Distribution Characteristics and Source of Dechloranes in Soil and Lichen of the Fildes Peninsula (Antarctica)
Abstract
:1. Introduction
2. Experimental Section
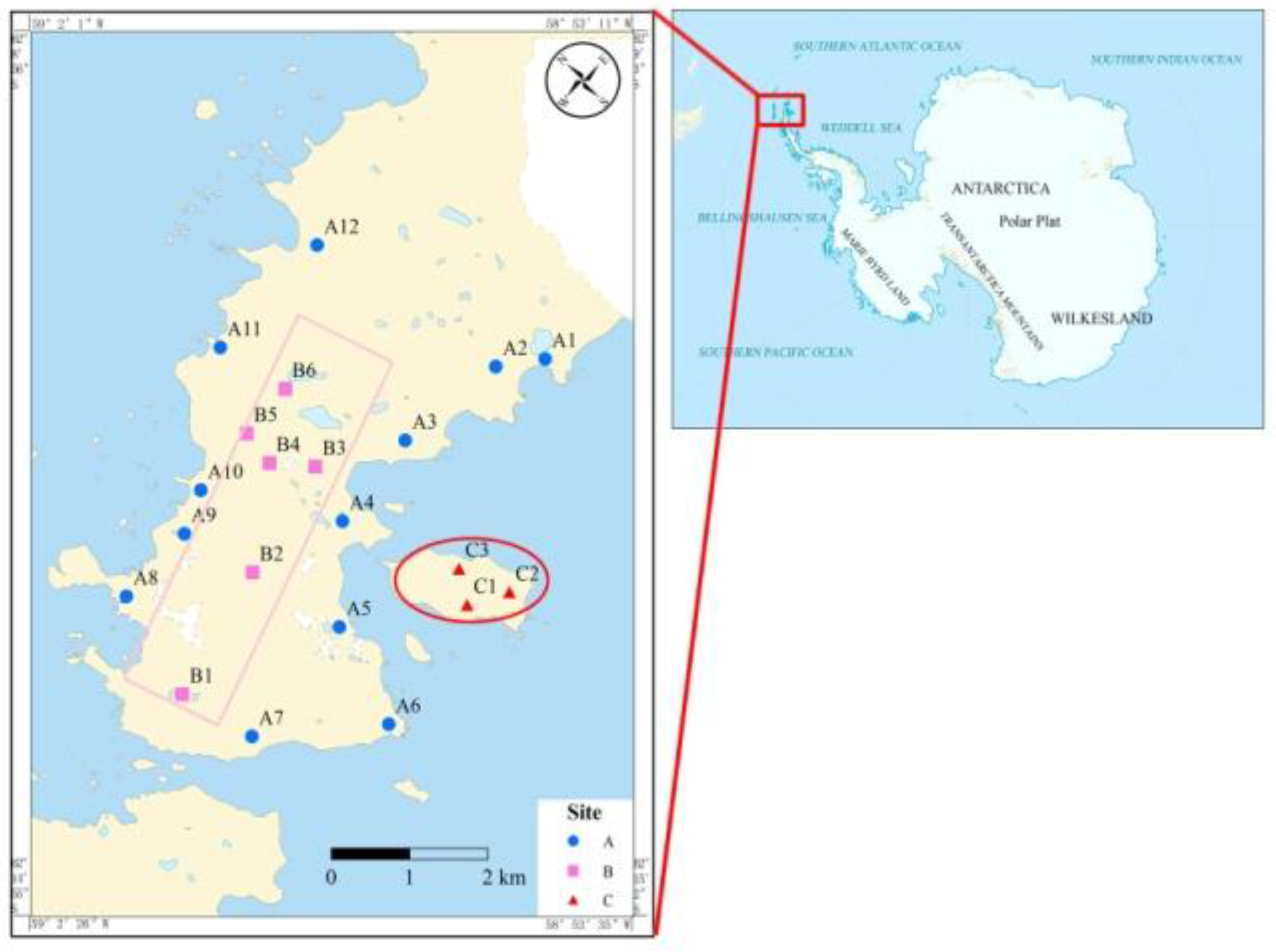
2.1. Sampling
2.2. Chemical Analysis
2.3. Quality Assurance and Quality Control
2.4. Statistics
3. Results and Discussion
3.1. Levels and Source of Decs in Soil and Lichen
3.2. Spatial Distribution of Dechloranes in Soil and Lichen
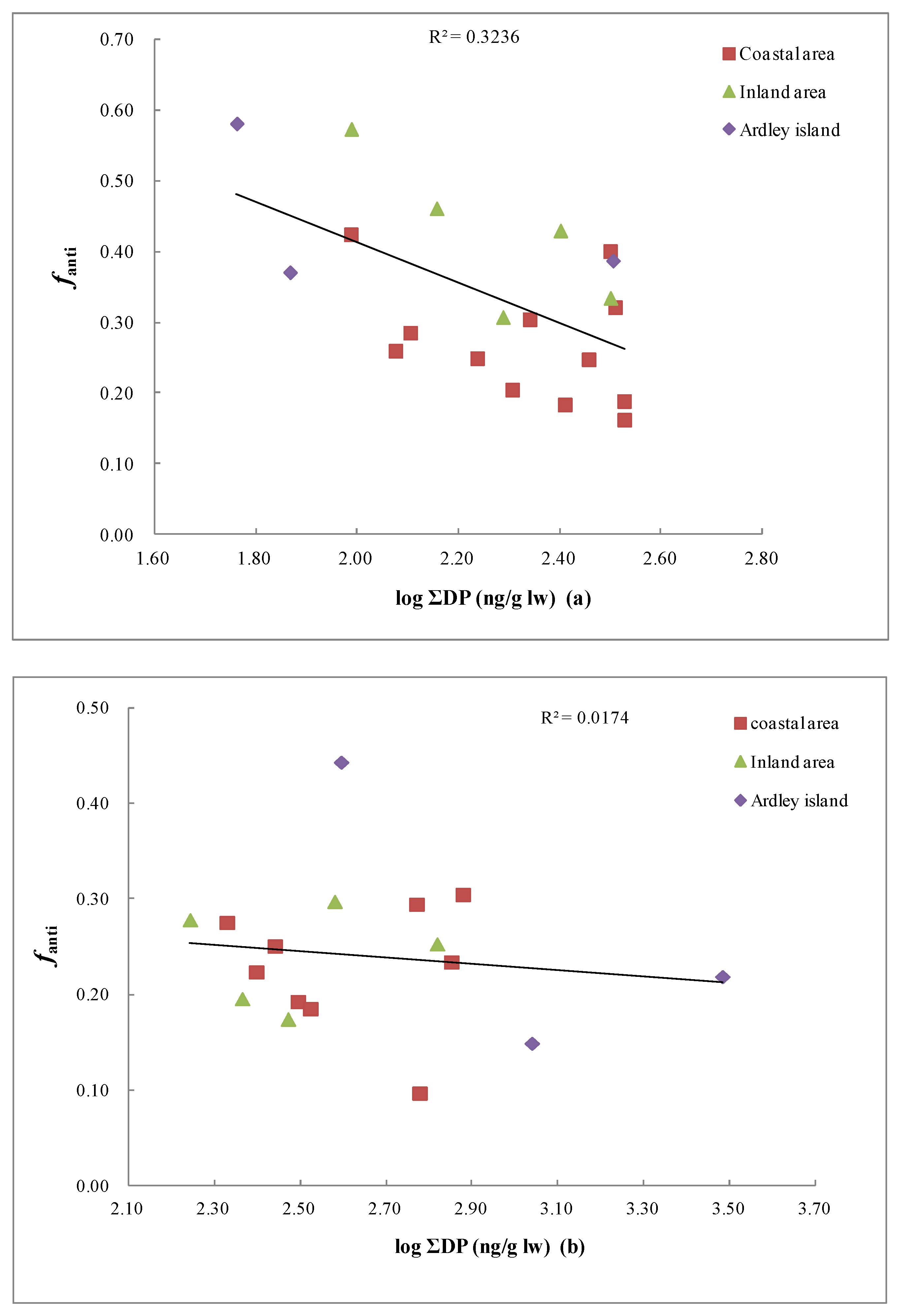
3.3. Fractional Abundance of DP Isomers
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Shen, L.; Reiner, E.J.; Helm, P.A.; Marvin, C.H.; Hill, B.; Zhang, X.M.; Macpherson, K.A.; Kolic, T.M.; Tomy, G.T.; Brindlet, L.D. Historic trends of Dechloranes 602, 603, 604, Dechlorane Plus and other norbornene derivatives and their bioaccumulation potential in Lake Ontario. Environ. Sci. Technol. 2011, 45, 3333–3340. [Google Scholar] [CrossRef] [PubMed]
- Feo, M.L.; Barón, E.; Eljarrat, E.; Barceló, D. Dechlorane Plus and related compounds in aquatic and terrestrial biota: A review. Anal. Bioanal. Chem. 2012, 404, 2625–2637. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.P.; Feng, Y.L.; Shoeib, M. Detection of Dechlorane Plus in residential indoor dust in the city of Ottawa, Canada. Environ. Sci. Technol. 2007, 41, 7694–7698. [Google Scholar] [CrossRef] [PubMed]
- Möller, A.; Xie, Z.Y.; Sturm, R.; Ebinghaus, R. Large-Scale Distribution of Dechlorane Plus in Air and Seawater from the Arctic to Antarctica. Environ. Sci. Technol. 2010, 44, 8977–8982. [Google Scholar] [CrossRef] [PubMed]
- Sverko, E.; Reiner, E.J.; Tomy, G.T.; Mccrindle, R.; Shen, L.; Arsenault, G.; Zaruk, D.; Macpherson, K.A.; Marvin, C.H.; Helm, P.A.; et al. Compounds structurally related to dechlorane plus in sediment and biota from Lake Ontario (Canada). Environ. Sci. Technol. 2010, 44, 574–579. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.J.; Tian, M.; Wang, J.; Shi, T.; Luo, Y.; Luo, X.J.; Mai, B.X. Dechlorane plus (DP) in air and plants at an electronic waste (e-waste) site in South China. Environ. Pollut. 2011, 159, 1290–1296. [Google Scholar] [CrossRef] [PubMed]
