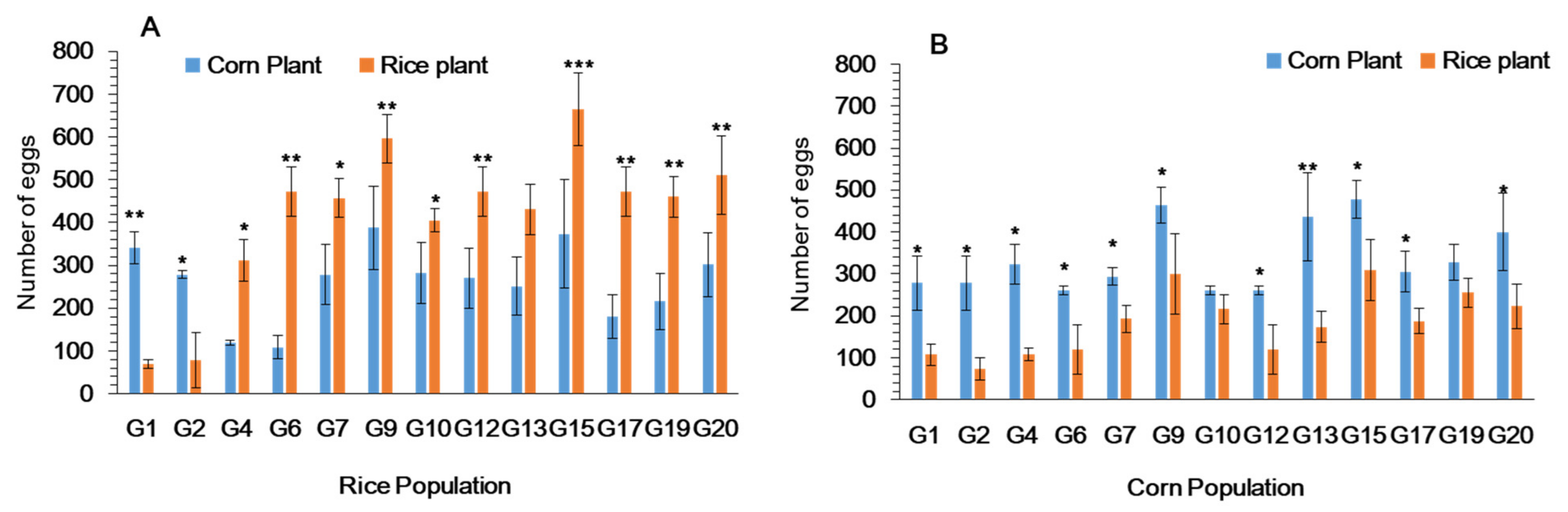
3.4. Female Oviposition Choice of Fall Armyworm in Cages
3.5. Host Plants Preference of Female for Oviposition Choice in Y-Tube Olfactometer
3.6. RNA Sequencing and de novo Assembly
3.7. Differential Expressed Genes (DEGs) When Reared on Rice and Corn Plants
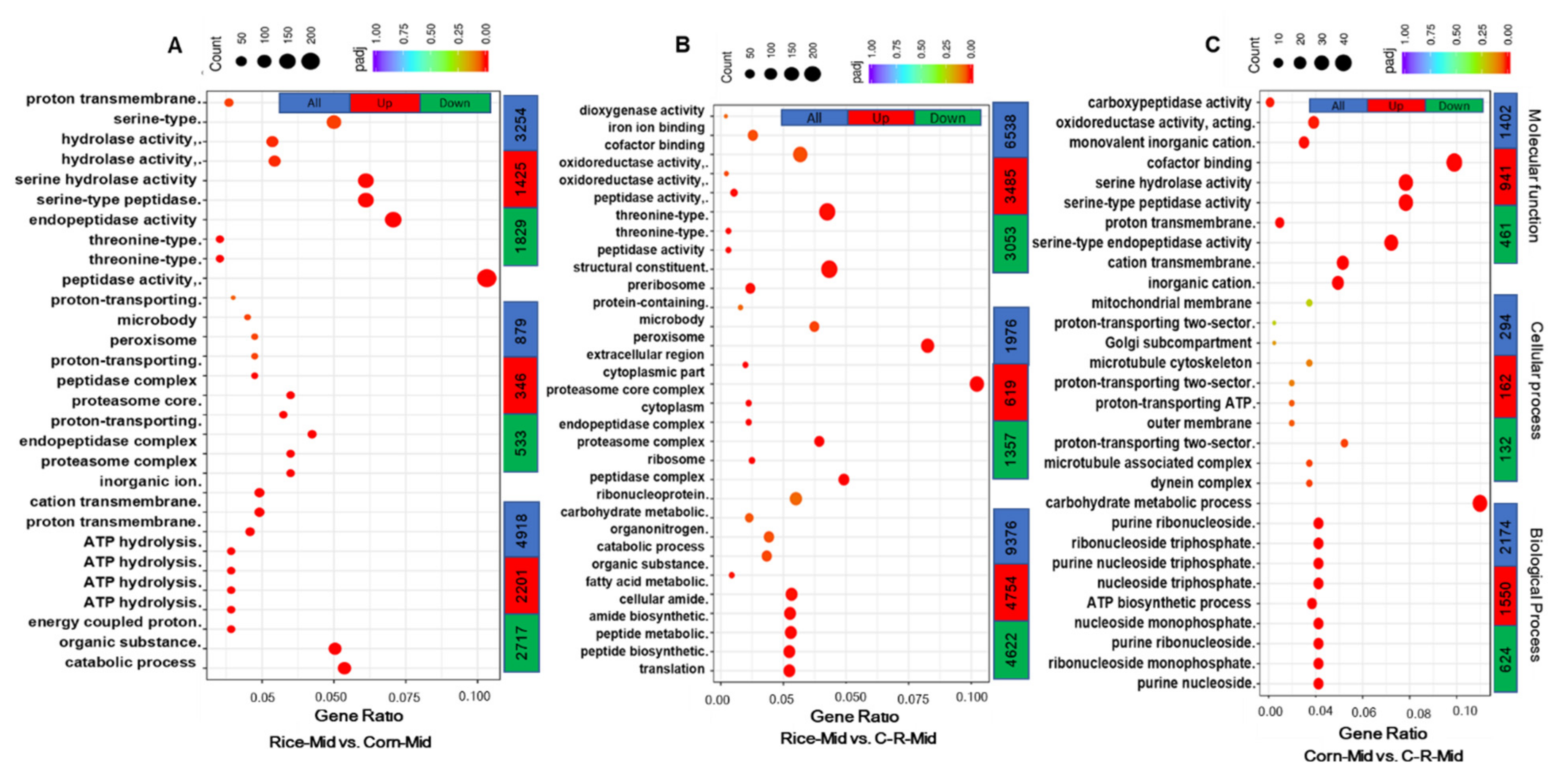
3.8. Gene Ontology (GO) Enrichment Analysis of Midgut
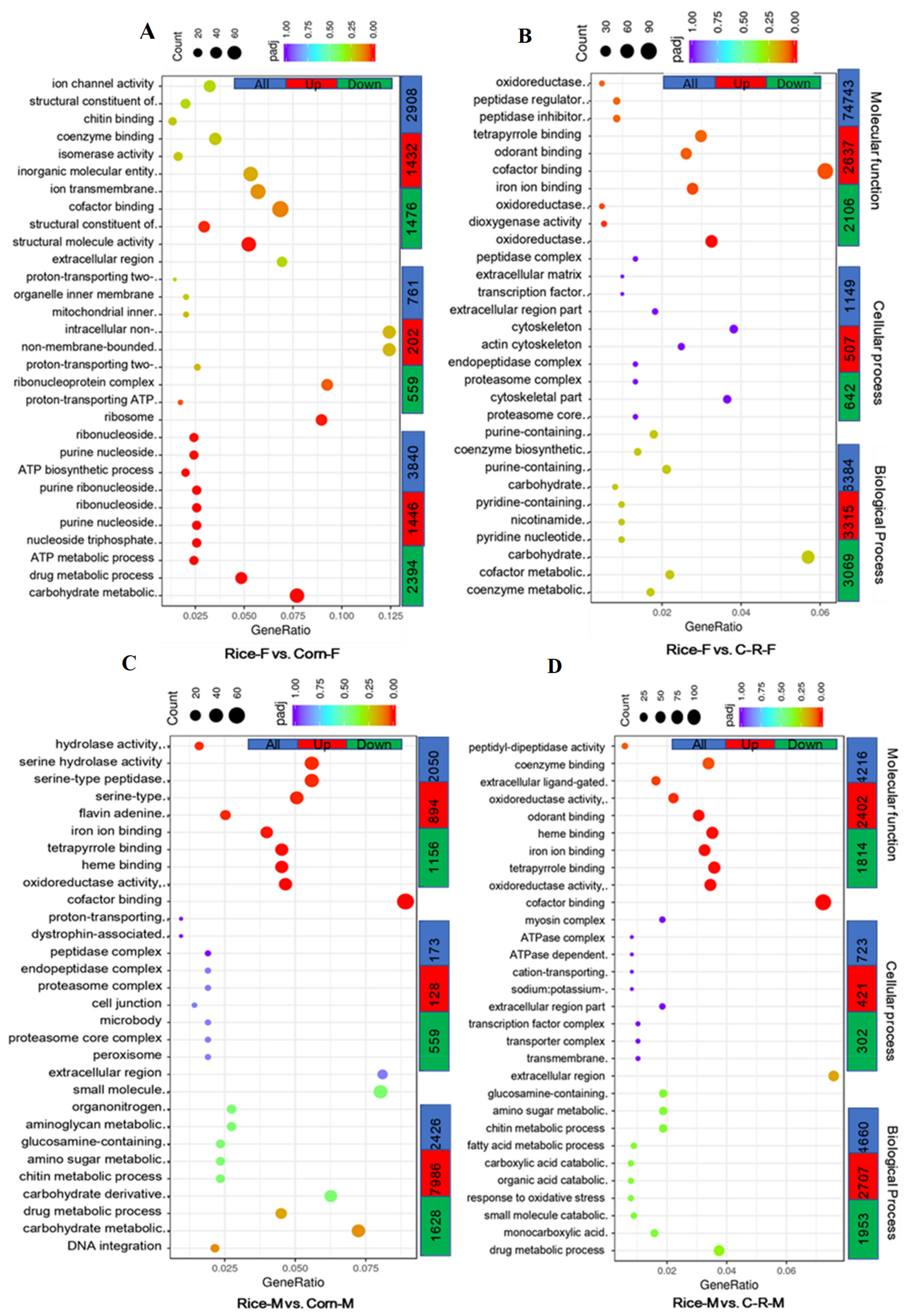
3.9. Gene Ontology (GO) Enrichment Analysis of Male and Female Antennae
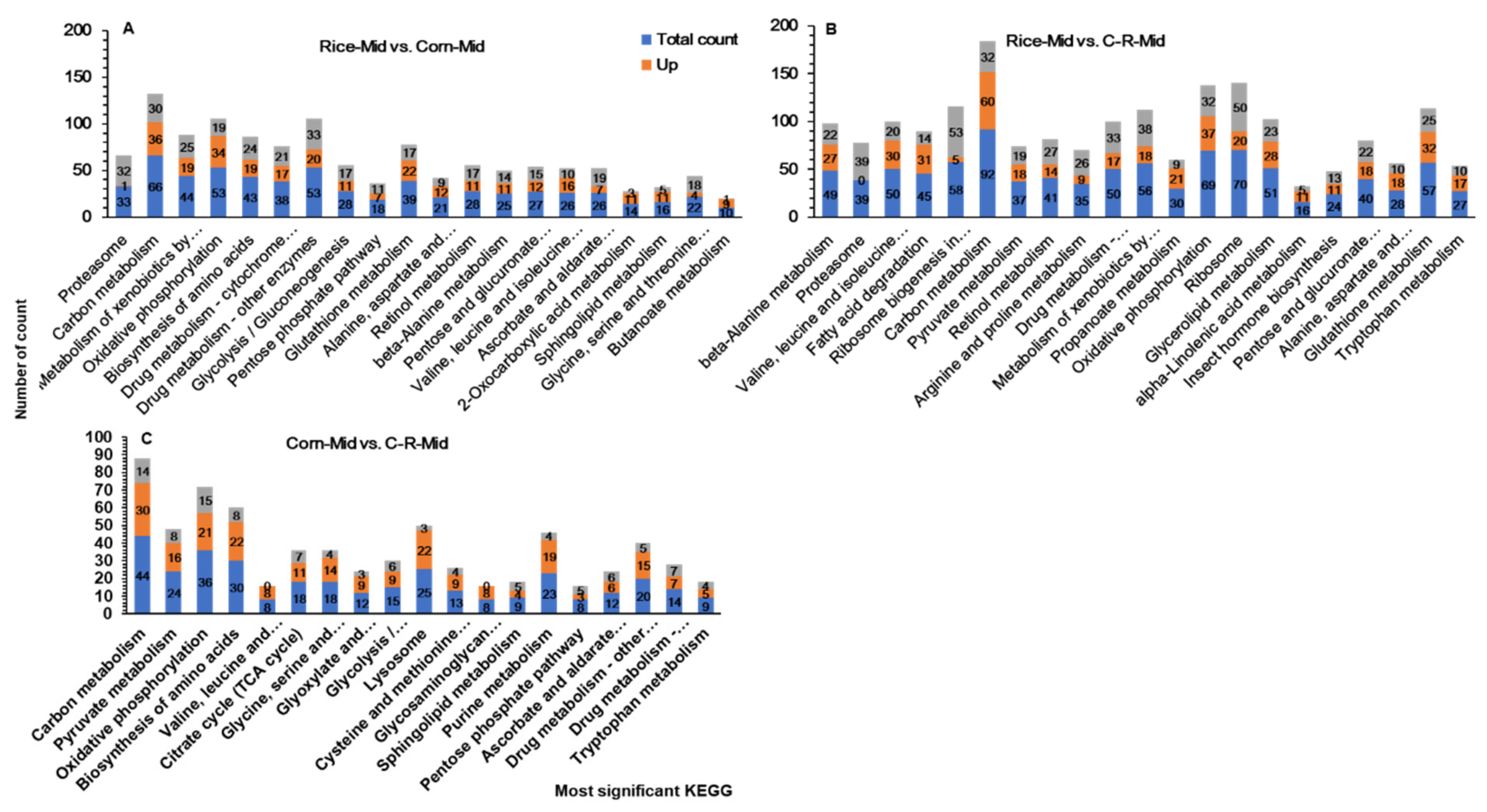
3.10. KEGG Pathway Enrichment Analysis of Midguts
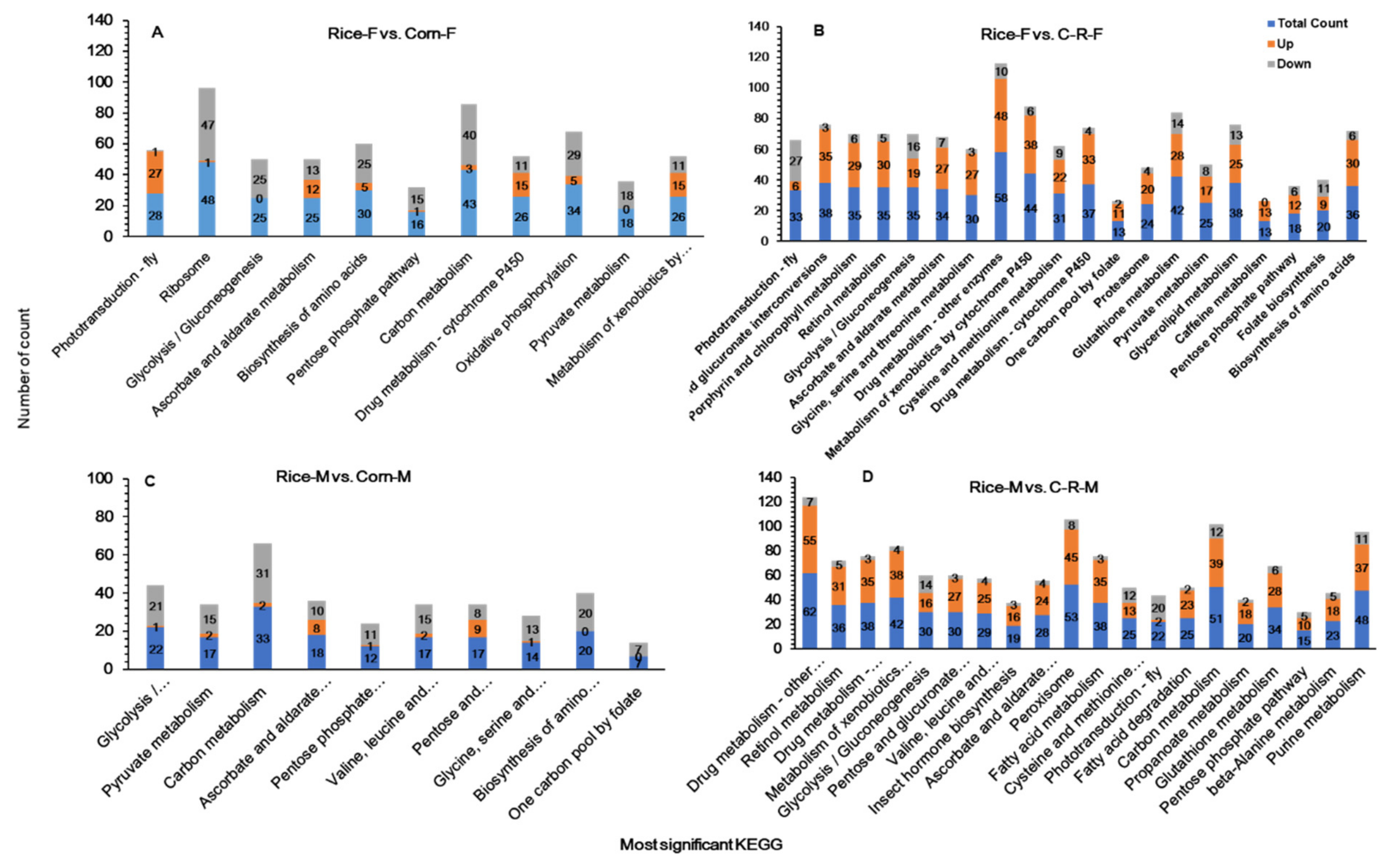
3.11. KEGG Pathway Enrichment Analysis of Male and Female Antennae
3.12. Validation of Transcriptome Data Using qPCR
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Aljbory, Z.; Chen, M.S. Indirect plant defense against insect herbivores: A review. Insect Sci. 2018, 25, 2–23. [Google Scholar] [CrossRef]
- Magalhães, S.; Fayard, J.; Janssen, A.; Carbonell, D.; Olivieri, I. Adaptation in a spider mite population after long-term evolution on a single host plant. J. Evol. Biol. 2007, 20, 2016–2027. [Google Scholar] [CrossRef]
- Agrawal, A.A. Host-range evolution: Adaptation and trade-offs in fitness of mites on alternative hosts. Ecology 2000, 81, 500–508. [Google Scholar] [CrossRef]
- Nylin, S.; Slove, J.; Janz, N. Host plant utilization, host range oscillations and diversification in nymphalid butterflies: A phylogenetic investigation. Evolution 2014, 68, 105–124. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Futuyma, D.J.; Agrawal, A.A. Macroevolution and the biological diversity of plants and herbivores. Proc. Natl. Acad. Sci. USA 2009, 106, 18054–18061. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matsubayashi, K.W.; Ohshima, I.; Nosil, P. Ecological speciation in phytophagous insects. Entomol. Exp. Appl. 2010, 164, 1–27. [Google Scholar] [CrossRef]
- Mullen, S.P.; Shaw, K.L. Insect speciation rules: Unifying concepts in speciation research. Annu. Rev. Entomol. 2014, 59, 339–361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bonsignore, C.P.; Laface, V.L.A.; Vono, G.; Marullo, R.; Musarella, C.M.; Spampinato, G. Threats posed to the rediscovered and rare Salvia ceratophylloides ard. (lamiaceae) by borer and seed feeder insect species. Diversity 2021, 13, 33. [Google Scholar] [CrossRef]
- Malausa, T.; Pélissié, B.; Piveteau, V.; Pélissier, C.; Bourguet, D.; Ponsard, S. Differences in oviposition behaviour of two sympatric sibling species of the genus Ostrinia. Bull. Entomol. Res. 2008, 98, 193–201. [Google Scholar] [CrossRef]
- Dimotsiou, O.C.; Andreadis, S.S.; Savopoulou-Soultani, M. Egg laying preference of Sesamia nonagrioides (Lepidoptera: Noctuidae) among primary and secondary hosts. Appl. Entomol. Zool. 2014, 49, 27–33. [Google Scholar] [CrossRef]
- Thorsteinson, A.J. Host Selection in Phytophagous Insects. Annu. Rev. Entomol. 1960, 5, 193–218. [Google Scholar] [CrossRef]
- Krenn, H.W.; Plant, J.D.; Szucsich, N.U. Mouthparts of flower-visiting insects. Arthropod Struct. Dev. 2005, 34, 1–40. [Google Scholar] [CrossRef]
- Cohen, M.B.; Schuler, M.A.; Berenbaum, M.R. A host-inducible cytochrome P-450 from a host-specific caterpillar: Molecular cloning and evolution. Proc. Natl. Acad. Sci. USA 1992, 89, 10920–10924. [Google Scholar] [CrossRef] [Green Version]
