Reference Intervals for Urinary Cotinine Levels and the Influence of Sampling Time and Other Predictors on Its Excretion Among Italian Schoolchildren
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population and Design
2.2. Sample Collection and Analytical Determinations
2.3. Covariates Gathered by the Questionnaires
- 1 = thinness of 1st, 2nd or 3rd degree;
- 2 = normal weight;
- 3 = overweight;
- 4 = obesity.
2.4. Statistical Analysis
3. Results
3.1. Characteristics of the Study Population
3.2. Reference Values of u-Cotinine Levels
3.3. The Contribution of the Covariates on u-Cotinine Levels
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- U.S. Department of Health and Human Services. The Health Consequences of Smoking: 50 Years of Progress: A Report of the Surgeon General; U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, Coordinating Center for Health Promotion, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health: Atlanta, GA, USA, 2014.
- Meeker, J.D.; Benedict, M.D. Infertility, pregnancy loss and adverse birth outcomes in relation to maternal secondhand tobacco smoke exposure. Curr. Womens Health Rev. 2013, 9, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Au, W.W. Children’s susceptibility to environmental toxicants. Int. J. Hyg. Environ. Health 2002, 205, 501–503. [Google Scholar] [CrossRef] [PubMed]
- Behbod, B.; Sharma, M.; Baxi, R.; Roseby, R.; Webster, P. Family and carer smoking control programmes for reducing children’s exposure to environmental tobacco smoke. Cochrane Database Syst. Rev. 2018, 31, CD001746. [Google Scholar] [CrossRef] [PubMed]
- Campo, L.; Polledri, E.; Bechtold, P.; Gatti, G.; Ranzi, A.; Lauriola, P.; Goldoni, C.A.; Bertazzi, P.A.; Fustinoni, S. Determinants of active and environmental exposure to tobacco smoke and upper reference value of urinary cotinine in not exposed individuals. Environ. Res. 2016, 148, 154–163. [Google Scholar] [CrossRef] [PubMed]
- Protano, C.; Andreoli, R.; Manini, P.; Guidotti, M.; Vitali, M. A tobacco-related carcinogen: Assessing the impact of smoking behaviours of cohabitants on benzene exposure in children. Tob. Control 2012, 21, 325–329. [Google Scholar] [CrossRef] [PubMed]
- Protano, C.; Andreoli, R.; Manini, P.; Vitali, M. How home-smoking habits affect children: A cross-sectional study using urinary cotinine measurement in Italy. Int. J. Public Health 2012, 57, 885–892. [Google Scholar] [CrossRef] [PubMed]
- Lupsa, I.R.; Nunes, B.; Ligocka, D.; Gurzau, A.E.; Jakubowski, M.; Casteleyn, L.; Aerts, D.; Biot, P.; Den Hond, E.; Castaño, A.; et al. Urinary cotinine levels and environmental tobacco smoke in mothers and children of Romania, Portugal and Poland within the European human biomonitoring pilot study. Environ. Res. 2015, 141, 106–117. [Google Scholar] [CrossRef] [PubMed]
- Andreoli, R.; Protano, C.; Manini, P.; De Palma, G.; Goldoni, M.; Petyx, M.; Rondinone, B.M.; Vitali, M.; Mutti, A. Association between environmental exposure to benzene and oxidative damage to nucleic acids in children. Med. Lav. 2012, 103, 324–337. [Google Scholar] [PubMed]
