Assessment of Exposure to Mycotoxins in Spanish Children through the Analysis of Their Levels in Plasma Samples
Abstract
:1. Introduction
2. Results and Discussion
2.1. Control of the Analytical Sequences
2.2. Plasma Samples
2.3. Chromatographic Results
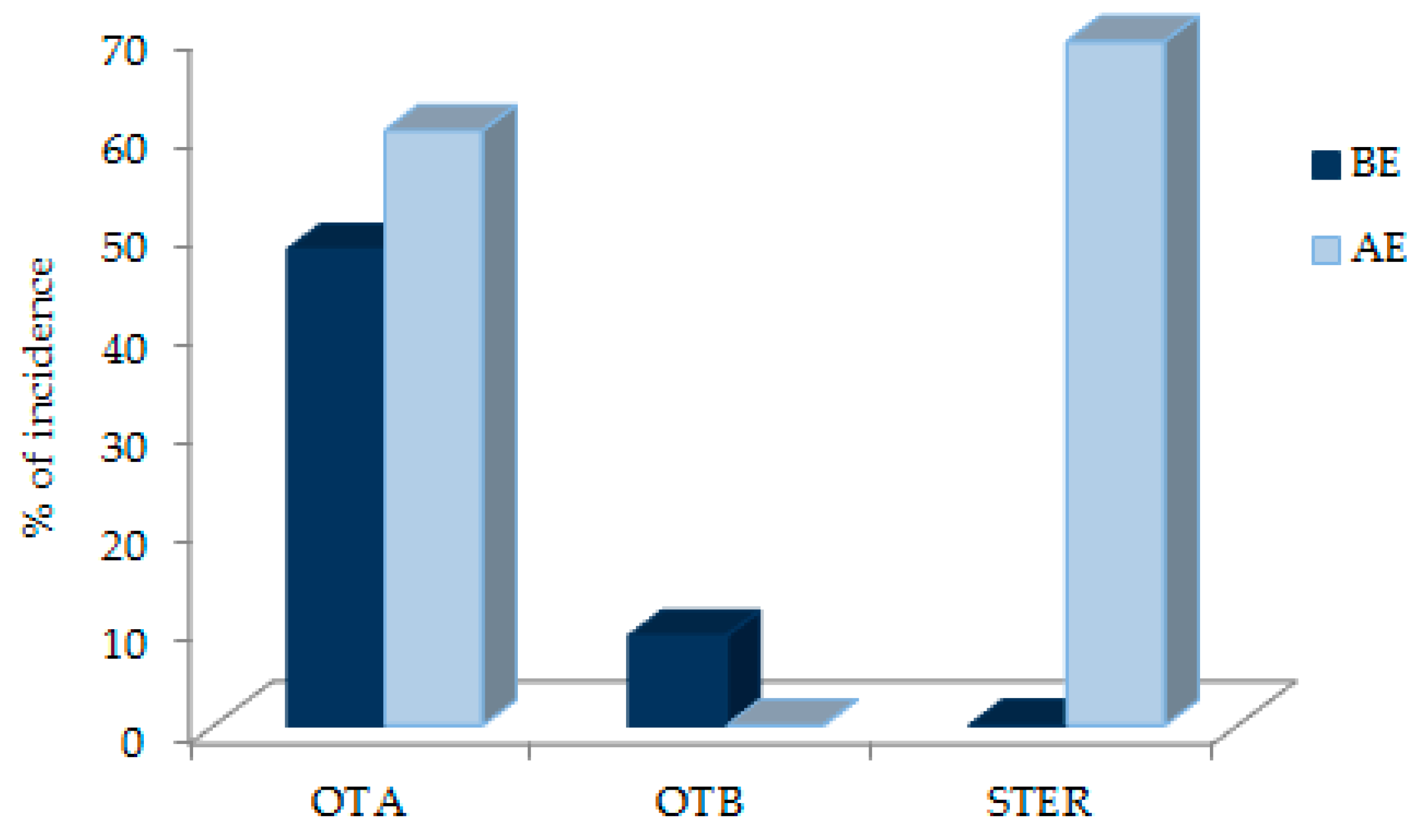
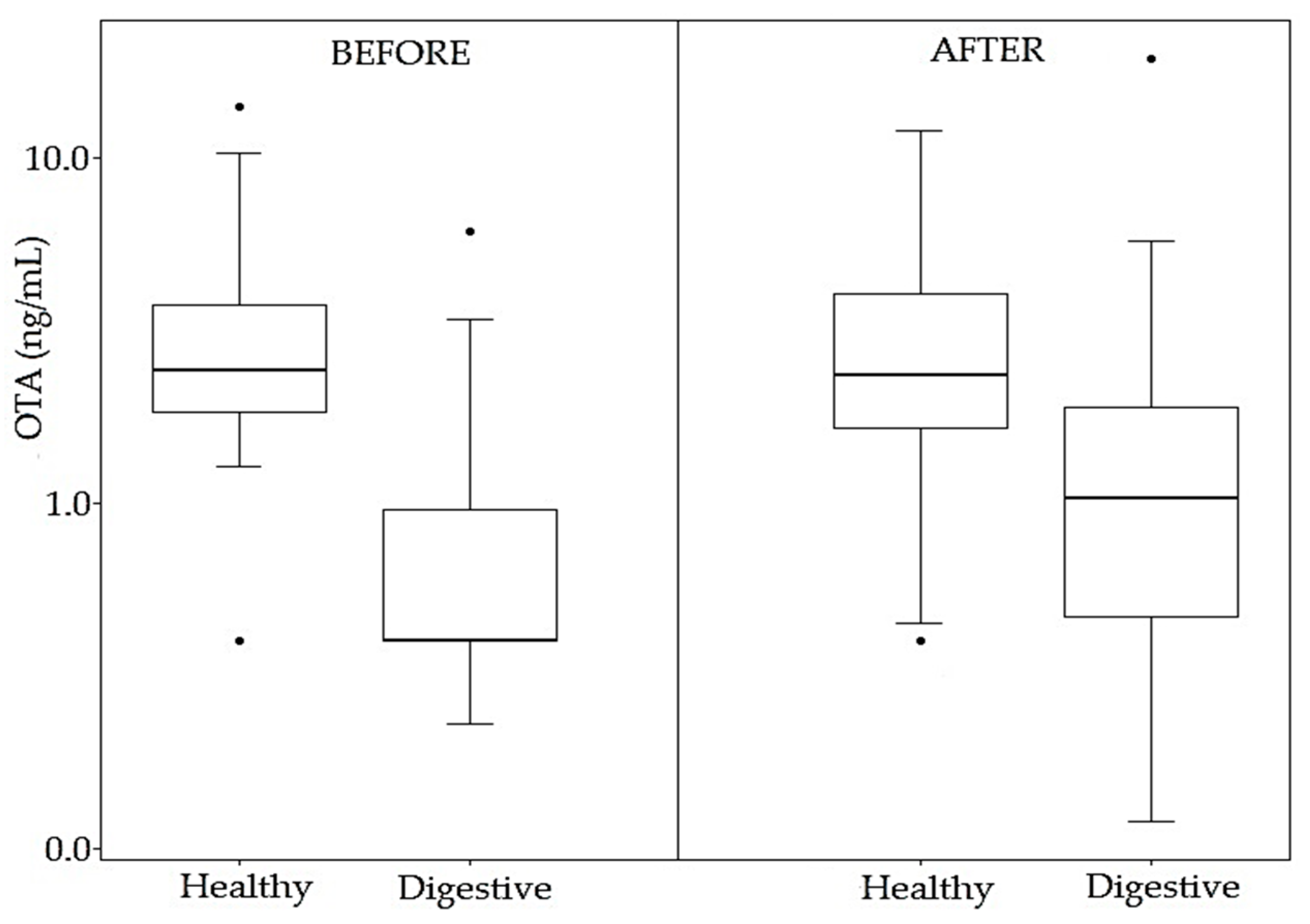
2.4. Mycotoxins in Samples
2.4.1. Results before Enzymatic Treatment
2.4.2. Results after Enzymatic Treatment
3. Conclusions
4. Materials and Methods
4.1. Subject Recruitment
4.2. Plasma Sample Collection
4.3. Sample Analysis
4.4. Control of the Analytical Sequences
4.5. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 15-ADON | 15-acetyldeoxynivalenol |
| 3-ADON | 3-acetyldeoxynivalenol |
| ACN | Acetonitrile |
| ADHD | Attention deficit hyperactivity disorder |
| AFB1 | Aflatoxin B1 |
| AFB2 | Aflatoxin B2 |
| AFG1 | Aflatoxin G1 |
| AFG2 | Aflatoxin G2 |
| AFM1 | Aflatoxin M1 |
| AFs | Aflatoxins |
| ASD | Autism spectrum disorder |
| CI | Confidence interval |
| DAS | Diacetoxyscirpenol |
| DOM-1 | Deepoxy-deoxynivalenol |
| DON | Deoxynivalenol |
| EDTA | Ethylendiaminetetraacetic acid |
| EMA | European Medicines Agency |
| ESI | Electrospray ionization |
| FBs | Fumonisins |
| FDA | Food and Drug Administration |
| FUS-X | Fusarenon-X |
| HT-2 | HT-2 toxin |
| IARC | International Agency for Research on Cancer |
| LC | Liquid chromatography |
| LC-MS/MS | Liquid chromatography–mass spectrometry |
| LOD | Limit of detection |
| LOQ | Limit of quantification |
| NEO | Neosolaniol |
| NIV | Nivalenol |
| OTA | Ochratoxin A |
| OTA-d5 | Ochratoxin A-(phenyl-d5) |
| OTB | Ochratoxin B |
| PAT | Patulin |
| PBS | Phosphate buffer solution |
| q | Transition of qualification |
| Q | Transition of quantification |
| QqQ | Triple quadrupole |
| RE | Relative error of the mean |
| RSD | Relative standard deviation |
| RT | Retention time |
| SD | Standard deviation |
| STER | Sterigmatocystin |
| T-2 | T-2 toxin |
| TCT | Trichothecenes |
| ZEA | Zearalenone |
References
- Peraica, M.; Richter, D.; Rašić, D. Mycotoxicoses in children. Arch. Ind. Hyg. Toxicol. 2014, 65, 347–363. [Google Scholar] [CrossRef] [Green Version]
- Marín, S.; Cano-Sancho, G.; Sanchis, V.; Ramos, A.J. The role of mycotoxins in the human exposome: Application of mycotoxin biomarkers in exposome-health studies. Food Chem. Toxicol. 2018, 121, 504–518. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raiola, A.; Tenore, G.C.; Manyes, L.; Meca, G.; Ritieni, A. Risk analysis of main mycotoxins occurring in food for children: An overview. Food Chem. Toxicol. 2015, 84, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Sherif, S.O.; Salama, E.E.; Abdel-Wahhab, M.A. Mycotoxins and child health: The need for health risk assessment. Int. J. Hyg. Environ. Health 2009, 212, 347–368. [Google Scholar] [CrossRef]
