Metagenome Techniques for Detection of Pathogens Causing Ocular Infection
Abstract
:1. Introduction
2. Results
2.1. Clinical Diagnosis
Reports of Representative Cases
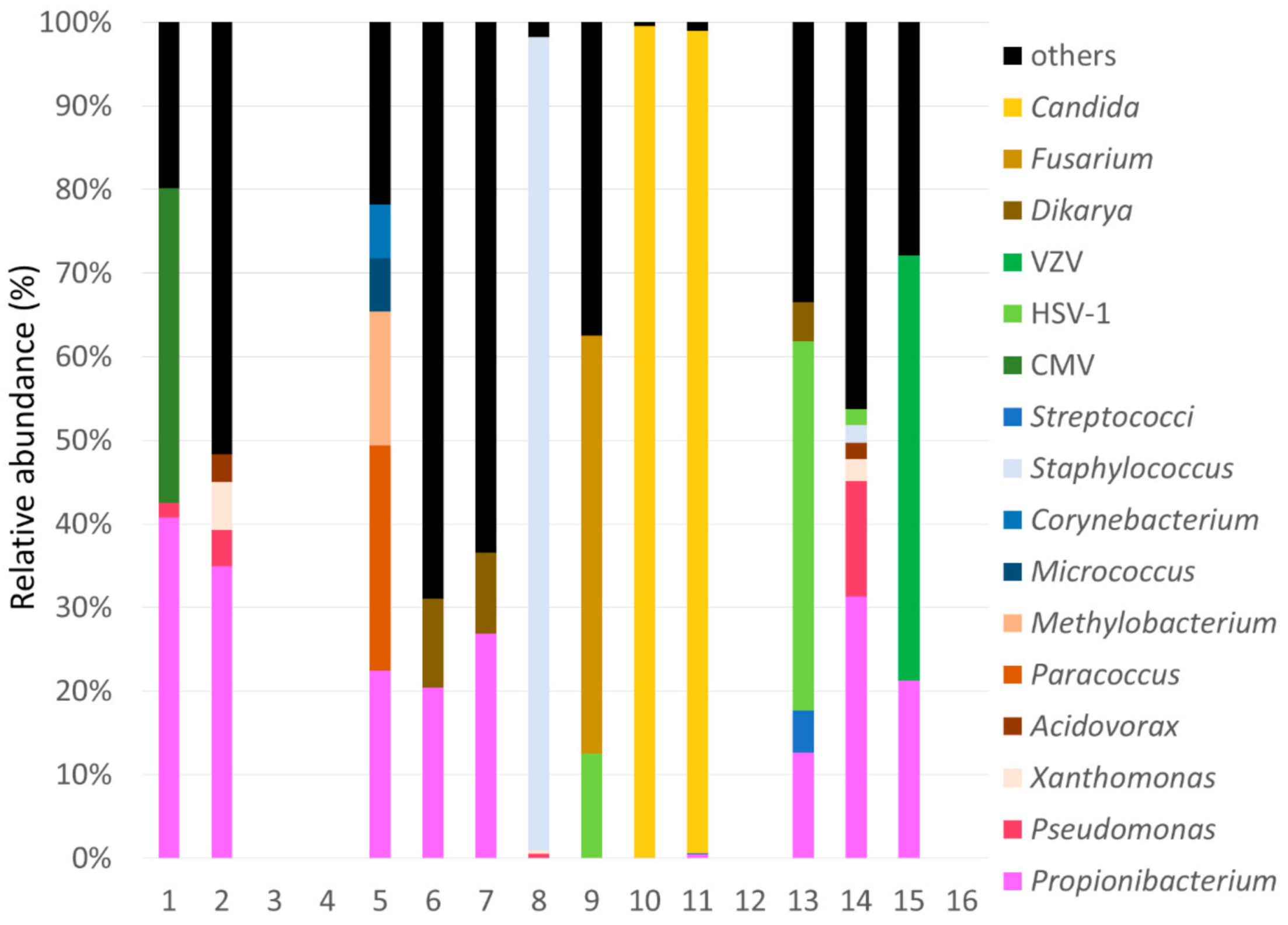
2.2. Summary of SMA
2.3. Comparison between SMA and Conventional Examination
3. Discussion
4. Materials and Methods
4.1. Microbiological Examination
4.2. PCR
4.3. Shotgun Metagenomic Sequencing Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PCR | Polymerase chain reaction |
| HSV | Herpes simplex virus |
| VZV | Varicella-zoster virus |
| CMV | Cytomegalovirus |
| NGS | Next-generation sequencing |
| SMA | Shotgun metagenomic analysis |
| DSAEK | Descemet’s stripping automated endothelial keratoplasty |
| TSPK | Thygeson’s superficial punctate keratitis |
| AH | Aqueous humor |
| CS | Corneal scraping |
| BRiSK | Biome representational in silico karyotyping |
References
- Kratz, A.; Levy, J.; Klemperer, I.; Lifshitz, T. Broth cultures yield vs traditional approach in the workup of infectious keratitis. Eye 2006, 20, 215–220. [Google Scholar] [CrossRef] [Green Version]
- Pakzad-Vaezi, K.; Levasseur, S.D.; Schendel, S.; Mark, S.; Mathias, R.; Roscoe, D.; Holland, S.P. The corneal ulcer one-touch study: A simplified microbiological specimen collection method. Am. J. Ophthalmol. 2015, 159, 37–43. [Google Scholar] [CrossRef]
- Inoue, T.; Ohashi, Y. Utility of real-time PCR analysis for appropriate diagnosis for keratitis. Cornea 2013, 32 (Suppl 1), S71–S76. [Google Scholar] [CrossRef]
- Knox, C.M.; Cevellos, V.; Dean, D. 16S ribosomal DNA typing for identification of pathogens in patients with bacterial keratitis. J. Clin. Microbiol. 1998, 36, 3492–3496. [Google Scholar] [CrossRef] [Green Version]
- Nakano, S.; Sugita, S.; Tomaru, Y.; Hono, A.; Nakamuro, T.; Kubota, T.; Takase, H.; Mochizuki, M.; Takahashi, M.; Shimizu, N. Establishment of Multiplex Solid-Phase Strip PCR Test for Detection of 24 Ocular Infectious Disease Pathogens. Invest. Ophthalmol. Vis. Sci. 2017, 58, 1553–1559. [Google Scholar] [CrossRef]
- Allcock, R.J.N.; Jennison, A.V.; Warrilow, D. Towards a Universal Molecular Microbiological Test. J. Clin. Microbiol. 2017, 55, 3175–3182. [Google Scholar] [CrossRef] [Green Version]