- Qiu, X.H.; Hites, R.A. Dechlorane Plus and other flame retardants in tree bark from the northeastern United States. Environ. Sci. Technol. 2008, 42, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Qi, H.; Liu, L.Y.; Jia, H.L.; Li, Y.F.; Ren, N.Q.; You, H.; Shi, X.Y.; Fan, L.L.; Ding, Y.S. Dechlorane plus in surficial water and sediment in a northeastern Chinese river. Environ. Sci. Technol. 2010, 44, 2305–2308. [Google Scholar] [CrossRef] [PubMed]
- Hoh, E.; Zhu, L.; Hites, R.A. Dechlorane Plus, a chlorinated flame retardant, in the Great Lakes. Environ. Sci. Technol. 2006, 40, 1184–1189. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Reiner, E.J.; MacPherson, K.A.; Kolic, T.M.; Helm, P.A.; Richman, L.A.; Marvin, C.H.; Burniston, D.A.; Hill, B.; Brindle, I.D.; et al. Dechloranes 602, 603, 604, Dechlorane Plus, and Chlordene Plus, a Newly Detected Analogue, in Tributary Sediments of the Laurentian Great Lakes. Environ. Sci. Technol. 2011, 45, 693–699. [Google Scholar] [CrossRef] [PubMed]
- Möller, A.; Xie, Z.Y.; Cai, M.H.; Zhong, G.C.; Huang, P.; Cai, M.G.; Sturm, R.; He, J.F.; Ebinghaust, R. Polybrominated Diphenyl Ethers vs Alternate Brominated Flame Retardants and Dechloranes from East Asia to the Arctic. Environ. Sic. Technol. 2011, 45, 6793–6799. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, W.L.; Liu, L.Y.; Qi, H.; Sun, D.Z.; Shen, J.M.; Wang, D.G.; Li, Y.F. Dechlorane Plus in multimedia in northeastern Chinese urban region. Environ. Int. 2011, 37, 66–70. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Iino, F.; Huang, J.; Lu, Y.; Yu, G.; Morita, M. Dechlorane Plus pollution and inventory in soil of Huai’an City, China. Chemosphere 2010, 80, 1285–1290. [Google Scholar] [CrossRef] [PubMed]
- Torre, A.D.L.; Sverko, E.; Alaee, M.; Martínez, M.Á. Concentrations and sources of Dechlorane Plus in sewage sludge. Chemosphere 2011, 82, 692–697. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Luo, X.J.; Wu, J.P.; Liu, J.; Wang, J.; Chen, S.J.; Mai, B.X. Contaminant pattern and bioaccumulation of legacy and emerging organohalogen pollutants in the aquatic biota from an E-waste recycling region in South China. Environ. Toxicol. Chem. 2010, 29, 852–859. [Google Scholar] [CrossRef] [PubMed]
