Acute Effects of Supervised Making Weight on Health Markers, Hormones and Body Composition in Muay Thai Fighters
Abstract
:1. Introduction
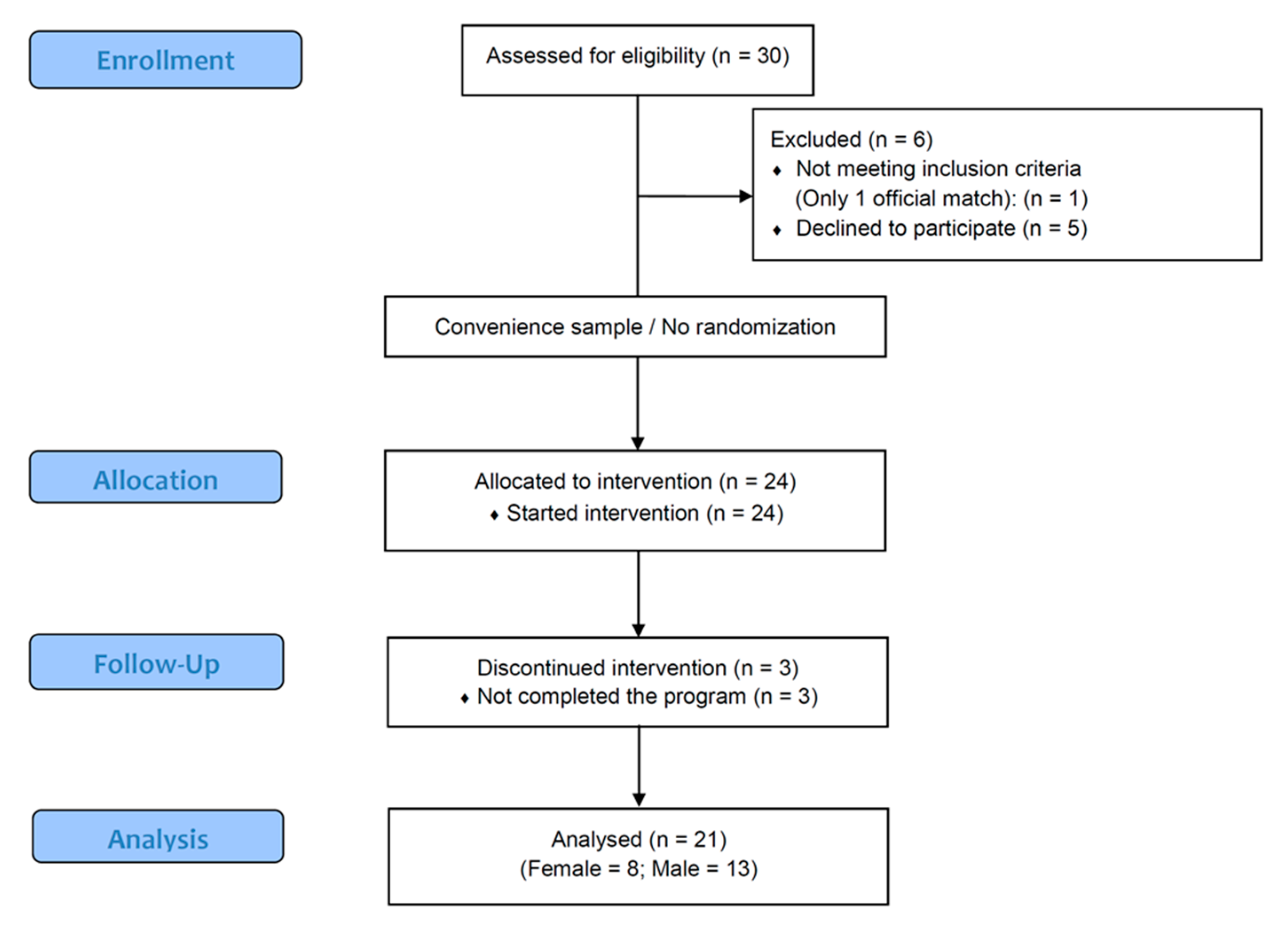
2. Methods
2.1. Trial Design
2.1.1. Participants
2.1.2. Intervention Procedures
Anthropometry
Nutrition Intervention
Exercise Regimen
2.2. Outcomes
2.2.1. Primary Outcome Measures
2.2.2. Secondary Outcome Measures
2.2.3. Sample Size
2.3. Statistical Methods
3. Results
3.1. Baseline Data
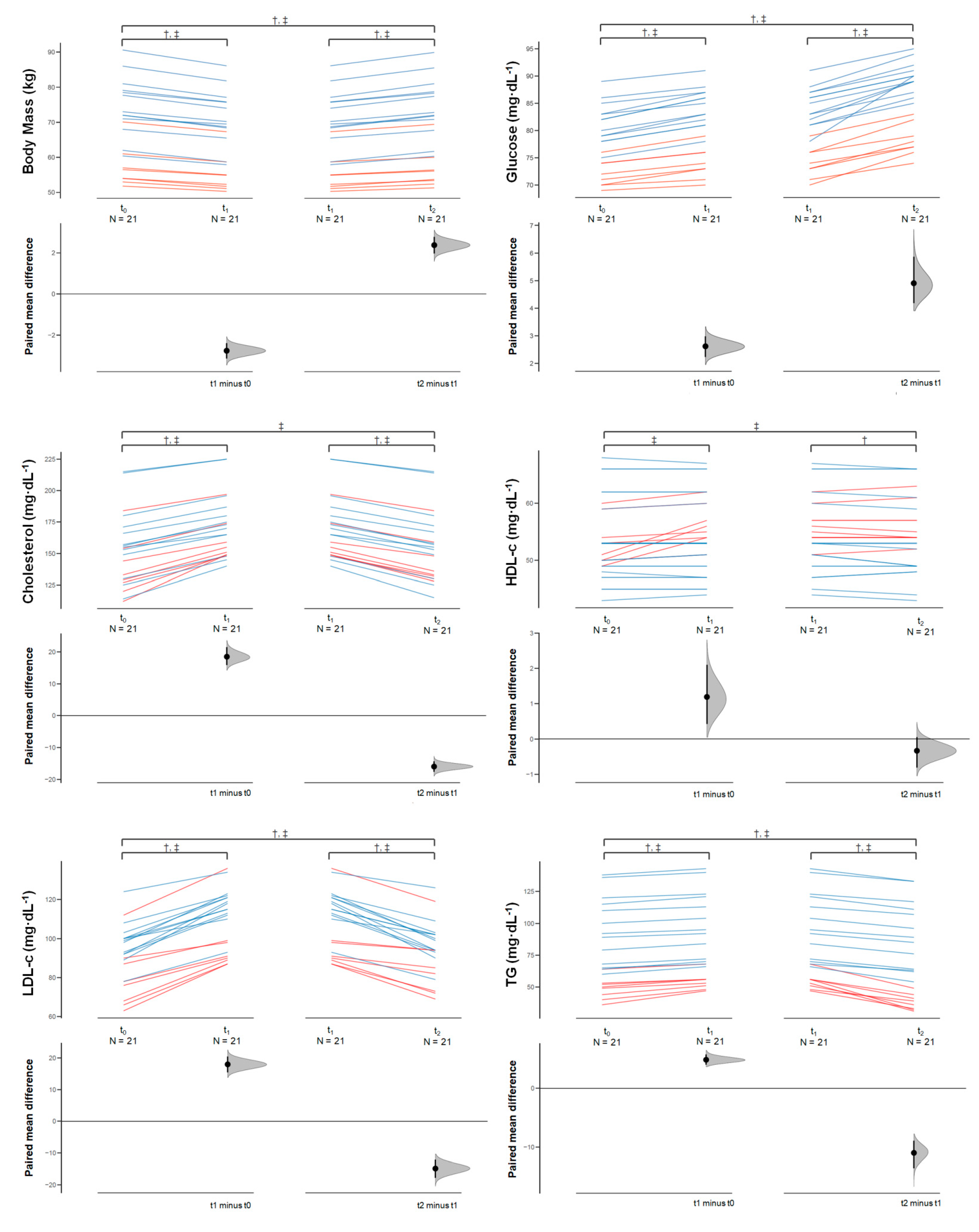
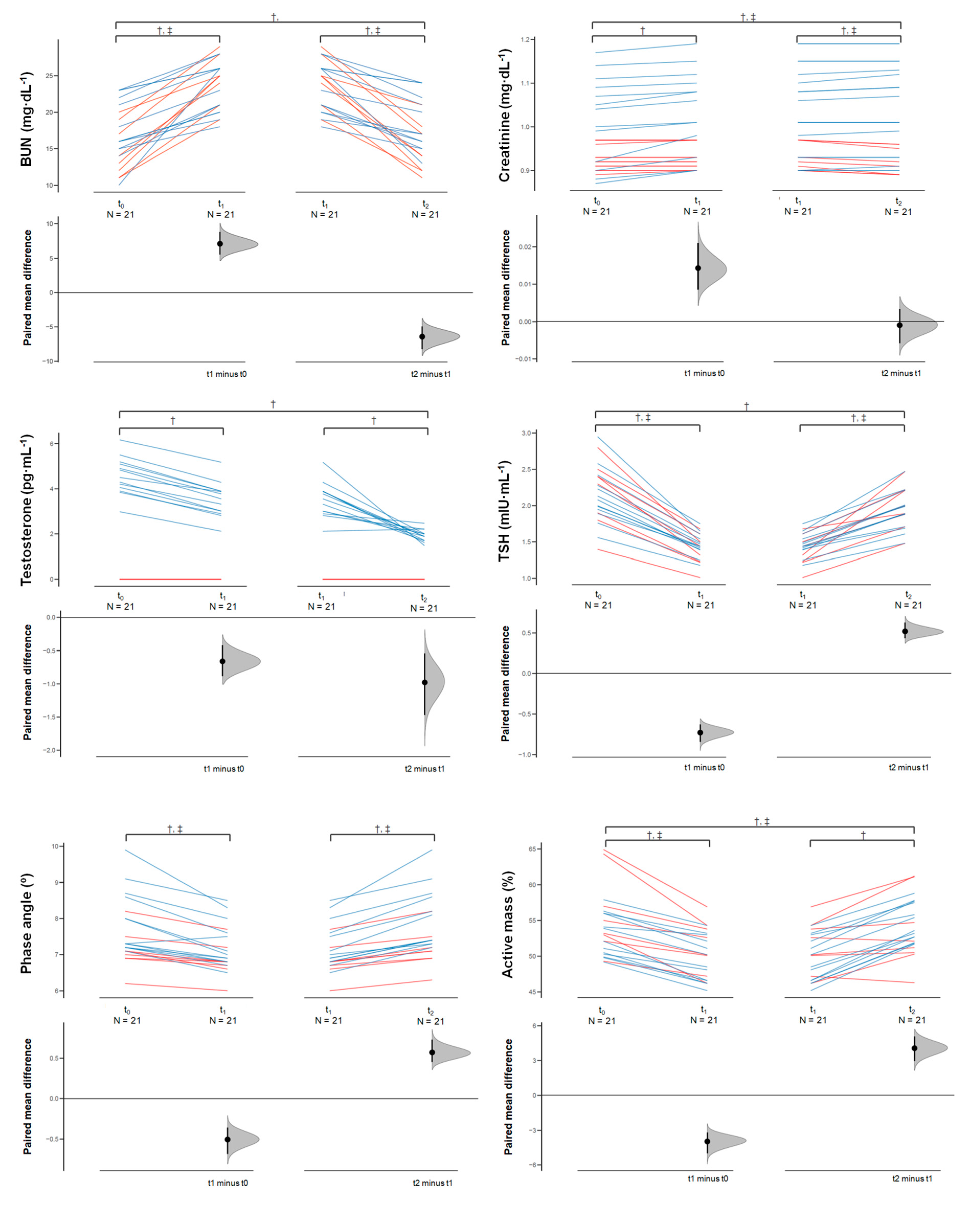
3.1.1. Outcomes
3.1.2. Rapid Weight Loss (Δ1)
3.1.3. Rapid Weight Gain (Δ2)
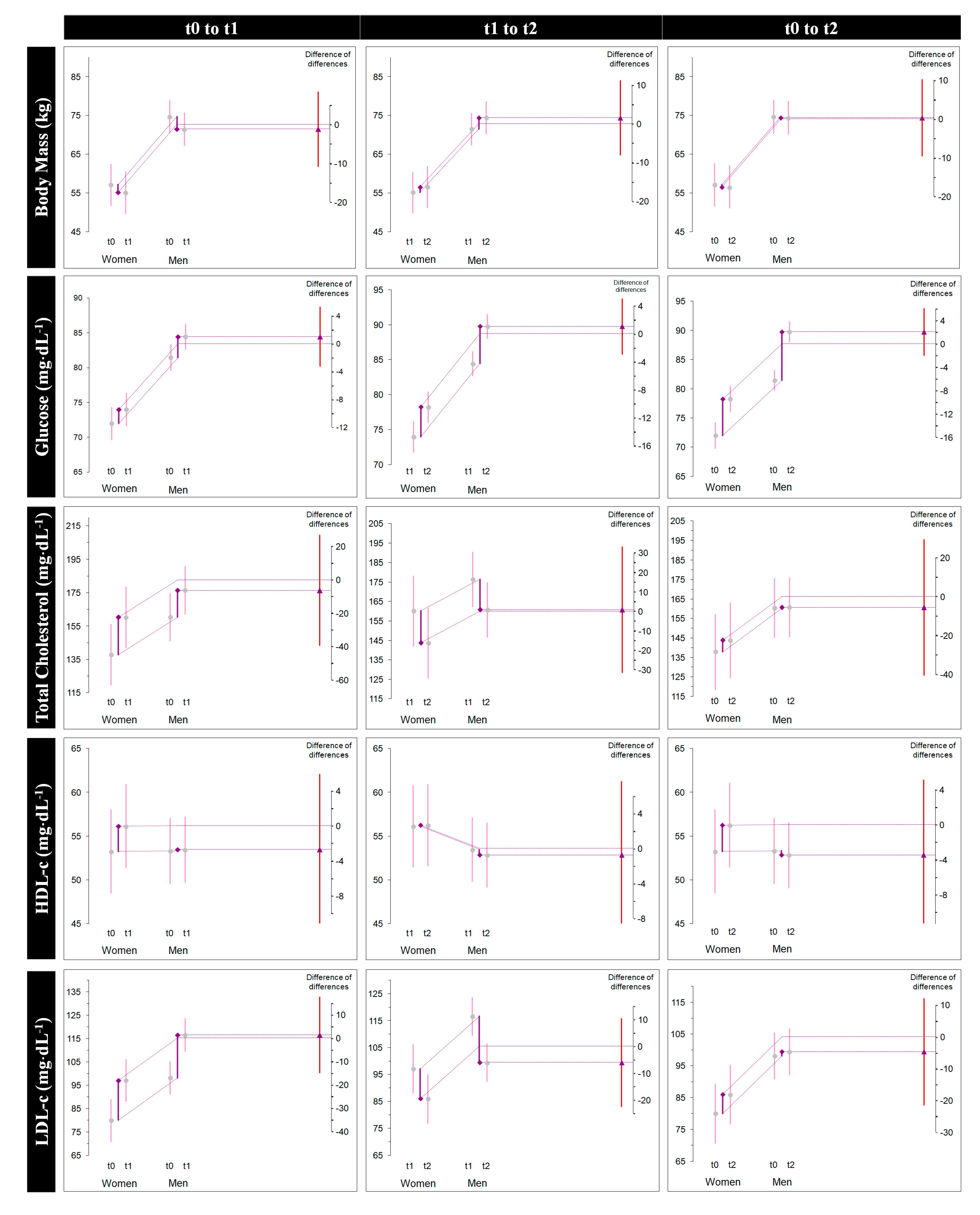
3.1.4. Overall Change (Δ3)
4. Discussion
5. Practical Applications
6. Limitations
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Barley, O.; Chapman, D.; Abbiss, C. The Current State of Weight-Cutting in Combat Sports. Sports 2019, 7, 123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khodaee, M.; Olewinski, L.; Shadgan, B.; Kiningham, R.R. Rapid Weight Loss in Sports with Weight Classes. Curr. Sports Med. Rep. 2015, 14, 435–441. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Horswill, C.A. Making Weight in Combat Sports. In Combat Sports Medicine; Springer: London, UK, 2009; pp. 21–39. [Google Scholar] [CrossRef]
- Hillier, M.; Sutton, L.; James, L.; Mojtahedi, D.; Keay, N.; Hind, K. High Prevalence and Magnitude of Rapid Weight Loss in Mixed Martial Arts Athletes. Int. J. Sport Nutr. Exerc. Metab. 2019, 29, 512. [Google Scholar] [CrossRef]
- Lenzi, J.L.; Teixeira, E.L.; de Jesus, G.; Schoenfeld, B.J.; de Salles Painelli, V. Dietary Strategies of Modern Bodybuilders during Different Phases of the Competitive Cycle. J. Strength Cond. Res. 2019. [Google Scholar] [CrossRef] [PubMed]
- Mota, J.A.; Nuckols, G.; Smith-Ryan, A.E. Nutritional Periodization. Strength Cond. J. 2019, 41, 69–78. [Google Scholar] [CrossRef]
