Two Different Approaches for Oral Administration of Voriconazole Loaded Formulations: Electrospun Fibers versus β-Cyclodextrin Complexes
Abstract
:1. Introduction
2. Results and Discussion
2.1. Morphology of Poly(ε-Caprolactone) (PCL) Fibrous Matrices Loaded and Unloaded with Voriconazole (VRCZ)
2.2. Morphology, Phase Solubility Studies, and Ultraviolet-Visible (UV-VIS) Spectroscopy of CD Complexes Containing VRCZ
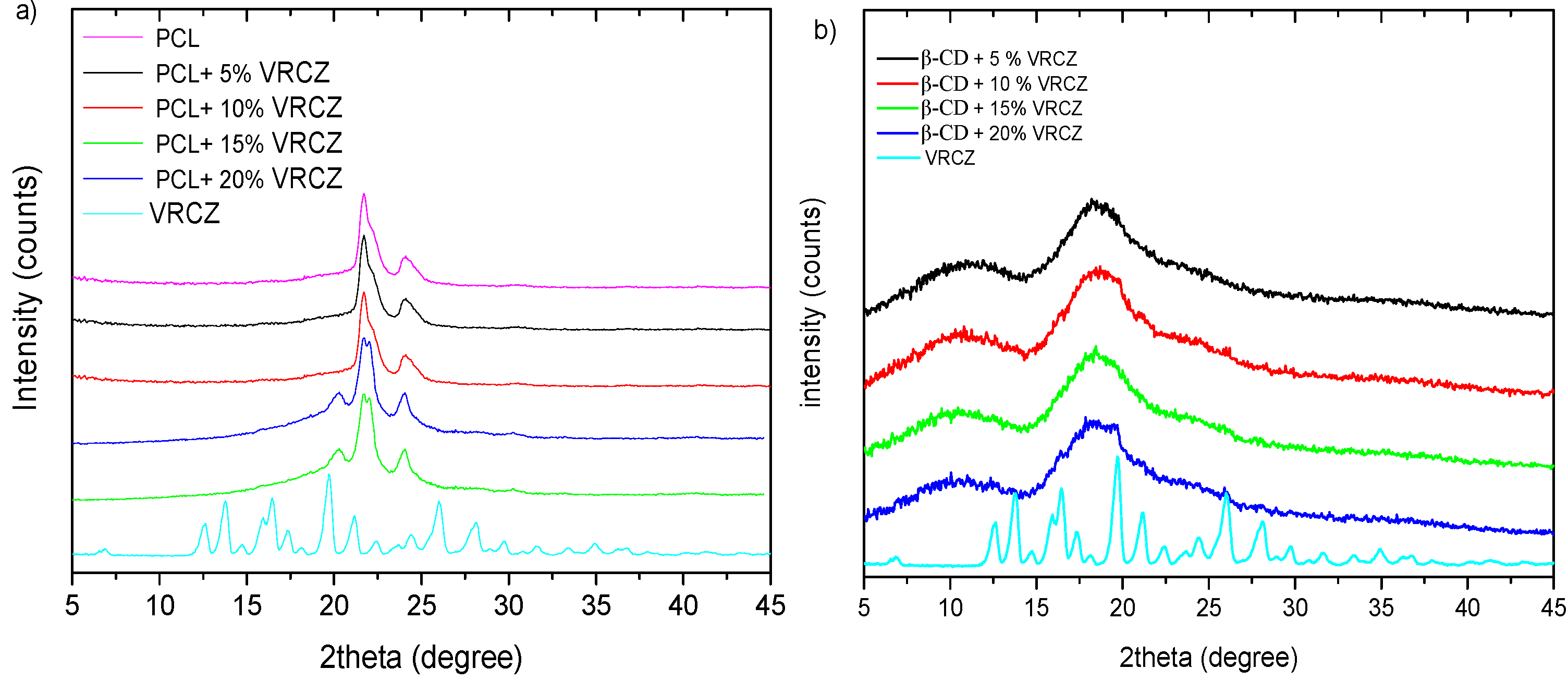
2.3. Characterization of the VRCZ Formulations Using X-ray Diffraction (XRD) Studies
2.4. Fourier-Transform Infrared (FT-IR) Spectroscopy Studies
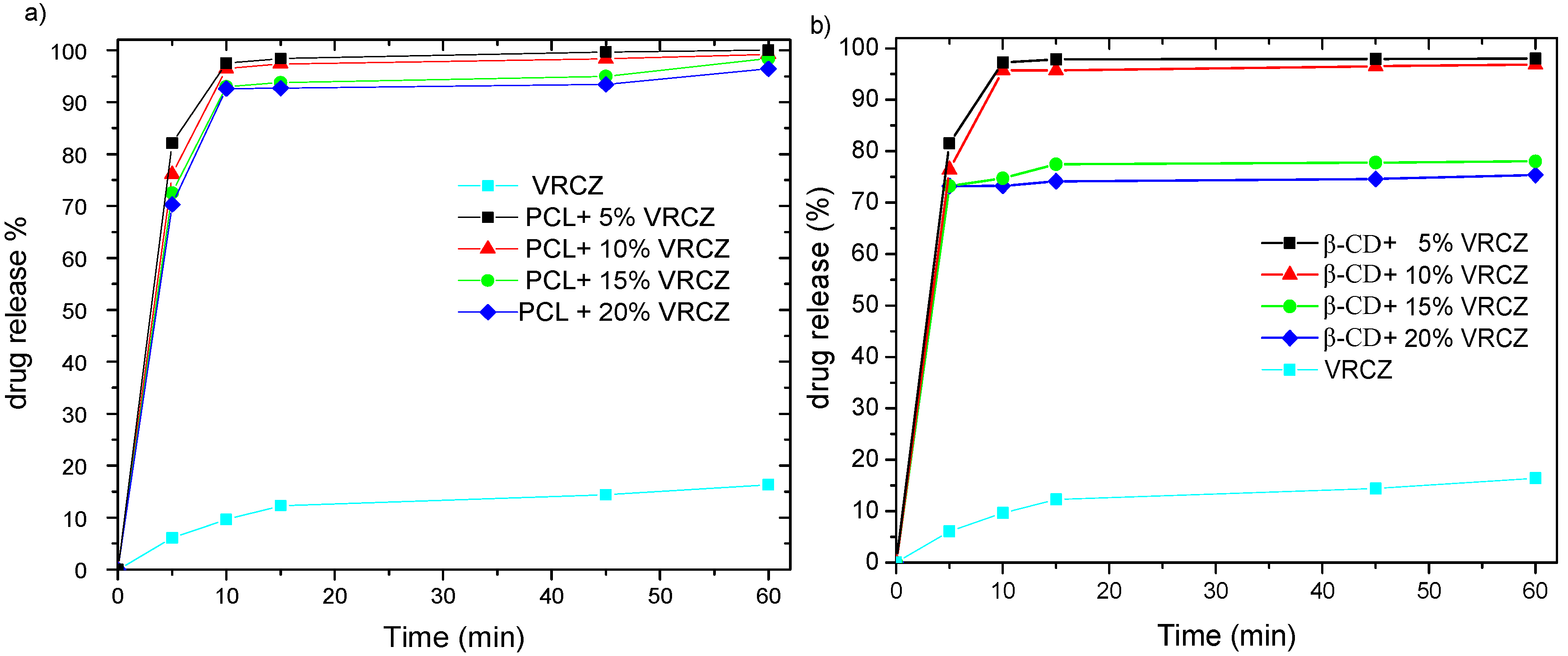
2.5. In Vitro Release Studies
2.6. Antifungal Activity of Formulations
3. Experimental Section
3.1. Materials
3.2. Fabrication of the Electrospun Fibers
3.3. Preparation of Inclusion Complex of VRCZ into β-CD via Freeze-Drying Method
3.4. Phase Solubility Studies for Inclusion Complexation
3.5. Characterization of Formulations
3.5.1. Surface Morphology
3.5.2. X-ray Diffraction Studies
3.5.3. FT-IR Spectroscopy
3.5.4. Evaluation of Drug Entrapment and Drug Loading
3.5.5. In Vitro Drug Release
3.5.6. Disk Diffusion Testing
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Zilberberg, M.D.; Shorr, A.F.; Kollef, M.H. Secular trends in candidemia-related hospitalization in the United States, 2000–2005. Infect. Control Hosp. Epidemiol. 2008, 29, 978–980. [Google Scholar] [CrossRef] [PubMed]
- Odds, F.C.; Brown, A.J.P.; Gow, N.A.R. Antifungal agents: Mechanisms of action. Trends Microbiol. 2003, 11, 272–279. [Google Scholar] [CrossRef]
- Ghannoum, M.A.; Rice, L.B. Antifungal agents: Mode of action, mechanisms of resistance, and correlation of these mechanisms with bacterial resistance. Clin. Microbiol. Rev. 1999, 12, 501–517. [Google Scholar] [PubMed]
- Dutcher, J.D. The discovery and development of amphotericin B. Dis. Chest 1968, 54, 296–298. [Google Scholar] [CrossRef] [PubMed]
- Sanati, H.; Belanger, P.; Fratti, R.; Ghannoum, M. A new triazole, voriconazole (UK-109,496), blocks sterol biosynthesis in candida albicans and candida krusei. Antimicrob. Agents Chemother. 1997, 41, 2492–2496. [Google Scholar] [PubMed]
- Kumar, S.S.; Thakuria, R.; Nangia, A. Pharmaceutical cocrystals and a nitrate salt of voriconazole. CrystEngComm 2014, 16, 4722. [Google Scholar] [CrossRef]
