Once Resin Composites and Dental Sealants Release Bisphenol-A, How Might This Affect Our Clinical Management?—A Systematic Review
Abstract
:1. Introduction
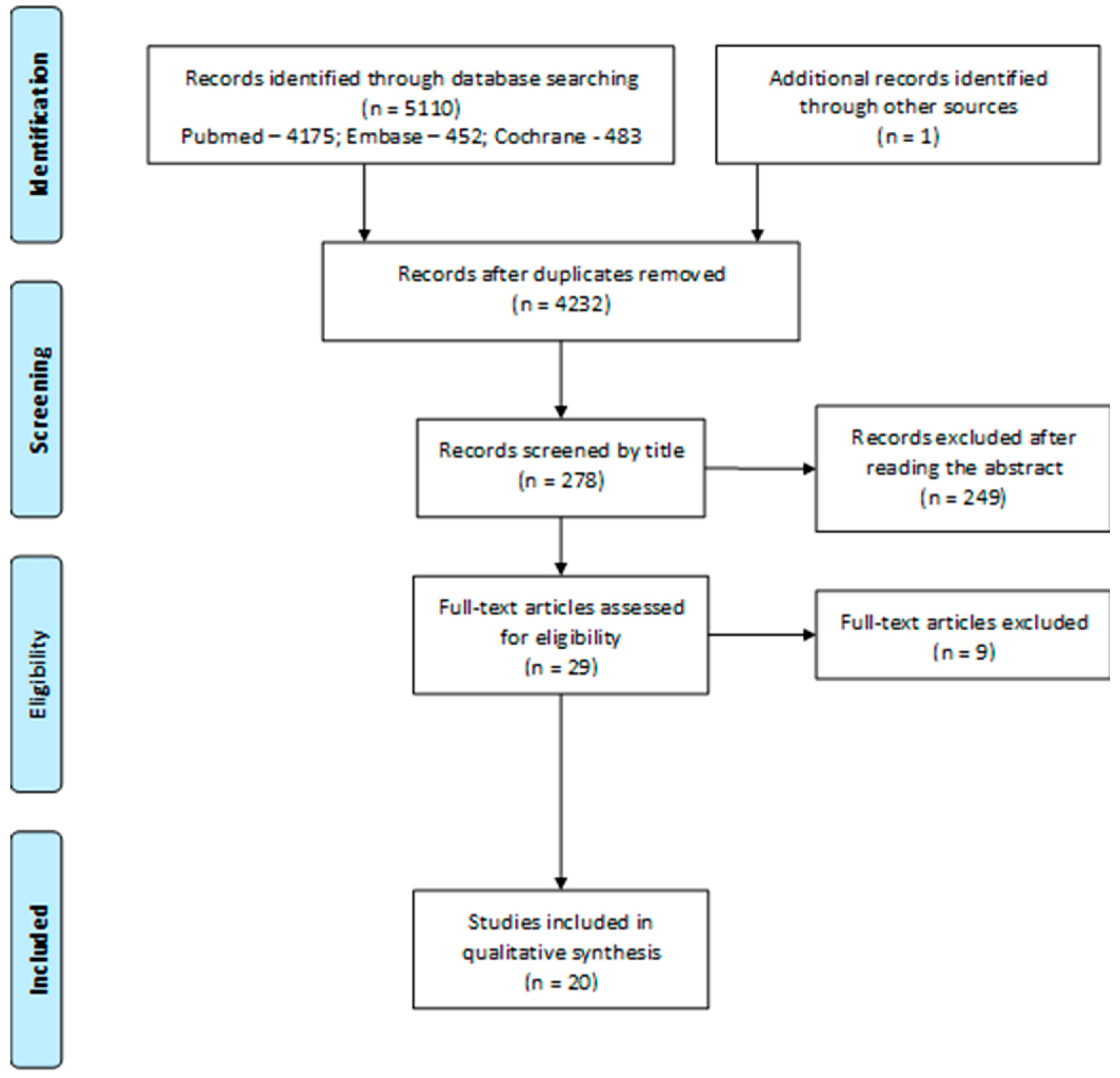
2. Materials and Methods
2.1. Search Strategy
2.2. Inclusion and Exclusion Criteria
2.3. Data Extraction
2.4. Quality Assessment
3. Results
3.1. Study Characteristics
3.2. Methodological Quality Assessment of Included Studies
4. Discussion
5. Conclusions
- (1)
- Resin composition used should be considered since some monomers have more estrogenic effects than others. Bis-GMA is preferred over Bis-DMA. However, the choice becomes difficult because most composite resins and sealants have several different monomers in their composition. The safety data sheets must be as complete as possible so that the percentage of each of the monomers can be evaluated and based on this information for the clinician to make the best choice.
- (2)
- Restorations or sealants must be done with a rubber dam to minimize their dissolution in the saliva.
- (3)
- To eliminate the last layer of resin unpolymerized by oxygen, a glycerin gel barrier must be placed prior to polymerization or alternatively surface polishing with a pumice or cotton applicator, or at least one air/water spray wash for 30 s should be carried out.
- (4)
- The patient should do a 30 s mouthwash after treatment because it is essential to introduce measures to dilute it for better patient safety.
- (5)
- Choose photopolymerizable composite resins instead of self-curing ones, for example in the application of an orthodontic appliance.
- (6)
- Special attention should be given to treatments in children, adolescents and pregnant women due to the high estrogenic and teratogenic level of BPA. For these patients, all the clinical precautions suggested should be taken simultaneously. For pregnant women, the postponement of treatment should be a clinical consideration, especially in the first trimester of pregnancy.
