Vitamin D3 Supplementation Reduces the Symptoms of Upper Respiratory Tract Infection during Winter Training in Vitamin D-Insufficient Taekwondo Athletes: A Randomized Controlled Trial
Abstract
:1. Introduction
2. Methods
2.1. Participants
2.2. Study Design
2.3. Physique and Body Composition
2.4. Winter Training
2.5. Serum 25OH(D) Concentration Analysis
2.6. SIgA and Lactoferrin Analysis
2.7. Upper Respiratory Tract Infection Symptoms
2.8. Statistical Analysis
3. Results
3.1. Changes in Body Composition Variables
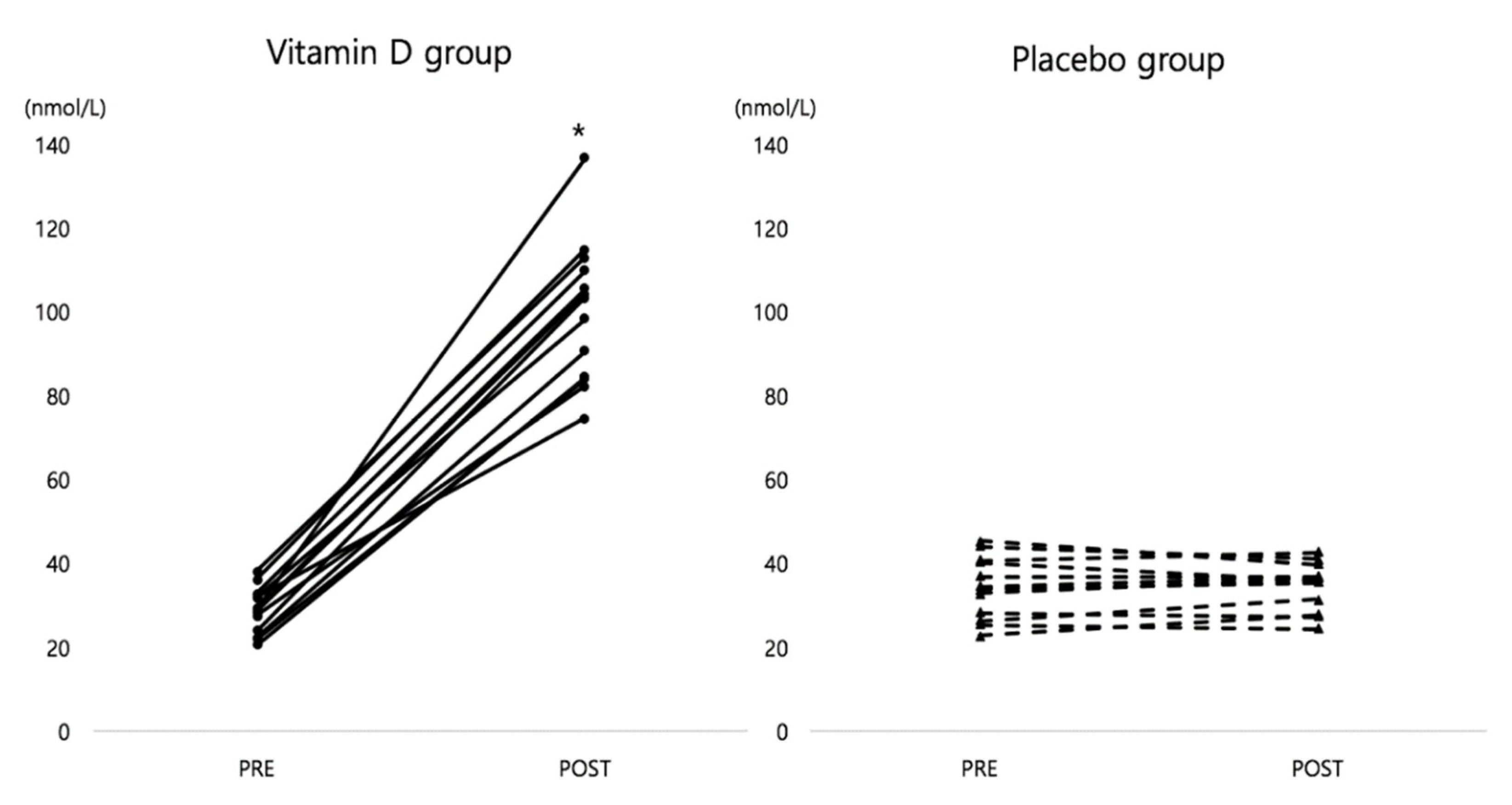
3.2. Change in Serum 25(OH)D Concentration
3.3. Changes in Salivary Immune Functions
3.4. Changes in URTI Symptoms
4. Discussion
4.1. Vitamin D Supplementation and Salivary Immune Functions
4.2. Vitamin D3 Supplementation and URTI Symptoms
5. Limitation of Study
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Da Silva Santos, J.F.; Valenzuela, T.H.; Franchini, E. Can different conditioning activities and rest intervals affect the acute performance of taekwondo turning kick? J. Strength Cond. Res. 2015, 29, 1640–1647. [Google Scholar] [CrossRef] [PubMed]
- Monks, L.; Seo, M.W.; Kim, H.B.; Jung, H.C.; Song, J.K. High-intensity interval training and athletic performance in Taekwondo athletes. J. Sports Med. Phys. Fit. 2017, 57, 1252–1260. [Google Scholar]
- Gleeson, M.; Williams, C. Intense exercise training and immune function. In Limits of Human Endurance; Karger Publishers: Basel, Switzerland, 2013; Volume 76, pp. 39–50. [Google Scholar]
- Walsh, N.P.; Oliver, S.J. Exercise, immune function and respiratory infection: An update on the influence of training and environmental stress. Immunol. Cell Biol. 2016, 94, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Nieman, D.C. Exercise, upper respiratory tract infection, and the immune system. Med. Sci. Sports Exerc. 1994, 26, 128–139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Engebretsen, L.; Soligard, T.; Steffen, K.; Alonso, J.M.; Aubry, M.; Budgett, R.; Dvorak, J.; Jegathesan, M.; Meeuwisse, W.H.; Mountjoy, M.; et al. Sports injuries and illnesses during the London Summer Olympic Games 2012. Br. J. Sports Med. 2013, 47, 407–414. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neville, V.J.; Molloy, J.; Brooks, J.H.; Speedy, D.B.; Atkinson, G. Epidemiology of injuries and illnesses in America’s Cup yacht racing. Br. J. Sports Med. 2006, 40, 304–312. [Google Scholar] [CrossRef] [PubMed]
- Soligard, T.; Steffen, K.; Palmer-Green, D.; Aubry, M.; Grant, M.E.; Meeuwisse, W.; Mountjoy, M.; Budgett, R.; Engebretsen, L. Sports injuries and illnesses in the Sochi 2014 Olympic Winter Games. Br. J. Sports Med. 2015, 49, 441–447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Owen, A.L.; Wong, D.P.; Dunlop, G.; Groussard, C.; Kebsi, W.; Dellal, A.; Morgans, R.; Zouhal, H. High-intensity training and salivary immunoglobulin responses in professional top-level soccer players: Effect of training intensity. J. Strength Cond. Res. 2016, 30, 2460–2469. [Google Scholar] [CrossRef] [PubMed]