- Franchini, E.; Brito, C.J.; Artioli, G.G. Weight loss in combat sports: Physiological, psychological and performance effects. J. Int. Soc. Sports Nutr. 2012, 9. [Google Scholar] [CrossRef] [Green Version]
- Matthews, J.J.; Stanhope, E.N.; Godwin, M.S.; Holmes, M.E.J.; Artioli, G.G. The Magnitude of Rapid Weight Loss and Rapid Weight Gain in Combat Sport Athletes Preparing for Competition: A Systematic Review. Int. J. Sport Nutr. Exerc. Metab. 2019, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Park, S.; Alencar, M.; Sassone, J.; Madrigal, L.; Ede, A. Self-reported methods of weight cutting in professional mixed-martial artists: How much are they losing and who is advising them? J. Int. Soc. Sports Nutr. 2019, 16. [Google Scholar] [CrossRef] [Green Version]
- Reale, R.; Slater, G.; Burke, L.M. Individualised dietary strategies for Olympic combat sports: Acute weight loss, recovery and competition nutrition. Eur. J. Sport Sci. 2017, 17, 727–740. [Google Scholar] [CrossRef]
- Lakicevic, N.; Roklicer, R.; Bianco, A.; Mani, D.; Paoli, A.; Trivic, T.; Ostojic, S.M.; Milovancev, A.; Maksimovic, N.; Drid, P. Effects of Rapid Weight Loss on Judo Athletes: A Systematic Review. Nutrients 2020, 12, 1220. [Google Scholar] [CrossRef]
- Crighton, B.; Close, G.L.; Morton, J.P. Alarming weight cutting behaviours in mixed martial arts: A cause for concern and a call for action. Br. J. Sports Med. 2016, 50, 446–447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berkovich, B.-E.; Stark, A.H.; Eliakim, A.; Nemet, D.; Sinai, T. Rapid Weight Loss in Competitive Judo and Taekwondo Athletes: Attitudes and Practices of Coaches and Trainers. Int. J. Sport Nutr. Exerc. Metab. 2019, 29, 532–538. [Google Scholar] [CrossRef]
- Dugonjić, B.; Krstulović, S.; Kuvačić, G. Rapid Weight Loss Practices in Elite Kickboxers. Int. J. Sport Nutr. Exerc. Metab. 2019, 29, 583–588. [Google Scholar] [CrossRef]