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans; Kujawa, M. Volume 56—Some Naturally Occurring Substances: Food Items and Constituents, Heterocyclic Aromatic Amines and Mycotoxins. In IARC Monographs on the Evaluation of Carcinogenic Risk to Humans; WHO: Geneva, Switzerland, 1994; p. 351. [Google Scholar]
- World Health Organization (WHO). Mycotoxins. Fact Sheets; WHO: Geneva, Switzerland, 2018. [Google Scholar]
- Bhat, R.; Rai, R.V.; Karim, A.A. Mycotoxins in Food and Feed: Present Status and Future Concerns. Compr. Rev. Food Sci. Food Saf. 2010, 9, 57–81. [Google Scholar] [CrossRef] [PubMed]
- Viegas, S.; Martins, C. The Usefulness of Human Biomonitoring in the Case of Mycotoxins Exposure Assessment. In Reference Module in Life Sciences; Elsevier: Amsterdam, The Netherlands, 2020; pp. 1–6. [Google Scholar]
- Al-Jaal, B.A.; Jaganjac, M.; Barcaru, A.; Horvatovich, P.; Latiff, A. Aflatoxin, fumonisin, ochratoxin, zearalenone and deoxynivalenol biomarkers in human biological fluids: A systematic literature review, 2001–2018. Food Chem. Toxicol. 2019, 129, 211–228. [Google Scholar] [CrossRef] [Green Version]
- Watson, S.; Moore, S.E.; Darboe, M.K.; Chen, G.; Tu, Y.-K.; Huang, Y.-T.; Eriksen, K.G.; Bernstein, R.M.; Prentice, A.M.; Wild, C.P.; et al. Impaired growth in rural Gambian infants exposed to aflatoxin: A prospective cohort study. BMC Public Health 2018, 18, 1247. [Google Scholar] [CrossRef]
- Shirima, C.P.; Kimanya, M.E.; Routledge, M.N.; Srey, C.; Kinabo, J.L.; Humpf, H.; Wild, C.P.; Tu, Y.-K.; Gong, Y.Y. A Pro-spective Study of Growth and Biomarkers of Exposure to Aflatoxin and fumonisin during early childhood in Tanzania. Child. Health 2015, 123, 173–179. [Google Scholar]
- Shirima, C.P.; Kimanya, M.E.; Kinabo, J.L.; Routledge, M.N.; Srey, C.; Wild, C.P.; Gong, Y.Y. Dietary exposure to aflatoxin and fumonisin among Tanzanian children as determined using biomarkers of exposure. Mol. Nutr. Food Res. 2013, 57, 1874–1881. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mitchell, N.J.; Hsu, H.-H.; Chandyo, R.K.; Shrestha, B.; Bodhidatta, L.; Tu, Y.-K.; Gong, Y.-Y.; Egner, P.A.; Ulak, M.; Groopman, J.D.; et al. Aflatoxin exposure during the first 36 months of life was not associated with impaired growth in Nepalese children: An extension of the MAL-ED study. PLoS ONE 2017, 12, e0172124. [Google Scholar] [CrossRef] [Green Version]