- Street, T.L.; Sanderson, N.D.; Atkins, B.L.; Brent, A.J.; Cole, K.; Foster, D.; McNally, M.A.; Oakley, S.; Peto, L.; Taylor, A.; et al. Molecular Diagnosis of Orthopedic-Device-Related Infection Directly from Sonication Fluid by Metagenomic Sequencing. J. Clin. Microbiol. 2017, 55, 2334–2347. [Google Scholar] [CrossRef] [Green Version]
- Wilson, M.R.; Suan, D.; Duggins, A.; Schubert, R.D.; Khan, L.M.; Sample, H.A.; Zorn, K.C.; Rodrigues Hoffman, A.; Blick, A.; Shingde, M.; et al. A novel cause of chronic viral meningoencephalitis: Cache Valley virus. Ann. Neurol. 2017, 82, 105–114. [Google Scholar] [CrossRef]
- Doan, T.; Pinsky, B.A. Current and future molecular diagnostics for ocular infectious diseases. Curr. Opin. Ophthalmol. 2016, 27, 561–567. [Google Scholar] [CrossRef]
- Doan, T.; Acharya, N.R.; Pinsky, B.A.; Sahoo, M.K.; Chow, E.D.; Banaei, N.; Budvytiene, I.; Cevallos, V.; Zhong, L.; Zhou, Z.; et al. Metagenomic DNA Sequencing for the Diagnosis of Intraocular Infections. Ophthalmology 2017, 124, 1247–1248. [Google Scholar] [CrossRef] [Green Version]
- Doan, T.; Wilson, M.R.; Crawford, E.D.; Chow, E.D.; Khan, L.M.; Knopp, K.A.; O’Donovan, B.D.; Xia, D.; Hacker, J.K.; Stewart, J.M.; et al. Illuminating uveitis: Metagenomic deep sequencing identifies common and rare pathogens. Genome Med. 2016, 8, 90. [Google Scholar] [CrossRef] [Green Version]
- Lee, A.Y.; Akileswaran, L.; Tibbetts, M.D.; Garg, S.J.; van Gelder, R.N. Identification of torque teno virus in culture-negative endophthalmitis by representational deep DNA sequencing. Ophthalmology 2015, 122, 524–530. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suzuki, T.; Ohashi, Y. Corneal endotheliitis. Semin Ophthalmol. 2008, 23, 235–240. [Google Scholar] [CrossRef]
- Gonzales, J.A.; Hinterwirth, A.; Shantha, J.; Wang, K.; Zhong, L.; Cummings, S.L.; Qian, Y.; Wilson, M.R.; Acharya, N.R.; Doan, T. Association of Ocular Inflammation and Rubella Virus Persistence. JAMA Ophthalmol. 2019, 137, 435–438. [Google Scholar] [CrossRef]