- Kasper, A.M.; Crighton, B.; Langan-Evans, C.; Riley, P.; Sharma, A.; Close, G.L.; Morton, J.P. Case Study: Extreme Weight Making Causes Relative Energy Deficiency, Dehydration, and Acute Kidney Injury in a Male Mixed Martial Arts Athlete. Int. J. Sport Nutr. Exerc. Metab. 2019, 29, 331–338. [Google Scholar] [CrossRef] [Green Version]
- Murugappan, K.R.; Cocchi, M.N.; Bose, S.; Neves, S.E.; Cook, C.H.; Sarge, T.; Shaefi, S.; Leibowitz, A. Case Study: Fatal Exertional Rhabdomyolysis Possibly Related to Drastic Weight Cutting. Int. J. Sport Nutr. Exerc. Metab. 2019, 29, 68–71. [Google Scholar] [CrossRef]
- Kreider, R.B. Essentials of Exercise & Sport Nutrition: Science to Practice; Lulu Press: Morrisville, CA, USA, 2019. [Google Scholar]
- Association, W.M. World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. Int. J. Bioeth. 2004, 15, 124–129. [Google Scholar]
- Murray, B.; Rosenbloom, C. Fundamentals of glycogen metabolism for coaches and athletes. Nutr. Rev. 2018, 76, 243–259. [Google Scholar] [CrossRef] [Green Version]
- Barbosa, T.M.; Carrasco-Marginet, M.; Castizo-Olier, J.; Rodríguez-Zamora, L.; Iglesias, X.; Rodríguez, F.A.; Chaverri, D.; Brotons, D.; Irurtia, A. Bioelectrical impedance vector analysis (BIVA) for measuring the hydration status in young elite synchronized swimmers. PLoS ONE 2017, 12, e0178819. [Google Scholar] [CrossRef] [Green Version]
- Di Vincenzo, O.; Marra, M.; Scalfi, L. Bioelectrical impedance phase angle in sport: A systematic review. J. Int. Soc. Sports Nutr. 2019, 16. [Google Scholar] [CrossRef] [Green Version]
- Cumming, G. Understanding the New Statistics; Routledge Taylor & Francis: New York, NY, USA, 2013. [Google Scholar] [CrossRef]
- Ho, J.; Tumkaya, T.; Aryal, S.; Choi, H.; Claridge-Chang, A. Moving beyond p values: Data analysis with estimation graphics. Nat. Methods 2019, 16, 565–566. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing [Computer Software]. Available online: https://www.R-project.org/ (accessed on 1 March 2020).