- (7)
- Do the smallest possible number of treatments in a single appointment to reduce the potential increase in BPA release. Do not perform more than four treatments per appointment, both restorations and sealants.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Joskow, R.; Barr, D.B.; Barr, J.R.; Calafat, A.M.; Needham, L.L.; Rubin, C. Exposure to bisphenol A from bis-glycidyl dimethacrylate–based dental sealants. J. Am. Dent. Assoc. 2006, 137, 353–362. [Google Scholar] [CrossRef]
- De Nys, S.; Putzeys, E.; Vervliet, P.; Covaci, A.; Boonen, I.; Elskens, M.; Vanoirbeek, J.; Godderis, L.; Van Meerbeek, B.; Van Landuyt, K.L.; et al. A novel high sensitivity UPLC-MS / MS method for the evaluation of bisphenol A leaching from dental materials. Sci. Rep. 2018, 8, 6981. [Google Scholar] [CrossRef] [PubMed]
- Azarpazhooh, A.; Main, P.A. Is There a Risk of Harm or Toxicity in the Placement of Pit and Fissure Sealant Materials? J. Can. Dent. Assoc. 2008, 74, 179–183. [Google Scholar] [PubMed]
- Atabek, D.; Alacam, A.; Berkkan, A. The Effect of Temperature on Bisphenol: An Elution from Dental Resins The Effect of Temperature on Bisphenol: An Elution from Dental Resins. J. Contemp. Dent. Pract. 2014, 15, 576–580. [Google Scholar] [CrossRef]
- Moreira, M.R.; Matos, L.G.; De Souza, I.D.; Brigante, T.A.V.; Queiroz, M.E.C.; Romano, F.L.; Nelson-Filho, P.; Matsumoto, M.A.N. Bisphenol A release from orthodontic adhesives measured in vitro and in vivo with gas chromatography. Am. J. Orthod. Dentofac. Orthop. 2017, 151, 477–483. [Google Scholar] [CrossRef]
- Han, D.-H.; Kim, M.-J.; Jun, E.-J.; Kim, J.-B. Salivary bisphenol-A levels due to dental sealant/resin: A case-control study in Korean children. J. Korean Med. Sci. 2012, 27, 1098–1104. [Google Scholar] [CrossRef]
- Trasande, L.; Attina, T.M.; Blustein, J. Association between urinary bisphenol A concentration and obesity prevalence in children and adolescents. JAMA 2012, 308, 1113–1121. [Google Scholar] [CrossRef]
- Hayden, C.; Bowler, J.O.; Chambers, S.; Freeman, R.; Humphris, G.; Richards, D.; Cecil, J.E. Obesity and dental caries in children: A systematic review and meta-analysis. Community Dent. Oral Epidemiol. 2013, 41, 289–308. [Google Scholar] [CrossRef] [PubMed]
- McKinney, C.; Rue, T.; Sathyanarayana, S.; Martin, M.; Seminario, A.L.; DeRouen, T. Dental sealants and restorations and urinary bisphenol A concentrations in children in the 2003–2004 National Health and Nutrition Examination Survey. J. Am. Dent. Assoc. 2015, 145, 745–750. [Google Scholar] [CrossRef]
- Fung, E.Y.; Ewoldsen, N.O.; St Germain, H.A.; Marx, D.B.; Miaw, C.L.; Siew, C.; Chou, H.N.; Gruninger, S.E.; Meyer, D.M. Pharmacokinetics of bisphenol A released from a dental sealant. J. Am. Dent. Assoc. 2000, 131, 51–58. [Google Scholar] [CrossRef]
- Fleisch, A.F.; Sheffield, P.E.; Chinn, C.; Edelstein, B.L.; Landrigan, P.J. Bisphenol A and Related Compounds in Dental Materials Abby. Pediatrics 2010, 126, 760–768. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA Statement for Reporting Systematic Reviews and Meta-Analyses of Studies That Evaluate Health Care Interventions: Explanation and Elaboration. Ann. Intern. Med. 2009, 151, 65–94. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Int. J. Surg. 2010, 8, 336–341. [Google Scholar] [CrossRef] [Green Version]
- De Carvalho, A.; Silva, V.; Grande, A. Avaliação do risco de viés de ensaios clínicos randomizados pela ferramenta da colaboração Cochrane. Rev. Diagnóstico Trat. 2013, 18, 38–44. [Google Scholar]
- Sterne, J.A.; Hernán, M.A.; Reeves, B.C.; Savović, J.; Berkman, N.D.; Viswanathan, M.; Henry, D.; Altman, D.G.; Ansari, M.T.; Boutron, I.; et al. ROBINS-I: A tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016, 355, i4919. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef] [Green Version]
- Kingman, A.; Hyman, J.; Masten, S.A.; Jayaram, B.; Smith, C.; Eichmiller, F.; Arnold, M.C.; Wong, P.A.; Schaeffer, J.M.; Solanki, S.; et al. Bisphenol A and other compounds in human saliva and urine associated with the placement of composite restorations. J. Am. Dent. Assoc. 2012, 143, 1292–1302. [Google Scholar] [CrossRef]
- Kang, Y.-G.; Kim, J.-Y.; Kim, J.; Won, P.-J.; Nam, J.-H. Release of bisphenol A from resin composite used to bond orthodontic lingual retainers. Am. J. Orthod. Dentofac. Orthop. 2011, 140, 779–789. [Google Scholar] [CrossRef]
- Zimmerman-Downs, J.M.; Shuman, D.; Stull, S.C.; Ratzlaff, R.E. Bisphenol A blood and saliva levels prior to and after dental sealant placement in adults. J. Dent. Hyg. JDH 2010, 84, 145–150. [Google Scholar]