- Spence, L.; Brown, W.J.; Pyne, D.B.; Nissen, M.D.; Sloots, T.P.; McCormack, J.G.; Locke, A.S.; Fricker, P.A. Incidence, etiology, and symptomatology of upper respiratory illness in elite athletes. Med. Sci. Sports Exerc. 2007, 39, 577–586. [Google Scholar] [CrossRef] [PubMed]
- MacKinnon, L.T. Special feature for the Olympics; effects of exercise on the immune system: Overtraining effects on immunity and performance in athletes. Immunol. Cell Biol. 2000, 78, 502–509. [Google Scholar] [CrossRef] [PubMed]
- He, C.S.; Handzlik, M.K.; Fraser, W.D.; Muhamad, A.S.; Preston, H.; Richardson, A.; Gleeson, M. Influence of vitamin D status on respiratory infection incidence and immune function during 4 months of winter training in endurance sport athletes. Exerc. Immunol. Rev. 2013, 19, 86–101. [Google Scholar] [PubMed]
- Schwellnus, M.; Soligard, T.; Alonso, J.M.; Bahr, R.; Clarsen, B.; Dijkstra, H.P.; Gabbett, T.J.; Gleeson, M.; Hägglund, M.; Hutchinson, M.R.; et al. How much is too much? (Part 2) International Olympic Committee consensus statement on load in sport and risk of illness. Br. J. Sports Med. 2016, 50, 1043–1052. [Google Scholar] [CrossRef] [PubMed]
- Soligard, T.; Schwellnus, M.; Alonso, J.M.; Bahr, R.; Clarsen, B.; Dijkstra, H.P.; Gabbett, T.J.; Gleeson, M.; Hägglund, M.; Hutchinson, M.R.; et al. How much is too much? (Part 1) International Olympic Committee consensus statement on load in sport and risk of injury. Br. J. Sports Med. 2016, 50, 1030–1041. [Google Scholar] [CrossRef] [PubMed]
- Gleeson, M.; Nieman, D.C.; Pedersen, B.K. Exercise, nutrition and immune function. J. Sports Sci. 2004, 22, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Walsh, N.P. Recommendations to maintain immune health in athletes. Eur. J. Sport Sci. 2018, 18, 820–831. [Google Scholar] [CrossRef] [PubMed]
- Dubnov-Raz, G.; Hemilä, H.; Cohen, A.H.; Rinat, B.; Choleva, L.; Constantini, N.W. Vitamin D supplementation and upper respiratory tract infections in adolescent swimmers: A randomized controlled trial. Pediatr. Exerc. Sci. 2015, 27, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, B.; Whiteley, R.; Farooq, A.; Chalabi, H. Vitamin D concentration in 342 professional football players and association with lower limb isokinetic function. J. Sci. Med. Sport 2014, 17, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Barcal, J.N.; Thomas, J.T.; Hollis, B.W.; Austin, K.J.; Alexander, B.M.; Larson-Meyer, D.E. Vitamin D and weight cycling: Impact on injury, illness, and inflammation in collegiate wrestlers. Nutrients 2016, 8, 775. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.C.; Seo, M.W.; Lee, S.; Jung, S.W.; Song, J.K. Correcting Vitamin D Insufficiency Improves Some, But Not All Aspects of Physical Performance during Winter Training in Taekwondo Athletes. Int. J. Sport Nutr. Exerc. Metab. 2018. [Google Scholar] [CrossRef] [PubMed]