- Cantón, P.E.; Bonning, B.C. Transcription and Activity of Digestive Enzymes of Nezara viridula Maintained on Different Plant Diets. Front. Physiol. 2020, 10, 1553. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Simon, J.C.; D’alençon, E.; Guy, E.; Jacquin-Joly, E.; Jaquiéry, J.; Nouhaud, P.; Peccoud, J.; Sugio, A.; Streiff, R. Genomics of adaptation to host-plants in herbivorous insects. Brief. Funct. Genom. 2015, 14, 413–423. [Google Scholar] [CrossRef] [PubMed]
- Engsontia, P.; Sangket, U.; Chotigeat, W.; Satasook, C. Molecular evolution of the odorant and gustatory receptor genes in lepidopteran insects: Implications for their adaptation and speciation. J. Mol. Evol. 2014, 79, 21–39. [Google Scholar] [CrossRef] [PubMed]
- Orsucci, M.; Audiot, P.; Dorkeld, F.; Pommier, A.; Vabre, M.; Gschloessl, B.; Rialle, S.; Severac, D.; Bourguet, D.; Streiff, R. Larval transcriptomic response to host plants in two related phytophagous lepidopteran species: Implications for host specialization and species divergence. BMC Genom. 2018, 19, 265. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.C.; Ozaki, K.; Makino, T.; Uchiyama, H.; Yajima, S.; Kawata, M. Evolution of gustatory receptor gene family provides insights into adaptation to diverse host plants in nymphalid butterflies. Genome Biol. Evol. 2018, 10, 1351–1362. [Google Scholar] [CrossRef]
- Liu, X.L.; Zhang, J.; Yan, Q.; Miao, C.L.; Han, W.K.; Hou, W.; Yang, K.; Hansson, B.S.; Peng, Y.C.; Guo, J.M.; et al. The Molecular Basis of Host Selection in a Crucifer-Specialized Moth. Curr. Biol. 2020, 30, 1–7. [Google Scholar] [CrossRef]
- Anton, S.; Rössler, W. Plasticity and modulation of olfactory circuits in insects. Cell Tissue Res. 2021, 383, 149–164. [Google Scholar] [CrossRef]
- Tsitoura, P.; Iatrou, K. Positive allosteric modulation of insect olfactory receptor function by ORco agonists. Front. Cell. Neurosci. 2016, 10, 275. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, H.; Kunwar, K.; Smith, D. Odorant receptor sensitivity modulation in Drosophila. J. Neurosci. 2017, 37, 9465–9473. [Google Scholar] [CrossRef] [Green Version]
- Wicher, D. Tuning insect odorant receptors. Front. Cell. Neurosci. 2018, 12, 94. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, D.; Shi, X.; Liu, D.; Yang, Y.; Shang, Z. Transcriptome profiling revealed potentially critical roles for digestion and defense-related genes in insects’ use of resistant host plants: A case study with sitobion avenae. Insects 2020, 11, 90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brattsten, L.B. Bioengineering of crop plants and resistant biotype evolution in insects: Counteracting coevolution. Arch. Insect Biochem. Physiol. 1991, 17, 253–267. [Google Scholar] [CrossRef]
- Wang, Y.; Cai, Q.N.; Zhang, Q.W.; Han, Y. Effect of the secondary substances from wheat on the growth and digestive physiology of cotton bollworm Helicoverpa armigera (Lepidoptera: Noctuidae). Eur. J. Entomol. 2006, 103, 255–258. [Google Scholar] [CrossRef] [Green Version]
- Wouters, F.C.; Blanchette, B.; Gershenzon, J.; Vassão, D.G. Plant defense and herbivore counter-defense: Benzoxazinoids and insect herbivores. Phytochem. Rev. 2016, 15, 1127–1151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Juma, G.; Le Ru, B.; Calatayud, P.-A. Assortments of Digestive Enzymes Induced in First Instar Larvae of Busseola fusca Feeding on Different Plants. Int. J. Insect Sci. 2019, 11, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Naseri, B.; Kouhi, D.; Razmjou, J.; Golizadeh, A. Digestive enzymatic activity and nutritional responses of Helicoverpa armigera (lepidoptera: Noctuidae) larvae fed various tomato cultivars. J. Econ. Entomol. 2014, 107, 1655–1661. [Google Scholar] [CrossRef]
