Disrupted Homeostatic Cytokines Expression in Secondary Lymph Organs during HIV Infection
Abstract
:1. Introduction
2. Results
2.1. Different Cytokines were Downregulated in Secondary Lymph Organs (SLOs) of Human Immunodeficiency Virus (HIV)-Infected Patients
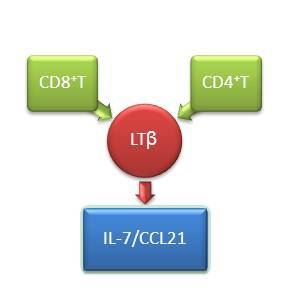
2.2. Lymphotoxin (LT)-β Plays a Critical Role in Regulating C–C Motif Chemokine Ligand (CCL12) and IL-7 Expression in Lymph Nodes of HIV-Infected Patients
2.3. Absence of CD4+T cells Have Little Effect on Cytokines Expression in SLOs
2.4. Elimination of Total T Cells Reduced LT-β Expression in SLOs
2.5. Depletion of Total T Cells Disrupts Cytokine Expression in SLOs
3. Discussion
4. Materials and Methods
4.1. Ethics Statement
4.2. Mice
4.3. Cell Migration in Vivo
4.4. Immunofluorescence Microscopy
4.5. Enzyme-Linked Immunosorbent Assay (ELISA)
4.6. Real-Time Quantitative Reverse Transcription Polymerase Chain Reaction (RT-qPCR)
4.7. Availability of Supporting Data
4.8. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| HIV | human immunodeficiency virus |
| SLOs | secondary lymph organs |
| LT-β | lymphotoxin (LT)-β |
| FRCs | fibroblastic reticular cells |
| FDCs | follicular dendritic cells |
| DCs | dendritic cells |
| CD4KO or CD8KO | CD4+T or CD8+T cells knockout |
References
- Volberding, P.A.; Deeks, S.G. Antiretroviral therapy and management of HIV infection. Lancet 2010, 376, 49–62. [Google Scholar] [CrossRef]
- Kelley, C.F.; Kitchen, C.M.; Hunt, P.W.; Rodriguez, B.; Hecht, F.M.; Kitahata, M.; Crane, H.M.; Willig, J.; Mugavero, M.; Saag, M.; et al. Incomplete peripheral CD4+ cell count restoration in HIV-infected patients receiving long-term antiretroviral treatment. Clin. Infect. Dis. 2009, 48, 787–794. [Google Scholar] [CrossRef] [PubMed]
- Schacker, T.W.; Nguyen, P.L.; Martinez, E.; Reilly, C.; Gatell, J.M.; Horban, A.; Bakowska, E.; Berzins, B.; van Leeuwen, R.; Wolinsky, S.; et al. Persistent abnormalities in lymphoid tissues of human immunodeficiency virus-infected patients successfully treated with highly active antiretroviral therapy. J. Infect. Dis. 2002, 186, 1092–1097. [Google Scholar] [CrossRef] [PubMed]
- Estes, J.D. Role of collagen deposition in lymphatic tissues and immune reconstruction during HIV-1 and SIV infections. Curr. HIV/AIDS Rep. 2009, 6, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Zeng, M.; Smith, A.J.; Wietgrefe, S.W.; Southern, P.J.; Schacker, T.W.; Reilly, C.S.; Estes, J.D.; Burton, G.F.; Silvestri, G.; Lifson, J.D.; et al. Cumulative mechanisms of lymphoid tissue fibrosis and T cell depletion in HIV-1 and SIV infections. J. Clin. Invest. 2011, 121, 998–1008. [Google Scholar] [CrossRef] [PubMed]
- Zeng, M.; Paiardini, M.; Engram, J.C.; Beilman, G.J.; Chipman, J.G.; Schacker, T.W.; Silvestri, G.; Haase, A.T. Critical role of CD4 T cells in maintaining lymphoid tissue structure for immune cell homeostasis and reconstitution. Blood 2012, 120, 1856–1867. [Google Scholar] [CrossRef] [PubMed]