- Groopman, J.D.; Egner, P.A.; Schulze, K.J.; Wu, L.S.-F.; Merrill, R.D.; Mehra, S.; Shamim, A.A.; Ali, H.; Shaikh, S.; Gernand, A.D.; et al. Aflatoxin exposure during the first 1000 days of life in rural South Asia assessed by aflatoxin B1-lysine albumin biomarkers. Food Chem. Toxicol. 2014, 74, 184–189. [Google Scholar] [CrossRef] [Green Version]
- Gong, Y.Y.; Wilson, S.; Mwatha, J.K.; Routledge, M.N.; Castelino, J.M.; Zhao, B.; Kimani, G.; Kariuki, H.C.; Vennervald, B.J.; Dunne, D.W.; et al. Aflatoxin Exposure May Contribute to Chronic Hepatomegaly in Kenyan School Children. Environ. Health Perspect. 2012, 120, 893–896. [Google Scholar] [CrossRef] [PubMed]
- Mahfuz, M.; Alam, M.A.; Fahim, S.M.; Gazi, A.; Raihan, M.J.; Hossain, M.; Egner, P.A.; Bessong, P.O.; Petri, W.A.; Groopman, J.D.; et al. Aflatoxin exposure in children living in Mirpur, Dhaka: Data from MAL-ED companion study. J. Expo. Sci. Environ. Epidemiol. 2019, 29, 655–662. [Google Scholar] [CrossRef] [Green Version]
- McMillan, A.; Renaud, J.B.; Burgess, K.M.; Orimadegun, A.E.; Akinyinka, O.O.; Allen, S.J.; Miller, J.D.; Reid, G.; Sumarah, M.W. Aflatoxin exposure in Nigerian children with severe acute malnutrition. Food Chem. Toxicol. 2018, 111, 356–362. [Google Scholar] [CrossRef]
- Shouman, B.O.; El Morsi, D.; Shabaan, S.; Abdel-Hamid, A.-H.; Mehrim, A. Aflatoxin B1 Level in Relation to Child’s Feeding and Growth. Indian J. Pediatr. 2012, 79, 56–61. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Mitchell, N.J.; Gratz, J.; Houpt, E.R.; Gong, Y.; Egner, P.A.; Groopman, J.D.; Riley, R.T.; Showker, J.L.; Svensen, E.; et al. Exposure to aflatoxin and fumonisin in children at risk for growth impairment in rural Tanzania. Environ. Int. 2018, 115, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Lombard, M.J. Mycotoxin exposure and infant and young child growth in Africa: What do we know? Ann. Nutr. Metab. 2014, 64, 42–52. [Google Scholar] [CrossRef]
- Ojuri, O.T.; Ezekiel, C.N.; Sulyok, M.; Ezeokoli, O.T.; Oyedele, O.A.; Ayeni, K.I.; Eskola, M.K.; Šarkanj, B.; HajšLová, J.; Adeleke, R.A.; et al. Assessing the mycotoxicological risk from consumption of complementary foods by infants and young children in Nigeria. Food Chem. Toxicol. 2018, 121, 37–50. [Google Scholar] [CrossRef]
