Rural Subsistence Maize Farming in South Africa: Risk Assessment and Intervention models for Reduction of Exposure to Fumonisin Mycotoxins
Abstract
:1. Introduction
2. Subsistence Maize Farming and Agricultural Practices in South Africa
- Formalising the rural market
- Increasing agricultural outputs from subsistence areas
- Encouraging agroprocessing operations within rural areas
- Enabling and providing linkages between producers and processors
- Promoting exports
3. Occurrence of Mycotoxins in Subsistence Crops in Southern Africa
4. The Impact of Varying Climate Conditions on Contamination of Crops with Mycotoxigenic Fungi and Mycotoxins
5. Mycotoxin Contamination and Risk Assessment
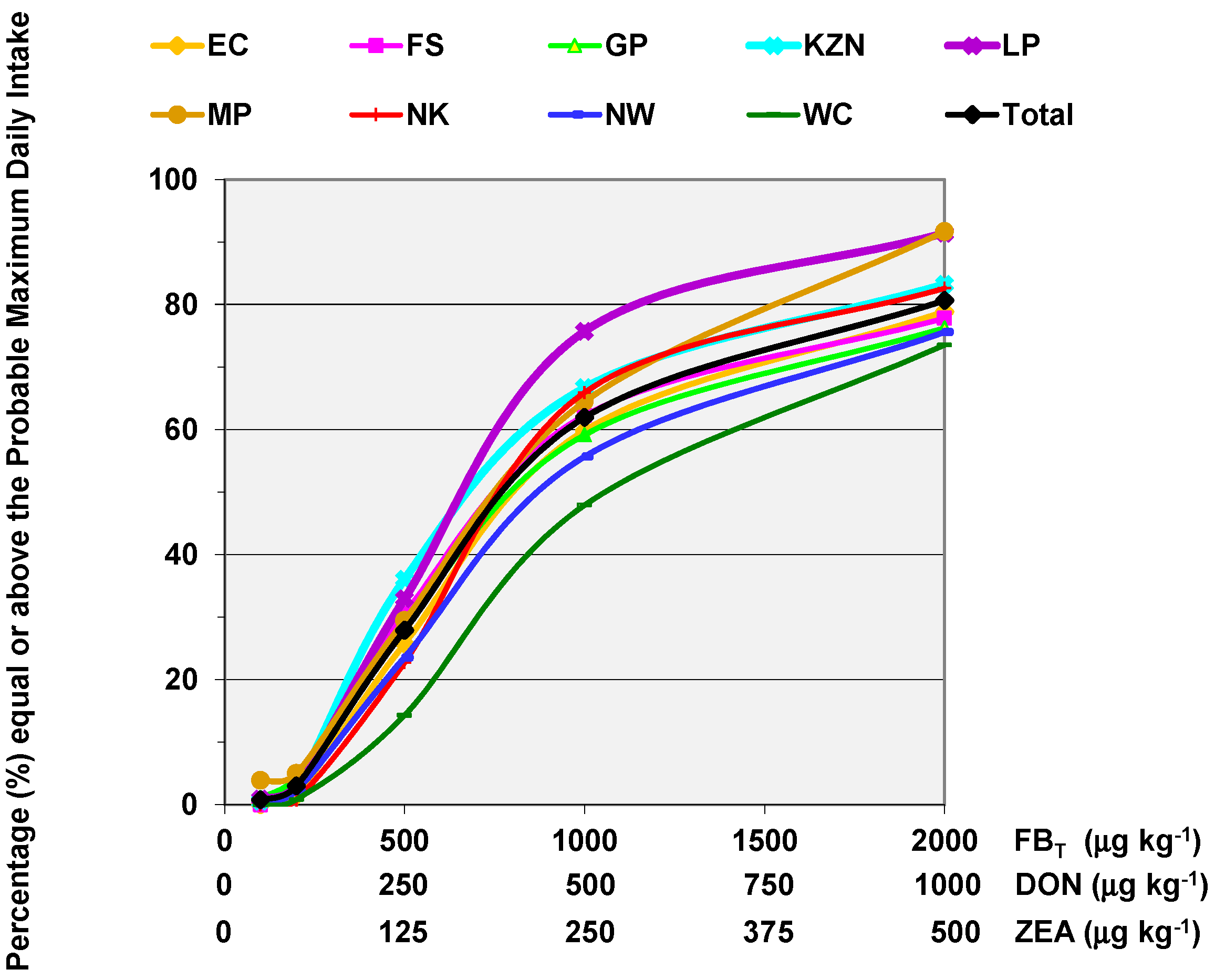
6. Mycotoxin Dietary Exposure in Rural Maize Subsistence Farming Communities
7. Simulation Models to Assess Mycotoxin Exposure
8. Biomarkers of Exposure
9. Intervention Models
9.1. Biologically Based Models for Control of Fusarium Growth and Fumonisin Production
9.1.1. Resistant and Transgenic Maize Cultivars
9.1.2. Transgenic Bt Maize
9.1.3. Biocontrol Microorganisms
9.1.4. Essential Oils and Antioxidants
9.1.5. Phyllosilicate Clay Minerals
9.1.6. Enzymatic Degradation
9.2. Community-Based Intervention Models
10. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Marasas, W.F.O.; Gelderblom, W.C.A.; Shephard, G.S.; Vismer, H.F. Mycotoxins: A global problem. In Mycotoxins: Detection Methods, Management, Public Health and Agricultural Trade; Leslie, J.F., Bandyopadhyay, R., Visconti, A., Eds.; CAB International: Oxfordshire, UK, 2008; pp. 29–39. ISBN 9781845930820. [Google Scholar]
- Wu, F. Mycotoxin risk assessment for the purpose of setting international regulatory standards. Environ. Sci. Technol. 2004, 38, 4049–4055. [Google Scholar] [CrossRef] [PubMed]
- Henry, S.H.; Bosch, X.; Torxell, T.C.; Bolger, P.M. Reducing liver cancer—Global control of aflatoxin. Science 1999, 286, 2453–2454. [Google Scholar] [CrossRef]
- Mitchell, N.J.; Bowers, E.; Hurburgh, C.; Wu, F. Potential economic losses to the USA corn industry from aflatoxin contamination. Food Addit. Contam. 2016, 33, 540–550. [Google Scholar] [CrossRef] [PubMed]
- Bankole, S.A.; Adebanjo, A. Mycotoxins in food in West Africa: Current situation and possibilities of controlling it. Afr. J. Biotechnol. 2003, 2, 254–263. [Google Scholar]
- Shephard, G.S. Impact of mycotoxins on human health in developing countries. Food Addit. Contam. 2008, 25, 146–151. [Google Scholar] [CrossRef] [Green Version]
- Schmale, D.; Munkvold, G. Mycotoxins in crops: A threat to human and domestic animal health. Plant Health Instructor 2009. Available online: https://www.apsnet.org/edcenter/disimpactmngmnt/topc/Mycotoxins/Pages/default.aspx (accessed on 22 February 2019). [CrossRef]
- Khlangwiset, P.; Wu, F. Costs and efficacy of public health interventions to reduce aflatoxin-induced human disease. Food Addit. Contam. 2010, 27, 998–1014. [Google Scholar] [CrossRef] [Green Version]
- Van der Fels-Klerx, H.J.; Liu, C.; Battilani, P. Modelling climate change impacts on mycotoxin contamination. World Mycotoxin J. 2016, 9, 717–726. [Google Scholar] [CrossRef] [Green Version]
- Marasas, W.F.O. Discovery and occurrence of the fumonisins: A historical perspective. Environ. Health Perspect. 2001, 109, 239–243. [Google Scholar]
- Marasas, W.F.O.; Gelderblom, W.C.A.; Shephard, G.S.; Vismer, H.F. Mycotoxicological research in South Africa 1910–2011. World Mycotoxin J. 2012, 5, 89–102. [Google Scholar] [CrossRef]
- Wilson, B.J.; Maronpot, R.R. Causative fungus agent of leukoencephalomalacia in equine animals. Vet. Rec. 1971, 88, 484–486. [Google Scholar] [CrossRef] [PubMed]
- Marasas, W.F.O.; Kellerman, T.S.; Pienaar, J.G.; Naude, T.W. Leukoencephalomalacia: A mycotoxicosis of equidae caused by Fusarium moniliforme Sheldon. Onderstepoort J. Vet. Res. 1976, 43, 113–122. [Google Scholar]
- Kriek, N.P.J.; Kellerman, T.S.; Marasas, W.F.O. A comparative study of the toxicity of Fusarium verticillioides (= F. moniliforme) to horses, primates, pigs, sheep and rats. Onderstepoort J. Vet. Res. 1981, 48, 129–131. [Google Scholar] [PubMed]
- Marasas, W.F.O.; Kriek, N.P.J.; Fincham, J.E.; Van Rensburg, S.J. Primary liver cancer and oesophageal basal cell hyperplasia in rats caused by Fusarium moniliforme. Int. J. Cancer 1984, 34, 383–387. [Google Scholar] [CrossRef] [PubMed]