- Hafeez, M.; Li, X.-W.; Zhang, J.-M.; Zhang, Z.-J.; Huang, J.; Wang, L.-K.; Khan, M.M.; Shah, S.; Fernández-Grandon, G.M.; Lu, Y.-B. Role of digestive protease enzymes and related genes in host plant adaptation of a polyphagous pest, Spodoptera frugiperda. Insect Sci. 2021, 28, 611–626. [Google Scholar] [CrossRef]
- Brito, L.O.; Lopes, A.R.; Parra, J.R.P.; Terra, W.R.; Silva-Filho, M.C. Adaptation of tobacco budworm Heliothis virescens to proteinase inhibitors may be mediated by the synthesis of new proteinases. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2001, 128, 365–375. [Google Scholar] [CrossRef]
- Pauchet, Y.; Wilkinson, P.; Vogel, H.; Nelson, D.R.; Reynolds, S.E.; Heckel, D.G.; Ffrench-Constant, R.H. Pyrosequencing the Manduca sexta larval midgut transcriptome: Messages for digestion, detoxification and defence. Insect Mol. Biol. 2010, 19, 61–75. [Google Scholar] [CrossRef]
- You, M.; Yue, Z.; He, W.; Yang, X.; Yang, G.; Xie, M.; Zhan, D.; Baxter, S.W.; Vasseur, L.; Gurr, G.M.; et al. A heterozygous moth genome provides insights into herbivory and detoxification. Nat. Genet. 2013, 45, 220–225. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pearce, S.L.; Clarke, D.F.; East, P.D.; Elfekih, S.; Gordon, K.H.J.; Jermiin, L.S.; McGaughran, A.; Oakeshott, J.G.; Papanikolaou, A.; Perera, O.P.; et al. Genomic innovations, transcriptional plasticity and gene loss underlying the evolution and divergence of two highly polyphagous and invasive Helicoverpa pest species. BMC Biol. 2017, 15, 63. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salehipourshirazi, G.; Bruinsma, K.; Ratlamwala, H.; Dixit, S.; Arbona, V.; Widemann, E.; Milojevic, M.; Jin, P.; Bensoussan, N.; Gómez-Cadenas, A.; et al. The generalist herbivore Tetranychus urticae (Koch) adapts to novel plant hosts through rapid evolution of metabolic resistance. bioRxiv 2020. [Google Scholar] [CrossRef] [Green Version]
- Celorio-mancera, M.D.L.P.; Wheat, C.W.; Vogel, H. Mechanisms of macroevolution: Polyphagous plasticity in butterfly larvae revealed by RNA-Seq. Mol. Ecol. 2013, 22, 4884–4895. [Google Scholar] [CrossRef]
- Xu, H.X.; Hong, Y.; Zhang, M.Z.; Wang, Y.L.; Liu, S.S.; Wang, X.W. Transcriptional responses of invasive and indigenous whiteflies to different host plants reveal their disparate capacity of adaptation. Sci. Rep. 2015, 5, 10774. [Google Scholar] [CrossRef] [Green Version]
- Pym, A.; Singh, K.S.; Nordgren, Å.; Davies, T.G.E.; Zimmer, C.T.; Elias, J.; Slater, R.; Bass, C. Host plant adaptation in the polyphagous whitefly, Trialeurodes vaporariorum, is associated with transcriptional plasticity and altered sensitivity to insecticides. BMC Genom. 2019, 20, 996. [Google Scholar] [CrossRef] [Green Version]
- Chang, X.Q.; Nie, X.P.; Zhang, Z.; Zeng, F.F.; Lv, L.; Zhang, S.; Wang, M.Q. De novo analysis of the oriental armyworm Mythimna separata antennal transcriptome and expression patterns of odorant-binding proteins. Comp. Biochem. Physiol. Part D Genom. Proteom. 2017, 22, 120–130. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Tang, K.Y.; Chen, X.F.; Tao, Y.; Jiang, H.B.; Wang, J.J. Comparative transcriptomic analysis reveals female-biased olfactory genes potentially involved in plant volatile-mediated oviposition behavior of Bactrocera dorsalis. BMC Genom. 2021, 22, 25. [Google Scholar] [CrossRef]