- Githang’A, D.; Anzala, O.; Mutegi, C.; Agweyu, A. The effects of exposures to mycotoxins on immunity in children: A systematic review. Curr. Probl. Pediatr. Adolesc. Health Care 2019, 49, 109–116. [Google Scholar] [CrossRef]
- Voth-Gaeddert, L.E.; Stoker, M.; Torres, O.; Oerther, D.B. Association of aflatoxin exposure and height-for-age among young children in Guatemala. Int. J. Environ. Health Res. 2018, 28, 280–292. [Google Scholar] [CrossRef]
- Soriano, J.M.; Rubini, A.; Morales-Suarez-Varela, M.; Merino-Torres, J.F.; Silvestre, D. Aflatoxins in organs and biological samples from children affected by kwashiorkor, marasmus and marasmic-kwashiorkor: A scoping review. Toxicon 2020, 185, 174–183. [Google Scholar] [CrossRef] [PubMed]
- Tesfamariam, K.; De Boevre, M.; Kolsteren, P.; Belachew, T.; Mesfin, A.; De Saeger, S.; Lachat, C. Dietary mycotoxins exposure and child growth, immune system, morbidity, and mortality: A systematic literature review. Crit. Rev. Food Sci. Nutr. 2020, 60, 3321–3341. [Google Scholar] [CrossRef] [PubMed]
- Etzel, R.A. What the Primary Care Pediatrician Should Know about Syndromes Associated with Exposures to Mycotoxins. Curr. Probl. Pediatr. Adolesc. Health Care 2006, 36, 282–305. [Google Scholar] [CrossRef]
- Guerre, P. Mycotoxin and Gut Microbiota Interactions. Toxins 2020, 12, 769. [Google Scholar] [CrossRef] [PubMed]
- Akbari, P.; Braber, S.; Varasteh, S.; Alizadeh, A.; Garssen, J.; Fink-Gremmels, J. The intestinal barrier as an emerging target in the toxicological assessment of mycotoxins. Arch. Toxicol. 2017, 91, 1007–1029. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Santis, B.; Raggi, M.E.; Moretti, G.; Facchiano, F.; Mezzelani, A.; Villa, L.; Bonfanti, A.; Campioni, A.; Rossi, S.; Camposeo, S.; et al. Study on the Association among Mycotoxins and other Variables in Children with Autism. Toxins 2017, 9, 203. [Google Scholar] [CrossRef]
- De Santis, B.; Brera, C.; Mezzelani, A.; Soricelli, S.; Ciceri, F.; Moretti, G.; Debegnach, F.; Bonaglia, M.C.; Villa, L.; Molteni, M.; et al. Role of mycotoxins in the pathobiology of autism: A first evidence. Nutr. Neurosci. 2019, 22, 132–144. [Google Scholar] [CrossRef]
- Duringer, J.; Fombonne, E.; Craig, M. No Association between Mycotoxin Exposure and Autism: A Pilot Case-Control Study in School-Aged Children. Toxins 2016, 8, 224. [Google Scholar] [CrossRef] [Green Version]
- Assunção, R.; Martins, C.; Vasco, E.; Jager, A.; Oliveira, C.; Cunha, S.C.; Fernandes, J.O.; Nunes, B.; Loureiro, S.; Alvito, P. Portuguese children dietary exposure to multiple mycotoxins—An overview of risk assessment under MYCOMIX project. Food Chem. Toxicol. 2018, 118, 399–408. [Google Scholar] [CrossRef]