- Protano, C.; Guidotti, M.; Manini, P.; Petyx, M.; La Torre, G.; Vitali, M. Benzene exposure in childhood: Role of living environments and assessment of available tools. Environ. Int. 2010, 36, 779–787. [Google Scholar] [CrossRef] [PubMed]
- Protano, C.; Andreoli, R.; Manini, P.; Vitali, M. Urinary trans, trans-muconic acid and S-phenylmercapturic acid are indicative of exposure to urban benzene pollution during childhood. Sci. Total Environ. 2012, 435–436, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Protano, C.; Andreoli, R.; Mutti, A.; Petti, S.; Vitali, M. Biomarkers of oxidative stress to nucleic acids: Background levels and effects of body mass index and life-style factors in an urban paediatric population. Sci. Total Environ. 2014, 500–501, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Protano, C.; Astolfi, M.L.; Canepari, S.; Vitali, M. Urinary levels of trace elements among primary school-aged children from Italy: The contribution of smoking habits of family members. Sci. Total Environ. 2016, 557–558, 378–385. [Google Scholar] [CrossRef] [PubMed]
- Protano, C.; Valeriani, F.; Macedonio, A.; Cammarota, F.; Romano Spica, V.; Orsi, G.B.; Vitali, M. Family-based social determinants and child health: A cross-sectional study. Pediatr. Int. 2017, 59, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Protano, C.; Canepari, S.; Astolfi, M.L.; De Meo, S.D.; Vitali, M. Urinary reference ranges and exposure profile for lithium among an Italian paediatric population. Sci. Total Environ. 2018, 619–620, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Manini, P.; De Palma, G.; Andreoli, R.; Poli, D.; Petyx, M.; Corradi, M.; Mutti, A.; Apostoli, P. Biological monitoring of low benzene exposure in Italian traffic policemen. Toxicol. Lett. 2008, 181, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Kroll, M.H.; Chesler, R.; Hagengruber, C.; Blank, D.W.; Kestner, J.; Rawe, M. Automated determination of urinary creatinine without sample dilution: Theory and practice. Clin. Chem. 1984, 32, 446–452. [Google Scholar]
- Cole, T.J.; Lobstein, T. Extended international (IOTF) body mass index cut-offs for thinness, overweight and obesity. Pediatr. Obes. 2012, 7, 284–294. [Google Scholar] [CrossRef] [PubMed]
- Frackiewicz, E.J.; Shiovitz, T.M.; Jhee, S.S. Ethnicity in Drug Development and Therapeutics; Cambridge University Press: Cambridge, UK, 2011. [Google Scholar]
- Andreoli, R.; Spatari, G.; Pigini, D.; Poli, D.; Banda, I.; Goldoni, M.; Riccelli, M.G.; Petyx, M.; Protano, C.; Vitali, M.; et al. Urinary biomarkers of exposure and of oxidative damage in children exposed to low airborne concentrations of benzene. Environ. Res. 2015, 142, 264–272. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, E.; Perez, R.; Hernandez, A.; Tejada, P.; Arteta, M.; Ramos, J.T. Factors and mechanisms for pharmacokinetic differences between pediatric population and adults. Pharmaceutics 2011, 3, 53–72. [Google Scholar] [CrossRef] [PubMed]