- Guerra, P.; Fernie, K.; Jiménez, B.; Pacepavicius, G.; Shen, L.; Reiner, E.; Eljarrat, E.; Barceló, D.; Alaee, M. Dechlorane Plus and related compounds in Peregrine Falcon (Falco peregrinus) eggs from Canada and Spain. Environ. Sci. Technol. 2011, 45, 1284–1290. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Wang, J.; Luo, X.J.; Tian, M.; He, L.Y.Y.; Yuan, J.G.; Mai, B.X.; Yang, Z. Y Dechlorane Plus in human hair from an e-waste recycling area in South China: Comparison with dust. Environ. Sci. Technol. 2010, 44, 9298–9303. [Google Scholar] [CrossRef] [PubMed]
- Ren, G.F.; Yu, Z.Q.; Ma, S.T.; Li, H.R.; Peng, P.; Sheng, G.Y.; Fu, J.M. Determination of Dechlorane Plus in serum from electronics dismantling workers in South China. Environ. Sci. Technol. 2009, 43, 9453–9457. [Google Scholar] [CrossRef] [PubMed]
- Sverko, E.; Tomy, G.T.; Reine, E.J.; Li, Y.Y.; Mcarry, B.E.; Arnot, J.A.; Law, R.J.; Hites, R.A. Dechlorane Plus and related compounds in the environment: A review. Environ. Sci. Technol. 2011, 45, 5088–5098. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.Q.; Wei, H.; Guo, J.H.; McLeod, C.; Li, A.; Sturchio, N.C. Historically and currently used dechloranes in the sediments of the Great Lakes. Environ. Sci. Technol. 2011, 45, 5156–5163. [Google Scholar] [CrossRef] [PubMed]
- Jia, H.L.; Sun, Y.Q.; Liu, X.J.; Yang, M.; Wang, D.G.; Qi, H.; Shen, L.; Sverko, E.; Reiner, E.J.; Li, Y.F. Concentration and bioaccumulation of dechlorane compounds in coastal environment of Northern China. Environ. Sci. Technol. 2011, 45, 2613–2618. [Google Scholar] [CrossRef] [PubMed]
- Tomy, G.T.; Pleskach, K.; Ismail, N.; Whittle, D.M.; Helm, P.A.; Sverko, E.; Zaruk, D.; Marvin, C.H. Isomer of dechlorane plus in Lake Winnipeg and Lake Ontario food webs. Environ. Sci. Technol. 2007, 41, 2249–2254. [Google Scholar] [CrossRef] [PubMed]
- Na, G.S.; Wei, W.; Zhou, S.Y.; Gao, H.; Ma, X.D.; Qiu, L.N.; Ge, L.K.; Bao, C.G.; Yao, Z.W. Distribution characteristics and indicator significance of Dechloranes in multi-matrices at Ny-Ålesund in the Arctic. J. Environ. Sci. 2015, 28, 8–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yao, Z.W.; Jiang, G.B.; Xu, H.Z. Distribution of organochlorine pesticides in seawater of the Bering and Chukchi Sea. Environ. Pollut. 2002, 116, 49–56. [Google Scholar] [PubMed]