- Assunção, R.; Martins, C.; Dupont, D.; Alvito, P. Patulin and ochratoxin A co-occurrence and their bioaccessibility in processed cereal-based foods: A contribution for Portuguese children risk assessment. Food Chem. Toxicol. 2016, 96, 205–214. [Google Scholar] [CrossRef]
- Sirot, V.; Fremy, J.-M.; Leblanc, J.-C. Dietary exposure to mycotoxins and health risk assessment in the second French total diet study. Food Chem. Toxicol. 2013, 52, 1–11. [Google Scholar] [CrossRef]
- Postupolski, J.; Starski, A.; Ledzion, E.; Kurpińska-Jaworska, J.; Szczęsna, M. Exposure assessment of infants and young chil-dren on selected Fusarium toxins. Rocz. Panstw. Zakl. Hig. 2019, 70, 5–14. [Google Scholar] [CrossRef]
- Brera, C.; Debegnach, F.; De Santis, B.; Di Ianni, S.; Gregori, E.; Neuhold, S.; Valitutti, F. Exposure assessment to mycotoxins in gluten-free diet for celiac patients. Food Chem. Toxicol. 2014, 69, 13–17. [Google Scholar] [CrossRef]
- Raiola, A.; Meca, G.; Mañes, J.; Ritieni, A. Bioaccessibility of Deoxynivalenol and its natural co-occurrence with Ochratoxin A and Aflatoxin B1 in Italian commercial pasta. Food Chem. Toxicol. 2012, 50, 280–287. [Google Scholar] [CrossRef] [Green Version]
- Oueslati, S.; Blesa, J.; Molto, J.C.; Ghorbel, A.; Mañes, J. Presence of mycotoxins in sorghum and intake estimation in Tunisia. Food Addit. Contam. Part A 2014, 31, 307–318. [Google Scholar] [CrossRef] [PubMed]
- Pallarés, N.; Carballo, D.; Ferrer, E.; Fernández-Franzón, M.; Berrada, H. Mycotoxin Dietary Exposure Assessment through Fruit Juices Consumption in Children and Adult Population. Toxins 2019, 11, 684. [Google Scholar] [CrossRef] [Green Version]
- Arce-López, B.; Lizarraga, E.; Vettorazzi, A.; González-Peñas, E. Human Biomonitoring of Mycotoxins in Blood, Plasma and Serum in Recent Years: A Review. Toxins 2020, 12, 147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arce-López, B.; Lizarraga, E.; Irigoyen, Á.; González-Peñas, E. Presence of 19 Mycotoxins in Human Plasma in a Region of Northern Spain. Toxins 2020, 12, 750. [Google Scholar] [CrossRef]
- Lemming, E.W.; Montes, A.M.; Schmidt, J.; Cramer, B.; Humpf, H.-U.; Moraeus, L.; Olsen, M. Mycotoxins in blood and urine of Swedish adolescents—Possible associations to food intake and other background characteristics. Mycotoxin Res. 2019, 36, 193–206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Erkekoǧlu, P.; Sabuncuoǧlu, S.; Aydin, S.; Şahin, G.; Giray, B. Determination of seasonal variations in serum ochratoxin A levels in healthy population living in some regions of Turkey by enzyme-linked immunosorbent assay. Toxicon 2010, 55, 507–513. [Google Scholar] [CrossRef]