- Sasaki, N.; Okuda, K.; Kato, T.; Kakishima, H.; Okuma, H.; Abe, K.; Tachino, H.; Tuchida, K.; Kubono, K. Salivary bisphenol-A levels detected by ELISA after restoration with composite resin. J. Mater. Sci. Mater. Med. 2005, 16, 297–300. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.-Y.; Kwon, H.; Choi, Y.-H.; Karmaus, W.; Merchant, A.T.; Song, K.-B.; Sakong, J.; Ha, M.; Hong, Y.-C.; Kang, D. Dental composite fillings and bisphenol A among children: A survey in South Korea. Int. Dent. J. 2012, 62, 65–69. [Google Scholar] [CrossRef]
- Maserejian, N.N.; Trachtenberg, F.L.; Wheaton, O.B.; Calafat, A.M.; Ranganathan, G.; Kim, H.-Y.; Hauser, R. Changes in urinary bisphenol A concentrations associated with placement of dental composite restorations in children and adolescents. J. Am. Dent. Assoc. 2016, 147, 620–630. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-H.; Yi, S.-K.; Kim, S.-Y.; Kim, J.-S.; Son, S.-A.; Jeong, S.-H.; Kim, J.-B. Salivary bisphenol A levels and their association with composite resin restoration. Chemosphere 2017, 172, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Berge, T.L.L.; Lygre, G.B.; Jönsson, B.A.G.; Lindh, C.H.; Björkman, L. Bisphenol A concentration in human saliva related to dental polymer-based fillings. Clin. Oral Investig. 2017, 21, 2561–2568. [Google Scholar] [CrossRef] [PubMed]
- Raghavan, A.S.; Sathyanarayana, H.P.; Kailasam, V.; Padmanabhan, S. Comparative evaluation of salivary bisphenol A levels in patients wearing vacuum-formed and Hawley retainers: An in-vivo study. Am. J. Orthod. Dentofac. Orthop. 2017, 151, 471–476. [Google Scholar] [CrossRef] [PubMed]
- Manoj, M.K.; Ramakrishnan, R.; Babjee, S.; Nasim, R. High-performance liquid chromatography analysis of salivary bisphenol A levels from light-cured and chemically cured orthodontic adhesives. Am. J. Orthod. Dentofac. Orthop. 2018, 154, 803–808. [Google Scholar] [CrossRef]
- Arenholt-Bindslev, D.; Breinholt, V.; Preiss, A.; Schmalz, G. Time-related bisphenol-A content and estrogenic activity in saliva samples collected in relation to placement of fissure sealants. Clin. Oral Investig. 1999, 3, 120–125. [Google Scholar] [CrossRef]
- Michelsen, V.B.; Kopperud, H.B.M.; Lygre, G.B.; Björkman, L.; Jensen, E.; Kleven, I.S.; Svahn, J.; Lygre, H. Detection and quantification of monomers in unstimulated whole saliva after treatment with resin-based composite fillings in vivo. Eur. J. Oral Sci. 2012, 120, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Olea, N.; Pulgar, R.; Pérez, P.; Olea-Serrano, F.; Rivas, A.; Novillo-Fertrell, A.; Pedraza, V.; Soto, A.M.; Sonnenschein, C. Estrogenicity of resin-based composites and sealants used in dentistry. Environ. Health Perspect. 1996, 104, 298–305. [Google Scholar] [CrossRef]
- Maserejian, N.N.; Shrader, P.; Brown, O.A.; Trachtenberg, F.L.; Soncini, J.; Hauser, R.; Shenker, B.J. Dental sealants and composite restorations and longitudinal changes in immune function markers in children. Int. J. Paediatr. Dent. 2014, 24, 215–225. [Google Scholar] [CrossRef]
- Trachtenberg, F.L.; Shrader, P.; Barregard, L.; Maserejian, N.N. Dental composite materials and renal function in children. Br. Dent. J. 2014, 216, E4. [Google Scholar] [CrossRef] [PubMed]
- Jalal, N.; Surendranath, A.R.; Pathak, J.L.; Yu, S.; Chung, C.Y. Bisphenol A (BPA) the mighty and the mutagenic. Toxicol. Rep. 2018, 5, 76–84. [Google Scholar] [CrossRef]
- Abasaeed, R.; Kranz, A.M.; Rozier, R.G. The impact of the Great Recession on untreated dental caries among kindergarten students in North Carolina. J. Am. Dent. Assoc. 2013, 144, 1038–1046. [Google Scholar] [CrossRef]
- Aligne, C.A.; Moss, M.E.; Auinger, P.; Weitzman, M. Association of pediatric dental caries with passive smoking. JAMA 2003, 289, 1258–1264. [Google Scholar] [CrossRef] [PubMed]
- Kloukos, D.; Sifakakis, I.; Voutsa, D.; Doulis, I.; Eliades, G.; Katsaros, C.; Eliades, T. BPA qualtitative and quantitative assessment associated with orthodontic bonding in vivo. Dent. Mater. 2015, 31, 887–894. [Google Scholar] [CrossRef] [Green Version]
- Kloukos, D.; Pandis, N.; Eliades, T. In vivo bisphenol-A release from dental pit and fissure sealants: A systematic review. J. Dent. 2013, 41, 659–667. [Google Scholar] [CrossRef]
- Rodriguez-Farre, E.; Testai, E.; Bruzell, E.M.; Jong, W.H.D. The Safety of Dental Amalgam and Alternative Dental Restoration Materials for Patients and Users. In Regulatory Toxicology and Pharmacology; Elsevier: Amsterdam, The Netherlands, 2015; ISBN 978-92-79-35605-6. [Google Scholar]
- Scientific Committee on Emerging and Newly Identified Health Risks. The Safety of the Use of Bisphenol A in Medical Devices; European Commission: Brussels, Belgium, 2015; ISBN 978-92-79-30133-9. [Google Scholar]