- Midega, C.A.O.; Pittchar, J.O.; Pickett, J.A.; Hailu, G.W.; Khan, Z.R. A climate-adapted push-pull system effectively controls fall armyworm, Spodoptera frugiperda (J E Smith), in maize in East Africa. Crop Prot. 2018, 105, 10–15. [Google Scholar] [CrossRef]
- Montezano, D.G.; Specht, A.; Sosa-Gómez, D.R.; Roque-Specht, V.F.; Sousa-Silva, J.C.; Paula-Moraes, S.V.; Peterson, J.A.; Hunt, T.E. Host Plants of Spodoptera frugiperda (Lepidoptera: Noctuidae) in the Americas. Afr. Entomol. 2018, 26, 286–300. [Google Scholar] [CrossRef] [Green Version]
- Pogue, M. A world revision of the genus Spodoptera Guenée (Lepidoptera: Noctuidae). Am. Entomol. Soc. 2002, 43, 1–102. [Google Scholar]
- Goergen, G.; Kumar, P.L.; Sankung, S.B.; Togola, A.; Tamò, M. First report of outbreaks of the fall armyworm spodoptera frugiperda (J E Smith) (Lepidoptera, Noctuidae), a new alien invasive pest in West and Central Africa. PLoS ONE 2016, 11, e0165632. [Google Scholar] [CrossRef] [Green Version]
- Stokstad, E. New crop pest takes Africa at lightning speed. Science 2017, 356, 473–474. [Google Scholar] [CrossRef]
- Ganiger, P.C.; Yeshwanth, H.M.; Muralimohan, K.; Vinay, N.; Kumar, A.R.V.; Chandrashekara, K. Occurrence of the new invasive pest, fall armyworm, Spodoptera frugiperda (J.E. Smith) (Lepidoptera: Noctuidae), in the maize fields of Karnataka, India. Curr. Sci. 2018, 115, 621. [Google Scholar] [CrossRef]
- Jing, D.P.; Guo, J.F.; Jiang, Y.Y.; Zhao, J.Z.; Sethi, A.; He, K.L.; Wang, Z.Y. Initial detections and spread of invasive Spodoptera frugiperda in China and comparisons with other noctuid larvae in cornfields using molecular techniques. Insect Sci. 2020, 27, 780–790. [Google Scholar] [CrossRef]
- Maino, J.L.; Schouten, R.; Overton, K.; Day, R.; Ekesi, S.; Bett, B.; Barton, M.; Gregg, P.C.; Umina, P.A.; Reynolds, O.L. Regional and seasonal activity predictions for fall armyworm in Australia. Curr. Res. Insect Sci. 2021, 1, 100010. [Google Scholar] [CrossRef]
- Levy, H.C.; Garcia-Maruniak, A.; Maruniak, J.E. Strain identification of Spodoptera frugiperda (Lepidoptera: Noctuidae) insects and cell line: PCR-RFLP of cytochrome oxidase C subunit I gene. Florida Entomol. 2003, 85, 186–190. [Google Scholar] [CrossRef]
- Clark, P.L.; Molina-Ochoa, J.; Martinelli, S.; Skoda, S.R.; Isenhour, D.J.; Lee, D.J.; Krumm, J.T.; Foster, J.E. Population variation of the fall armyworm, Spodoptera frugiperda, in the Western Hemisphere. J. Insect Sci. 2007, 7, 5. [Google Scholar] [CrossRef] [Green Version]
- Dumas, P.; Legeai, F.; Lemaitre, C.; Scaon, E.; Orsucci, M.; Labadie, K.; Gimenez, S.; Clamens, A.L.; Henri, H.; Vavre, F.; et al. Spodoptera frugiperda (Lepidoptera: Noctuidae) host-plant variants: Two host strains or two distinct species? Genetica 2015, 143, 305–316. [Google Scholar] [CrossRef] [Green Version]
- Nagoshi, R.N.; Dhanani, I.; Asokan, R.; Mahadevaswamy, H.M.; Kalleshwaraswamy, C.M.; Sharanabasappa; Meagher, R.L. Genetic characterization of fall armyworm infesting South Africa and India indicate recent introduction from a common source population. PLoS ONE 2019, 14, e0217755. [Google Scholar] [CrossRef] [Green Version]
- Carroll, M.J.; Schmelz, E.A.; Meagher, R.L.; Teal, P.E.A. Attraction of Spodoptera frugiperda larvae to volatiles from herbivore-damaged maize seedlings. J. Chem. Ecol. 2006, 32, 1911–1924. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–498. [Google Scholar] [CrossRef] [PubMed]