- Camarço, N.F.; Sousa Neto, I.V.; Nascimento, D.C.; Almeida, J.A.; Vieira, D.C.L.; Rosa, T.S.; Pereira, G.B.; Prestes, J. Salivary nitrite content, cognition and power in Mixed Martial Arts fighters after rapid weight loss: A case study. J. Clin. Transl. Res. 2016, 2, 63–69. [Google Scholar] [PubMed]
- Matthews, J.J.; Nicholas, C. Extreme Rapid Weight Loss and Rapid Weight Gain Observed in UK Mixed Martial Arts Athletes Preparing for Competition. Int. J. Sport Nutr. Exerc. Metab. 2017, 27, 122–129. [Google Scholar] [CrossRef] [PubMed]
- Coswig, V.S.; Miarka, B.; Pires, D.A.; da Silva, L.M.; Bartel, C.; Del Vecchio, F.B. Weight Regain, but not Weight Loss, Is Related to Competitive Success in Real-Life Mixed Martial Arts Competition. Int. J. Sport Nutr. Exerc. Metab. 2019, 29, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Coswig, V.S.; Fukuda, D.H.; Del Vecchio, F.B. Rapid Weight Loss Elicits Harmful Biochemical and Hormonal Responses in Mixed Martial Arts Athletes. Int. J. Sport Nutr. Exerc. Metab. 2015, 25, 480–486. [Google Scholar] [CrossRef]
- Artioli, G.G.; Iglesias, R.T.; Franchini, E.; Gualano, B.; Kashiwagura, D.B.; Solis, M.Y.; Benatti, F.B.; Fuchs, M.; Lancha Junior, A.H. Rapid weight loss followed by recovery time does not affect judo-related performance. J. Sports Sci. 2010, 28, 21–32. [Google Scholar] [CrossRef]
- Filaire, E.; Maso, F.; Degoutte, F.; Jouanel, P.; Lac, G. Food restriction, performance, psychological state and lipid values in judo athletes. Int. J. Sports Med. 2001, 22, 454–459. [Google Scholar] [CrossRef] [Green Version]
- Rhi, S.; Lee, J.; Lee, Y.; Kim, K. Changes of body composition, physical fitness, and blood variables according to the short-term weight reduction in college ssireum athletes. J. Exerc. Rehabil. 2019, 15, 74–77. [Google Scholar] [CrossRef] [Green Version]
- Knapik, K.; Sieron, K.; Wojtyna, E.; Onik, G.; Romuk, E.; Birkner, E.; Stanek, A.; Kawczyk-Krupka, A.; Plinta, R.; Sieron, A. Precompetitional Weight Reduction Modifies Prooxidative-Antioxidative Status in Judokas. Oxid. Med. Cell. Longev. 2019, 2019, 2164698. [Google Scholar] [CrossRef] [Green Version]
- Finaud, J.; Degoutte, F.; Scislowski, V.; Rouveix, M.; Durand, D.; Filaire, E. Competition and food restriction effects on oxidative stress in judo. Int. J. Sports Med. 2006, 27, 834–841. [Google Scholar] [CrossRef]
- Gounden, V.; Bhatt, H.; Jialal, I. Renal Function Tests. Available online: https://0-www-ncbi-nlm-nih-gov.brum.beds.ac.uk/books/NBK507821/ (accessed on 20 June 2020).
- Edelstein, C.L. Biomarkers of acute kidney injury. Adv. Chronic Kidney Dis. 2008, 15, 222–234. [Google Scholar] [CrossRef] [Green Version]
- Manoeuvrier, G.; Bach-Ngohou, K.; Batard, E.; Masson, D.; Trewick, D. Diagnostic performance of serum blood urea nitrogen to creatinine ratio for distinguishing prerenal from intrinsic acute kidney injury in the emergency department. BMC Nephrol. 2017, 18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Connor, J.; Egan, B. Prevalence, Magnitude and Methods of Rapid Weight Loss Reported by Male Mixed Martial Arts Athletes in Ireland. Sports 2019, 7, 206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karila, T.; Sarkkinen, P.; Marttinen, M.; Seppälä, T.; Mero, A.; Tallroth, K. Rapid Weight Loss Decreases Serum Testosterone. Int. J. Sports Med. 2008, 29, 872–877. [Google Scholar] [CrossRef] [PubMed]
- Yildirim, I. Associations among Dehydration, Testosterone and Stress Hormones in Terms of Body Weight Loss before Competition. Am. J. Med. Sci. 2015, 350, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Degoutte, F.; Jouanel, P.; Bègue, R.J.; Colombier, M.; Lac, G.; Pequignot, J.M.; Filaire, E. Food Restriction, Performance, Biochemical, Psychological, and Endocrine Changes in Judo Athletes. Int. J. Sports Med. 2006, 27, 9–18. [Google Scholar] [CrossRef]
- Joseph-Bravo, P.; Gutiérrez-Mariscal, M.; Jaimes-Hoy, L.; Charli, J.-L. Thyroid Axis and Energy Balance: Focus on Animals and Implications for Humankind. In Handbook of Famine, Starvation, and Nutrient Deprivation; Springer Nature: Cham, Switzerland, 2017; pp. 1–28. [Google Scholar] [CrossRef]