- Jaskiewicz, K.; Van Rensburg, S.J.; Marasas, W.F.O.; Gelderblom, W.C.A. Carcinogenicity of Fusarium moniliforme culture material in rats. J. Natl. Cancer Inst. 1987, 78, 321–325. [Google Scholar] [PubMed]
- Marasas, W.F.O.; Van Rensburg, S.J.; Mirocha, C.J. Incidence of Fusarium species and the mycotoxins, deoxynivalenol and zearalenone, in corn produced in esophageal cancer areas in Transkei. J. Agric. Food Chem. 1979, 27, 1108–1112. [Google Scholar] [CrossRef]
- Marasas, W.F.O.; Wehner, F.C.; Van Rensburg, S.J.; Van Schalkwyk, D.J. Mycoflora of corn produced in human oesophageal cancer areas in Transkei, southern Africa. Phytopathology 1981, 71, 792–796. [Google Scholar] [CrossRef]
- Zhen, Y.Z. Isolation and culture of fungi from the cereals in 5 high and 3 low in incidence counties of esophageal cancer in Henan Province (China). Zhonghua Zhongliu Zazhi 1984, 6, 27–29. [Google Scholar]
- Marasas, W.F.O.; Kellerman, T.S.; Gelderblom, W.C.A.; Coetzer, J.A.W.; Thiel, P.G.; Van der Lugt, J.J. Leukoencephalomalacia in a horse induced by fumonisin B1 isolated from Fusarium moniliforme. Onderstepoort J. Vet. Res. 1988, 55, 197–203. [Google Scholar]
- Harrison, L.R.; Colvin, B.M.; Greene, J.T.; Newman, L.E.; Cole, J.R. Pulmonary edema and hydrothorax in swine produced by fumonisin B1, a toxic metabolite of Fusarium moniliforme. J. Vet. Diagn. Invest. 1990, 2, 217–221. [Google Scholar] [CrossRef]
- Gelderblom, W.C.A.; Kriek, N.P.J.; Marasas, W.F.O.; Thiel, P.G. Toxicity and carcinogenicity of the Fusarium moniliforme metabolite, fumonisin B1, in rats. Carcinogenesis 1991, 12, 1247–1251. [Google Scholar] [CrossRef] [PubMed]
- Howard, P.C.; Eppley, R.M.; Stack, M.E.; Warbritton, A.; Voss, K.E.; Lorentzen, R.J.; Kovach, R.M.; Bucci, T. Fumonisin B1 carcinogenicity in a two-year feeding study using F344 rats and B6C3F1 mice. Environ. Health Perspect. 2001, 1091, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Marasas, W.F.O.; Riley, R.L.; Hendricks, K.A.; Stevens, V.L.; Sadler, T.W.; Gelineau-van Waes, J.; Missmer, S.A.; Valverde, J.C.; Torres, O.L.; Gelderblom, W.C.A.; et al. Fumonisins disrupt sphingolipid metabolism, folate transport and development of neural crest cells in embryo culture and in vivo: A potential risk factor for human neural tube defects among populations consuming fumonisin-contaminated maize. J. Nutr. 2004, 134, 711–716. [Google Scholar] [CrossRef] [PubMed]
- Shephard, G.S. Chromatographic determination of the fumonisin mycotoxins. J. Chromatogr. A 1998, 815, 31–39. [Google Scholar] [CrossRef]
- Rheeder, J.P.; Marasas, W.F.O.; Thiel, P.G.; Sydenham, E.W.; Shephard, G.S.; Van Schalkwyk, D.J. Fusarium moniliforme and fumonisins in corn in relation to human esophageal cancer in Transkei. Phytopathology 1992, 82, 353–357. [Google Scholar] [CrossRef]
- Zhang, H.; Nagashima, H.; Goto, T. Natural occurrence of mycotoxins in corn, samples from high and low risk areas for human esophageal cancer in China. Mycotoxins 1997, 44, 29–35. [Google Scholar] [CrossRef]
- Mismer, S.A.; Suarez, L.; Felkener, M.; Wang, E.; Merrill, A.H.; Rothman, K.J.; Hendricks, K.A. Exposure to fumonisins and the occurrence of neural tube defects along the Texas-Mexico border. Environ. Health Perspect. 2006, 114, 237–241. [Google Scholar] [CrossRef]
- Sun, G.; Wang, S.; Hu, X.; Su, J.; Huang, T.; Yu, J.; Tang, L.; Gao, W.; Wang, J.-S. Fumonisin B1 contamination of home-grown corn in high-risk areas for esophageal and liver cancer in China. Food Addit. Contam. 2007, 24, 181–185. [Google Scholar] [CrossRef]
- Wang, J.; Zhou, Y.; Liu, W.; Zhu, X.; Du, L.; Wang, Q. Fumonisin level in corn-based food and feed from Linxian County, a high-risk area for esophageal cancer in China. Food Chem. 2008, 106, 241–246. [Google Scholar] [CrossRef]
- Van der Westhuizen, L.; Shephard, G.S.; Gelderblom, W.C.A.; Torres, O.A.; Riley, R.T. Fumonisin biomarkers in maize eaters and implications for human disease. World Mycotoxin J. 2013, 6, 223–232. [Google Scholar] [CrossRef]
- United States National Toxicology Program (US NTP). Regulatory Actions for Year 2001. Available online: https://ntp.niehs.nih.gov/pubhealth/impact/2000s/2001/index.html (accessed on 22 February 2019).
- World Health Organization (WHO). Fumonisin B1. In Safety Evaluation of Certain Mycotoxins in Food; WHO Food Additives Series: 47; 2001; pp. 103–279. Available online: http://www.inchem.org/documents/jecfa/jecmono/v47je01.htm (accessed on 22 February 2019).