- Bikiaris, D.N. Solid dispersions, Part I: Recent evolutions and future opportunities in manufacturing methods for dissolution rate enhancement of poorly water-soluble drugs. Expert Opin. Drug Deliv. 2011, 8, 1501–1519. [Google Scholar] [CrossRef] [PubMed]
- Bikiaris, D.N. Solid dispersions, Part II: New strategies in manufacturing methods for dissolution rate enhancement of poorly water-soluble drugs. Expert Opin. Drug Deliv. 2011, 8, 1663–1680. [Google Scholar] [CrossRef] [PubMed]
- Aleem, O.; Kuchekar, B.; Pore, Y.; Late, S. Effect of β-cyclodextrin and hydroxypropyl β-cyclodextrin complexation on physicochemical properties and antimicrobial activity of cefdinir. J. Pharm. Biomed. Anal. 2008, 47, 535–540. [Google Scholar] [CrossRef] [PubMed]
- Goonoo, N.; Bhaw-Luximon, A.; Jhurry, D. Drug loading and release from electrospun biodegradable nanofibers. J. Biomed. Nanotechnol. 2014, 10, 2173–2199. [Google Scholar] [CrossRef] [PubMed]
- Veras, F.F.; Roggia, I.; Pranke, P.; Pereira, C.N.; Brandelli, A. Inhibition of filamentous fungi by ketoconazole-functionalized electrospun nanofibers. Eur. J. Pharm. Sci. 2016, 84, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, N.; Kundu, S.C. Electrospinning: A fascinating fiber fabrication technique. Biotechnol. Adv. 2010, 28, 325–347. [Google Scholar] [CrossRef] [PubMed]
- Bognitzki, M.; Frese, T.; Steinhart, M.; Greiner, A.; Wendorff, J.H.; Schaper, A.; Hellwig, M.; Pivsa-Art, W.; Pivsa-Art, S.; Fujii, K.; et al. Preparation of fibers with nanoscaled morphologies: Electrospinning of polymer blends. J. Control. Release 2002, 24, 4977–4985. [Google Scholar] [CrossRef]
- Taepaiboon, P.; Rungsardthong, U.; Supaphol, P. Drug-loaded electrospun mats of poly(vinyl alcohol) fibres and their release characteristics of four model drugs. Nanotechnology 2006, 2317. [Google Scholar] [CrossRef]
- Woodruff, M.A.; Hutmacher, D.W. The return of a forgotten polymer—Polycaprolactone in the 21st century. Prog. Polym. Sci. 2010, 35, 1217–1256. [Google Scholar] [CrossRef] [Green Version]
- Patra, S.N.; Easteal, A.J.; Bhattacharyya, D. Parametric study of manufacturing poly(lactic) acid nanofibrous mat by electrospinning. J. Mater. Sci. 2009, 44, 647–654. [Google Scholar] [CrossRef]
- Zamani, M.; Prabhakaran, M.P.; Ramakrishna, S. Advances in drug delivery via electrospun and electrosprayed nanomaterials. Int. J. Nanomed. 2013, 8, 2997–3017. [Google Scholar]