- Berry, D.J.; Hesketh, K.; Power, C.; Hyppönen, E. Vitamin D status has a linear association with seasonal infections and lung function in British adults. Br. J. Nutr. 2011, 106, 1433–1440. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, P.T.; Stenger, S.; Li, H.; Wenzel, L.; Tan, B.H.; Krutzik, S.R.; Ochoa, M.T.; Schauber, J.; Wu, K.; Meinken, C.; et al. Toll-like receptor triggering of a vitamin D-mediated human antimicrobial response. Science 2016, 311, 1770–1773. [Google Scholar] [CrossRef] [PubMed]
- Autier, P.; Mullie, P.; Macacu, A.; Dragomir, M.; Boniol, M.; Coppens, K.; Pizot, C.; Boniol, M. Effect of vitamin D supplementation on non-skeletal disorders: A systematic review of meta-analyses and randomised trials. Lancet Diabetes Endocrinol. 2017, 5, 986–1004. [Google Scholar] [CrossRef]
- Martineau, A.R.; Jolliffe, D.A.; Hooper, R.L.; Greenberg, L.; Aloia, J.F.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; Ginde, A.A.; et al. Vitamin D supplementation to prevent acute respiratory tract infections: Systematic review and meta-analysis of individual participant data. BMJ 2017, 356, i6583. [Google Scholar] [CrossRef] [PubMed]
- He, C.S.; Fraser, W.D.; Tang, J.; Brown, K.; Renwick, S.; Rudland-Thomas, J.; Teah, J.; Tangueray, E.; Gleeson, M. The effect of 14 weeks of vitamin D3 supplementation on antimicrobial peptides and proteins in athletes. J. Sports Sci. 2016, 34, 67–74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seo, M.W.; Jung, H.C.; Song, J.K.; Kim, H.B. Effect of 8 weeks of pre-season training on body composition, physical fitness, anaerobic capacity, and isokinetic muscle strength in male and female collegiate taekwondo athletes. J. Exerc. Rehabil. 2015, 11, 101–107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Institute of Medicine. Dietary Reference Intakes for Calcium and Vitamin D; The National Academic Press: Washington, DC, USA, 2011. [Google Scholar]
- Jung, H.C.; Song, J.K. Decreased abdominal fat and improved bone metabolism after taekwondo training in obese adolescents. Kinesiology 2018, 50, 79–88. [Google Scholar] [CrossRef]
- Obasi, C.N.; Brown, R.L.; Barrett, B.P. Item reduction of the Wisconsin Upper Respiratory Symptom Survey (WURSS-21) leads to the WURSS-11. Qual. Life Res. 2014, 23, 1293–1298. [Google Scholar] [CrossRef] [PubMed]
- Fritz, C.O.; Morris, P.E.; Richler, J.J. Effect size estimates: Current use, calculations, and interpretation. J. Exp. Psychol. Gen. 2012, 141, 2–18. [Google Scholar] [CrossRef] [PubMed]
- Neville, V.; Gleeson, M.; Folland, J.P. Salivary IgA as a risk factor for upper respiratory infections in elite professional athletes. Med. Sci. Sports Exerc. 2008, 40, 1228–1236. [Google Scholar] [CrossRef] [PubMed]
- Mazanec, M.B.; Nedrud, J.G.; Kaetzel, C.S.; Lamm, M.E. A three-tiered view of the role of IgA in mucosal defense. Immunol. Today 1993, 14, 430–435. [Google Scholar] [CrossRef]