- Slimani, M.; Paravlic, A.H.; Chaabene, H.; Davis, P.; Chamari, K.; Cheour, F. Hormonal responses to striking combat sports competition: A systematic review and meta-analysis. Biol. Sport 2018. [Google Scholar] [CrossRef]
- Vera, F.M.; Manzaneque, J.M.; Carranque, G.A.; Rodríguez-Peña, F.M.; Sánchez-Montes, S.; Blanca, M.J. Endocrine Modulation in Long-Term Karate Practitioners. Evid. Based Complement. Alternat. Med. 2018, 2018, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Morales-Rosillo, D.G.; Petro, J.L.; Bonilla, D.A. Civilizing process, violence, and combat sports; integrative review of boxing and mixed martial arts. Ricyde. Rev. Int. Cienc. Deporte 2019, 15, 413–432. [Google Scholar] [CrossRef]
- Roklicer, R.; Lakicevic, N.; Stajer, V.; Trivic, T.; Bianco, A.; Mani, D.; Milosevic, Z.; Maksimovic, N.; Paoli, A.; Drid, P. The effects of rapid weight loss on skeletal muscle in judo athletes. J. Transl. Med. 2020, 18. [Google Scholar] [CrossRef]
- Pettersson, S.; Berg, C.M. Hydration Status in Elite Wrestlers, Judokas, Boxers, and Taekwondo Athletes on Competition Day. Int. J. Sport Nutr. Exerc. Metab. 2014, 24, 267–275. [Google Scholar] [CrossRef]
- Jetton, A.M.; Lawrence, M.M.; Meucci, M.; Haines, T.L.; Collier, S.R.; Morris, D.M.; Utter, A.C. Dehydration and Acute Weight Gain in Mixed Martial Arts Fighters before Competition. J. Strength Cond. Res. 2013, 27, 1322–1326. [Google Scholar] [CrossRef] [PubMed]
- De Crée, C. Effects of Rapid Reduction of Body Mass (“Weight-Cutting”) on Performance Indices and Proneness to Injury in jūdōka. A Critical Appraisal from a Historical, Gender-Comparative and Coaching Perspective. OAJ Exerc. Sports Med. 2017, 1, 1–33. [Google Scholar]
- Cannataro, R.; Caroleo, M.C.; Fazio, A.; La Torre, C.; Plastina, P.; Gallelli, L.; Lauria, G.; Cione, E. Ketogenic Diet and microRNAs Linked to Antioxidant Biochemical Homeostasis. Antioxidants 2019, 8, 269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zirpoli, H.; Caputo, M.; Carraturo, A.; Torino, G.; Fazio, A.; Attya, M.; Rastrelli, L.; Tecce, M.F. Selective action of human sera differing in fatty acids and cholesterol content on in vitro gene expression. J. Cell Biochem. 2012, 113, 815–823. [Google Scholar] [CrossRef] [PubMed]


| Variable | (SD) (Women n = 8) | 95% CI (min, max) | (SD) (Men n = 13) | 95% CI (min, max) |
|---|---|---|---|---|
| Age (years) | 24.62 (5.42) | 20.09, 29.15 | 26.53 (6.35) | 22.69, 30.38 |
| Stature (m) | 163.00 (3.11) | 160.39, 165.60 | 177.07 (4.80) | 174.17, 179.97 |
| Body mass (kg) | 57.17 (5.96) | 52.19, 62.15 | 74.71 (8.68) | 69.46, 79.96 |
| BMI (kg∙m−2) | 21.54 (2.51) | 19.44, 23.65 | 22.74 (1.89) | 21.60, 23.89 |
| Glucose (mg/dL) | 72.00 (2.44) | 69.95, 74.04 | 81.46 (3.82) | 79.15, 83.77 |
| Total Cholesterol (mg/dL) | 137.87 (22.78) | 118.82, 156.92 | 160.38 (30.21) | 142.12, 178.64 |
| HDL-c (mg/dL) | 53.25 (4.20) | 49.73, 56.76 | 53.30 (8.00) | 48.47, 58.14 |
| LDL-c (mg/dL) | 80.00 (16.13) | 66.51, 93.48 | 98.15 (10.76) | 91.64, 104.66 |
| TG (mg/dL) | 48.50 (8.65) | 41.26, 49.50 | 95.07 (27.26) | 78.60, 111.55 |
| BUN (mg/dL) | 14.62 (3.58) | 11.62, 17.62 | 17.84 (4.18) | 15.32, 20.37 |
| Creatinine (mg/dL) | 0.93 (0.03) | 0.90, 0.95 | 1.01 (0.10) | 0.95, 1.07 |
| Testosterone (pg/mL) | No measure | No measure | 4.57 (0.82) | 4.07, 5.07 |
| TSH (mIU/mL) | 2.18 (0.45) | 1.80, 2.56 | 2.14 (0.36) | 1.92, 2.35 |
| Whole-body phase angle (°) | 7.11 (0.56) | 6.63, 7.58 | 7.91 (0.90) | 7.36, 8.46 |
| Body fat (%) | 17.73 (2.05) | 16.02, 19.45 | 11.46 (1.30) | 10.67, 12.25 |
| Active tissue mass (%) | 56.10 (5.69) | 51.33, 60.86 | 52.85 (2.98) | 51.05, 54.65 |
| Total body water (%) | 58.87 (5.41) | 54.35, 63.39 | 62.31 (2.26) | 60.94, 63.68 |
| Intracellular water (%) | 60.26 (2.31) | 58.32, 62.20 | 61.73 (2.10) | 60.46, 63.00 |
| Extracellular water (%) | 39.73 (2.31) | 37.79, 41.67 | 38.26 (2.10) | 36.99, 39.53 |
| Baseline | RWL | RWG | Comparison of the Means | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Variable | Group | t0 (SD) | t1 (SD) | t2 (SD) | Δ1 (SD) [95% CI] | dunb δ [95% CI] | Δ2 (SD) [95% CI] | dunb δ [95% CI] | Δ3 (SD) [95% CI] | dunb δ [95% CI] |