- Zamani, M.; Morshed, M.; Varshosaz, J.; Jannesari, M. Controlled release of metronidazole benzoate from poly epsilon-caprolactone electrospun nanofibers for periodontal diseases. Eur. J. Pharm. Biopharm. 2010, 75, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Spasova, M.; Paneva, D.; Manolova, N.; Radenkov, P.; Rashkov, I. Electrospun chitosan-coated fibers of poly(l-lactide) and poly(l-lactide)/poly(ethylene glycol): Preparation and characterization. Macromol. Biosci. 2008, 8, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Natu, M.V.; de Sousa, H.C.; Gil, M.H. Effects of drug solubility, state and loading on controlled release in bicomponent electrospun fibers. Int. J. Pharm. 2010, 397, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Fahr, A.; Liu, X. Drug delivery strategies for poorly water-soluble drugs. Expert Opin. Drug Deliv. 2007, 4, 403–416. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Guo, Q.X. The driving forces in the inclusion complexation of cyclodextrins. J. Incl. Phenom. Macrocycl. Chem. 2002, 42, 1–14. [Google Scholar] [CrossRef]
- Loftsson, T.; Brewster, M.E. Pharmaceutical applications of cyclodextrins. 1. Drug solubilization and stabilization. J. Pharm. Sci. 1996, 85, 1017–1025. [Google Scholar] [CrossRef] [PubMed]
- Salústio, P.J.; Pontes, P.; Conduto, C.; Sanches, I.; Carvalho, C.; Arrais, J.; Marques, H.M.C. Advanced technologies for oral controlled release: Cyclodextrins for oral controlled release. AAPS PharmSciTech 2011, 12, 1276–1292. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, G.M.; Le, K.H.T.; Giannitelli, S.M.; Lee, Y.J.; Rainer, A.; Trombetta, M. Electrospinning of PCL/PVP blends for tissue engineering scaffolds. J. Mater. Sci. Mater. Med. 2013, 24, 1425–1442. [Google Scholar] [CrossRef] [PubMed]
- Luong-Van, E.; Grøndahl, L.; Chua, K.N.; Leong, K.W.; Nurcombe, V.; Cool, S.M. Controlled release of heparin from poly(epsilon-caprolactone) electrospun fibers. Biomaterials 2006, 27, 2042–2050. [Google Scholar] [CrossRef] [PubMed]
- Pillay, V.; Dott, C.; Choonara, Y.E.; Tyagi, C.; Tomar, L.; Kumar, P.; Toit, L.C.; Ndesendo, V.M.K. A review of the effect of processing variables on the fabrication of electrospun nanofibers for drug delivery applications. J. Nanomater. 2013, 2013. [Google Scholar] [CrossRef]
- Sahoo, R.; Jocob, S.; Jayshree, P.; Manirao, R. Blend effect of PCL on antimicrobial activity of different molecular. Eur. Sci. J. 2014, 10, 36–47. [Google Scholar]