- De Paula Freitas, B.; Ventura, C.V.; Maia, M.; Belfort, R., Jr. Zika virus and the eye. Curr. Opin. Ophthalmol. 2017, 28, 595–599. [Google Scholar] [CrossRef] [PubMed]
- Drancourt, M.; Berger, P.; Terrada, C.; Bodaghi, B.; Conrath, J.; Raoult, D.; LeHoang, P. High prevalence of fastidious bacteria in 1520 cases of uveitis of unknown etiology. Medicine 2008, 87, 167–176. [Google Scholar] [CrossRef]
- Kanis, M.J.; Oosterheert, J.J.; Lin, S.; Boel, C.H.; Ekkelenkamp, M.B. Corneal graft rejection complicated by Paracoccus yeei infection in a patient who had undergone a penetrating keratoplasty. J. Clin. Microbiol. 2010, 48, 323–325. [Google Scholar] [CrossRef] [Green Version]
- Goto, H.; Usui, Y.; Umazume, A.; Uchida, K.; Eishi, Y. Propionibacterium acnes as a possible pathogen of granuloma in patients with ocular sarcoidosis. Br. J. Ophthalmol. 2017, 101, 1510–1513. [Google Scholar] [CrossRef]
- Nagata, K.; Eishi, Y.; Uchida, K.; Yoneda, K.; Hatanaka, H.; Yasuhara, T.; Nagata, M.; Sotozono, C.; Kinoshita, S. Immunohistochemical Detection of Propionibacterium acnes in the Retinal Granulomas in Patients with Ocular Sarcoidosis. Sci. Rep. 2017, 7, 15226. [Google Scholar] [CrossRef] [Green Version]
- Yasuhara, T.; Tada, R.; Nakano, Y.; Tei, M.; Mochida, C.; Kamei, M.; Kinoshita, S. The presence of Propionibacterium spp. in the vitreous fluid of uveitis patients with sarcoidosis. Acta Ophthalmol. Scand. 2005, 83, 364–369. [Google Scholar] [CrossRef]
- Li, Z.; Breitwieser, F.P.; Lu, J.; Jun, A.S.; Asnaghi, L.; Salzberg, S.L.; Eberhart, C.G. Identifying Corneal Infections in Formalin-Fixed Specimens Using Next Generation Sequencing. Invest Ophthalmol. Vis. Sci. 2018, 59, 280–288. [Google Scholar] [CrossRef] [PubMed]
- Boisjoly, H.M.; Pavan-Langston, D.; Kenyon, K.R.; Baker, A.S. Superinfections in herpes simplex keratitis. Am. J. Ophthalmol. 1983, 96, 354–361. [Google Scholar] [CrossRef]
- Thygeson, P. Superficial punctate keratitis. J. Am. Med. Assoc. 1950, 144, 1544–1549. [Google Scholar] [CrossRef] [PubMed]
- Thygeson, P. Clinical and laboratory observations on superficial punctate keratitis. Am. J. Ophthalmol. 1966, 61, 1344–1349. [Google Scholar] [CrossRef]