- Yu, Z.Q.; Lu, S.Y.; Gao, S.T.; Wang, J.Z.; Li, H.R.; Zeng, X.Y.; Sheng, G.Y.; Fu, J.M. Levels and isomer profiles of Dechlorane Plus in the surface soils from e-waste recycling areas and industrial areas in South China. Environ. Pollut. 2010, 158, 2920–2925. [Google Scholar] [CrossRef] [PubMed]
- Syed, J.H.; Malik, R.N.; Li, J.; Wang, Y.; Xu, Y.; Zhang, G.; Jones, K.C. Levels, profile and distribution of Dechloran Plus (DP) and polybrominated diphenylethers (PBDEs) in the environment of Pakistan. Chemosphere 2013, 93, 1646–1653. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.Y.; Hites, R.A. Brominated flame retardants in tree bark from North America. Environ. Sci. Technol. 2006, 40, 3711–3716. [Google Scholar] [CrossRef] [PubMed]
- Simonich, S.L.; Hites, R.A. Relationships between socioeconomic indicators and concentrations of organochlorine pesticides in tree bark. Environ. Sci. Technol. 1997, 31, 999–1003. [Google Scholar] [CrossRef]
- Hermanson, M.H.; Hites, R.A. Polychlorinated biphenyls in tree bark. Environ. Sci. Technol. 1990, 24, 666–671. [Google Scholar] [CrossRef]
- Chen, J.W.; Zhao, H.M.; Gao, L.N.; Henkelmann, B.; Schramm, K.W. Atmospheric PCDD/F and PCB levels implicated by pine (Cedrusdeodara) needles at Dalian, China. Environ. Pollut. 2006, 144, 510–515. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhang, G.; Jones, K.C.; Li, X.D.; Peng, X.Z.; Qi, S.H. Compositional fractionation of polycyclic aromatic hydrocarbons (PAHs) in mosses (Hypnum plumaeformae WILS.) from the northern slope of Nanling Mountains, South China. Atmosp. Environ. 2005, 39, 5490–5499. [Google Scholar] [CrossRef]
- Wang, L.; Jia, H.L.; Liu, X.J.; Yang, M.; Hong, W.J.; Sun, Y.Q.; Qi, H.; Song, W.W.; Lin, J.; Li, Y.F. Dechloranes in a river in northeastern China: Spatial trends in multi-matrices and bio-accumulation in fish (Enchelyopuselongatus). Ecotoxicol. Environ. Saf. 2012, 84, 262–267. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, C.; Iglesias, K.; Bícego, M.C.; Taniguchi, S.; Sasaki, S.T.; Kandratavicius, N.; Venturini, N. Hydrocarbons in soil and meltwater stream sediments near Artigas Antarctic Research Station: Origin, sources and levels. Antarct. Sci. 2018, 30, 170–182. [Google Scholar] [CrossRef]