- Lee, K.H.; Kim, H.Y.; Khil, M.S.; Ra, Y.M.; Lee, D.R. Characterization of nano-structured poly(ε-caprolactone) nonwoven mats via electrospinning. Polymer 2003, 44, 1287–1294. [Google Scholar] [CrossRef]
- Beachley, V.; Wen, X. Effect of electrospinning parameters on the nanofiber diameter and length. Mater. Sci. Eng. C 2009, 29, 663–668. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Zhu, L.; White, K.; Branford-white, C. Electrospun nanofiber-based drug delivery systems. Pharm. Nanotechnol. 2009, 1, 67–75. [Google Scholar] [CrossRef]
- Wei, J.-D.; Tseng, H.; Chen, E.T.-H.; Hung, C.-H.; Liang, Y.-C.; Sheu, M.-T.; Chen, C.-H.; Goh, Y.-F.; Shakir, I.; Hussain, R.; et al. Electrospun fibers for tissue engineering, drug delivery, and wound dressing. J. Biomed. Mater. Res. A 2012, 48, 3027–3054. [Google Scholar]
- Kenawy, E.-R.; Abdel-Hay, F.I.; El-Newehy, M.H.; Wnek, G.E. Processing of polymer nanofibers through electrospinning as drug delivery systems. Mater. Chem. Phys. 2009, 113, 296–302. [Google Scholar] [CrossRef]
- Kenawy, E.R.; Bowlin, G.L.; Mansfield, K.; Layman, J.; Simpson, D.G.; Sanders, E.H.; Wnek, G.E. Release of tetracycline hydrochloride from electrospun poly(ethylene-co-vinylacetate), poly(lactic acid), and a blend. J. Control. Release 2002, 81, 57–64. [Google Scholar] [CrossRef]
- Sambasevam, K.; Mohamad, S.; Sarih, N.; Ismail, N. Synthesis and characterization of the inclusion complex of β-cyclodextrin and azomethine. Int. J. Mol. Sci. 2013, 14, 3671–3682. [Google Scholar] [CrossRef] [PubMed]
- Brewster, M.E.; Loftsson, T. Cyclodextrins as pharmaceutical solubilizers. Adv. Drug Deliv. Rev. 2007, 59, 645–666. [Google Scholar] [CrossRef] [PubMed]
- Higuchi, T.; Connors, K.A. Phase-solubility techniques. In Advances in Analytical Chemistry and Instrumentation; Jonh Wiley & Sons, Inc.: Hoboken, NJ, USA, 1965; pp. 117–212. [Google Scholar]
- Jahangiri, A.; Barzegar-Jalali, M.; Garjani, A.; Javadzadeh, Y.; Hamishehkar, H.; Asadpour-Zeynali, K.; Adibkia, K. Evaluation of physicochemical properties and in vivo efficiency of atorvastatin calcium/ezetimibe solid dispersions. Eur. J. Pharm. Sci. 2016, 82, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Siafaka, P.I.; Barmpalexis, P.; Lazaridou, M.; Papageorgiou, G.Z.; Koutris, E.; Karavas, E.; Kostoglou, M.; Bikiaris, D.N. Controlled release formulations of risperidone antipsychotic drug in novel aliphatic polyester carriers: Data analysis and modelling. Eur. J. Pharm. Biopharm. 2015, 94, 473–484. [Google Scholar] [CrossRef] [PubMed]