- Małkiewicz, K.; Turło, J.; Marciniuk-kluska, A.; Grzech-leśniak, K.; Gąsior, M.; Kluska, M. Release of bisphenol A and its derivatives from orthodontic adhesive systems available on the European market as a potential health risk factor. Ann. Agric. Environ. Med. 2015, 22, 172–177. [Google Scholar] [CrossRef] [Green Version]
- Purushothaman, D.; Kailasam, V.; Chitharanjan, A.B. Bisphenol A release from orthodontic adhesives and its correlation with the degree of conversion. Am. J. Orthod. Dentofac. Orthop. 2015, 147, 29–36. [Google Scholar] [CrossRef]
- Sunitha, C.; Kailasam, V.; Padmanabhan, S.; Chitharanjan, A.B. Bisphenol A release from an orthodontic adhesive and its correlation with the degree of conversion on varying light-curing tip distances. Am. J. Orthod. Dentofac. Orthop. 2011, 140, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Eliades, T.; Voutsa, D.; Sifakakis, I.; Makou, M.; Katsaros, C. Release of bisphenol-A from a light-cured adhesive bonded to lingual fixed retainers. Am. J. Orthod. Dentofac. Orthop. 2011, 139, 192–195. [Google Scholar] [CrossRef]
- Becher, R.; Wellendorf, H.; Sakhi, A.K.; Samuelsen, J.T.; Thomsen, C.; Bølling, A.K.; Kopperud, H.M. Presence and leaching of bisphenol a (BPA ) from dental materials. Acta Biomater. Odontol. Scand. 2018, 4, 56–62. [Google Scholar] [CrossRef]
- Coelho, A.J.M. Endocrine disruptors and dental materials: Health implications associated with their use in Brazil. Cad. Saude Publica 2002, 18, 505–509. [Google Scholar] [CrossRef]
- Stoker, C.; Rey, F.; Rodriguez, H.; Ramos, J.G.; Sirosky, P.; Larriera, A.; Luque, E.H.; Muñoz-de-Toro, M. Sex reversal effects on Caiman latirostris exposed to environmentally relevant doses of the xenoestrogen bisphenol A. Gen. Comp. Endocrinol. 2003, 133, 287–296. [Google Scholar] [CrossRef]
- Markey, C.M.; Wadia, P.R.; Rubin, B.S.; Sonnenschein, C.; Soto, A.M. Long-Term Effects of Fetal Exposure to Low Doses of the Xenoestrogen Bisphenol-A in the Female Mouse Genital Tract1. Biol. Reprod. 2005, 72, 1344–1351. [Google Scholar] [CrossRef] [Green Version]
- Muñoz-de-Toro, M.; Markey, C.M.; Wadia, P.R.; Luque, E.H.; Rubin, B.S.; Sonnenschein, C.; Soto, A.M. Perinatal Exposure to Bisphenol-A Alters Peripubertal Mammary Gland Development in Mice. Endocrinology 2005, 146, 4138–4147. [Google Scholar] [CrossRef] [Green Version]
- Maffini, M.V.; Rubin, B.S.; Sonnenschein, C.; Soto, A.M. Endocrine disruptors and reproductive health: The case of bisphenol-A. Mol. Cell. Endocrinol. 2006, 254–255, 179–186. [Google Scholar] [CrossRef]
- Nakamura, D.; Yanagiba, Y.; Duan, Z.; Ito, Y.; Okamura, A.; Asaeda, N.; Tagawa, Y.; Li, C.; Taya, K.; Zhang, S.-Y.; et al. Bisphenol A may cause testosterone reduction by adversely affecting both testis and pituitary systems similar to estradiol. Toxicol. Lett. 2010, 194, 16–25. [Google Scholar] [CrossRef]
- Tanaka, M.; Kawamoto, T.; Matsumoto, H. Distribution of 14C-bisphenol A in pregnant and newborn mice. Dent. Mater. 2010, 26, e181–e187. [Google Scholar] [CrossRef] [PubMed]
- Darmani, H.; Al-Hiyasat, A.S. The effects of BIS-GMA and TEG-DMA on female mouse fertility. Dent. Mater. 2006, 22, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Schwengberg, S.; Bohlen, H.; Kleinsasser, N.; Kehe, K.; Seiss, M.; Walther, U.I.; Hickel, R.; Reichl, F.X. In vitro embryotoxicity assessment with dental restorative materials. J. Dent. 2005, 33, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Arossi, G.A.; Lehmann, M.; Dihl, R.R.; Reguly, M.L.; de Andrade, H.H.R. Induced DNA Damage by Dental Resin Monomers in Somatic Cells. Basic Clin. Pharmacol. Toxicol. 2010, 106, 124–129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arossi, G.A.; Dihl, R.R.; Lehmann, M.; Cunha, K.S.; Reguly, M.L.; de Andrade, H.H.R. In vivo genotoxicity of dental bonding agents. Mutagenesis 2008, 24, 169–172. [Google Scholar] [CrossRef] [PubMed]
- Durner, J.; Dębiak, M.; Bürkle, A.; Hickel, R.; Reichl, F.-X. Induction of DNA strand breaks by dental composite components compared to X-ray exposure in human gingival fibroblasts. Arch. Toxicol. 2011, 85, 143–148. [Google Scholar] [CrossRef]
- Lee, H.N.; Pokorny, C.D.; Law, S.; Pratt, M.; Sasseville, D.; Storrs, F.J. Cross-reactivity among epoxy acrylates and bisphenol F epoxy resins in patients with bisphenol A epoxy resin sensitivity. Am. J. Contact Dermat. 2002, 13, 108–115. [Google Scholar] [PubMed]
- Lyapina, M.; Krasteva, A.; Dencheva, M.; Tzekova, M.; Nikolov, G.; Yaneva-Deliverska, M.; Kisselova-Yaneva, A. Pilot study of contact sensitization to rubber allergens and bisphenol A amongst dental students. Int. J. Occup. Med. Environ. Health 2017, 30, 397–405. [Google Scholar] [CrossRef] [PubMed]
| Author/Year | Type of Study | Sample | Type of Treatment | Object of Study | BPA Sources/Materials | BPA Evaluation Methods | Follow- Up | Results | Notes |
|---|---|---|---|---|---|---|---|---|---|