- Sheck, A.L.; Gould, F. The genetic basis of differences in growth and behavior of specialist and generalist herbivore species: Selection on hybrids of Heliothis virescens and Heliothis subflexa (Lepidoptera). Evolution 1996, 50, 831–841. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.; Kennedy, G.G.; Gould, F. Genetic analysis of larval survival and larval growth of two populations of Leptinotarsa decemlineata on tomato. Entomol. Exp. Appl. 2001, 99, 143–155. [Google Scholar] [CrossRef] [Green Version]
- Agrawal, A.A. Ecology: Phenotypic plasticity in the interactions and evolution of species. Science 2001, 294, 321–326. [Google Scholar] [CrossRef] [Green Version]
- Jermy, T. The Role of Experience in the Host Selection of Phytophagous Insects. In Perspectives in Chemoreception and Behavior; Chapman, R.F., Bernays, E.A., Stoffolano, J.G., Eds.; Springer: New York, NY, USA, 1987. [Google Scholar]
- Papaj, D.R.; Prokopy, R.J. Ecological and evolutionary aspects of learning in phytophagous insects. Annu. Rev. Entomol. 1989, 34, 315–350. [Google Scholar] [CrossRef]
- Ning, S.Y.; Yang, H.Y.; Fan, D.S.; Feng, J.N. Influence of larval experience on preference of a subterranean insect Delia antiqua on Allium hosts. J. Appl. Entomol. 2018, 142, 263–271. [Google Scholar] [CrossRef]
- Carlsson, M.A.; Anderson, P.; Hartlieb, E.; Hansson, B.S. Experience-dependent modification of orientational response to olfactory cues in larvae of Spodoptera littoralis. J. Chem. Ecol. 1999, 25, 2445–2454. [Google Scholar] [CrossRef]
- Fry, J.D. Evolutionary adaptation to host plants in a laboratory population of the phytophagous mite Tetranychus urticae Koch. Oecologia 1989, 81, 559–565. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dermauw, W.; Wybouw, N.; Rombauts, S.; Menten, B.; Vontas, J.; Grbić, M.; Clark, R.M.; Feyereisen, R.; Van Leeuwen, T. A link between host plant adaptation and pesticide resistance in the polyphagous spider mite Tetranychus urticae. Proc. Natl. Acad. Sci. USA 2013, 110, E113–E122. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jeschke, V.; Zalucki, J.M.; Raguschke, B.; Gershenzon, J.; Heckel, D.G.; Zalucki, M.P.; Vassão, D.G. So much for glucosinolates: A generalist does survive and develop on brassicas, but at what cost? Plants 2021, 10, 962. [Google Scholar] [CrossRef] [PubMed]
- Zalucki, J.M.; Heckel, D.G.; Wang, P.; Kuwar, S.; Vassão, D.G.; Perkins, L.; Zalucki, M.P. A generalist feeding on brassicaceae: It does not get any better with selection. Plants 2021, 10, 954. [Google Scholar] [CrossRef]
- Silva-Brandão, K.L.; Horikoshi, R.J.; Bernardi, D.; Omoto, C.; Figueira, A.; Brandão, M.M. Transcript expression plasticity as a response to alternative larval host plants in the speciation process of corn and rice strains of Spodoptera frugiperda. BMC Genom. 2017, 18, 792. [Google Scholar] [CrossRef]
- Roy, A.; Walker, W.B.; Vogel, H.; Chattington, S.; Larsson, M.C.; Anderson, P.; Heckel, D.G.; Schlyter, F. Diet dependent metabolic responses in three generalist insect herbivores Spodoptera spp. Insect Biochem. Mol. Biol. 2016, 71, 91–105. [Google Scholar] [CrossRef]
- Groot, A.T.; Marr, M.; Heckel, D.G.; SchÖfl, G. The roles and interactions of reproductive isolation mechanisms in fall armyworm (Lepidoptera: Noctuidae) host strains. Ecol. Entomol. 2010, 35, 105–118. [Google Scholar] [CrossRef]
- Wang, D.; Zhou, L.; Wang, Q.; Ding, J. Plant Chemistry Determines Host Preference and Performance of an Invasive Insect. Front. Plant Sci. 2020, 11, 594663. [Google Scholar] [CrossRef] [PubMed]