- Estes, J.D. Pathobiology of HIV/SIV-associated changes in secondary lymphoid tissues. Immunol. Rev. 2013, 254, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Bajenoff, M.; Egen, J.G.; Koo, L.Y.; Laugier, J.P.; Brau, F.; Glaichenhaus, N.; Germain, R.N. Stromal cell networks regulate lymphocyte entry, migration, and territoriality in lymph nodes. Immunity 2006, 25, 989–1001. [Google Scholar] [CrossRef] [PubMed]
- Malhotra, D.; Fletcher, A.L.; Turley, S.J. Stromal and hematopoietic cells in secondary lymphoid organs: Partners in immunity. Immunol. Rev. 2013, 251, 160–176. [Google Scholar] [CrossRef] [PubMed]
- Den Haan, J.M.; Mebius, R.E.; Kraal, G. Stromal cells of the mouse spleen. Front. Immunol. 2012, 3. [Google Scholar] [CrossRef]
- Aguzzi, A.; Kranich, J.; Krautler, N.J. Follicular dendritic cells: Origin, phenotype, and function in health and disease. Trends Immunol. 2014, 35, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Mueller, S.N.; Hosiawa-Meagher, K.A.; Konieczny, B.T.; Sullivan, B.M.; Bachmann, M.F.; Locksley, R.M.; Ahmed, R.; Matloubian, M. Regulation of homeostatic chemokine expression and cell trafficking during immune responses. Science 2007, 317, 670–674. [Google Scholar] [CrossRef] [PubMed]
- Siegert, S.; Luther, S.A. Positive and negative regulation of T cell responses by fibroblastic reticular cells within paracortical regions of lymph nodes. Front. Immunol. 2012, 3. [Google Scholar] [CrossRef] [PubMed]
- Comerford, I.; Harata-Lee, Y.; Bunting, M.D.; Gregor, C.; Kara, E.E.; McColl, S.R. A myriad of functions and complex regulation of the CCR7/CCL19/CCL21 chemokine axis in the adaptive immune system. Cytokine Growth Factor Rev. 2013, 24, 269–283. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Liu, L.; Gao, J.; Yang, Y.; Hu, C.; Guo, B.; Zhu, B. T lymphocytes maintain structure and function of fibroblastic reticular cells via lymphotoxin (LT)-B. BMC Immunol. 2014, 15. [Google Scholar] [CrossRef] [PubMed]
- Bonizzi, G.; Karin, M. The two NF-κB activation pathways and their role in innate and adaptive immunity. Trends Immunol. 2004, 25, 280–288. [Google Scholar] [CrossRef] [PubMed]
- Schneider, K.; Potter, K.G.; Ware, C.F. Lymphotoxin and light signaling pathways and target genes. Immunol. Rev. 2004, 202, 49–66. [Google Scholar] [CrossRef] [PubMed]
- Tumanov, A.V.; Kuprash, D.V.; Nedospasov, S.A. The role of lymphotoxin in development and maintenance of secondary lymphoid tissues. Cytokine Growth Factor Rev. 2003, 14, 275–288. [Google Scholar] [CrossRef]
- Chai, Q.; Onder, L.; Scandella, E.; Gil-Cruz, C.; Perez-Shibayama, C.; Cupovic, J.; Danuser, R.; Sparwasser, T.; Luther, S.A.; Thiel, V.; et al. Maturation of lymph node fibroblastic reticular cells from myofibroblastic precursors is critical for antiviral immunity. Immunity 2013, 38, 1013–1024. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Smith, A.J.; Schacker, T.W.; Carlis, J.V.; Duan, L.; Reilly, C.S.; Haase, A.T. Microarray analysis of lymphatic tissue reveals stage-specific, gene expression signatures in HIV-1 infection. J. Immunol. 2009, 183, 1975–1982. [Google Scholar] [CrossRef] [PubMed]
- Bajenoff, M.; Glaichenhaus, N.; Germain, R.N. Fibroblastic reticular cells guide T lymphocyte entry into and migration within the splenic T cell zone. J. Immunol. 2008, 181, 3947–3954. [Google Scholar] [CrossRef] [PubMed]