| Kingman A et al., 2013 [17] | RCT | n = 172 (264 teeth = 120 mg resin/patient) Mean age— 43.9 ± 1.1 years | Composite restorations | Saliva (n = 151) Urine (n = 171) | G1—BPA G2—BPAHPE G3—TEGDMA G4—BADGE G5—Bis-DMA G6—Bis-GMA | Liquid Chromatography System Mass Spectrometer (Limits of detection: personnel derived statistically valid lower reporting limits) | T0—0–<1h (n = 151) T1—1–8 h (n = 44) T2—9–30 h (n = 107) | Saliva (ng/mL): T0; T1; T2 G1—0.43; 0.64; 0.41 G2—0.98; 120.50; 1.28 G3—0.70; 4.67; 0.75 G4—1.34; 1.30; 1.43 G5—0.62; 0.66; 0.58 G6—3.09; 198.65; 3.24 Urine (ng/mL): T0; T1; T2 G1—1.75; 1.05; 2.38 G2—1.09; 1.25; 1.10 G3—0.47; 0.50; 0.46 G4—0.61; 0.62; 0.62 G5—0.26; 0.23; 0.31 G6—3.24; 2.68; 3.60 | Lack of information on the dental adhesives used. Rubber dam effect on BPA concentrations was studied. Statistically significant (before/after) with BPAHPE and Bis-GMA |
| Kang, Y et al., 2011 [18] | RCT | n = 22 Mean age—21.5 years | Bonding of lingual retainers | Saliva Urine | Adper Single Bond2 + G1—Filtek Flow (Bis-GMA+TEGMA) G2—Z250 Universal Restorative (Bis-GMA + BIS-DEMA) | Liquid Chromatography System (Limit of Detection: 0.5 ng/mL) | T0—Before treatment T1—30 min* T2—1 day T3—1 week T4—1 month | Saliva (ng/mL): (n = 20) T0: G1 (0 ± 0.0000); G2 (0.8389 ± 2.2685) (n = 20) T1: G1 (2.3211 ± 2.2000); G2 (7.2676 ± 68186) (n = 19) T2: G1 (0.5525 ± 1.5627); G2 (0.3684 ± 1.2217) (n = 19) T3: G1 (0.0914 ± 0.2743); G2 (0.8502 ± 2.3258) (n = 20) T4: G1 (0 ± 0.0000); G2 (0 ± 0.0000) Urine (ng/mL): (n = 22) T0: G1 (0.7974 ± 1.6509); G2 (0.3284 ± 0.7064) (n = 22) T2: G1 (0.5897 ± 1.1459); G2 (4.1116 ± 6.3120) (n = 19) T3: G1 (0.6987 ± 1.0267); G2 (3.3291 ± 6.2734) (n = 22) T4: G1 (2.8113 ± 4.0100); G2 (0.7988 ± 1.5626) | *Only saliva was collected after 30 min. Water irrigation/pumice effect on BPA concentration was studied. |
| Zimmerman-Downs, J. et al., 2010 [19] | RCT | n = 30 (18–40 years) | Dental sealants | Saliva Blood | Delton® Pit & Fissure Sealant—Light Cure Opaque 91.2% ADM, 1% EDA G1—Low-dose G2—High-dose | BPA Enzyme Linked ImmunoSorbent Assay (ELISA) (Quantitative analysis ranges from 0.05 to 10 μg/L (ppb)) | T0—1 h before treatment After treatment— T1—1–3 h T2—3–24 h | Baseline Salivary BPA (both groups): 0.07–6.00 ng/mL T1—Low dose: 3.98 ng/mL; High dose: 9.08 ng/mL T2—significant decrease to baseline values No statistically significant difference between mean salivary BPA concentration levels in low or high-dose groups at 1 h prior (p = 0.4328) or 24 h post (p = 0.3283). | Low-dose group: One occlusal sealant application. High-dose group: Four occlusal sealant applications Blood serum did not contain BPA at any point in this study. |
| Sasaki et al., 2005 [20] | RCT | n = 21 | Composite restoration | Saliva | G2—Z 100 (Bis-GMA/TEGDMA); Toughwell (Bis-GMA) Beautifil (Bis-GMA/TEGDMA); Xeno CFII (Bis-GMA); Prodigy (Bis-GMA/TEGDMA); Cleafil ST (Bis-GMA/TEGDMA) G1—Progress (UDMA/TEGDMA); Palfique; Matafil Flo (UDMA/TEGDMA); Unifil S(UDMA); | BPA ELISA “EIKEN” Kit (Quantitative analysis ranges from 0.05 to 10 μg/L (ppb)) | T0—Before treatment T1—immediately after treatment | G1 T0—0.3–2.0 ng/mL (mean 0.87 ± 0.69 ng/mL); T1—21.0–60.1 ng/mL (mean 32.1 ± 16.27 ng/mL); After gargling—1.6–4.7 ng/mL (3.1 ± 1.47 ng/mL) G2 mean <40 ng/mL or lower | Water effect on BPA concentration was studied. |
| Chung et al., 2012 [21] | RCT | n = 495 (8–9 years) | Resin, sealant and resin composites | Urine | Without information | Classification by the number of composite resins and sealant surfaces (0, 1–5, 6–10 and 11+) Liquid Chromatography System (Creatinine-adjusted urinary BPA) (Limit of Detection: Without information) | Without information | Mean of surfaces—10.07 ± 8.44 The mean creatinine-adjusted urinary BPA concentration was 2.08 ± 3.81 μg/g creatinine; Children with 11 or more composite resin surfaces—2.67 μg creatinine Resin—0 surfaces (−0.65); 6–10 surfaces (−0.43); +11 surfaces (1.02) μg/g Sealants—0 surfaces (0.22); 6–10 surfaces (−0.63); +11 surfaces (9.13) μg/g Resin composites—0 surfaces (0.06); 6–10 surfaces (−0.49); +11 surfaces (2.68) μg/g | |
| Fung et al., 2000 [10] | RCT | n = 40 (20–55 years age) | Dental sealant | Saliva Blood | Delton Opaque Light cure Pit and fissure sealant | High-pressure liquid chromatography (HPLC) (Limit of Detection: 5 ppb) | Baseline—before treatment After treatment— 1 h, 3 h, 1 day, 3 days and 5 days | Low-dose—8 mg dental sealant on 1 surface High-dose—32 mg of sealant (8 mg on each four surfaces) BPA in some saliva specimens (5.8–105.6 ppb) collected at 1 h and 3 h. The BPA was not detectable beyond 3 h or in any of the serum specimens. For the 1- and 3-h saliva samples, the BPA concentration in the high-dose (32 mg) group was significantly greater than in the low-dose (8 mg) group (p < 0.05). | In the high-dose group, there was a significant decrease in saliva BPA concentrations from 1 to 3 h (p < 0.01). |