- Guarino, V.; Cirillo, V.; Taddei, P.; Alvarez-Perez, M.A.; Ambrosio, L. Tuning size scale and crystallinity of PCL electrospun fibres via solvent permittivity to address hMSC response. Macromol. Biosci. 2011, 11, 1694–1705. [Google Scholar] [CrossRef] [PubMed]
- Owens, P.K.; Fell, A.F.; Coleman, M.W.; Berridge, J.C. Complexation of voriconazole stereoisomers with neutral and anionic derivatised cyclodextrins. J. Incl. Phenom. Macrocycl. Chem. 2000, 38, 133–151. [Google Scholar] [CrossRef]
- Nanaki, S.G.; Koutsidis, I.A.; Koutri, I.; Karavas, E.; Bikiaris, D. Miscibility study of chitosan/2-hydroxyethyl starch blends and evaluation of their effectiveness as drug sustained release hydrogels. Carbohydr. Polym. 2012, 87, 1286–1294. [Google Scholar] [CrossRef]
- Karavas, E.; Georgarakis, E.; Bikiaris, D. Adjusting drug release by using miscible polymer blends as effective drug carries. J. Therm. Anal. Calorim. 2006, 84, 125–133. [Google Scholar] [CrossRef]
- Dott, C.; Tyagi, C.; Tomar, L.K.; Choonara, Y.E.; Kumar, P.; du Toit, L.C.; Pillay, V. A mucoadhesive electrospun nanofibrous matrix for rapid oramucosal drug delivery. J. Nanomater. 2013, 2013, 1–19. [Google Scholar] [CrossRef]
- Yu, D.-G.; Shen, X.-X.; Branford-White, C.; White, K.; Zhu, L.-M.; Bligh, S.W.A. Oral fast-dissolving drug delivery membranes prepared from electrospun polyvinylpyrrolidone ultrafine fibers. Nanotechnology 2009, 20, 055104. [Google Scholar] [CrossRef] [PubMed]
- Nagy, Z.K. Electrospun water soluble polymer mat for ultrafast release of Donepezil HCl. Polym. Lett. 2010, 4, 763–772. [Google Scholar] [CrossRef] [Green Version]
- Hirayama, F. Cyclodextrin-based controlled drug release system. Adv. Drug Deliv. Rev. 1999, 36, 125–141. [Google Scholar] [CrossRef]
- Gaharwar, A.K.; Mihaila, S.M.; Kulkarni, A.A.; Patel, A.; Di, A.; Reis, R.L.; Gomes, M.E.; van Blitterswijk, C.; Moroni, L.; Khademhosseini, A. Amphiphilic beads as depots for sustained drug release integrated into fi brillar scaffolds. J. Control. Release 2014, 187, 66–73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jenkinson, H.F.; Douglas, L.J. Interactions between Candida species and bacteria in mixed infections. In Polymicrobial Diseases; Brogden, K.A., Guthmiller, J.M., Eds.; ASM Press: Washington, DC, USA, 2002. [Google Scholar]