- Benowitz, N.L.; Hukkanen, J.; Jacob, P., 3rd. Nicotine chemistry, metabolism, kinetics and biomarkers. Handb. Exp. Pharmacol. 2009, 192, 29–60. [Google Scholar] [CrossRef]
- Ino, T.; Shibuya, T.; Saito, K.; Inaba, Y. Relationship between body mass index of offspring and maternal smoking during pregnancy. Int. J. Obes. (Lond.) 2012, 36, 554–558. [Google Scholar] [CrossRef] [PubMed]
- Robinson, O.; Martínez, D.; Aurrekoetxea, J.J.; Estarlich, M.; Somoano, A.F.; Íñiguez, C.; Santa-Marina, L.; Tardón, A.; Torrent, M.; Sunyer, J.; et al. The association between passive and active tobacco smoke exposure and child weight status among Spanish children. Obesity 2016, 24, 1767–1777. [Google Scholar] [CrossRef] [PubMed]
- Kermah, D.; Shaheen, M.; Pan, D.; Friedman, T.C. Association between secondhand smoke and obesity and glucose abnormalities: Data from the National Health and Nutrition Examination Survey (NHANES 1999–2010). BMJ Open Diabetes Res. Care 2017, 5, e000324. [Google Scholar] [CrossRef] [PubMed]
- Scherer, G.; Krämer, U.; Meger-Kossien, I.; Riedel, K.; Heller, W.D.; Link, E.; Gostomzyk, J.G.; Ring, J.; Behrendt, H. Determinants of children’s exposure to environmental tobacco smoke (ETS): A study in Southern Germany. J. Expo. Anal. Environ. Epidemiol. 2004, 14, 284–292. [Google Scholar] [CrossRef] [PubMed]
- Orton, S.; Jones, L.L.; Cooper, S.; Lewis, S.; Coleman, T. Predictors of children’s second-hand smoke exposure at home: A systematic review and narrative synthesis of the evidence. PLoS ONE 2014, 9, e112690. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yang, M.; Huang, Z.; Tian, L.; Niu, L.; Xiao, S. Urinary cotinine concentrations in preschool children showed positive associations with smoking fathers. Acta Paediatr. 2017, 106, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Protano, C.; Vitali, M. The new danger of third-hand smoke: Why passive smoking does not stop at secondhand smoke. Environ. Health Perspect. 2011, 119, A422. [Google Scholar] [CrossRef] [PubMed]
- Vitali, M.; Protano, C. How relevant are fathers who smoke at home to the passive smoking exposure of their children? Acta Paediatr. 2017, 106, 74. [Google Scholar] [CrossRef] [PubMed]
| Variable | Descriptives in % If Not Stated Otherwise (N) | |
|---|---|---|
| Age | Mean 8.60 (standard deviation 1.38) | |
| Gender | Male | 54.7 (N = 180) |
| Female | 45.3 (149) | |
| Exposure to environmental tobacco smoke | Not exposed | 63.6 (N = 208) |
| exposed | 36.4 (N = 119) | |
| Ponderal status according to body mass index | Thinness of 1st, 2nd or 3rd degree | 9.2 (N = 26) |
| Normal weight | 67.7 (N = 191) | |
| Overweight | 18.4 (N = 52) | |
| Obesity | 4.6 (N = 13) | |
| Maternal education (years) | Basic (≤9 years) | 14.1 (N = 46) |
| Upper secondary (≤14 years) | 44.8 (N = 146) | |
| Tertiary/higher (≥17 years) | 41.1 (N = 134) | |
| Paternal education (years) | Basic (≤9 years) | 20.7 (N = 66) |
| Upper secondary (≤14 years) | 41.4 (N = 132) | |