- International Agency for Research on Cancer (IARC). Fumonisin B1. In IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, Some Traditional Herbal Medicines, Some Mycotoxins, Naphthalene and Styrene; IARC Working Group on the Evaluation of Carcinogenic Risks to Humans; IARC Press: Lyon, France, 2002; Volume 82, pp. 301–366. ISBN 9283212827. [Google Scholar]
- South Africa Foodstuffs, Cosmetics and Disinfectants Act (54/1972): Regulations Governing Tolerances for fungus-produced toxins in Foodstuffs: Amendment. In Government Gazette; 5 September 2016; No 40250, Government Notice number 987. Available online: https://www.gov.za/sites/default/files/gcis_document/201609/40250gon987.pdf (accessed on 22 February 2019).
- Steyn, N.P.; Nel, J.H.; Casey, A. Secondary data analysis of dietary surveys undertaken in South Africa in order to determine usual food consumption of the population. Public Health Nutr. 2003, 6, 631–644. [Google Scholar] [CrossRef]
- Shephard, G.S.; Marasas, W.F.O.; Burger, H.-M.; Somdyala, N.I.M.; Rheeder, J.P.; Van der Westhuizen, L.; Gatyeni, P.; Van Schalkwyk, D.J. Exposure assessment for fumonisins in the former Transkei region of South Africa. Food Addit. Contam. 2007, 24, 621–629. [Google Scholar] [CrossRef] [PubMed]
- Shephard, G.S.; Van der Westhuizen, L.; Sewram, V. Biomarkers of exposure to fumonisin mycotoxins: A review. Food Addit. Contam. 2007, 24, 1196–1201. [Google Scholar] [CrossRef] [PubMed]
- Ncube, E.; Flett, B.C.; Waalwijk, C.; Viljoen, A. Fusarium spp. and levels of fumonisins in maize produced by subsistence farmers in South Africa. S. Afr. J. Sci. 2011, 107, 33–39. [Google Scholar] [CrossRef]
- Chiona, M.; Ntawuruhunga, P.; Benesi, I.R.M.; Matumba, L.; Moyo, C.C. Aflatoxins contamination in processed cassava in Malawi and Zambia. Afr. J. Food Nutr. Agric. Dev. 2014, 14, 8809–8820. [Google Scholar]
- Probst, C.; Bandyopadhyay, P.J.; Cotty, P.J. Diversity of aflatoxin-producing fungi and their impact on food safety in sub-Saharan Africa. Int. J. Food Microbiol. 2014, 174, 113–122. [Google Scholar] [CrossRef]
- Hove, M.; De Boevre, M.; Lachat, C.; Jacxsens, L.; Nyanga, L.K.; De Saeger, S. Occurrence and risk assessment of mycotoxins in subsistence farmed maize from Zimbabwe. Food Control 2016, 69, 36–44. [Google Scholar] [CrossRef]
- Mwalwayo, D.S.; Thole, B. Prevalence of aflatoxin and fumonisins (B1 + B2) in maize consumed in rural Malawi. Toxicol. Rep. 2016, 3, 173–179. [Google Scholar] [CrossRef]
- Njoroge, S.M.C.; Matumba, L.; Kanenga, K.; Siambi, M.; Waliyar, F.; Maruwo, J.; Monyo, E.S. A case for regular aflatoxin monitoring in peanut butter in Sub-Saharan Africa: Lessons from a 3-year survey in Zambia. J. Food Prot. 2016, 79, 795–800. [Google Scholar] [CrossRef]
- Aliber, M.; Hart, T.G.B. Should subsistence agriculture be supported as a strategy to address rural food insecurity? Agrekon 2019, 48, 434–458. [Google Scholar] [CrossRef]
- Burger, H.-M.; Lombard, M.J.; Shephard, G.S.; Danster-Christians, N.; Gelderblom, W.C.A. Development and evaluation of a sensitive mycotoxin risk assessment model (MYCORAM). Toxicol. Sci. 2014, 141, 387–397. [Google Scholar] [CrossRef] [PubMed]
- Ncube, E. Mycotoxin Levels in Subsistence Farming Systems in South Africa. Master’s Thesis, Stellenbosch University, Stellenbosch, South Africa, 2008. [Google Scholar]
- Wilke, A.L.; Bronson, C.R.; Tomas, A.; Munkvold, G.P. Seed transmission of Fusarium verticillioides in maize plants grown under three different temperature regimes. Plant Dis. 2007, 91, 1109–1115. [Google Scholar] [CrossRef] [PubMed]
- Munkvold, G.P.; Desjardins, A.E. Fumonisins in maize. Can we reduce their occurrence? Plant Dis. 1997, 81, 556–565. [Google Scholar] [CrossRef] [PubMed]
- Fandohan, P.; Hell, K.; Marasas, W.F.O.; Wingfield, M.J. Infection of maize by Fusarium species and contamination with fumonisin in Africa. Afr. J. Biotechnol. 2003, 2, 570–579. [Google Scholar]
- Pray, C.E.; Rheeder, J.P.; Gouse, M.; Volkwyn, Y.; Van der Westhuizen, L.; Shephard, G.S. Bt maize and fumonisin reduction in South Africa: Potential health impacts. In Genetically Modified Crops in Africa: Economic and Policy Lessons from Countries South of the Sahara; Falck-Zepeda, J., Gruere, G., Sithole-Niang, I., Eds.; International Food Policy Research Institute (IFPRI): Washington, DC, USA, 2013; pp. 43–57. ISBN 9780896297951. [Google Scholar]
- Department of Rural Development and Land Reform (DRDLR). Rural Development Plans for the Eastern Cape Province and the Identified District Municipalities within the Province. Available online: http://www.ruraldevelopment.gov.za/phocadownload/SPLUMB/Dev_Plans2017/Eastern-Cape/2015-12-report3v1-ortambo-dm.pdf (accessed on 22 February 2019).