- Trevizani, T.H.; Petti, M.A.V.; Ribeiro, A.P.; Corbisier, T.N.; Figueira, R.C.L. Heavy metal concentrations in the benthic trophic web of Martel Inlet, Admiralty Bay (King George Island, Antarctica). Mar. Pollut. Bull. 2018, 130, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Klánová, J.; Matykiewiczová, N.; Máčka, Z.; Prošek, P.; Láska, K.; Klán, P. Persistent organic pollutants in soils and sediments from James Ross Island, Antarctica. Environ. Pollut. 2008, 152, 416–423. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.Q.; Zhang, S.J.; Li, X.H.; Luo, D.X.; Jing, C.Y. Dechloranes in lichens from the southeast Tibetan Plateau: Evidence of long-range atmospheric transport. Chemosphere 2016, 144, 446–451. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, J.T.; Choi, Y.J.; Barghi, M.; Yoon, Y.J.; Kim, J.H.; Kim, J.H.; Chang, Y.Y. Occurrence and distribution of old and new halogenated flame retardants in mosses and lichens from the South Shetland Islands, Antarctica. Environ. Pollut. 2018, 235, 302–311. [Google Scholar] [CrossRef] [PubMed]
- Qiu, X.H.; Marvin, C.H.; Hites, R.A. Dechlorane plus and other flame retardants in a sediment core from Lake Ontario. Environ. Sci. Technol. 2007, 41, 6014–6019. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.P.; Zhang, Y.; Luo, X.J.; Wang, J.; Chen, S.J.; Guan, Y.T.; Mai, B.X. Isomer-specific bioaccumulation and trophic transfer of dechlorane plus in the freshwater food web from a highly contaminated site, South China. Environ. Sci. Technol. 2010, 44, 606–611. [Google Scholar] [CrossRef] [PubMed]
- Xian, Q.M.; Siddique, S.; Li, T.; Feng, Y.L.; Takser, L.; Zhu, J.P. Sources and environmental behavior of dechlorane plus-a review. Environ. Int. 2011, 37, 1273–1284. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.X.; Xu, X.R.; Hao, Q.; Luo, X.J.; Ruan, W.; Zhang, Z.W.; Zhang, Q.; Zou, F.S.; Mai, B.X. Species-specific accumulation of halogenated flame retardants in eggs of terrestrial birds from an ecological station in the Pearl River Delta, South China. Chemosphere 2014, 95, 442–447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tomy, G.T.; Thomas, C.R.; Zidane, T.M.; Murison, K.E.; Pleskach, K.; Hare, J.; Sverko, E.D. Examination of isomer specific bioaccumulation parameters and potential in vivo hepatic metabolites of syn-and anti-Dechlorane Plus isomers in juvenile rainbow trout (Oncorhynchus mykiss). Environ. Sci. Technol. 2008, 42, 5562–5567. [Google Scholar] [CrossRef] [PubMed]