- Awmack, C.S.; Leather, S.R. Host plant quality and fecundity in herbivorous insects. Annu. Rev. Entomol. 2002, 47, 817–844. [Google Scholar] [CrossRef]
- Horgan, F.G.; Quiring, D.T.; Lagnaoui, A.; Pelletier, Y. Life Histories and Fitness of Two Tuber Moth Species Feeding on Native Andean Potatoes. Neotrop. Entomol. 2012, 41, 333–340. [Google Scholar] [CrossRef]
- Tammaru, T.; Javois, J. Responses of ovipositing moths (Lepidoptera: Geometridae) to host plant deprivation: Life-history aspects and implications for population dynamics. Environ. Entomol. 2000, 29, 1002–1010. [Google Scholar] [CrossRef]
- García-Barros, E.; Fartmann, T. Butterfly oviposition: Sites, behaviour and modes. In Ecology of Butterflies in Europe; Cambridge: Cambridge, UK, 2009. [Google Scholar]
- Davis, J.M.; Stamps, J.A. The effect of natal experience on habitat preferences. Trends Ecol. Evol. 2004, 19, 411–416. [Google Scholar] [CrossRef]
- Anderson, P.; Sadek, M.M.; Larsson, M.; Hansson, B.S.; Thöming, G. Larval host plant experience modulates both mate finding and oviposition choice in a moth. Anim. Behav. 2013, 85, 1169–1175. [Google Scholar] [CrossRef]
- Thöming, G.; Larsson, M.C.; Hansson, B.S.; Anderson, P. Comparison of plant preference hierarchies of male and female moths and the impact of larval rearing hosts. Ecology 2013, 94, 1744–1752. [Google Scholar] [CrossRef] [PubMed]
- Moreau, J.; Rahme, J.; Benrey, B.; Thiery, D. Larval host plant origin modifies the adult oviposition preference of the female European grapevine moth Lobesia botrana. Naturwissenschaften 2008, 95, 317–324. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Videla, M.; Valladares, G.; Salvo, A. Differential effects of experience on feeding and ovipositing preferences of a polyphagous leafminer. Entomol. Exp. Appl. 2010, 137, 184–192. [Google Scholar] [CrossRef]
- Janz, N.; Söderlind, L.; Nylin, S. No effect of larval experience on adult host preferences in Polygonia c-album (Lepidoptera: Nymphalidae): On the persistence of Hopkins’ host selection principle. Ecol. Entomol. 2009, 34, 50–57. [Google Scholar] [CrossRef]
- Firempong, S.; Zalucki, M.P. Host plant selection by Helicoverpa armigera (Lepidoptera: Noctuidae): The role of some herbivore attributes. Aust. J. Zool. 1991, 39, 343–350. [Google Scholar] [CrossRef]
- Petrén, H.; Gloder, G.; Posledovich, D.; Wiklund, C.; Friberg, M. Innate preference hierarchies coupled with adult experience, rather than larval imprinting or transgenerational acclimation, determine host plant use in Pieris rapae. Ecol. Evol. 2021, 11, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Barron, A.B. The life and death of Hopkins’ host-selection principle. J. Insect Behav. 2001, 14, 725–737. [Google Scholar] [CrossRef]
- Kaissling, K.E.; Rospars, J.P. Dose-response relationships in an olfactory flux detector model revisited. Chem. Senses 2004, 29, 529–531. [Google Scholar] [CrossRef]
- Berdegué, M.; Reitz, S.R.; Trumble, J.T. Host plant selection and development in Spodoptera exigua: Do mother and offspring know best? Entomol. Exp. Appl. 1998, 89, 57–64. [Google Scholar] [CrossRef]
- Tao, X.Y.; Xue, X.Y.; Huang, Y.P.; Chen, X.Y.; Mao, Y.B. Gossypol-enhanced P450 gene pool contributes to cotton bollworm tolerance to a pyrethroid insecticide. Mol. Ecol. 2012, 21, 4371–4385. [Google Scholar] [CrossRef]
- Yu, Q.Y.; Fang, S.M.; Zhang, Z.; Jiggins, C.D. The transcriptome response of Heliconius melpomene larvae to a novel host plant. Mol. Ecol. 2016, 25, 4850–4865. [Google Scholar] [CrossRef] [PubMed]