| BM (kg) | All | 68.0 (11.5) | 65.2 (10.8) | 67.6 (11.6) | −2.7 (0.9) [−3.1, −2.3] * | −0.237 [−0.329, −0.163] | 2.3 (0.9) [1.9, 2.8] * | 0.204 [0.136, 0.286] | −0.3 (0.2) [−0.5, −0.2] * | −0.032 [−0.047, −0.019] |
| Female | 57.1 (5.9) | 55.1 (5.6) | 56.5 (5.8) | −2.0 (0.4) [−2.3, −1.6] * | −0.309 [−0.533, −0.162] | 1.4 (0.4) [1.0, 1.7] * | 0.221 [0.112, 0.384] | −0.5 (0.2) [−0.7, −0.4] * | −0.088 [−0.155, −0.042] | |
| Male | 74.7 (8.6) | 71.4 (8.1) | 74.4 (8.5) | −3.2 (0.8) [−3.7, −2.7] * | −0.358 [−0.541, −0.222] | 2.9 (0.7) [2.5, 3.4] * | 0.354 [0.205, 0.500] | −0.2 (0.1) [−0.3, −0.1] * | −0.028 [−0.047, −0.013] | |
| Glucose (mg/dL−1) | All | 77.8 (5.7) | 80.4 (6.1) | 85.3 (6.3) | 2.6 (0.9) [2.1, 3.0] * | 0.424 [0.289, 0.591] | 4.9 (1.9) [4.0, 5.8] * | 0.753 [0.505, 1.056] | 7.5 (2.2) [6.5, 8.5] * | 1.190 [0.824, 1.645] |
| Female | 72.0 (2.4) | 74.0 (2.9) | 78.2 (3.0) | 2.0 (0.7) [1.3, 2.6] * | 0.658 [0.311, 1.163] | 4.2 (1.2) [3.1, 5.3] * | 1.271 [0.636, 2.217] | 6.2 (1.3) [5.0, 7.4] * | 2.022 [1.058, 3.489] | |
| Male | 81.4 (3.8) | 84.4 (3.5) | 89.7 (2.8) | 3.0 (0.8) [2.5, 3.4] * | 0.764 [0.470, 1.158] | 5.3 (2.2) [3.9, 6.6] * | 1.547 [0.888, 2.400] | 8.3 (2.2) [6.9, 9.6] * | 2.303 [0.672, 3.981] | |
| Total Cholesterol (mg/dL) | All | 151.8 (29.2) | 170.2 (24.5) | 154.2 (27.3) | 18.4 (6.4) [15.5, 21.4] * | 0.659 [0.449, 0.917] | −16.0 (3.8) [−17.7, −14.2] * | −0593 [−0.815, −0.416] | 2.4 (4.0) [0.6, 4.3] * | 0.084 [0.020, 0.153] |
| Female | 137.8 (22.7) | 160.2 (17.1) | 143.7 (19.3) | 22.3 (7.6) [15.9, 28.7] * | 0.986 [0.479, 1.730] | −16.5 (3.3) [−19.3, −13.6] * | −0.802 [−1.381, −0.423] | 5.8 (4.9) [1.7, 10.0] * | 0.247 [0.057, 0.488] | |
| Male | 160.3 (30.2) | 176.4 (26.8) | 160.7 (30.1) | 16.0 (4.3) [13.4, 18.7] * | 0.526 [0.324, 0.798] | −15.6 (4.2) [−18.2, −13.1] * | −0.515 [−0.780, −0.317] | 0.38 (0.65) [−0.008, 0.77] NS | 0.012 [0.000, 0.025] | |
| HDL-c (mg/dL) | All | 53.2 (6.6) | 54.4 (6.5) | 54.1 (6.6) | 1.9 (2.0) [0.27, 2.10] * | 0.173 [0.036, 0.320] | −0.33 (0.9) [−0.77, 0.10] NS | −0.049 [−0.115, 0.015] | 0.85 (2.2) [−0.15, 1.86] NS | 0.124 [−0.021, 0.275] |
| Female | 53.2 (4.2) | 56.1 (3.5) | 56.2 (3.8) | 2.8 (2.3) [0.9, 4.8] * | 0.659 [0.160, 1.296] | 0.12 (0.83) [−0.57, 0.82] NS | 0.030 [−0.125, 0.191] | 3.0 (2.1) [1.2, 4.7] * | 0.662 [0.203, 1.264] | |
| Male | 53.3 (8.0) | 53.4 (7.8) | 52.8 (7.7) | 0.15 (0.68) [−0.26, 0.57] NS | 0.018 [−0.029, 0.067] | −0.61 (0.96) [−1.19, −0.03] * | −0.074 [−0.152, −0.004] | −0.46 (0.7) [−0.93, 0.007] NS | −0.055 [−0.116, 0.001] | |
| LDL-c (mg/dL) | All | 91.2 (15.5) | 109.1 (15.5) | 94.3 (14.5) | 17.9 (5.9) [15.2, 20.6] * | 1.111 [0.762, 1.543] | −14.8 (6.7) [−17.9, −11.7] * | −0.951 [−1.344, −0.625] | 3.09 (2.5) [1.9, 4.2] * | 0.198 [0.108, 0.300] |
| Female | 80.0 (16.1) | 97.1 (16.3) | 86.0 (16.4) | 17.1 (6.1) [12.02, 22.2] * | 0.936 [0.450, 1.647] | −11,1 (5.9) [−16.1, −6.1] * | −0.603 [−1.101, −0.242] | 6.0 (1.3) [4.9, 7.0] * | 0.327 [0.172, 0.565] | |
| Male | 98.1 (10.7) | 116.6 (9.3) | 99.46 (10.9) | 18.4 (5.9) [14.8, 22.0] * | 1.710 [1.031, 2.611] | −17.1 (6.3) [−20.9, −13.3] * | −1.574 [−2.420, −0.931] | 1.3 (0.8) [0.7, 1.8] * | 0.113 [0.055, 0.183] | |
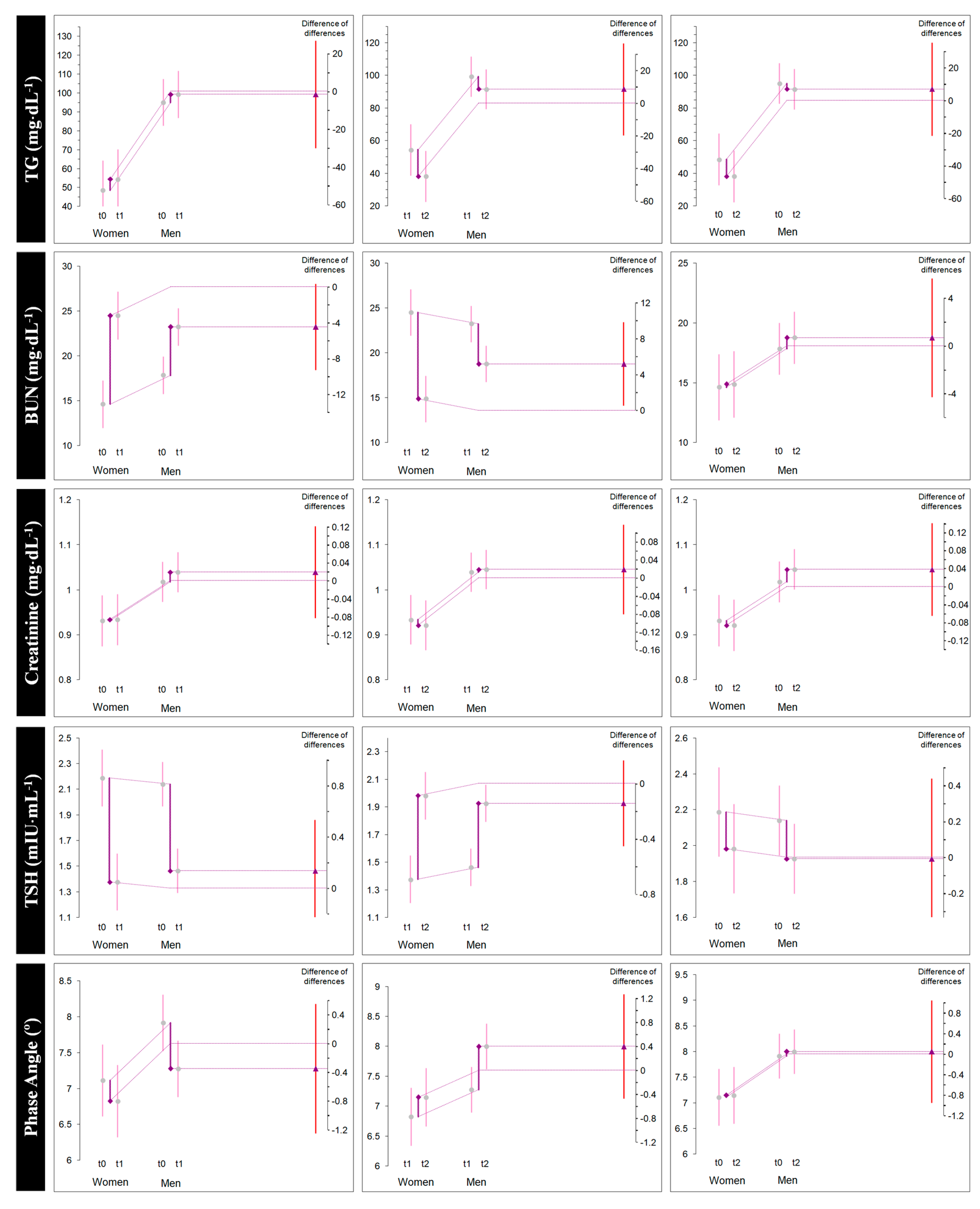
| TG (mg/dL) | All | 77.3 (31.7) | 82.1 (30.9) | 71.1 (34.1) | 4.8 (2.0) [3.9, 5.7] * | 0.149 [0.099, 0.210] | −11.0 (5.3) [−13.4, −8.5] * | −0.325 [−0.462, −0.211] | −6.1 (5.5) [−8.6, −3.6] * | −0.179 [−0.277, −0.093] |
| Female | 48.5 (8.6) | 54.3 (6.5) | 38.1 (6.3) | 5.8 (2.6) [3.6, 8.1] * | 0.679 [0.297, 1.220] | −16.2 (5.0) [−20.4, −12.0] * | −2.237 [−3.908, −1.111] | −10.3 (7.1) [−16.3, −4.3] * | −1.215 [−2.307, −0.387] | |
| Male | 95.0 (27.2) | 99.3 (27.0) | 91.5 (27.1) | 4.2 (1.2) [3.4, 4.9] * | 0.146 [0.089, 0.221] | −7.7 (1.9) [−8.9, −6.5] * | −0.268 [−0.405, −0.166] | −3.5 (1.1) [−4.2, −2.8] * | −0.122 [−0.186, −0.074] | |
| BUN (mg/dL) | All | 16.6 (4.1) | 23.7 (3.4) | 17.28 (4.1) | 7.0 (3.7) [5.3, 8.8] * | 1.781 [1.134, 2.552] | −6.4 (3.8) [−8.1, −4.6] * | −1.626 [−2.356, −1.007] | 0.6 (0.8) [0.2, 1.0] * | 0.154 [0.058, 0.259] |
| Female | 14.6 (3.5) | 24.5 (3.2) | 14.8 (3.4) | 9.8 (2.4) [7.8, 11.9] * | 2.548 [1.313, 4.413] | −9.6 (2.8) [−11.9, −7.2] * | −2.522 [−4.393, −1.268] | 0.2 (0.7) [−0.3. 0.8] NS | 0.063 [−0.075, 0.212] | |