| Tertiary/higher (≥17 years) | 37.9 (N = 121) |
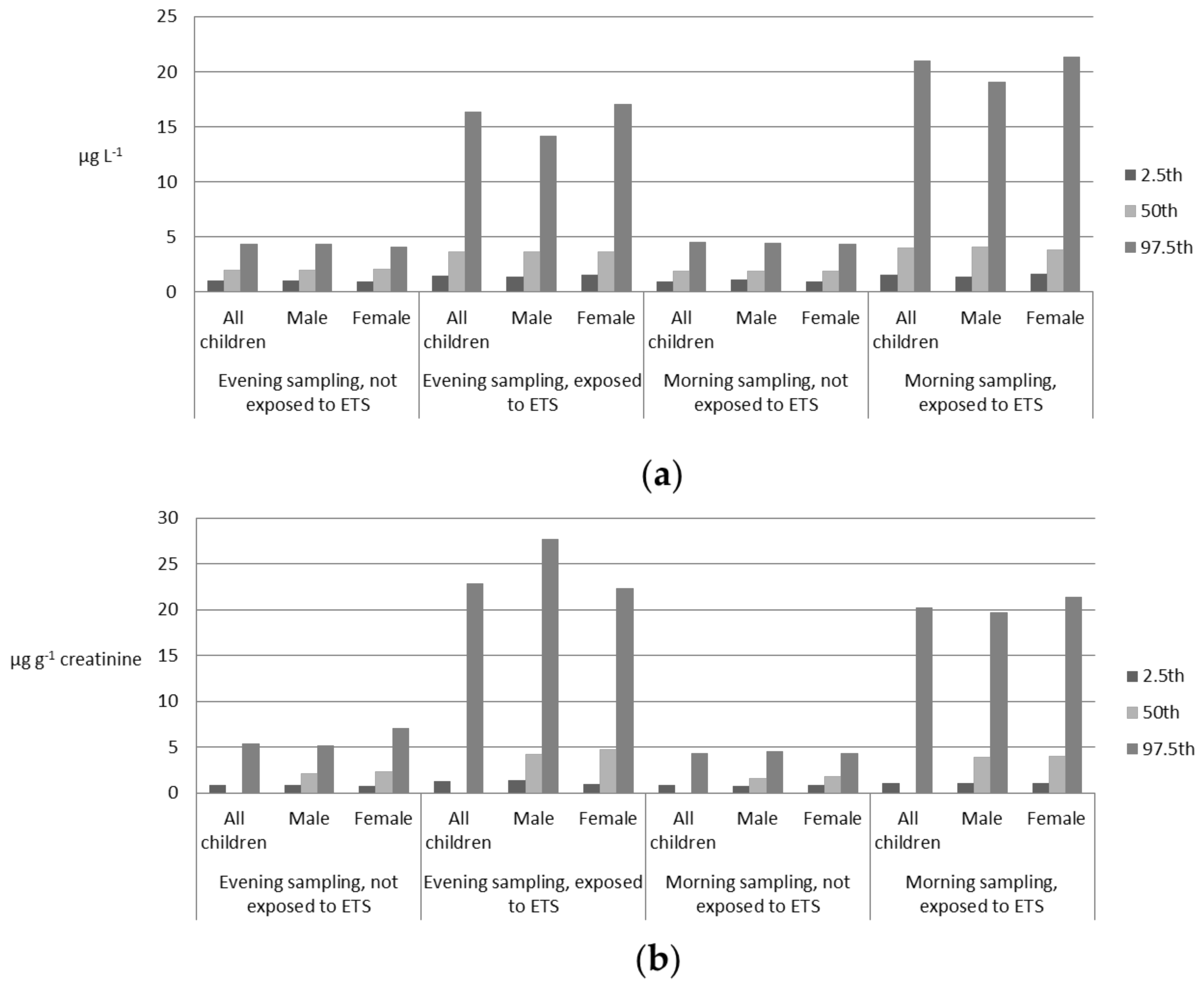
| Variable | 2.5th | 50th | 97.5th | |
|---|---|---|---|---|
| Evening sampling Not exposed to ETS | All children | 0.98 | 1.97 1,7 | 4.29 |
| Male | 0.98 | 1.92 2 | 4.36 | |
| Female | 0.93 | 2.03 2 | 4.04 | |
| Evening sampling Exposed to ETS | All children | 1.39 | 3.66 1,8 | 16.34 |
| Male | 1.34 | 3.63 3 | 14.14 | |
| Female | 1.52 | 3.66 3 | 17.03 | |
| Morning sampling Not exposed to ETS | All children | 0.91 | 1.83 4,7 | 4.50 |
| Male | 1.04 | 1.83 5 | 4.39 | |
| Female | 0.86 | 1.83 5 | 4.36 | |
| Morning sampling Exposed to ETS | All children | 1.49 | 3.97 4,8 | 20.95 |
| Male | 1.35 | 4.05 6 | 19.07 | |
| Female | 1.61 | 3.78 6 | 21.36 | |
| Variable | 2.5th | 50th | 97.5th | |
|---|---|---|---|---|
| Evening sampling Not exposed to ETS | All children | 0.85 | 2.20 1,7 | 5.41 |
| Male | 0.89 | 2.09 2 | 5.18 | |
| Female | 0.79 | 2.32 2 | 7.08 | |
| Evening sampling Exposed to ETS | All children | 1.33 | 4.35 1,8 | 22.88 |
| Male | 1.38 | 4.27 3 | 27.75 | |
| Female | 0.98 | 4.74 3 | 22.37 | |
| Morning sampling Not exposed to ETS | All children | 0.84 | 1.75 4,7 | 4.39 |
| Male | 0.80 | 1.65 5 | 4.55 | |
| Female | 0.84 | 1.83 5 | 4.39 | |
| Morning sampling Exposed to ETS | All children | 1.07 | 3.93 4,8 | 20.18 |
| Male | 1.05 | 3.89 6 | 19.67 | |
| Female | 1.06 | 4.05 6 | 21.37 | |
| Variable | u-Creatinine Spearman’s rho (p-Value) | Age Spearman’s rho (p-Value) | |
|---|---|---|---|
| Evening samples Not exposed to ETS | u-cotinine | 0.291 (<0.000) | −0.004 (0.959) |
| u-creatinine | 0.131 (0.059) | ||
| Morning samples Not exposed to ETS | u-cotinine | 0.301 (0.001) | −0.057 (0.418) |
| u-creatinine | 0.125 (0.073) | ||
| Evening samples Exposed to ETS | u-cotinine | 0.199 (0.030) | 0.086 (0.363) |
| u-creatinine | 0.172 (0.066) | ||
| Morning samples Exposed to ETS | u-cotinine | 0.084 (0.362) | 0.121 (0.196) |
| u-creatinine | 0.172 (0.066) | ||
| Variable | Evening Samples Median [IQR] GM [GSD] 1 | p-Value | Morning Samples Median [IQR] GM [GSD] | p-Value | |
|---|---|---|---|---|---|
| Ponderal status according to BMI | Thinness of 1st, 2nd or 3rd degree and normal weight | 2.17 [1.52] 2.35 [0.53] | 0.001 | 2.00 [0.58] 2.34 [0.59] | 0.009 |
| Overweight and obesity | 2.73 [2.23] 3.09 [0.68] | 2.77 [2.93] 2.96 [0.74] | |||
| Maternal education (years) | Until to upper secondary (≤14 years) | 2.46 [2.39] 2.89 [0.65] | <0.001 | 2.40 [2.91] 2.92 [0.73] | <0.001 |
| Tertiary/higher (≥17 years) | 2.07 [2.09] 2.14 [0.47] | 1.93 [0.81] 2.06 [0.49] | |||
| Paternal education (years) | Until to upper secondary (≤14 years) | 2.44 [2.44] 2.88 [0.67] | <0.001 | 2.35 [2.82] 2.88 [0.75] | <0.001 |
| Tertiary/higher (≥17 years) | 2.09 [1.04] 2.10 [0.41] | 1.95 [0.81] 2.03 [0.41] |
| Variable | Evening Samples Median [IQR] GM [GSD] 1 | p-Value | Morning Samples Median [IQR] GM [GSD] | p-Value | |
|---|---|---|---|---|---|