- Udomkun, P.; Wiredu, A.N.; Nagle, M.; Bandyopadhyay, R.; Müller, J.; Vanlauwe, B. Mycotoxins in Sub-Saharan Africa: Present situation, socio-economic impact, awareness, and outlook. Food Control 2017, 72, 110–122. [Google Scholar] [CrossRef]
- Kimanya, M.E. The health impacts of mycotoxins in the eastern Africa region. Curr. Opin. Food Sci. 2015, 6, 7–11. [Google Scholar] [CrossRef]
- International Agency for Research on Cancer (IARC). Mycotoxin Control in Low- and Middle Income Countries; Wild, C.P., Miller, J.D., Groopman, J.D., Eds.; WHO Press, World Health Organisation: Geneva, Switzerland, 2015; ISBN 9789283225102. [Google Scholar]
- Mupunga, I.; Lebelo, S.L.; Mngqawa, P.; Rheeder, J.P.; Katerere, D.R. Natural occurrence of aflatoxins in peanuts and peanut butter from Bulawayo, Zimbabwe. J. Food Prot. 2014, 77, 1814–1818. [Google Scholar] [CrossRef]
- Matumba, L.; Sulyok, M.; Monjerezi, M.; Biswick, T.; Krska, R. Fungal metabolites diversity in maize and associated human dietary exposures relate to micro-climatic patterns in Malawi. World Mycotoxin J. 2014, 8, 269–282. [Google Scholar] [CrossRef]
- Matumba, L.; Monjerezi, M.; Biswick, T.; Mwatseteza, J.; Makumba, W.; Kamangira, D.; Mtukuso, A. A survey of the incidence and level of aflatoxin contamination in a range of locally and imported processed foods on Malawian retail market. Food Control 2014, 39, 87–91. [Google Scholar] [CrossRef]
- Shephard, G.S.; Van der Westhuizen, L.; Katerere, D.R.; Herbst, M.; Pineiro, M. Preliminary exposure assessment of deoxynivalenol and patulin in South Africa. Mycotoxin Res. 2010, 26, 181–185. [Google Scholar] [CrossRef] [PubMed]
- Misihairabgwi, J.M.; Ezekiel, C.N.; Sulyok, M.; Shephard, G.S.; Krska, R. Mycotoxin contamination of foods in Southern Africa: A 10-year review (2007–2016). Crit. Rev. Food Sci. Nutr. 2017, 59, 43–58. [Google Scholar] [CrossRef] [PubMed]
- Shephard, G.S.; Burger, H.-M.; Rheeder, J.P.; Alberts, J.F.; Gelderblom, W.C.A. The effectiveness of regulatory maximum levels for fumonisin mycotoxins in commercial and subsistence maize crops in South Africa. Food Control 2019, 97, 77–80. [Google Scholar] [CrossRef]
- Katerere, D.R.; Shephard, G.S.; Faber, M. Infant malnutrition and chronic aflatoxicosis in Southern Africa: Is there a link? Int. J. Food Safety Nutr. Public Health 2008, 1, 127–135. [Google Scholar] [CrossRef]
- Rheeder, J.P.; Van der Westhuizen, L.; Imrie, G.; Shephard, G.S. Fusarium species and fumonisins in subsistence maize in the former Transkei region, South Africa: A multi-year study in rural villages. Food Addit. Contam. 2016, 9, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Kruger, A.C.; Shongwe, S. Temperature trends in South Africa: 1960–2003. Int. J. Climatol. 2004, 24, 1929–1945. [Google Scholar] [CrossRef]
- Magan, N.; Medina, A.; Aldred, D. Possible climate-change effects on mycotoxins contamination of food crops pre- and postharvest. Plant Pathol. 2011, 60, 150–163. [Google Scholar] [CrossRef]
- Battilani, P.; Rossi, V.; Pietri, A. Modelling Fusarium verticillioides infection and fumonisin synthesis in maize ears. Asp. Appl. Biol. 2003, 68, 91–100. [Google Scholar]
- Food and Agriculture Organization (FAO); World Health Organization (WHO). Environmental Health Criteria 240, Principles and Methods for the Risk Assessment of Chemicals in Food; FAO, WHO: Geneva, Switzerland, 2009; ISBN 9789241572408. [Google Scholar]
- Fryer, M.; Collins, C.D.; Ferrier, H.; Colvile, R.N.; Nieuwenhuijsen, M.J. Human exposure modelling for chemical risk assessment: A review of current approaches and research policy implications. Environ. Sci. Policy 2006, 9, 261–274. [Google Scholar] [CrossRef]
- Parmar, B.; Miller, P.F.; Burt, R. Stepwise approaches for estimating the intakes of chemicals in food. Regul. Toxicol. Pharmacol. 1997, 26, 44–51. [Google Scholar] [CrossRef]
- Lambe, J. The use of food consumption data in assessments of exposure to food chemicals including the application of probabilistic modelling. Proc. Nutr. Soc. 2002, 61, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Hart, A.; Smith, G.C.; Macarthur, R.; Rose, M. Application of uncertainty analysis in assessing dietary exposure. Toxicol. Lett. 2003, 140, 437–442. [Google Scholar] [CrossRef]