- Antualpa, K.; Aoki, M.S.; Moreira, A. Intensified training period increases salivary IgA responses but does not affect the severity of upper respiratory tract infection symptoms in prepuberal rhythmic gymnasts. Pediat. Exerc. Sci. 2018, 30, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Legrand, D.; Elass, E.; Pierce, A.; Mazurier, J. Lactoferrin and host defence: An overview of its immuno-modulating and anti-inflammatory properties. Biometals 2004, 17, 225–229. [Google Scholar] [CrossRef] [PubMed]
- Kunz, H.; Bishop, N.C.; Spielmann, G.; Pistillo, M.; Reed, J.; Ograjsek, T.; Park, Y.; Mehta, S.K.; Pierson, D.L.; Simpson, R.J. Fitness level impacts salivary antimicrobial protein responses to a single bout of cycling exercise. Eur. J. Appl. Physiol. 2015, 115, 1015–1027. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Glimvall, P.; Wickstro, C.; Jansson, H. Elevated levels of salivary lactoferrin, a marker for chronic periodontitis? J. Period. Res. 2012, 47, 655–660. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bergman, P.; Norlin, A.C.; Hansen, S.; Rekha, R.S.; Agerberth, B.; Björkhem-Bergman, L.; Ekström, L.; Lindh, J.D.; Andersson, J. Vitamin D3 supplementation in patients with frequent respiratory tract infections: A randomised and double-blind intervention study. BMJ Open 2012, 2, e001663. [Google Scholar] [CrossRef] [PubMed]
- Camargo, C.A.; Ganmaa, D.; Frazier, A.L.; Kirchberg, F.F.; Stuart, J.J.; Kleinman, K.; Sumberzui, N.; Rich-Edwards, J.W. Randomized trial of vitamin D supplementation and risk of acute respiratory tract infection in Mongolia. Pediatrics 2012, 130, e561–e567. [Google Scholar] [CrossRef] [PubMed]
- Laaksi, I.; Ruohola, J.P.; Mattila, V.; Auvinen, A.; Ylikomi, T.; Pihlajamäki, H. Vitamin D supplementation for the prevention of acute respiratory tract infection: A randomized, double-blinded trial among young Finnish men. J. Infect. Dis. 2010, 202, 809–814. [Google Scholar] [CrossRef] [PubMed]
- Murdoch, D.R.; Slow, S.; Chambers, S.T.; Jennings, L.C.; Stewart, A.W.; Priest, P.C.; Florkowski, C.M.; Livesey, J.H.; Camargo, C.A.; Scragg, R. Effect of vitamin D3 supplementation on upper respiratory tract infections in healthy adults: The VIDARIS randomized controlled trial. JAMA 2012, 308, 1333–1339. [Google Scholar] [CrossRef] [PubMed]
- De Koning, L.; Al-Turkmani, M.R.; Berg, A.H.; Shkreta, A.; Law, T.; Kellogg, M.D. Variation in clinical vitamin D status by DiaSorin Liaison and LC-MS/MS in the presence of elevated 25-OH vitamin D2. Clin. Chim. Acta 2013, 415, 54–58. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.W.; Jian, W.; Sullivan, S.; Sankaran, B.; Edom, R.W.; Weng, N.; Sharkey, D. Development and validation of an LC-MS/MS based method for quantification of 25 hydroxyvitamin D2 and 25 hydroxyvitamin D3 in human serum and plasma. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2014, 961, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Binkley, N.; Krueger, D.C.; Morgan, S.; Wiebe, D. Current status of clinical 25-hydroxyvitamin D measurement: An assessment of between-laboratory agreement. Clin. Chim. Acta 2010, 411, 1976–1982. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wagner, D.; Hanwell, H.E.; Vieth, R. An evaluation of automated methods for measurement of serum 25-hydroxyvitamin D. Clin. Biochem. 2009, 42, 1549–1556. [Google Scholar] [CrossRef] [PubMed]