| Ponderal status according to BMI | Thinness of 1st, 2nd or 3rd degree and normal weight | 2.55 [2.05] 2.71 [0.61] | 0.131 | 2.02 [1.71] 2.24 [0.62] | 0.004 |
| Overweight and obesity | 2.86 [3.02] 3.17 [0.77] | 2.83 [2.76] 2.96 [0.73] | |||
| Maternal education (years) | Until to upper secondary (≤14 years) | 2.97 [3.06] 3.16 [0.75] | 0.002 | 2.47 [2.86] 2.77 [0.73] | 0.001 |
| Tertiary/higher (≥17 years) | 2.43 [1.60] 2.48 [0.53] | 1.88 [1.44] 2.06 [0.52] | |||
| Paternal education (years) | Until to upper secondary (≤14 years) | 2.91 [3.07] 3.17 [0.74] | 0.002 | 2.41 [2.70] 2.74 [0.74] | 0.001 |
| Tertiary/higher (≥17 years) | 2.41 [1.51] 2.39 [0.51] | 1.88 [1.46] 1.969 [0.45] |
| Independent Variable | B (Regression Coefficient) | Standard Error | (Regression Standardized Coefficient) | p-Value | Adjusted R2 | |
|---|---|---|---|---|---|---|
| Evening samples | Constant | 0.358 | 0.078 | - | <0.001 | 0.380 |
| Exposure to ETS | 0.726 | 0.059 | 0.594 | <0.001 | ||
| u-creatinine | 0.304 | 0.071 | 0.207 | <0.001 | ||
| Morning samples | Constant | 0.311 | 0.089 | - | 0.001 | 0.451 |
| Exposure to ETS | 0.852 | 0.061 | 0.641 | <0.001 | ||
| u-creatinine | 0.259 | 0.075 | 0.158 | 0.001 |
| Independent Variable | B (Regression Coefficient) | Standard Error | (Regression Standardized Coefficient) | p-Value | Adjusted R2 | |
|---|---|---|---|---|---|---|
| Evening samples | Constant | 0.954 | 0.168 | - | <0.001 | 0.131 |
| Ponderal status according to BMI | 0.272 | 0.131 | 0.202 | 0.041 | ||
| Paternal education (years) | −0.423 | 0.162 | −0.258 | 0.010 | ||
| u-creatinine | 0.429 | 0.169 | 0.253 | 0.013 | ||
| Morning samples | Constant | 1.112 | 0.209 | - | <0.001 | 0.089 |
| Ponderal status according to BMI | 0.239 | 0.143 | 0.169 | 0.039 | ||
| Paternal education (years) | −0.536 | 0.176 | −0.013 | 0.003 | ||
| u-creatinine | 0.386 | 0.177 | 0.224 | 0.031 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Protano, C.; Andreoli, R.; Mutti, A.; Manigrasso, M.; Avino, P.; Vitali, M. Reference Intervals for Urinary Cotinine Levels and the Influence of Sampling Time and Other Predictors on Its Excretion Among Italian Schoolchildren. Int. J. Environ. Res. Public Health 2018, 15, 817. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph15040817
Protano C, Andreoli R, Mutti A, Manigrasso M, Avino P, Vitali M. Reference Intervals for Urinary Cotinine Levels and the Influence of Sampling Time and Other Predictors on Its Excretion Among Italian Schoolchildren. International Journal of Environmental Research and Public Health. 2018; 15(4):817. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph15040817
Chicago/Turabian StyleProtano, Carmela, Roberta Andreoli, Antonio Mutti, Maurizio Manigrasso, Pasquale Avino, and Matteo Vitali. 2018. "Reference Intervals for Urinary Cotinine Levels and the Influence of Sampling Time and Other Predictors on Its Excretion Among Italian Schoolchildren" International Journal of Environmental Research and Public Health 15, no. 4: 817. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph15040817