- Connell, P.P.; O’Reilly, J.; Coughlan, S.; Collum, L.M.; Power, W.J. The role of common viral ocular pathogens in Thygeson’s superficial punctate keratitis. Br. J. Ophthalmol. 2007, 91, 1038–1041. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reinhard, T.; Roggendorf, M.; Fengler, I.; Sundmacher, R. PCR for varicella zoster virus genome negative in corneal epithelial cells of patients with Thygeson’s superficial punctate keratitis. Eye 2004, 18, 304–305. [Google Scholar] [CrossRef]
- Fukui, Y.; Aoki, K.; Okuma, S.; Sato, T.; Ishii, Y.; Tateda, K. Metagenomic analysis for detecting pathogens in culture-negative infective endocarditis. J. Infect. Chemother. 2015, 21, 882–884. [Google Scholar] [CrossRef]
- Li, H.; Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 2009, 25, 1754–1760. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Handsaker, B.; Wysoker, A.; Fennell, T.; Ruan, J.; Homer, N.; Marth, G.; Abecasis, G.; Durbin, R. Genome Project Data Processing, S. The Sequence Alignment/Map format and SAMtools. Bioinformatics 2009, 25, 2078–2079. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huson, D.H.; Mitra, S.; Ruscheweyh, H.J.; Weber, N.; Schuster, S.C. Integrative analysis of environmental sequences using MEGAN4. Genome Res. 2011, 21, 1552–1560. [Google Scholar] [CrossRef] [Green Version]
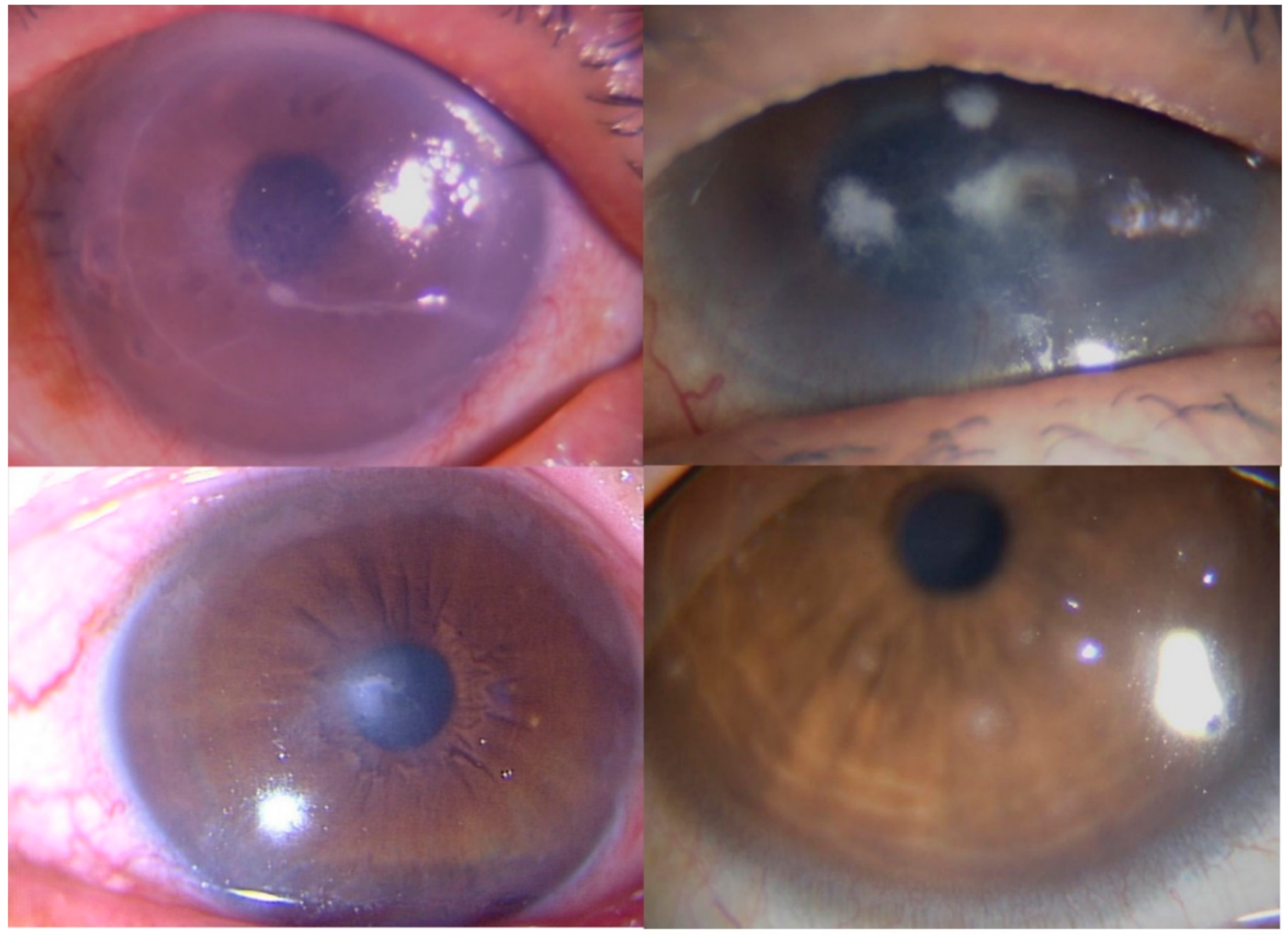

| Case | Age | Gender | Conventional Examination | Clinical Diagnosis |
|---|---|---|---|---|
| 1 | 78 | M | Positive for CMV-DNA | CMV endotheliitis |
| 2 | 67 | M | Positive for CMV-DNA | CMV endotheliitis |
| 3 | 14 | M | Negative | Acute anterior uveitis |
| 4 | 73 | M | Negative | Iridocyclitis |
| 5 | 61 | F | Negative | Endophthalmitis |
| 6 | 23 | F | Negative | Acanthamoeba keratitis |
| 7 | 36 | M | Culture of Propionibacterium spp. | Acanthamoeba keratitis |
| 8 | 47 | F | Culture of S. haemolyticus | Bacterial keratitis |
| 9 | 45 | F | Culture of Fusarium spp. | Fungal keratitis |
| 10 | 91 | F | Culture of C. parapsilosis | Fungal keratitis |
| 11 | 80 | F | Culture of C. albicans | Fungal keratitis |
| 12 | 30 | F | Culture of Propionibacterium spp. | Fungal keratitis |
| 13 | 75 | F | Negative | HSV keratitis |