| Maserejian et al., 2016 [22] | RCT | n = 91 (Age—3–17 years) 1 s—43.9% 2 s—25.3% 3 s—17.6% 4 s—8.8% 6 s—2.2% 8 s—2.2% | Adhesive Resin composite Dental sealant | Urine | G1—resin restoration with adhesive and composite (69.2%) + fissure sealant if needed (38.5%). Z100 restorative (Bis-GMA) + Optibond bonding agent (Bis-GMA) + Embrace (Bis-GMA) | Solid-phase extraction-high performance liquid chromatography isotope-dilution tandem mass spectrometry (Limit of Detection: 0.1 ng/mL) | Baseline—before treatment—T0 After treatment— 24 h—T1 14 days—T2 6 months—T3 | (0 to 3 surfaces) ng/mL T0–T1(n = 89)—3.33 ± 3.84 to 5.04 ± 9.94 (+51.4%) T0–T2 (n = 81)—3.45 ± 3.97to 2.95 ± 4.09 (−14.5%) (3 to 5 surfaces) ng/mL T0–T1 (n = 26)—3.45 ± 3.35 to 2.86 ± 3.62 (−17.1%) T0–T2 (n = 15)—3.21 ± 2.36 to 2.93 ± 3.55 (−0.4%) (+6 surfaces) ng/mL T0–T1 (n = 3)—1.03 ± 0.53 to 3 ± 2.91 (+191.3%) T0–T2 (n = 5)—1.91 ± 1.38 to 1.64 ± 1.28 (−14.1%) T0–T3 (n = 77)—3.07 ± 3.01 to 3.36 ± 4.66 (+9.4%) | In 51.6% participants rubber dam was used. |
| McKinney et al., 2014 [9] | RCT | n = 1001 (Age—6–19 years) | Resin-based dental sealants and composites | Urine | Bisphenol A-glycidyl methacrylate | Without information | Without information | Lowest quartile had BPA concentrations of 0.3–1.9 ng/mL. Highest quartile had mean BPA concentrations of 7.3 to 149 ng/mL Children with 7–16 sealants—BPA +25%; +10 sealants— +11% higher (BPA) Children with 7–42 restorations had (BPA) 20% higher | It is not possible to conclude on the increase of urinary concentrations of BPA after the placement of sealants or restorations, nor is the time or sources of other exposures to BPA known |
| Lee et al., 2017 [23] | RCT | n = 30 (Mean age—40) | Composite Resin | saliva | Filtek Z350 XT | Ecologiena® supersensitive BPA ELISA Kit (Quantitative analysis ranges from 0.05 to 10 μg/L (ppb)) | Before treatment—T0; After treatment: 5 min—T1 and 7 days—T2 | BPA (ug/L) in saliva T0 n = 30, Mean—0.15 ± 0.42 (0 teeth—n = 20, Mean—0.18 ± 0.51; 1 tooth or more—n = 10, Mean—0.09 ± 0.09) T1 n = 30 Mean—3.64 ± 2.32 (1 tooth—n = 13, Mean—2.67 ± 2.32; 2 teeth or more—n = 17, Mean—4.38 ± 2.10) T2 n = 30 Mean—0.59 ± 1.27 (1 tooth—n = 13, Mean—0.32 ± 0.36; 2 Teeth or more—n = 17, Mean—0.79 ± 1.65) | The level of salivary BPA was not significantly influenced by the number of teeth or surfaces of teeth previously treated with the filling of composite resin. |
| Han et al., 2012 [6] | Case-control study | n = 302 n—62 with >4 surfaces; 62 controls Ages: Control—10.13 ± 2.14 Ages Experimental group—10.03 ± 2.09 | Dental sealant/resin | saliva | Without information | Ecologiena® supersensitive BPA ELISA Kit (Quantitative analysis ranges from 0.05 to 10 μg/L (ppb)) | Without information | 0 surface (n = 62 teeth) BPA, mean ± SD 0.42 ± 0.38 >4 surfaces (n = 62 teeth) BPA, mean ± SD 0.90 ± 1.80 Children with four or more surfaces with sealants or composite resin had higher BPA salivary values p = 0.239; after adjusting for confounding variables p = 0.026. Salivary BPA level was in the range of doses detectable and there may be a relationship between salivary BPA level and dental sealant/resin in Korean children. | Age, gender, salivary flow rate, salivary buffer capacity, snack frequency and brushing frequency were selected as confounding factors. |
| Moreira et al., 2017 [5] | RCT | n = 20 (Age 12–18) | Adhesive and composite resin | Urine Saliva | Transbond XT system (adhesive and resin) | Gas Chromatograph mass spectrometer (Limit of Detection: Without information) | T0—before treatment: T1—30 min. T2—24 h T3—1 week T4—1 month | BPA in saliva (ng.g−1): T0—0.56 ± 0.06 T1—1.04 ± 0.28 (p < 0.05) T2—0.64 ± 0.21 T3—0.76 ± 0.33 T4—0.61 ± 0.16 BPA in urine (ng.g−1): T0—2.17 ± 0.93 T1—5.04 ± 2.47 (p < 0.05) T2—4.22 ± 2.07 (p < 0.05) T3—3.05 ± 1.61 T4—2.17 ± 0.93 | Bonding brackets with the Transbond XT orthodontic adhesive system resulted in an increase in BPA levels in saliva and urine. The levels were significant, but lower than the reference dose for daily intake and decreased with time. |
| Berge et al., 2017 [24] | RCT | n = 40 (Age 20–35) Test G-20 (with six or more restorations) Control G-20 | Resin composite | Saliva | G Test—with six or more restorations) G Control—without intervention Score 1—class I and V restorations Score 2—small class II, III and IV restorations Score 3—extensive class II | Triple quadrupole linear ion trap mass spectrometer coupled to a liquid chromatography system (Limit of Detection: 0.1 ng/mL) | Without information | BPA total (ng/mL): G Test—0.11 (p = 0.302) G Control—no detection BPA free (ng/mL): G Test—0.12 (p = 0.044) G Control—no detection. Total BPA concentration was higher but not significantly higher in G test compared to G control There was no significant correlation between the size and number rest. and free BPA. | The presence of restorative material based on dental polymers was associated with the slightly elevated concentration of free BPA in saliva. |