- Thiel, P.G.; Marasas, W.F.O.; Sydenham, E.W.; Shephard, G.S.; Gelderblom, W.C.A. The implications of naturally occurring levels of fumonisins in corn for human and animal health. Mycopathologia 1992, 117, 3–9. [Google Scholar] [CrossRef]
- Lombard, M.J.; Steyn, N.; Burger, H.-M.; Charlton, K.; Senekal, M.; Gelderblom, W.C.A. A proposed method to determine fumonisin exposure from maize consumption in a rural South African population using a culturally appropriate food frequency questionnaire. Public Health Nutr. 2012, 17, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Lombard, M.J.; Steyn, N.; Burger, H.-M.; Charlton, K.; Senekal, M. A food photograph series for identifying portion sizes of culturally specific dishes in rural areas with high incidence of oesophageal cancer. Nutrients 2013, 5, 3118–3130. [Google Scholar] [CrossRef] [PubMed]
- Shephard, G.S.; Van der Westhuizen, L.; Gatyeni, P.M.; Somdyala, N.I.; Burger, H.-M.; Marasas, W.F.O. Fumonisin mycotoxins in traditional Xhosa maize beer in South Africa. J. Agric. Food Chem. 2005, 53, 9634–9637. [Google Scholar] [CrossRef] [PubMed]
- Burger, H.-M.; Lombard, M.J.; Shephard, G.S.; Rheeder, J.P.; Van der Westhuizen, L.; Gelderblom, W.C.A. Dietary fumonisin exposure in a rural population of South Africa. Food Chem. Toxicol. 2010, 48, 2103–2108. [Google Scholar] [CrossRef]
- Gelderblom, W.C.A.; Riedel, S.; Burger, H.-M.; Abel, S.; Marasas, W.F.O. Carcinogenesis by the fumonisins: Mechanisms, risk analyses and implications. In Food Contaminants, Mycotoxins and Food Allergens; ACS Symposium Series 1001; Sianta, D.P., Trucksess, M.W., Scot, P.M., Herman, E.M., Eds.; ACS: Washington, DC, USA, 2008; pp. 80–95. [Google Scholar]
- Gelderblom, W.; Marasas, W. Controversies in fumonisin mycotoxicology and risk assessment. Hum. Exp. Toxicol. 2012, 31, 215–235. [Google Scholar] [CrossRef]
- Paustenbach, D.; Galbraith, D. Biomonitoring and biomarkers: Exposure assessment will never be the same. Environ. Health Perspect. 2006, 114, 1143–1149. [Google Scholar] [CrossRef]
- Van der Westhuizen, L.; Shephard, G.S.; Rheeder, J.P.; Burger, H.-M. Individual fumonisin exposure and sphingoid base levels in rural populations consuming maize in South Africa. Food Chem. Toxicol. 2010, 48, 1698–1703. [Google Scholar] [CrossRef]
- Gong, Y.Y.; Torres-Sanchez, L.; Lopez-Carrillo, L.; Jian, H.P.; Sutcliffe, A.E.; White, K.L.; Humpf, H.U.; Turner, P.C.; Wild, C.P. Association between tortilla consumption and human urinary fumonisin B1 levels in a Mexican population. Cancer Epidemiol. Biomarkers Prev. 2008, 17, 688–694. [Google Scholar] [CrossRef] [PubMed]
- Van der Westhuizen, L.; Shephard, G.S.; Burger, H.-M.; Rheeder, J.P.; Gelderblom, W.C.; Wild, C.P.; Gong, Y.Y. Fumonisin B1 as a urinary biomarker of exposure in a maize intervention study among South African subsistence farmers. Cancer Epidemiol. Biomarkers Prev. 2011, 20, 483–489. [Google Scholar] [CrossRef] [PubMed]
- Shephard, G.S.; Burger, H.-M.; Gambacorta, L.; Gong, Y.Y.; Krska, R.; Rheeder, J.P.; Solfrizzo, M.; Srey, C.; Sulyok, M.; Visconti, A.; et al. Multiple mycotoxin exposure determined by urinary biomarkers in rural subsistence farmers in the former Transkei, South Africa. Food Chem. Toxicol. 2013, 62, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Wild, C.P.; Gong, Y.Y. Mycotoxins and human disease: A largely ignored global health issue. Carcinogenesis 2010, 31, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Alabouvette, C.; Olivain, C.; Migheli, Q.; Steinberg, C. Microbiological control of soil-borne phytopathogenic fungi with special emphasis on wilt-inducing Fusarium oxysporum. New Phytol. 2009, 184, 529–544. [Google Scholar] [CrossRef]
- He, J.; Zhou, T. Patented techniques for detoxification of mycotoxins in feeds and food matrices. Rec. Pat. Food Nutr. Agric. 2010, 2, 96–104. [Google Scholar] [CrossRef]
- Larkin, R.P.; Fravel, D.R. Efficacy of various fungal and bacterial biocontrol organisms for control of Fusarium wilt of tomato. Plant Dis. 1998, 82, 1022–1028. [Google Scholar] [CrossRef]
- Schatzmayr, G.; Zehner, F.; Täubel, M.; Schatzmayr, D.; Klimitsch, A.; Loibner, A.P.; Binder, E.M. Microbiologicals for deactivating mycotoxins. Mol. Nutr. Food Res. 2006, 50, 543–551. [Google Scholar] [CrossRef]
- da Cruz Cabral, L.; Pinto, V.F.; Patriarca, A. Application of plant derived compounds to control fungal spoilage and mycotoxin production in foods. Int. J. Food Microbiol. 2013, 166, 1–14. [Google Scholar] [CrossRef]