- Levy, Y.; Lacabaratz, C.; Weiss, L.; Viard, J.-P.; Goujard, C.; Lelievre, J.-D.; Boue, F.; Molina, J.-M.; Rouzioux, C.; Avettand-Fenoel, V.; et al. Enhanced T cell recovery in HIV-1-infected adults through IL-7 treatment. J. Clin. Invest. 2009, 119, 997–1007. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fry, T.J.; Mackall, C.L. The many faces of IL-7: From lymphopoiesis to peripheral T cell maintenance. J. Immunol. 2005, 174, 6571–6576. [Google Scholar] [CrossRef] [PubMed]
- Onder, L.; Narang, P.; Scandella, E.; Chai, Q.; Iolyeva, M.; Hoorweg, K.; Halin, C.; Richie, E.; Kaye, P.; Westermann, J.; et al. IL-7-producing stromal cells are critical for lymph node remodeling. Blood 2012, 120, 4675–4683. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Liu, L.; Guo, B.; Zhu, B. Regulation of adaptive immune responses by guiding cell movements in the spleen. Front. Microbiol. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Zhao, L.; Wan, Y.Y.; Zhu, B. Mechanism of action of IL-7 and its potential applications and limitations in cancer immunotherapy. Int. J. Mol. Sci. 2015, 16, 10267–10280. [Google Scholar] [CrossRef] [PubMed]
- Soudja, S.M.; Henri, S.; Mello, M.; Chasson, L.; Mas, A.; Wehbe, M.; Auphan-Anezin, N.; Leserman, L.; van den Eynde, B.; Schmitt-Verhulst, A.M. Disrupted lymph node and splenic stroma in mice with induced inflammatory melanomas is associated with impaired recruitment of t and dendritic cells. PLoS ONE 2011, 6, e22639. [Google Scholar] [CrossRef] [PubMed]
- Tsitsiou, E.; Williams, A.E.; Moschos, S.A.; Patel, K.; Rossios, C.; Jiang, X.; Adams, O.-D.; Macedo, P.; Booton, R.; Gibeon, D.; et al. Transcriptome analysis shows activation of circulating CD8+T cells in patients with severe asthma. J. Allergy. Clin. Immunol. 2012, 129, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.J.; Toledo, C.M.; Wietgrefe, S.W.; Duan, L.; Schacker, T.W.; Reilly, C.S.; Haase, A.T. The immunosuppressive role of IL-32 in lymphatic tissue during HIV-1 infection. J. Immunol. 2011, 186, 6576–6584. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Schacker, T.; Carlis, J.; Beilman, G.; Nguyen, P.; Haase, A.T. Functional genomic analysis of the response of HIV-1-infected lymphatic tissue to antiretroviral therapy. J. Infect. Dis. 2004, 189, 572–582. [Google Scholar] [CrossRef] [PubMed]




© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, L.; Gao, J.; Li, Y.; Liu, L.; Yang, Y.; Guo, B.; Zhu, B. Disrupted Homeostatic Cytokines Expression in Secondary Lymph Organs during HIV Infection. Int. J. Mol. Sci. 2016, 17, 413. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms17030413
Zhao L, Gao J, Li Y, Liu L, Yang Y, Guo B, Zhu B. Disrupted Homeostatic Cytokines Expression in Secondary Lymph Organs during HIV Infection. International Journal of Molecular Sciences. 2016; 17(3):413. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms17030413
Chicago/Turabian StyleZhao, Lintao, Jianbao Gao, Yan Li, Lina Liu, Yang Yang, Bo Guo, and Bo Zhu. 2016. "Disrupted Homeostatic Cytokines Expression in Secondary Lymph Organs during HIV Infection" International Journal of Molecular Sciences 17, no. 3: 413. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms17030413