| Raghavan et al., 2017 [25] | RCT | n = 45 | Retainer post-fixed orthodontic | Saliva | G1—Vacuum-formed retainer G2—Hawley retainer fabricated by heat cure G3—Hawley retainer fabricated by chemical cure | HPLC (Limit of Detection: Without information) | Before placement—T0 After placement— T1—1 h T2—7 days T3—30 days | BPA levels greater in G1 and G3 G1—increase BPA from T0 to T1 (+1.20 ppm); increase BPA from T1 to T2 (+1.18 ppm); decrease BPA from T2 to T3 (−2.18 ppm) G2 and G3—increase BPA from T0 to T1; decrease BPA from T1 to T2; increase BPA from T2 to T3 | |
| Joskow et al., 2006 [1] | Prospective Cohort | n = 15 (86 teeth nG1 = 30; nG2 = 56) | Dental sealants (7.36 mg/sealant) (40.35 mg/patient) | Saliva Urine | G1—Helioseal F G2—Delton Light Cure Opaque | Gas chromatograph-high resolution mass spectrometer (Limit of Detection: 0.1 ng/mL) | T0—Pretreatment T1—Immediately after treatment T2—1 h after treatment | G1—BPA levels 5.5 μg G2—BPA levels 110 μg Saliva (ng/mL) G1 (T0—0.22 ± 0.03; T1—0.54 ± 0.45; T2—0.21 ± 0.03) G2 (T0—0.34 ± 0.19; T1—42.8 ± 28.9; T2—7.86 ± 12.73) Urine (ng/mL) G1 (T0—2.12 ± 0.93; T1—7.26 ± 13.5; T2—2.06 ± 1.04) G2 (T0—2.6 ± 1.4; T1—27.3 ± 39.1; T2—7.34 ± 3.81) | Delton Light Cure Opaque is a sealant without the ADA seal of Acceptance |
| Manoj et al., 2018 [26] | RCT | n = 4 (Age—13–30 years) | Adhesive and resin composites (brackets bonded) | Saliva | G1—Unite no-mix adhesive G2—Transbond Xt light cure adhesive | HPLC/mass spectrometry method (Limit of Detection: 0.5 ng/mm3) | T0—before treatment T1—30 min. T2—1 day T3—1 week T4—1 month | G1—μg/mL T0—0.0 ± 0.0; T1—19.6 ± 8.0; T2—5.0 ± 1.3; T3—4.0 ± 1.3; T4—1.2 ± 0.8 G2—μg/mL T0—0.0 ± 0.0; T1—11.2 ± 4.2; T2—3.1 ± 1.0; T3—2.0 ± 1.0; T4—0.6 ± 0.32 | |
| Arenholt-Bindslev et al., 1999 [27] | RCT | n = 8 (Age—20–23 years) | Fissures sealants | Saliva | G1—Visio-Seal G2—Delton LC pit and fissure sealant Clear | HPLC (Limit of Detection: 0.1 ppm; Quantitation limit: 0.3 ppm) Estrogenic assay (spectrophotometrically) | T0—before treatment T1—1 min. T2—1 h T3—24 h | G1—ppm T0, T1, T2, T3—≤0.1 G2—ppm T0—≤0.1; T1—1.43; T2, T3—≤0.1 Dose-range relevant—maximum effect level 1.56 ppm) | BPA present in saliva after treatment with Delton LC. After 1 h neither BPA nor estrogenic activity could be detected. |
| Michelsen et al., 2012 [28] | RCT | n = 10 (Mean age— 54.5 ± 4.1) | Resin composites + Resin Adhesive (score size 0–3) (use rubber Dam) | Saliva | Filtek Z250 (TEGDMA 1%–5%, Bis-GMA 1%–5%, Bis-EMA 5%–10%, and UDMA 5%–10%) Scotchbond 1 (Bis-GMA 10%–30%, HEMA 5%–25%, and dimethacrylates 7%–28%) | Gas Chromatography combined with mass spectrometer Liquid Chromatography combined with mass spectrometer (Mass range Detection: 5–350 m/z) | T0—before treatment T1—10 min. T2—24 h T3—7 days | T1— HEMA—0.068 μg/mL−1 TEGDMA—0.00 μg/mL−1 Bis-GMA—2.149 μg/mL−1 UDMA—0.188 μg/mL−1 T0, T2 and T3—0.00 μg/mL | Patients were also asked not to use lipstick or lip balm, not to chew chewing gum, and not to eat pastilles or candy before sampling. |
| Olea et al., 1996 [29] | RCT | n = 18 (Range age—18–25) | Fissures sealants | Saliva | Bis-GMA (50 mg/Patient) | HPLC Gas Chromatography with Mass Spectrometer (Limit of Detection: Without information) | T0—before treatment T1—1 h | T1—90–931 μg/mL | unpolymerized material collected during 1 h after treatment never exceeded 2% of the total of sealant |
| Maserejian et al., 2014 [30] | Prospective Cohort | n = 534 (Age 6–10) | G1 Sealant G2 Preventive resin G3 Resto. on primary teeth G4 Resto on permanent teeth | Blood | G1 Ultraseal XT (bisGMA, diurethane dimethacrylate) G2 Revolution (bisGMA) G3 Dyract AP compomer (UDMA, trimethacrylate resins) G4 Z100 composite 3M ESPE (St. Paul, MN, USA) bisGMA, TEGDMA) G5 amalgam | Flow Cytometry (immune function) (i) white blood cell enumeration, (ii) T cell responsiveness, (iii) B cell responsiveness, and (iv) neutrophil and monocyte responsiveness. | T0—before treatment T1—5–7 days T2—6 months T3—12 months T4—18 months T5—5 years | Positive association—non-flowable BisGMA-based composites—changes in B cell activation (indicating increased activation), was present at 6 months and 1 year, but not at the 5-year visit. BisGMA-based flowable (sealant or non-flowable composites increased, monocyte and neutrophil functions were decreased at 6 months and 1 year, but not at year 5. | |