- Alberts, J.F.; van Zyl, W.H.; Gelderblom, W.C.A. Biologically based methods for control of fumonisin-producing Fusarium species and reduction of the fumonisins. Front. Microbiol. 2016, 7, 548. [Google Scholar] [CrossRef]
- Duvick, J.; Rood, T.; Maddox, J.; Gilliam, J. Detoxification of mycotoxins in planta as a strategy for improving grain quality and disease resistance: Identification of fumonisin-degrading microbes from maize. In Molecular Genetics of Host-Specific Toxins in Plant Disease, Developments in Plant Pathology; Kohmoto, K., Yoder, O.C., Eds.; Springer International Publishing AG: Basel, Switzerland, 1998; Volume 13, pp. 369–381. ISBN 9789401152181. [Google Scholar]
- Cleveland, T.E.; Dowd, P.F.; Desjardins, A.E.; Bhatnagar, D.; Cotty, P.J. United States Department of Agriculture-Agricultural Research Service research on pre-harvest prevention of mycotoxins and mycotoxigenic fungi in US crops. Pest Manag. Sci. 2003, 59, 629–642. [Google Scholar] [CrossRef]
- Lanubile, A.; Pasini, L.; Marocco, A. Differential gene expression in kernels and silks of maize lines with contrasting levels of ear rot resistance after Fusarium verticillioides infection. J. Plant Phys. 2010, 167, 1398–1406. [Google Scholar]
- Brown, D.W.; Butchko, R.A.E.; Proctor, R.H. Fusarium genomic resources: Tools to limit crop diseases and mycotoxin contamination. Mycopathologia 2006, 162, 191–199. [Google Scholar] [CrossRef]
- Desjardins, A.E.; Proctor, R.H. Molecular biology of Fusarium mycotoxins. Int. J. Food Microbiol. 2007, 119, 47–50. [Google Scholar] [CrossRef]
- Betz, F.S.; Hammond, B.G.; Fuchs, R.L. Safety and advantages of Bacillus thuringiensis- protected plants to control insect pests. Regul. Toxicol. Pharm. 2000, 32, 156–173. [Google Scholar] [CrossRef] [PubMed]
- Hammond, B.G.; Campbell, K.W.; Pilcher, C.D.; Degooyer, T.A.; Robinson, A.E.; Mcmillen, B.L.; Spangler, S.M.; Riordan, S.G.; Rice, L.G.; Richard, J.L. Lower fumonisin mycotoxin levels in the grain of Bt corn grown in the United States in 2000–2002. J. Agric. Food Chem. 2004, 52, 1390–1397. [Google Scholar] [CrossRef]
- Wu, F. Mycotoxin reduction in Bt corn: Potential economic, health, and regulatory impacts. Transgenic Res. 2006, 15, 277–289. [Google Scholar] [CrossRef] [PubMed]
- Gouse, M.; Kirsten, J.F.; Van Der Walt, W.J. Bt Cotton and Bt Maize: An Evaluation of Direct and Indirect Impact on the Cotton and Maize Farming Sectors in South Africa. Commissioned report to the Department of Agriculture: Directorate BioSafety, Pretoria, South Africa. 2008. Available online: https://scholar.google.co.za/citations?user=ZVzSBB0AAAAJ&hl=en#d=gs_md_cita-d&u=%2Fcitations%3Fview_op%3Dview_citation%26hl%3Den%26user%3DZVzSBB0AAAAJ%26citation_for_view%3DZVzSBB0AAAAJ%3AUeHWp8X0CEIC%26tzom%3D-120 (accessed on 26 February 2019).
- Rheeder, J.P.; Vismer, H.F.; Van der Westhuizen, L.; Imrie, G.; Gatyeni, P.; Thomas, D.; Shephard, G.S.; Marasas, W.F.O.; Flett, B. Effect of Bt corn hybrids on insect damage, incidence of fumonisin-producing Fusarium species and fumonisin levels in South Africa. Phytopathology 2005, 95, S88. [Google Scholar]
- Alberts, J.F.; Lilly, M.; Rheeder, J.P.; Burger, H.M.; Shephard, G.S.; Gelderblom, W.C.A. Technological and community-based methods to reduce mycotoxin exposure. Food Control 2017, 73, 101–109. [Google Scholar] [CrossRef]
- Fischer, K.; Van den Berg, J.; Mutengwa, C. Is Bt maize effective in improving South African smallholder agriculture? S. Afr. J. Sci. 2015, 111, 01–02. [Google Scholar] [CrossRef]
- Dalie, D.K.; Deschamps, A.M.; Atanasova-Penichon, V.; Richard-Forget, F. Potential of Pediococcus pentosaceus (L006) isolated from maize leaf to suppress fumonisin-producing fungal growth. J. Food Prot. 2010, 73, 1129–1137. [Google Scholar] [CrossRef] [PubMed]
- Yates, I.E.; Meredith, F.; Smart, W.; Bacon, C.W.; Jaworski, A.J. Trichoderma viride suppresses fumonisin B1 production by Fusarium moniliforme. J. Food Prot. 1999, 62, 1326–1332. [Google Scholar] [CrossRef] [PubMed]
- Cavaglieri, L.; Passone, A.; Etcheverry, M. Screening procedures for selecting rhizobacteria with biocontrol effects upon Fusarium verticillioides growth and fumonisin B1 production. Res. Microbiol. 2004, 155, 747–754. [Google Scholar] [CrossRef] [PubMed]
- United States Food and Drug Administration (US FDA). Generally Recognized as Safe (GRAS) Substances (SGOCS) Database. Available online: https://www.fda.gov/food/generally-recognized-safe-gras/gras-substances-scogs-database (accessed on 18 March 2019).