| Sampling Area | Sample Number | Soil (pg/g dw) | Sample Number | Lichen (pg/g dw) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 602 | 603 | 604 | DP | 602 | 603 | 604 | DP | |||||
| Coastal area | 12 | FD (%) | 33 | 25 | 75 | 100 | 9 | FD (%) | 22 | 22 | 78 | 100 |
| Range | nd–7.04 | nd–43.10 | nd–483.22 | 97.18–336.42 | Range | nd–1.72 | nd–62.91 | nd–309.70 | 213.29–757.12 | |||
| Mean | 2.31 ± 2.01 | 21.01 ± 10.89 | 187.85 ± 147.60 | 232.58 ± 88.96 | Mean | 0.65 ± 0.44 | 18.09 ± 23.90 | 69.64 ± 93.81 | 449.32 ± 213.26 | |||
| Inland area | 5 | FD (%) | 40 | 60 | 40 | 100 | 6 | FD (%) | 17 | 33 | 83 | 100 |
| Range | nd–5.56 | nd–74.25 | nd–116.85 | 97.37–316.08 | Range | nd–3.56 | nd–24.74 | nd–130.76 | 175–659.24 | |||
| Mean | 2.49 ± 2.01 | 38.20 ± 25.02 | 52.89 ± 38.94 | 200.54 ± 86.48 | Mean | 0.97 ± 1.27 | 11.27 ± 8.28 | 70.16 ± 46.29 | 337.47 ± 171.90 | |||
| Ardley island | 3 | FD (%) | 67 | 0 | 67 | 100 | 3 | FD (%) | 33 | 0 | 100 | 100 |
| Range | nd–3.05 | nd | nd–293.87 | 57.86–319.71 | Range | nd–1.47 | nd | 142.14–543.75 | 393.80–3048.91 | |||
| Mean | 2.33 ± 1.06 | nd | 136.73 ± 139.39 | 150.43 ± 146.82 | Mean | 0.79 ± 0.59 | nd | 294.26 ± 217.79 | 1513.03 ± 1375.72 | |||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gao, H.; Na, G.; Yao, Y.; Li, R.; Gao, Y.; Zhang, Z.; Yao, Z. Distribution Characteristics and Source of Dechloranes in Soil and Lichen of the Fildes Peninsula (Antarctica). Int. J. Environ. Res. Public Health 2018, 15, 2312. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph15102312
Gao H, Na G, Yao Y, Li R, Gao Y, Zhang Z, Yao Z. Distribution Characteristics and Source of Dechloranes in Soil and Lichen of the Fildes Peninsula (Antarctica). International Journal of Environmental Research and Public Health. 2018; 15(10):2312. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph15102312
Chicago/Turabian StyleGao, Hui, Guangshui Na, Yao Yao, Ruijing Li, Yuhang Gao, Zhifeng Zhang, and Ziwei Yao. 2018. "Distribution Characteristics and Source of Dechloranes in Soil and Lichen of the Fildes Peninsula (Antarctica)" International Journal of Environmental Research and Public Health 15, no. 10: 2312. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph15102312