| Male | 17.8 (4.1) | 23.2 (3.5) | 18.7 (3.8) | 5.3 (3.4) [3.2, 7.4] * | 1.298 [0.643, 2.105] | −4.4 (2.9) [−6.2, −2.6] * | −1.118 [−1.821, −0.547] | 0.9 (0.8) [0.4, 1.4] * | 0.214 [0.078, 0.372] | |
| Creatinine (mg/dL) | All | 0.98 (0.09) | 0.99 (0.09) | 0.99 (0.09) | 0.01 (0.01) [0.007, 0.020] * | 0.151 [0.071, 0.239] | −0.01 (0.01) [−0.005, 0.004] NS | −0.010 [−0.059, 0.039] | 0.01 (0.02) [0.003, 0.020] * | 0.136 [0.028, 0.251] |
| Female | 0.93 (0.03) | 0.93 (0.03) | 0.92 (0.03) | 0.002 (0.004) [−0.001, 0.006] NS | 0.070 [−0.032, 0.185] | −0.01 (0.004) [−0.016, −0.008] * | −0.353 [−0.622, −0.168] | −0.01 (0.005) [−0.01, −0.005] * | −0.281 [−0.513, −0.113] | |
| Male | 1.01 (0.10) | 1.03 (0.09) | 1.04 (0.09) | 0.02 (0.01) [0.013, 0.030] * | 0.208 [0.102, 0.338] | 0.006 (0.006) [0.002, 0.010] * | 0.061 [0.019, 0.110] | 0.02 (0.01) [0.018, 0.037] * | 0.265 [0.139, 0.424] | |
| Testosterone (pg/mL) | Male | 4.57 (0.82) | 3.50 (0.77) | 1.92 (0.27) | −1.06 (0.2) [−1.1, −0.9] * | −1.245 [−1.871, −0.787] | −1.5 (0.9) [−2.1, −0.9] * | −2.545 [−4.114, −1.277] | −2.6 (1.0) [−3.2, −2.0] * | −4.018 [−6.200, −2.346] |
| TSH (mIU/mL) | All | 2.15 (0.38) | 1.43 (0.18) | 1.94 (0.28) | −0.72 (0.2) [−0.8, −0.6] * | −2.304 [−3.208, −1.570] | 0.51 (0.2) [0.4, 0.6] * | 2.093 [1.385, 2.951] | −0.21 (0.2) [−0.3, −0.1] * | −0.595 [−0.970, −0.255] |
| Female | 2.18 (0.45) | 1.37 (0.22) | 1.98 (0.31) | −0.81 (0.2) [−1.0, −0.5] * | −2.022 [−3.554, −0.976] | 0.6 (0.3) [0.3, 0.8] * | 1.974 [0.819, 3.582] | −0.20 (0.3) [−0.5, 0.1] NS | −0.468 [−1.236, 0.214] | |
| Male | 2.14 (0.36) | 1.46 (0.15) | 1.92 (0.27) | −0.67 (0.2) [−0.8, −0.5] * | −2.271 [−3.477, −1.358] | 0.46 (0.1) [0.3, 0.5] * | 1.964 [1.187, 2.996] | −0.21 (0.1) [−0.2, −0.1] * | −0.622 [−0.985, −0.335] | |
| Whole body phase angle (°) | All | 7.60 (0.87) | 7.10 (0.61) | 7.67 (0.84) | −0.5 (0.3) [−0.6, −0.3] * | −0.642 [−0.962, −0.363] | 0.5 (0.3) [0.4, 0.7] * | 0.739 [0.464, 1.066] | 0.06 (0.27) [−0.05, 0.19] NS | 0.074 [−0.061, 0.214] |
| Female | 7.11 (0.56) | 6.82 (0.48) | 7.15 (0.54) | −0.2 (0.1) [−0.4, −0.1] * | −0.482 [−0.875, −0.200] | 0.3 (0.08) [0.2, 0.3] * | 0.559 [0.285, 0.971] | 0.03 (0.14) [−0.08, 0.15] NS | 0.060 [−0.113, 0.243] | |
| Male | 7.91 (0.90) | 7.27 (0.64) | 8.00 (0.85) | −0.6 (0.4) [−0.8, −0.3] * | −0.761 [−1.245, −0.367] | 0.7 (0.3) [0.5, 0.9] * | 0.893 [0.506, 1.392] | 0.08 (0.33) [−0.11, 0.28] NS | 0.090 [−0.116, 0.304] | |
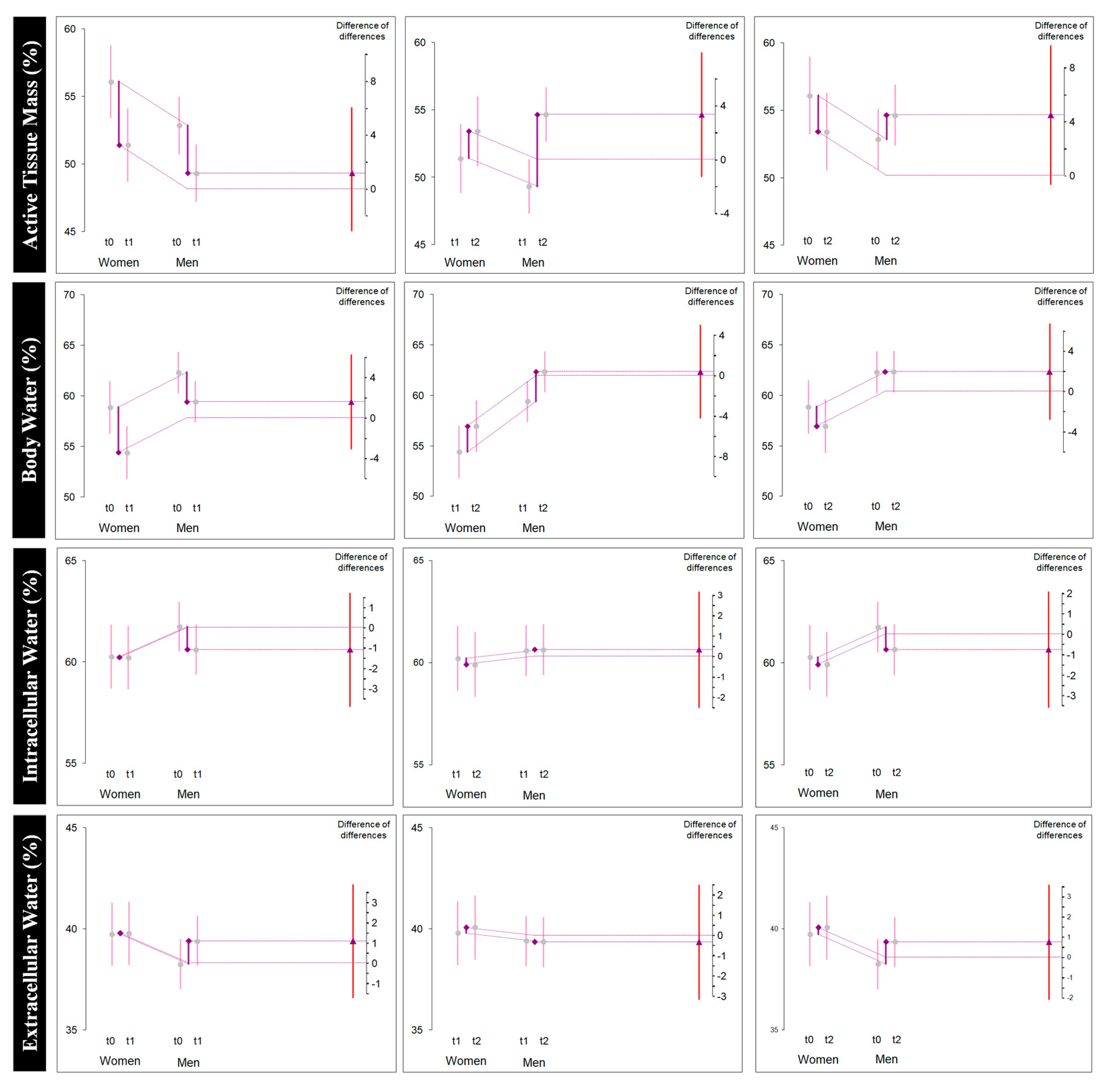
| Active tissue mass (%) | All | 54.0 (4.3) | 50.1 (3.4) | 54.1 (3.7) | −3.9 (2.1) [−4.9, −2.9] * | −0.969 [−1.390, −0.616] | 4.0 (2.4) [2.9, 5.1] * | 1.085 [0.667, 1.577] | 0.1 (2.3) [−0.9, 1.1] NS | 0.023 [−0.216, 0.265] |
| Female | 56.1 (5.6) | 51.4 (3.6) | 53.4 (5.3) | −4.6 (3.1) [−7.2, −2.0] * | −0.870 [−1.640, −0.290] | 2.0 (2.7) [−0.2, 4.3] NS | 0.392 [−0.045, 0.905] | −2.6 (0.6) [−3.2, −2.0] * | −0.432 [−0.749, −0.222] | |
| Male | 52.8 (2.9) | 49.3 (3.1) | 54.6 (2.6) | −3.5 (1.1) [−4.2, −2.8] * | −1.076 [−1.644, −0.647] | 5.3 (1.1) [4.6, 6.0] * | 1.728 [1.083, 2.603] | 1.8 (0.5) [1.4, 2.1] * | 0.604 [0.639, 0.917] | |
| Body water (%) | All | 61.0 (4.0) | 57.5 (4.2) | 60.3 (4.4) | −3.4 (1.2) [−4.0, −2.9] * | −0.810 [−1.128, −0.551] | 2.8 (1.0) [2.3, 3.2] * | 0.616 [0.416, 0.861] | −0.7 (1.0) [−1.2, −0.2] * | −0.157 [−0.281, −0.042] |
| Female | 58.8 (5.4) | 54.4 (5.2) | 56.9 (5.3) | −4.4 (0.8) [−5.2, −3.7] * | −0.749 [−1.288, −0.397] | 2.5 (1.0) [1.6, 3.4] * | 0.435 [0.198, 0.774] | −1.9 (0.7) [−2.4, −1.3] * | −0.315 [−0.555, −0.150] | |
| Male | 62.3 (2.2) | 59.4 (2.0) | 62.3 (2.2) | −2.8 (1.0) [−3.5, −2.2] * | −1.256 [−1.928, −0.746] | 2.9 (1.0) [2.3, 3.5] * | 1.291 [0.766, 1.982] | 0.04 (0.29) [−0.1, 0.2] NS | 0.019 [−0.052, 0.093] | |
| Intracellular water (%) | All | 61.1 (2.2) | 60.4 (2.1) | 60.3 (2.2) | −0.72 (0.6) [−1.0, −0.4] * | −0.317 [−0.487, −0.167] | −0.08 (0.4) [−0.2, 0.1] NS | −0.036 [−0.126, 0.053] | −0.8 (0.4) [−1.01, −0.5] * | −0.347 [−0.502, −0.217] |
| Female | 60.2 (2.3) | 60.2 (2.3) | 59.9 (2.4) | −0.05 (0.05) [−0.09, −0.005] * | −0.019 [−0.040, −0.002] | −0.28 (0.1) [−0.3, −0.1] * | −0.107 [−0.192, −0.048] | −0.3 (0.1) [−0.4, −0.2] * | −0.127 [−0.227, −0.056] | |