- Bacon, C.W.; Yates, I.E.; Hinton, D.M.; Meredith, F. Biological control of Fusarium moniliforme in maize. Environ. Health Perspect. 2001, 109, 325–332. [Google Scholar] [PubMed]
- Samapundo, S.; De Meulenaer, B.; Osei-Nimoh, D.; Lamboni, Y.; Debevere, J.; Devlieghere, F. Can phenolic compounds be used for the protection of corn from fungal invasion and mycotoxin contamination during storage? Food Microbiol. 2007, 24, 465–473. [Google Scholar] [CrossRef] [PubMed]
- Etcheverry, M.; Torres, A.; Ramirez, M.L.; Chulze, S.; Magan, N. In vitro control of growth and fumonisin production by Fusarium verticillioides and F. proliferatum using antioxidants under different water availability and temperature regimes. J. Appl. Microbiol. 2002, 92, 624–632. [Google Scholar] [CrossRef] [PubMed]
- Reynoso, M.M.; Torres, A.M.; Ramirez, M.L.; Rodrigues, M.I.; Chulze, S.; Magan, N. Efficacy of antioxidant mixtures on growth, fumonisin production and hydrolytic enzyme production by Fusarium verticillioides and F. proliferatum in vitro on maize-based media. Mycol. Res. 2002, 106, 1093–1099. [Google Scholar] [CrossRef]
- Torres, A.M.; Ramirez, M.L.; Arroyo, M.; Chulze, S.N.; Magan, N. Potential use of antioxidants for control of growth and fumonisin production by Fusarium verticillioides and Fusarium proliferatum on whole maize grain. Int. J. Food Microbiol. 2003, 83, 319–324. [Google Scholar] [CrossRef]
- Phillips, T.D.; Sarr, A.B.; Grant, P.G. Selective chemisorption and detoxification of aflatoxins by phyllosilicate clay. Nat. Toxins 1995, 3, 204–213. [Google Scholar] [CrossRef]
- Phillips, T.D.; Afriyie-Gyawu, E.; Williams, J.; Huebner, H.; Ankrah, N.-A.; Ofori-Adjei, D.; Jolly, P.; Johnson, N.; Taylor, J.; Marroquin-Cardona, A.; et al. Reducing human exposure to aflatoxin through the use of clay: A review. Food Addit. Contam. 2008, 25, 134–145. [Google Scholar] [CrossRef]
- Aly, S.E.; Abdel-Galil, M.M.; Abdel-Wahhab, M.A. Application of adsorbent agents technology in the removal of aflatoxin B1 and fumonisin B1 from malt extract. Food Chem. Toxicol. 2004, 42, 1825–1831. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, N.J.; Xue, K.S.; Lin, S.; Marroquin-Cardona, A.; Brown, K.A.; Elmore, S.E.; Tang, L.; Romoser, A.; Gelderblom, W.C.A.; Wang, J.; et al. Calcium montmorillonite clay reduces AFB1 and FB1 biomarkers in rats exposed to single and co-exposures of aflatoxin and fumonisin. J. Appl. Toxicol. 2013, 34, 795–804. [Google Scholar] [CrossRef] [PubMed]
- Gelderblom, W.C.A.; Cawood, M.E.; Snyman, S.D.; Vleggaar, R.; Marasas, W.F.O. Structure-activity relationships of fumonisins in short-term carcinogenesis and cytotoxicity assays. Food Chem. Toxicol. 1993, 31, 407–414. [Google Scholar] [CrossRef]
- Alberts, J.F.; Engelbrecht, Y.; Steyn, P.S.; Holzapfel, W.H.; van Zyl, W.H. Biological degradation of aflatoxin B1 by Rhodococcus erythropolis cultures. Int. J. Food Microbiol. 2006, 109, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Alberts, J.F.; Gelderblom, W.C.; Botha, A.; van Zyl, W.H. Degradation of aflatoxin B1 by fungal laccase enzymes. Int. J. Food Microbiol. 2009, 135, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Heinl, S.; Hartinger, D.; Thamhesl, M.; Kunz-Vekiru, E.; Krska, R.; Schatzmayr, G.; Moll, W.; Grabherr, R. Degradation of fumonisin B1 by the consecutive action of two bacterial enzymes. J. Biotechnol. 2010, 145, 120–129. [Google Scholar] [CrossRef]
- Minto, R.E.; Townsend, C.A. Enzymology and molecular biology of aflatoxin biosynthesis. Chem. Rev. 1997, 97, 2537–2555. [Google Scholar] [CrossRef]
- Mishra, H.N.; Das, C. A review on biological control and metabolism of aflatoxin. Critical Rev. Food Sci. Nutr. 2003, 43, 245–264. [Google Scholar] [CrossRef]
- Wang, J.; Ogata, M.; Hirai, H.; Kawagishi, H. Detoxification of aflatoxin B1 by manganese peroxidase from the white rot fungus Phanerochaete sordida YK-624. FEMS Microbiol. Lett. 2011, 314, 164–169. [Google Scholar] [CrossRef]
- Yehia, R.S. Aflatoxin detoxification by manganese peroxidase purified from Pleurotus ostreatus. Braz. J. Microbiol. 2014, 45, 127–133. [Google Scholar] [CrossRef]
- Wu, Y.; Lu, F.; Jiang, H.; Tan, C.; Yao, C.; Liu, D. The furofuran-ring selectivity, hydrogen peroxide-production and low Km value are the three elements for highly effective detoxification of aflatoxin oxidase. Food Chem. Toxicol. 2015, 76, 125–131. [Google Scholar] [CrossRef]
- Duvick, J.; Rood, T.; Wang, X. Fumonisin Detoxification Enzymes. Patent No US5716820, 10 February 1998. [Google Scholar]
- Blackwell, B.A.; Gilliam, J.T.; Savard, M.E.; Miller, D.; Duvick, J.P. Oxidative deamination of hydrolysed fumonisin B1 (AP1) by cultures of Exophiala spinifera. Nat. Toxins 1999, 7, 31–38. [Google Scholar] [CrossRef]