- Loftsson, T.; Hreinsdóttir, D.; Másson, M. Evaluation of cyclodextrin solubilization of drugs. Int. J. Pharm. 2005, 302, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Pathak, A.; Jain, N.; Joshi, R. Antibiogram of Candida species isolated from mono and multi-species oral Candidal carriage using disk diffusion method. Saudi J. Heal. Sci. 2012, 1, 132. [Google Scholar] [CrossRef]
- Srinivas, P.; Sreeja, K. Formulation and evaluation of voriconazole loaded nanosponges for oral and topical delivery. Int. J. Drug Dev. Res. 2013, 5, 55–69. [Google Scholar]








| Sample | Particle Size (μm) |
|---|---|
| β-CD + 5 wt % VRCZ | 2.14 ± 0.75 |
| β-CD + 10 wt % VRCZ | 3.42 ± 1.56 |
| β-CD + 15 wt % VRCZ | 12.02 ± 4.9 |
| β-CD + 20 wt % VRCZ | 25.26 ± 8.8 |
| Sample | Water Uptake (%) | Morphology after Dissolution (HCl 1N) |
|---|---|---|
| PCL | 14.578 ± 0.903 |  |
| PCL + 5% VRCZ | 15.789 ± 0.688 |  |
| PCL + 10% VRCZ | 15.2723 ± 1.114 |  |
| PCL + 15% VRCZ | 15. 645 ± 1.235 |  |
| PCL + 20% VRCZ | 14.306 ± 1.908 |  |
| VRCZ Concentration | Zone Inhibition Diameter (cm) | ||
|---|---|---|---|
| Control | β-CD Complexes | PCL Fibers | |
| 5% | 4.0 ± 0.3 | 4.1 ± 0.2 | 4.0 ± 0.3 |
| 10% | 4.6 ± 0.1 | 5.2 ± 0.1 | 4.8 ± 0.2 |
| 15% | 4.8 ± 0.2 | 5.4 ± 0.2 | 5.2 ± 0.3 |
| 20% | 5.0 ± 0.2 | 5.4 ± 0.3 | 5.4 ± 0.1 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Siafaka, P.I.; Üstündağ Okur, N.; Mone, M.; Giannakopoulou, S.; Er, S.; Pavlidou, E.; Karavas, E.; Bikiaris, D.N. Two Different Approaches for Oral Administration of Voriconazole Loaded Formulations: Electrospun Fibers versus β-Cyclodextrin Complexes. Int. J. Mol. Sci. 2016, 17, 282. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms17030282
Siafaka PI, Üstündağ Okur N, Mone M, Giannakopoulou S, Er S, Pavlidou E, Karavas E, Bikiaris DN. Two Different Approaches for Oral Administration of Voriconazole Loaded Formulations: Electrospun Fibers versus β-Cyclodextrin Complexes. International Journal of Molecular Sciences. 2016; 17(3):282. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms17030282
Chicago/Turabian StyleSiafaka, Panoraia I., Neslihan Üstündağ Okur, Mariza Mone, Spyridoula Giannakopoulou, Sevda Er, Eleni Pavlidou, Evangelos Karavas, and Dimitrios N. Bikiaris. 2016. "Two Different Approaches for Oral Administration of Voriconazole Loaded Formulations: Electrospun Fibers versus β-Cyclodextrin Complexes" International Journal of Molecular Sciences 17, no. 3: 282. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms17030282