- Nieto, C.H.D.; Granero, A.M.; Zon, M.A.; Fernández, H. Sterigmatocystin: A mycotoxin to be seriously considered. Food Chem. Toxicol. 2018, 118, 460–470. [Google Scholar] [CrossRef] [PubMed]
- International Agency for Research on Cancer. Overall Evaluations of Carcinogenicity: An Updating of IARC Monographs Volumes 1 to 42; WHO: Geneva, Switzerland, 1987; Volume 46. [Google Scholar]
- Arce-López, B.; Lizarraga, E.; Flores-Flores, M.; Irigoyen, Á.; González-Peñas, E. Development and validation of a methodology based on Captiva EMR-lipid clean-up and LC-MS/MS analysis for the simultaneous determination of mycotoxins in human plasma. Talanta 2020, 206, 120193. [Google Scholar] [CrossRef]
- Food and Drug Administration. Bioanalytical Method Validation Guidance for Industry; Food and Drug Administration: Silver Spring, MD, USA, 2018; Volume 1043.
- European Commission. Commission Decision of 12 August 2002 implementing Council Directive 96/23/EC concerning the performance of analytical methods and the interpretation of results (2002/657/EC). Off. J. Eur. Communities 2002, 221, 8–36. [Google Scholar]
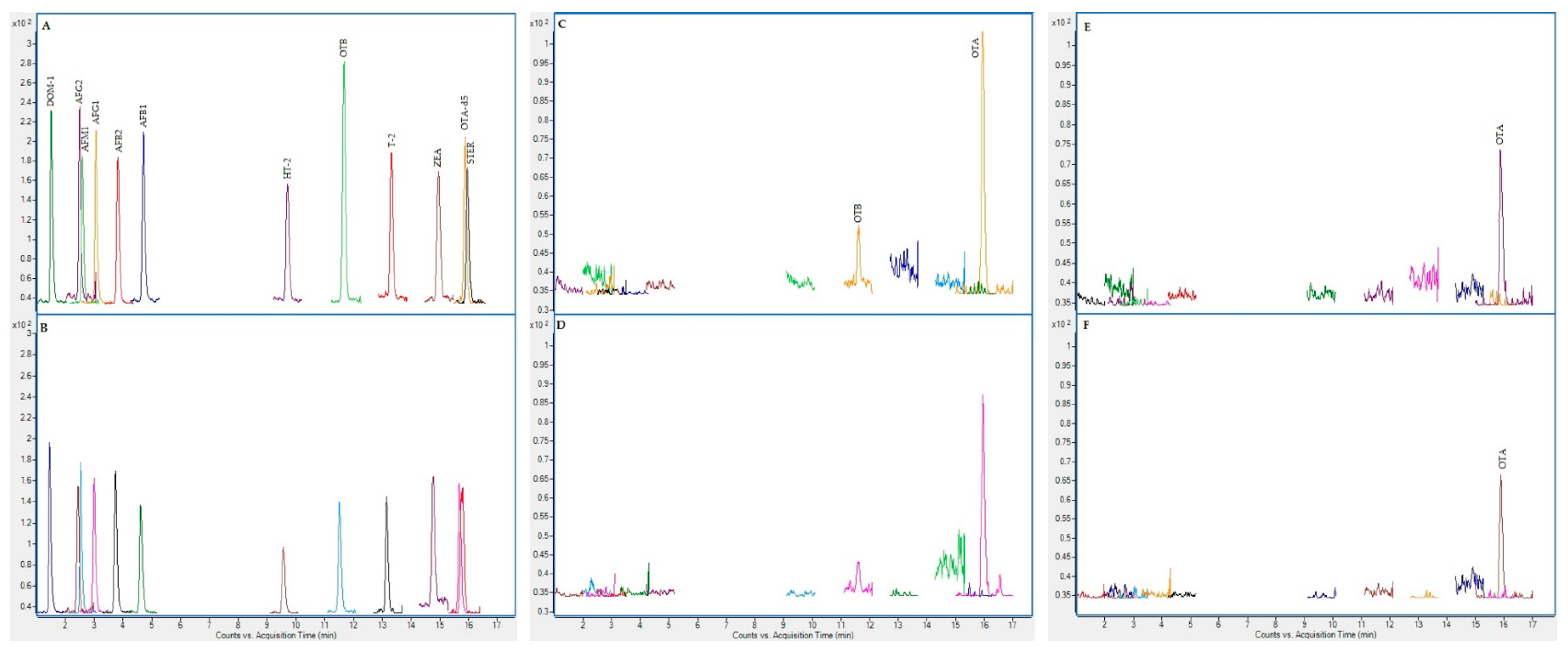

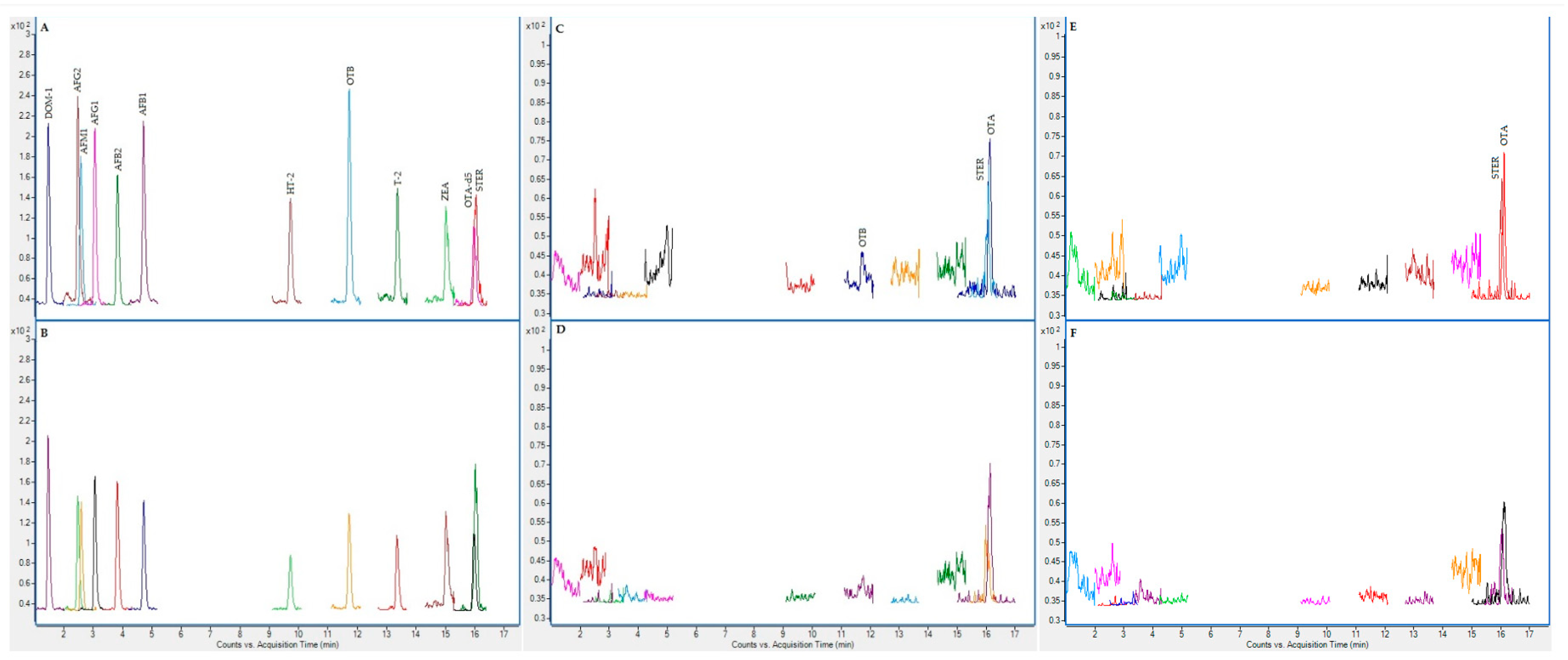
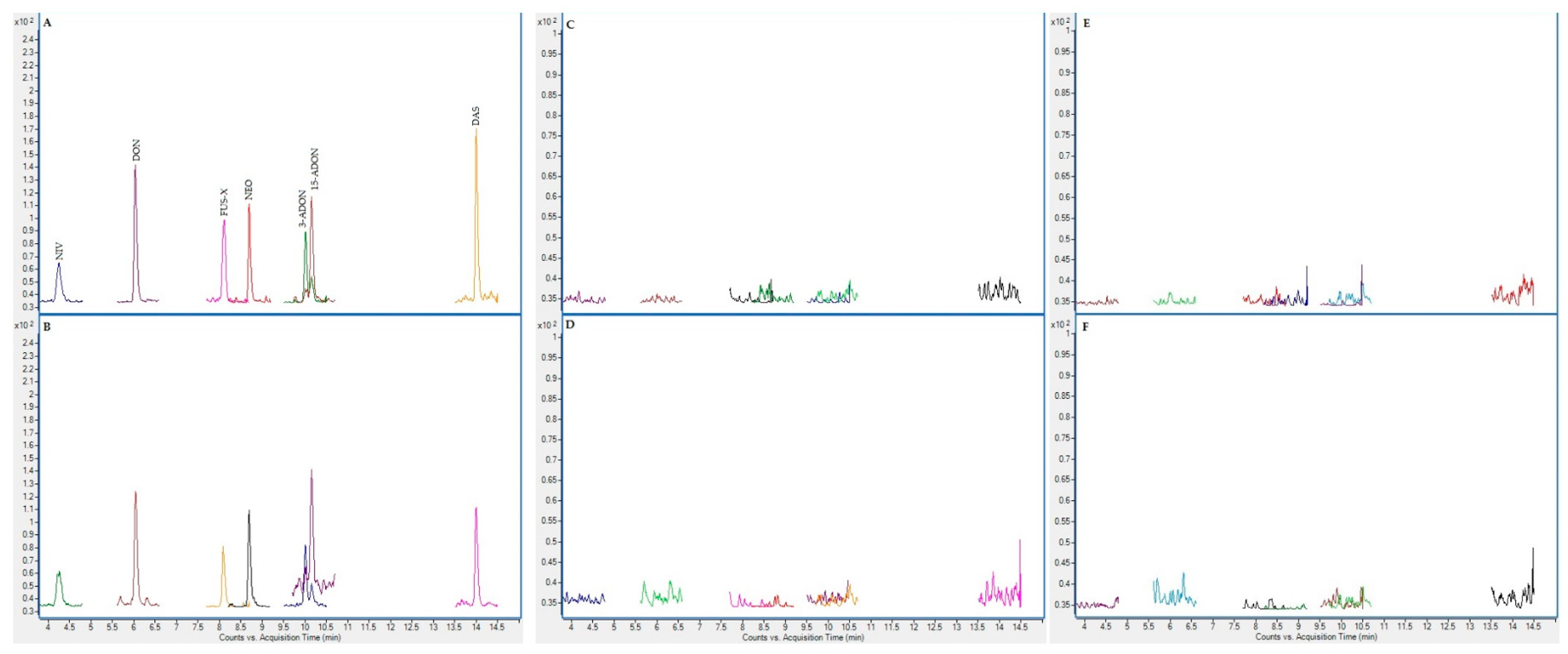

| OTA | OTB | STER | ||||
|---|---|---|---|---|---|---|
| BE | AE | BE | AE | AE | ||
| Calibrators | q/Q ratio | 77.41 ± 4.09 | 78.50 ± 5.71 | 44.74 ± 2.23 | 46.02 ± 1.45 | 95.07 ± 12.87 |
| RT (min) | 15.90 ± 0.22 | 16.04 ± 0.07 | 11.68 ± 0.17 | 11.79 ± 0.06 | 16.10 ± 0.07 | |
| Samples | q/Q ratio | 73.69 ± 7.63 | 78.94 ± 9.22 | 43.65 ± 3.99 | 46.06 ± 6.35 | 97.17 ± 12.15 |
| RT (min) | 15.96 ± 0.13 | 16.18 ± 0.06 | 11.60 ± 0.08 | 11.74 ± 0.04 | 16.10 ± 0.06 | |
| Health Status | Gender | n | Range (Years) | Mean ± SD (Years) | Total Mean ± SD (Years) |
|---|---|---|---|---|---|
| Healthy | Girls | 28 | 6–15 | 11.2 ± 2.4 | 10.6 ± 2.8 |
| Boys | 12 | 4–13 | 9.1 ± 3.1 | ||
| ADHD | Girls | 1 | 6 | 6 | 9.7 ± 3.3 |
| Boys | 6 | 7–14 | 10.3 ± 3.1 | ||
| ASD | Girls | 0 | n.s. | n.s. | 6.5 ± 2.1 |
| Boys | 2 | 5–8 | 6.5 ± 2.1 | ||
| Digestive | Girls | 15 | 2–16 | 8.1 ± 4.4 | 9.0 ± 4.4 |
| Boys | 15 | 4–15 | 10.3 ± 3.9 |
| Healthy Girls | BE | AE | Healthy Boys | BE | AE | Patient Girls | BE | AE | Patient Boys | BE | AE | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample | Age | OTA | OTB | OTA | STER | Sample | Age | OTA | OTB | OTA | STER | Sample | Age | OTA | OTA | STER | Sample | Age | OTA | OTA | STER |