- Hartinger, D.; Schwartz, H.; Hametner, C.; Schatzmayr, G.; Haltrich, D.; Moll, W.D. Enzyme characteristics of aminotransferase FumI of Sphingopyxis sp. MTA144 for deamination of hydrolyzed fumonisin B1. Appl. Microbiol. Biotechnol. 2011, 91, 757–768. [Google Scholar] [CrossRef]
- Oswald, I.P.; Grenier, B.; Schatzmayr, G.; Moll, W. Enzymatic detoxification of mycotoxins: Hydrolysis of fumonisin B1 strongly reduced the toxicity for piglets. In World Nutrition Forum, Nutri Economics: Balancing Global Nutrition and Productivity; Binder, E.M., Ed.; Anytime Publishing: Leicestershire, UK, 2012; pp. 263–271. ISBN 0957372124. [Google Scholar]
- Moll, D.; Hartinger, D.; Grießler, K.; Binder, E.M.; Schatzmayr, G. Method for the Production of an Additive for the Enzymatic Decomposition of Mycotoxins, Additive, and Use Thereof. Patent No US8703460B2, 4 October 2011. [Google Scholar]
- Fandohan, P.; Zoumenou, D.; Hounhouigan, D.J.; Marasas, W.F.; Wingfield, M.J.; Hell, K. Fate of aflatoxins and fumonisins during the processing of maize into food products in Benin. Int. J. Food Microbiol. 2005, 98, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Afolabi, C.G.; Bandyopadhyay, R.; Leslie, J.F.; Ekpo, E.J.A. Effect of sorting on incidence and occurrence of fumonisins and Fusarium verticillioides on maize from Nigeria. J. Food Prot. 2006, 69, 2019–2023. [Google Scholar] [CrossRef] [PubMed]
- Kimanya, M.E.; De Meulenaer, B.; Tiisekwa, B.; Ndomondo-Sigonda, M.; Devlieghere, F.; Van Camp, J.; Kolsteren, P. Co-occurrence of fumonisins with aflatoxins in home-stored maize for human consumption in rural villages of Tanzania. Food Addit. Contam. 2008, 25, 1353–1364. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van der Westhuizen, L.; Shephard, G.S.; Rheeder, J.P.; Burger, H.-M.; Gelderblom, W.C.A.; Wild, C.P.; Gong, Y.Y. Simple intervention method to reduce fumonisin exposure in a subsistence maize-farming community in South Africa. Food Addit. Contam. 2010, 27, 1582–1588. [Google Scholar] [CrossRef] [PubMed]
- Matumba, L.; Monjerezi, M.; Kankwamba, H.; Njoroge, S.M.C.; Ndilowe, P.; Kabuli, H.; Kambewa, D.; Njapau, H. Knowledge, attitude, and practices concerning presence of molds in foods among members of the general public in Malawi. Mycotoxin Res. 2015, 32, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Desjardins, A.E.; Manandhar, G.; Plattner, R.D.; Maragos, C.M.; Shrestha, K.; McCormick, S.P. Occurrence of Fusarium species and mycotoxins in Nepalese maize and wheat and the effect of traditional processing methods on mycotoxin levels. J. Agric. Food Chem. 2000, 48, 1377–1383. [Google Scholar] [CrossRef]
- Fandohan, P.; Ahouansou, R.; Houssou, P.; Hell, K.; Marasas, W.F.O.; Wingfield, M.J. Impact of mechanical shelling and dehulling on Fusarium infection and fumonisin contamination in maize. Food Addit. Contam. 2006, 23, 415–421. [Google Scholar] [CrossRef]
- Sydenham, E.W.; Van der Westhuizen, L.; Stockenström, S.; Shephard, G.S.; Thiel, P.G. Fumonisin-contaminated maize: Physical treatment for the partial decontamination of bulk shipments. Food Addit. Contam. 1994, 11, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Bullerman, L.B.; Bianchini, A. Stability of mycotoxins during food processing. Int. J. Food Microbiol. 2007, 119, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Van der Westhuizen, L.; Shephard, G.S.; Rheeder, J.P.; Burger, H.-M.; Gelderblom, W.C.A.; Wild, C.P.; Gong, Y.Y. Optimising sorting and washing of home-grown maize to reduce fumonisin contamination under laboratory-controlled conditions. Food Control. 2011, 22, 396–400. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alberts, J.; Rheeder, J.; Gelderblom, W.; Shephard, G.; Burger, H.-M. Rural Subsistence Maize Farming in South Africa: Risk Assessment and Intervention models for Reduction of Exposure to Fumonisin Mycotoxins. Toxins 2019, 11, 334. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins11060334
Alberts J, Rheeder J, Gelderblom W, Shephard G, Burger H-M. Rural Subsistence Maize Farming in South Africa: Risk Assessment and Intervention models for Reduction of Exposure to Fumonisin Mycotoxins. Toxins. 2019; 11(6):334. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins11060334
Chicago/Turabian StyleAlberts, Johanna, John Rheeder, Wentzel Gelderblom, Gordon Shephard, and Hester-Mari Burger. 2019. "Rural Subsistence Maize Farming in South Africa: Risk Assessment and Intervention models for Reduction of Exposure to Fumonisin Mycotoxins" Toxins 11, no. 6: 334. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins11060334