- Nieman, D.C.; Henson, D.A.; Austin, M.D.; Sha, W. Upper respiratory tract infection is reduced in physically fit and active adults. Br. J. Sports Med. 2011, 45, 987–992. [Google Scholar] [CrossRef] [PubMed]
- Cox, A.J.; Gleeson, M.; Pyne, D.B.; Callister, R.; Hopkins, W.G.; Fricker, P.A. Clinical and laboratory evaluation of upper respiratory symptoms in elite athletes. Clin. J. Sport Med. 2008, 18, 438–445. [Google Scholar] [CrossRef] [PubMed]



| Variables | Group | Pre | Post | F-value | ||
|---|---|---|---|---|---|---|
| G | T | G × T | ||||
| Body weight (kg) | Vitamin D | 78.6 ± 3.28 | 76.7 ± 2.98 + | 1.788 | 27.768 * | 2.080 |
| Placebo | 72.5 ± 2.84 | 71.5 ± 2.77 + | ||||
| Lean body mass (kg) | Vitamin D | 64.2 ± 2.12 | 64.1 ± 2.02 | 3.147 | 1.011 | 1.257 |
| Placebo | 58.7 ± 1.98 | 59.3 ± 2.02 | ||||
| Fat mass (kg) | Vitamin D | 9.9 ± 1.19 | 9.3 ± 1.16 + | 0.086 | 6.916 * | 0.000 |
| Placebo | 9.5 ± 0.82 | 8.9 ± 0.67 + | ||||
| BFP (%) | Vitamin D | 12.7 ± 0.99 | 11.8 ± 0.93 + | 0.237 | 39.894 * | 0.106 |
| Placebo | 13.4 ± 0.67 | 12.3 ± 0.55 + | ||||
| Variables | Vitamin D | Placebo | U-value | p-value |
|---|---|---|---|---|
| URTI symptom (score/day) | 7.7 ± 1.06 | 13.0 ± 1.60 | 10.5 | 0.011 |
| QOL (score/day) | 7.0 ±1.00 | 9.7 ± 2.27 | 18.0 | 0.093 |
| Total URTI symptom (score/day) | 14.7 ± 1.64 | 22.7 ± 2.27 | 11.0 | 0.015 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jung, H.C.; Seo, M.-W.; Lee, S.; Kim, S.W.; Song, J.K. Vitamin D3 Supplementation Reduces the Symptoms of Upper Respiratory Tract Infection during Winter Training in Vitamin D-Insufficient Taekwondo Athletes: A Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2018, 15, 2003. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph15092003
Jung HC, Seo M-W, Lee S, Kim SW, Song JK. Vitamin D3 Supplementation Reduces the Symptoms of Upper Respiratory Tract Infection during Winter Training in Vitamin D-Insufficient Taekwondo Athletes: A Randomized Controlled Trial. International Journal of Environmental Research and Public Health. 2018; 15(9):2003. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph15092003
Chicago/Turabian StyleJung, Hyun Chul, Myong-Won Seo, Sukho Lee, Sung Woo Kim, and Jong Kook Song. 2018. "Vitamin D3 Supplementation Reduces the Symptoms of Upper Respiratory Tract Infection during Winter Training in Vitamin D-Insufficient Taekwondo Athletes: A Randomized Controlled Trial" International Journal of Environmental Research and Public Health 15, no. 9: 2003. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph15092003