| 14 | 85 | M | Negative | HSV keratitis |
| 15 | 21 | M | Negative | VZV keratitis |
| 16 | 37 | M | Negative | Thygeson’s keratitis |
| Case | Sample for Metagenome | Reads of Original Genome | After Genome Subtracted | Causative Pathogen in SMA | |
|---|---|---|---|---|---|
| Reads | (%) | ||||
| 1 | AH | 1,656,432 | 27,298 | 1.6 | P: CMV |
| 2 | AH | 1,453,184 | 212,481 | 14.6 | Not Pathogen |
| 3 | AH | 3,900,195 | 7082 | 0.18 | Not detected |
| 4 | AH | 3,012,251 | 4733 | 0.16 | Not detected |
| 5 | VH | 2,071,858 | 6686 | 0.32 | P: Paracoccus spp. |
| 6 | CS | 2,015,171 | 16,480 | 0.8 | Not Pathogen |
| 7 | CS | 1,899,108 | 17,814 | 0.9 | Not Pathogen |
| 8 | CS | 2,258,598 | 18,911 | 0.84 | D: Staphylococcus spp. |
| 9 | CS | 993,734 | 7977 | 0.8 | D: Fusarium spp.P: HSV-1 |
| 10 | CS | 2,309,392 | 13,807 | 0.6 | D: Candida spp. |
| 11 | CS | 2,230,667 | 1,288,610 | 57.77 | D: Candida spp. |
| 12 | CS | 2,245,163 | 3493 | 0.16 | Not detected |
| 13 | CS | 1,974,298 | 16,033 | 0.8 | P: HSV-1 |
| 14 | CS | 1,362,016 | 31,877 | 2.3 | P: HSV-1 |
| 15 | CS | 2,510,419 | 21,955 | 0.9 | D: VZV |
| 16 | CS | 1,605,632 | 8146 | 0.51 | Not detected |
| Conventional Methods | ||||
|---|---|---|---|---|
| Positive | Negative | Total | ||
| SMA | Positive | 5 * | 4 | 9 |
| Negative | 1 | 6 | 7 | |
| Total | 6 | 10 | 16 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kobayashi, T.; Suzuki, T.; Okajima, Y.; Aoki, K.; Ishii, Y.; Tateda, K.; Hori, Y. Metagenome Techniques for Detection of Pathogens Causing Ocular Infection. Reports 2021, 4, 6. https://0-doi-org.brum.beds.ac.uk/10.3390/reports4010006
Kobayashi T, Suzuki T, Okajima Y, Aoki K, Ishii Y, Tateda K, Hori Y. Metagenome Techniques for Detection of Pathogens Causing Ocular Infection. Reports. 2021; 4(1):6. https://0-doi-org.brum.beds.ac.uk/10.3390/reports4010006
Chicago/Turabian StyleKobayashi, Tatsuhiko, Takashi Suzuki, Yukinobu Okajima, Kotaro Aoki, Yoshikazu Ishii, Kazuhiro Tateda, and Yuichi Hori. 2021. "Metagenome Techniques for Detection of Pathogens Causing Ocular Infection" Reports 4, no. 1: 6. https://0-doi-org.brum.beds.ac.uk/10.3390/reports4010006