- Herde, M.; Howe, G.A. Host plant-specific remodeling of midgut physiology in the generalist insect herbivore Trichoplusia ni. Insect Biochem. Mol. Biol. 2014, 50, 58–67. [Google Scholar] [CrossRef]
- Orsucci, M.; Moné, Y.; Audiot, P.; Gimenez, S.; Nhim, S.; Naït-Saïdi, R.; Frayssinet, M.; Dumont, G.; Pommier, A.; Boudon, J.P.; et al. Transcriptional plasticity evolution in two strains of Spodoptera frugiperda (Lepidoptera: Noctuidae) feeding on alternative host-plants. bioRxiv 2018. [Google Scholar] [CrossRef] [Green Version]
- De la Paz Celorio-Mancera, M.; Heckel, D.G.; Vogel, H. Transcriptional analysis of physiological pathways in a generalist herbivore: Responses to different host plants and plant structures by the cotton bollworm, Helicoverpa armigera. Entomol. Exp. Appl. 2012, 144, 123–133. [Google Scholar] [CrossRef]
- Waniek, P.J.; Araújo, C.A.C.; Momoli, M.M.; Azambuja, P.; Jansen, A.M.; Genta, F.A. Serine carboxypeptidases of Triatoma brasiliensis (Hemiptera, Reduviidae): Sequence characterization, expression pattern and activity localization. J. Insect Physiol. 2014, 63, 9–20. [Google Scholar] [CrossRef]
- Hou, Z.; Wei, C. De novo comparative transcriptome analysis of a rare cicada, with identification of candidate genes related to adaptation to a novel host plant and drier habitats. BMC Genom. 2019, 20, 182. [Google Scholar] [CrossRef]
- Eyres, I.; Jaquiéry, J.; Sugio, A.; Duvaux, L.; Gharbi, K.; Zhou, J.J.; Legeai, F.; Nelson, M.; Simon, J.C.; Smadja, C.M.; et al. Differential gene expression according to race and host plant in the pea aphid. Mol. Ecol. 2016, 25, 4197–4215. [Google Scholar] [CrossRef]
- Heidel-Fischer, H.M.; Vogel, H. Molecular mechanisms of insect adaptation to plant secondary compounds. Curr. Opin. Insect Sci. 2015, 8, 8–14. [Google Scholar] [CrossRef]
- Zhong, H.; Li, F.; Chen, J.; Zhang, J.; Li, F. Comparative transcriptome analysis reveals host-associated differentiation in Chilo suppressalis (Lepidoptera: Crambidae). Sci. Rep. 2017, 7, 13778. [Google Scholar] [CrossRef]
- Terra, W.R.; Ferreira, C. Biochemistry and Molecular Biology of Digestion. In Insect Molecular Biology and Biochemistry; Cambridge: Cambridge, UK, 2012. [Google Scholar]
- Spit, J.; Holtof, M.; Badisco, L.; Vergauwen, L.; Vogel, E.; Knapen, D.; Vanden Broeck, J. Transcriptional Analysis of the Adaptive Digestive System of the Migratory Locust in Response to Plant Defensive Protease Inhibitors. Sci. Rep. 2016, 6, 32460. [Google Scholar] [CrossRef] [Green Version]
- Del Campo, M.L.; Miles, C.I.; Schroe, F.C.; Mueller, C.; Booker, R.; Renwick, J.A. Host recognition by the tobacco hornworm is mediated by a host plant compound. Nature 2001, 411, 186–189. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhang, H.; Guan, R.; Miao, X. Identification of differential expression genes associated with host selection and adaptation between two sibling insect species by transcriptional profile analysis. BMC Genom. 2013, 14, 582. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qiu, L.; He, L.; Tan, X.; Zhang, Z.; Wang, Y.; Li, X.; He, H.; Ding, W.; Li, Y. Identification and phylogenetics of Spodoptera frugiperda chemosensory proteins based on antennal transcriptome data. Comp. Biochem. Physiol. Part D Genom. Proteom. 2020, 130, 323–332. [Google Scholar] [CrossRef]
- Israni, B.; Wouters, F.C.; Luck, K.; Seibel, E.; Ahn, S.J.; Paetz, C.; Reinert, M.; Vogel, H.; Erb, M.; Heckel, D.G.; et al. The Fall Armyworm Spodoptera frugiperda Utilizes Specific UDP-Glycosyltransferases to Inactivate Maize Defensive Benzoxazinoids. Front. Physiol. 2020, 11, 604754. [Google Scholar] [CrossRef]