| Male | 61.7 (2.1) | 60.6 (2.0) | 60.6 (2.1) | −1.13 (0.4) [−1.39, −0.88] * | −0.509 [−0.783, −0.300] | 0.046 (0.5) [−0.2, 0.3] NS | 0.021 [−0.117, 0.160] | −1.0 (0.3) [−1.2, −0.8] * | −0.484 [−0.737, −0.294] | |
| Extracellular water (%) | All | 38.8 (2.2) | 39.5 (2.1) | 39.6 (2.2) | 0.7 (0.6) [0.4, 1.0] * | 0.317 [0.167, 0.487] | 0.08 (0.45) [−0.1, 0.2] NS | 0.036 [−0.053, 0.126] | 0.80 (0.46) [0.5, 1.0] * | 0.347 [0.217, 0.502] |
| Female | 39.7 (2.3) | 39.7 (2.3) | 40.0 (2.4) | 0.05 (0.05) [0.005, 0.09] * | 0.019 [0.002, 0.040] | 0.28 (0.12) [0.1, 0.3] * | 0.107 [0.048, 0.192] | 0.33 (0.15) [0.2, 0.4] * | 0.127 [0.056, 0.227] | |
| Male | 38.2 (2.1) | 39.4 (2.0) | 39.3 (2.1) | 1.1 (0.4) [0.8, 1.3] * | 0.509 [0.300, 0.783] | −0.04 (0.5) [−0.3, 0.2] NS | −0.021 [−0.160, 0.117] | 1.09 (0.33) [0.8, 1.2] * | 0.484 [0.294, 0.737] | |
| Diff-In-Diff from t0 to t1 (Δ1) | Diff-In-Diff from t1 to t2 (Δ2) | Diff-In-Diff from t0 to t2 (Δ3) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Variable | Mean (Male Δ1—Female Δ1 | DID | 95% CI | p | Mean (Male Δ2—Female Δ2 | DID | 95% CI | p | Mean (Male Δ3—Female Δ3 | DID | 95% CI | p |
| BM (kg) | −3.22–−2.01 | −1.21 | −10.94, 8.52 | 0.803 | 2.96–1.42 | 1.53 | −8.12, 11.2 | 0.749 | −0.26–−0.58 | 0.32 | −9.63, 10.28 | 0.948 |
| Glucose (mg/dL) | 3.00–2.00 | 1.00 | −3.31, 5.30 | 0.641 | 5.30–4.25 | 1.05 | −2.96, 5.07 | 0.597 | 8.30–6.25 | 2.05 | −2.00, 6.12 | 0.312 |
| Total Cholesterol (mg/dL) | 16.07–22.37 | −6.29 | −39.50, 26.90 | 0.703 | −15.69–−16.50 | 0.80 | −31.68, 33.29 | 0.960 | 0.38–5.87 | −5.49 | −40.47, 29.49 | 0.752 |
| HDL-c (mg/dL) | 0.15–2.87 | −2.72 | −11.36, 5.91 | 0.527 | −0.61–−0.12 | −0.74 | −9.19, 7.71 | 0.860 | −0.46–3.00 | −3.46 | −12.10, 5.17 | 0.422 |
| LDL-c (mg/dL) | 18.46–17.125 | 1.33 | −15.03, 17.70 | 0.870 | −17.15–−11.13 | −6.02 | −22.54, 10.48 | 0.464 | 1.30–6.00 | −4.69 | −21.57, 12.18 | 0.577 |
| TG (mg/dL) | 4.23–5.87 | −1.64 | −30.07, 26.78 | 0.907 | −7.76–−16.25 | 8.48 | −19.72, 36.68 | 0.546 | −3.53–−10.38 | 6.83 | −21.62, 35.29 | 0.630 |
| BUN (mg/dL) | 5.38–9.87 | −4.49 | −9.28, 0.30 | 0.066 | −4.46–−9.62 | 5.16 * | 0.51, 9.80 | 0.03 | 0.92–0.25 | 0.67 | −4.29, 5.64 | 0.785 |
| Creatinine (mg/dL) | 0.02–0.0025 | 0.019 | −0.08, 0.12 | 0.709 | 0.006–−0.013 | 0.018 | −0.08, 0.11 | 0.706 | 0.027–−0.01 | 0.037 | −0.06, 0.14 | 0.463 |
| TSH (mIU/mL) | −0.67–−0.81 | −0.13 | −0.26, 0.53 | 0.496 | 0.46–0.60 | −0.143 | −0.45, 0.16 | 0.356 | −0.213–−0.205 | −0.008 | −0.45, 0.43 | 0.971 |
| Whole body phase angle (°) | −0.63–−0.28 | −0.35 | −1.25, 0.55 | 0.436 | 0.72–0.32 | −0.398 | −0.47, 1.27 | 0.362 | 0.084–0.037 | 0.047 | −0.95, 1.04 | 0.924 |
| Active tissue mass (%) | −3.52–−4.68 | 1.16 | −3.71, 6.04 | 0.632 | 5.33–2.01 | 3.318 | −1.30, 7.94 | 0.154 | 1.80–2.67 | 4.482 | −0.68, 9.64 | 0.087 |
| Body water (%) | −2.89–−4.47 | 1.58 | −3.11, 6.27 | 0.499 | 2.93–2.57 | 0.36 | −4.28, 5.02 | 0.875 | 0.046–−1.900 | 1.946 | −2.85, 6.71 | 0.414 |
| Intracellular water (%) | −1.13–−0.05 | −1.08 | −3.89, 1.72 | 0.438 | 0.046–−0.28 | 0.333 | −2.51, 3.17 | 0.813 | −1.092–−0.337 | −0.755 | −3.59, 2.08 | 0.594 |
| Extracellular water (%) | 1.13–0.05 | 1.08 | −1.72, 3.89 | 0.438 | −0.046–0.28 | −0.333 | −3.17, 2.51 | 0.813 | 1.092–0.337 | 0.755 | −2.08, 3.59 | 0.594 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cannataro, R.; Cione, E.; Gallelli, L.; Marzullo, N.; Bonilla, D.A. Acute Effects of Supervised Making Weight on Health Markers, Hormones and Body Composition in Muay Thai Fighters. Sports 2020, 8, 137. https://0-doi-org.brum.beds.ac.uk/10.3390/sports8100137
Cannataro R, Cione E, Gallelli L, Marzullo N, Bonilla DA. Acute Effects of Supervised Making Weight on Health Markers, Hormones and Body Composition in Muay Thai Fighters. Sports. 2020; 8(10):137. https://0-doi-org.brum.beds.ac.uk/10.3390/sports8100137
Chicago/Turabian StyleCannataro, Roberto, Erika Cione, Luca Gallelli, Natale Marzullo, and Diego A. Bonilla. 2020. "Acute Effects of Supervised Making Weight on Health Markers, Hormones and Body Composition in Muay Thai Fighters" Sports 8, no. 10: 137. https://0-doi-org.brum.beds.ac.uk/10.3390/sports8100137