| PS2 | 13 | 2.9 | 1.6 | PS1 | 12 | 4.7 | 0.5 a | 1.9 | 1.2 | PD2 | 16 | 2.9 | 1.6 | PD1 | 12 | 1.2 | |||||
| PS3 | 11 | 2.3 | 1.2 | 1.5 | PS5 | 4 | 4 | 4.7 | 2.5 | PD3 | 4 | 34.2 | 19.3 | 1.7 | PD5 | 6 | 0.5 | 3.4 | 2.1 | ||
| PS4 | 10 | 2.8 | 1.9 | 1.3 | PS6 | 6 | 6 | 2.6 | 0.9 | PD4 | 6 | 0.5 | 0.7 | 1 | PD6 | 8 | 0.5 | 1.3 | |||
| PS7 | 9 | 2.1 | 1.5 | PS9 | 13 | 2.6 | 0.4 | 2.3 | 1.1 | PD9 | 14 | 2 | 1.9 | 1.6 | PD7 | 15 | 1.1 | 1.4 | |||
| PS8 | 12 | 1.5 | 3.8 | 1.2 | PS15 | 10 | 2.7 | 1.6 | 1.2 | PD10 | 3 | 0.5 | 1.9 | PD8 | 15 | 1.9 | 1.3 | ||||
| PS10 | 12 | 1.5 | 1.4 | PS23 | 11 | 2.4 | 2.9 | 1.5 | PD12 | 13 | 2.2 | 2.9 | 1.8 | PD11 | 8 | 1.8 | 1.8 | ||||
| PS11 | 6 | 3.9 | 0.7 | 11.9 | 1.6 | PS25 | 10 | 1.8 | 2 | 1.3 | PD13 | 7 | 2.2 | 1.8 | PD15 | 5 | 1.2 | 1.7 | |||
| PS12 | 10 | 1.9 | 1.9 | 2.5 | PS27 | 7 | 2.3 | 4 | 2 | PD14 | 9 | 1.5 | 1.4 | PD16 | 7 | 0.9 | 1.9 | ||||
| PS13 | 12 | 3.9 | 2.6 | 1.6 | PS28 | 9 | 4.3 | 0.5 | 4.9 | 1 | PD17 | 9 | 1.3 | 1.6 | PD18 | 5 | 1.3 | 2.4 | |||
| PS14 | 13 | 1.6 | 1.6 | PS31 | 4 | 2.4 | 0.5 | 2 | 1.3 | PD19 | 9 | 0.7 | 1.6 | PD20 | 13 | 0.8 | |||||
| PS16 | 15 | 3.4 | 4.1 | 1.5 | PS39 | 12 | 1.3 | 2.5 | 1.3 | PD22 | 9 | 3.4 | 1.5 | PD21 | 12 | 1 | 1.1 | ||||
| PS17 | 8 | 1.7 | 1.7 | 1.4 | PS40 | 11 | 2.4 | 2.5 | 1.3 | PD24 | 3 | 1.5 | PD23 | 10 | 6.1 | 5.8 | 1.5 | ||||
| PS18 | 9 | 2.4 | 2.1 | 1.2 | PD25 | 5 | 0.8 | 1.3 | PD26 | 4 | 0.7 | 1.6 | |||||||||
| PS19 | 11 | 1.9 | 1.2 | 1.5 | PD28 | 2 | 1.3 | 1.4 | 1.4 | PD27 | 12 | 2.2 | 1.9 | ||||||||
| PS21 | 12 | 5.3 | 9.1 | 1.6 | PD29 | 13 | 0.6 | 1 | PD30 | 15 | 1.6 | 1.4 | |||||||||
| PS22 | 14 | 2.2 | 7 | 1.4 | PA1 | 5 | 1.4 | ||||||||||||||
| PS24 | 14 | 2 | 1.1 | 0.9 | PA2 | 8 | 1.1 | 1.9 | 1.5 | ||||||||||||
| PS26 | 7 | 3.7 | 0.5 | 2.7 | 1.4 | PT5 | 6 | PT1 | 0.7 | 1.3 | |||||||||||
| PS29 | 14 | 14.1 | 0.6 | 8.9 | 1.5 | PT2 | 0.6 | 1.3 | |||||||||||||
| PS30 | 9 | 3 | 6.6 | 2 | PT3 | 14 | 0.6 | 1.4 | |||||||||||||
| PS32 | 8 | 1.8 | 0.9 | PT4 | 8 | 1 | 1.7 | ||||||||||||||
| PS33 | 10 | 2.9 | 1 | PT6 | 14 | 1.1 | 2 | ||||||||||||||
| PS34 | 14 | 2.1 | 4 | 1.7 | PT7 | 8 | 0.5 | 0.9 | 1.2 | ||||||||||||
| PS35 | 14 | 9.6 | 0.6 | 7.8 | 1.4 | 7 | |||||||||||||||
| PS36 | 11 | 1.6 | 0.4 | 11 | |||||||||||||||||
| PS37 | 14 | 10.2 | 0.8 | 7.2 | 1.6 | ||||||||||||||||
| PS38 | 11 | 2.8 | 1.7 | 1.1 | |||||||||||||||||
| PS41 | 10 | 2 | 1.3 | 0.9 | |||||||||||||||||