| Feeding Condition | No of 1st Instar Larvae | No of 12-Day Larvae | No Pupae | No of Emerged Adults | Larval Duration (Days) | Overall Survival (%) |
|---|---|---|---|---|---|---|
| Corn-Pop on rice (G-1) | 400 | 167 (41.75%) | 76 (24.25%) | 47 (58.08%) | 21.87 (18.25–24.26) | 20.25 |
| Corn-pop on corn-(G-20) | 327 | 287 (87.76%) | 202 (70.80%) | 172 (85.15%) | 18.97 (16.26–20.53) | 52.59 |
| Corn-pop on rice-(G-20) | 350 | 321 (91.71%) | 297(92.52%) | 283 (95.29%) | 17.03 (14.24–19.78) | 80.86 |
| Classification | Candidate Genes | Number of DEGs | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Rice-Mid vs. Corn-Mid | Rice-Mid vs. C-R-Mid | Corn-Mid vs. C-R-Mid | ||||||||
| Total | Up | Down | Total | Up | Down | Total | Up | Down | ||
| Digestion | Trypsin | 56 | 24 | 32 | 102 | 45 | 57 | 31 | 23 | 8 |
| carboxypeptidase | 10 | 7 | 3 | 21 | 20 | 1 | 11 | 9 | 2 | |
| Lipase | 15 | 10 | 5 | 34 | 29 | 5 | 5 | 3 | 2 | |
| Alpha amylase | 7 | 3 | 4 | 7 | 6 | 1 | 2 | 2 | 0 | |
| cysteine protease | 3 | 0 | 3 | 15 | 5 | 8 | 1 | 1 | 0 | |
| serine protease | 4 | 2 | 2 | 8 | 2 | 6 | 0 | 0 | 0 | |
| Trypsin Inhibitor | 8 | 6 | 2 | 12 | 10 | 2 | 2 | 2 | 0 | |
| Detoxification | P450s | 25 | 17 | 7 | 56 | 34 | 22 | 15 | 7 | 8 |
| CEs | 26 | 18 | 8 | 43 | 32 | 11 | 6 | 4 | 2 | |
| GSTs | 14 | 11 | 3 | 14 | 4 | 10 | 1 | 2 | ||
| UGTs | 18 | 5 | 13 | 30 | 6 | 24 | 4 | 3 | 1 | |
| ABC transporters | 7 | 4 | 3 | 26 | 18 | 8 | 0 | 0 | 0 | |
| Ribosomal | Ribosomal protein | 3 | 0 | 3 | 23 | 2 | 21 | 1 | 0 | 1 |
| Candidate Genes | Number of DEGs | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Rice-F vs. Corn F | Rice-F vs. C-R-F | Rice-M vs. Corn-M | Rice-M vs. C-R-M | |||||||||
| Total | Up | Down | Total | Up | Down | Total | Up | Down | Total | Up | Down | |
| Insect pheromone-binding family | 9 | 1 | 8 | 7 | 4 | 3 | 10 | 0 | 10 | 5 | 3 | 2 |
| PBP/GOBP family | 4 | 3 | 1 | 25 | 21 | 4 | 6 | 0 | 6 | 4 | 0 | 4 |
| Olfactory receptor | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Odorant receptor | 6 | 4 | 2 | 22 | 22 | 0 | 4 | 4 | 0 | 14 | 3 | |
| Chemosensory receptor | 1 | 1 | 0 | 2 | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hafeez, M.; Li, X.; Ullah, F.; Zhang, Z.; Zhang, J.; Huang, J.; Khan, M.M.; Chen, L.; Ren, X.; Zhou, S.; et al. Behavioral and Physiological Plasticity Provides Insights into Molecular Based Adaptation Mechanism to Strain Shift in Spodoptera frugiperda. Int. J. Mol. Sci. 2021, 22, 10284. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms221910284
Hafeez M, Li X, Ullah F, Zhang Z, Zhang J, Huang J, Khan MM, Chen L, Ren X, Zhou S, et al. Behavioral and Physiological Plasticity Provides Insights into Molecular Based Adaptation Mechanism to Strain Shift in Spodoptera frugiperda. International Journal of Molecular Sciences. 2021; 22(19):10284. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms221910284
Chicago/Turabian StyleHafeez, Muhammad, Xiaowei Li, Farman Ullah, Zhijun Zhang, Jinming Zhang, Jun Huang, Muhammad Musa Khan, Limin Chen, Xiaoyun Ren, Shuxing Zhou, and et al. 2021. "Behavioral and Physiological Plasticity Provides Insights into Molecular Based Adaptation Mechanism to Strain Shift in Spodoptera frugiperda" International Journal of Molecular Sciences 22, no. 19: 10284. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms221910284