| Trachtenberg et al., 2014 [31] | Prospective Cohort | n = 534 (Age 6–10) | G1 Sealant G2 Preventive resin G3 Resto. on primary teeth G4 Resto on permanent teeth | Urine Blood | G1 Ultraseal XT (bisGMA, diurethane dimethacrylate) G2 Revolution (bisGMA) G3 Dyract AP compomer (UDMA, trimethacrylate resins) G4 Z100 composite 3M ESPE (St. Paul, MN, USA) bisGMA, TEGDMA) G5 amalgam | Flow Cytometry (renal function) Gamma-glutamyl transpeptidase (gamma-GT) Albumin and N-acetyl-β-D-glucosaminidase (NAG) | T0—before treatment T1—5–7 days T2—6 months T3—12 months T4—18 months T5—5 years | 5 years Composite restorations on permanent teeth was 10.4 ± 17.0 surface-years (range 0–15.1), to compomer restorations on primary teeth was 11.8 ± 18.1 surface-years (range 0–16.7), and to flowable composite sealants and PRRs was 39.9 ± 21.1 surface-years (range 25–54). There was no evidence that composite treatments were associated with impaired renal function. | OR of high albumin excretion decreased with increased exposure to composite restorations on permanent teeth OR of high NAG decreased with increased exposure to dental sealants and PRRs |
| Random Sequence Generation (Selection Bias) | Allocation Concealment(Selection Bias) | Blinding of Participants and Personnel (Performance Bias) | Blinding of Outcome Assessment (Detection Bias) | Incomplete Outcome Data(Attrition Bias) | Selective Reporting(Reporting Bias) | Other Bias | |
|---|---|---|---|---|---|---|---|
 |  |  |  |  |  |  | Kingman et al., 2012 [17] |
 |  |  |  |  |  |  | Kang et al., 2011 [18] |
 |  |  |  |  |  |  | Zimmerman-Downs et al., 2010 [19] |
 |  |  |  |  |  |  | Sasaki et al., 2005 [20] |
 |  |  |  |  |  |  | Chung et al., 2012 [21] |
 |  |  |  |  |  |  | Fung et al., 2000 [10] |
 |  |  |  |  |  |  | Maserejian et al., 2016 [22] |
 |  |  |  |  |  |  | McKinney et al., 2014 [9] |
 |  |  |  |  |  |  | Lee et al., 2017 [23] |
 |  |  |  |  |  |  | Moreira et al., 2017 [5] |
 |  |  |  |  |  |  | Berge et al., 2017 [24] |
 |  |  |  |  |  |  | Raghavan et al., 2017 [25] |
 |  |  |  |  |  |  | Manoj et al., 2018 [26] |
 |  |  |  |  |  |  | Arenholt-Bindslev et al., 1999 [27] |
 |  |  |  |  |  |  | Michelsen et al., 2012 [28] |
 |  |  |  |  |  |  | Olea et al., 1996 [29] |
 Low risk of bias;
Low risk of bias;  Unclear risk of bias;
Unclear risk of bias;  High risk of bias.
High risk of bias.| Pre-Intervention | At Intervention | Post-Intervention | Non-Randomized Studies of Interventions | ||||
|---|---|---|---|---|---|---|---|
| Bias Due to Confounding | Bias in Selection of Participants into the Study | Bias in Classification of Interventions | Bias Due to Deviations from Intended interventions | Bias Due to Missing Data | Bias in Measurement of Outcomes | Bias in Selection of the Reported Result | |
| Y/PY/PN/N/NI | Y/PY/PN/N/NI | Y/PY/PN/N/NI | Y/PY/PN/N/NI | Y/PY/PN/N/NI | Y/PY/PN/N/NI | Y/PY/PN/N/NI | Han et al., 2012 [6] |
| Y/PY/PN/N/NI | Y/PY/PN/N/NI | Y/PY/PN/N/NI | Y/PY/PN/N/NI | Y/PY/PN/N/NI | Y/PY/PN/N/NI | Y/PY/PN/N/NI | Joskow et al., 2006 [1] |
| Y/PY/PN/N/NI | Y/PY/PN/N/NI | Y/PY/PN/N/NI | Y/PY/PN/N/NI | Y/PY/PN/N/NI | Y/PY/PN/N/NI | Y/PY/PN/N/NI | Maserejian et al., 2014 [30] |
| Y/PY/PN/N/NI | Y/PY/PN/N/NI | Y/PY/PN/N/NI | Y/PY/PN/N/NI | Y/PY/PN/N/NI | Y/PY/PN/N/NI | Y/PY/PN/N/NI | Trachtenberg et al., 2014 [31] |
| Low risk | Low risk | Moderate risk | Low risk | Moderate risk | Moderate risk | Low-risk | RISK OF BIAS JUDGEMENTS |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paula, A.B.; Toste, D.; Marinho, A.; Amaro, I.; Marto, C.-M.; Coelho, A.; Marques-Ferreira, M.; Carrilho, E. Once Resin Composites and Dental Sealants Release Bisphenol-A, How Might This Affect Our Clinical Management?—A Systematic Review. Int. J. Environ. Res. Public Health 2019, 16, 1627. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph16091627
Paula AB, Toste D, Marinho A, Amaro I, Marto C-M, Coelho A, Marques-Ferreira M, Carrilho E. Once Resin Composites and Dental Sealants Release Bisphenol-A, How Might This Affect Our Clinical Management?—A Systematic Review. International Journal of Environmental Research and Public Health. 2019; 16(9):1627. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph16091627
Chicago/Turabian StylePaula, Anabela Baptista, Debbie Toste, Alfredo Marinho, Inês Amaro, Carlos-Miguel Marto, Ana Coelho, Manuel Marques-Ferreira, and Eunice Carrilho. 2019. "Once Resin Composites and Dental Sealants Release Bisphenol-A, How Might This Affect Our Clinical Management?—A Systematic Review" International Journal of Environmental Research and Public Health 16, no. 9: 1627. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph16091627