| Health Status | Gender | n | % Positives | Mean ± SD (ng/mL) | Median (ng/mL) | Max Value (ng/mL) | |
|---|---|---|---|---|---|---|---|
| BE | Healthy | Girls | 28 | 89.3 | 3.27 ± 3.10 | 2.27 | 14.07 |
| Boys | 12 | 100 | 3.08 ± 1.37 | 2.50 | 6.03 | ||
| Total | 40 | 92.5 | 3.21 ± 2.69 | 2.42 | 14.07 | ||
| Digestive disorder | Girls | 15 | 46.7 | 1.10 ± 1.07 | 0.40 | 3.39 | |
| Boys | 15 | 26.7 | 0.89 ± 1.47 | 0.40 | 6.07 | ||
| Total | 30 | 36.7 | 0.99 ± 1.27 | 0.40 | 6.07 | ||
| AE | Healthy | Girls | 28 | 89.3 | 3.49 ± 3.15 | 2.07 | 11.92 |
| Boys | 12 | 100 | 2.82 ± 1.11 | 2.50 | 4.69 | ||
| Total | 40 | 92.5 | 3.29 ± 2.70 | 2.36 | 11.92 | ||
| Digestive disorder | Girls | 15 | 80 | 2.33 ± 4.75 | 0.75 | 19.27 | |
| Boys | 15 | 73.3 | 1.46 ± 1.48 | 1.14 | 5.75 | ||
| Total | 30 | 76.7 | 1.90 ± 3.48 | 1.04 | 19.27 |
| Health Status | Gender | n | % Positives | Mean ± SD (ng/mL) | Median (ng/mL) | Max Value (ng/mL) |
|---|---|---|---|---|---|---|
| Healthy | Girls | 28 | 96.4 | 1.43 ± 0.34 | 1.44 | 2.51 |
| Boys | 12 | 100 | 1.36 ± 0.46 | 1.26 | 2.54 | |
| Total | 40 | 97.5 | 1.41 ± 0.38 | 1.41 | 2.54 | |
| Digestive Disorders | Boys | 15 | 100 | 1.51 ± 0.25 | 1.57 | 1.91 |
| Girls | 15 | 100 | 1.55 ± 0.41 | 1.48 | 2.35 | |
| Total | 30 | 100 | 1.53 ± 0.33 | 1.54 | 2.35 |
| Column | Ascentis Express C18, 2.7 μm, 150 × 2.1 mm (Supelco Analytical, St. Louis, MO, USA) at 45 °C |
| Mobile Phase | Mixture of A (5 mM ammonium formate and 0.1% formic acid in water) and B (5 mM ammonium formate and 0.1% formic acid in 95:5 methanol/water) in gradient conditions |
| Flow Rate | 0.4 mL/min |
| Volume of Injection | 20 μL |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arce-López, B.; Lizarraga, E.; López de Mesa, R.; González-Peñas, E. Assessment of Exposure to Mycotoxins in Spanish Children through the Analysis of Their Levels in Plasma Samples. Toxins 2021, 13, 150. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins13020150
Arce-López B, Lizarraga E, López de Mesa R, González-Peñas E. Assessment of Exposure to Mycotoxins in Spanish Children through the Analysis of Their Levels in Plasma Samples. Toxins. 2021; 13(2):150. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins13020150
Chicago/Turabian StyleArce-López, Beatriz, Elena Lizarraga, Reyes López de Mesa, and Elena González-Peñas. 2021. "Assessment of Exposure to Mycotoxins in Spanish Children through the Analysis of Their Levels in Plasma Samples" Toxins 13, no. 2: 150. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins13020150





