Characterization of FLOWERING LOCUS C Homologs in Apple as a Model for Fruit Trees
Abstract
:1. Introduction
2. Results
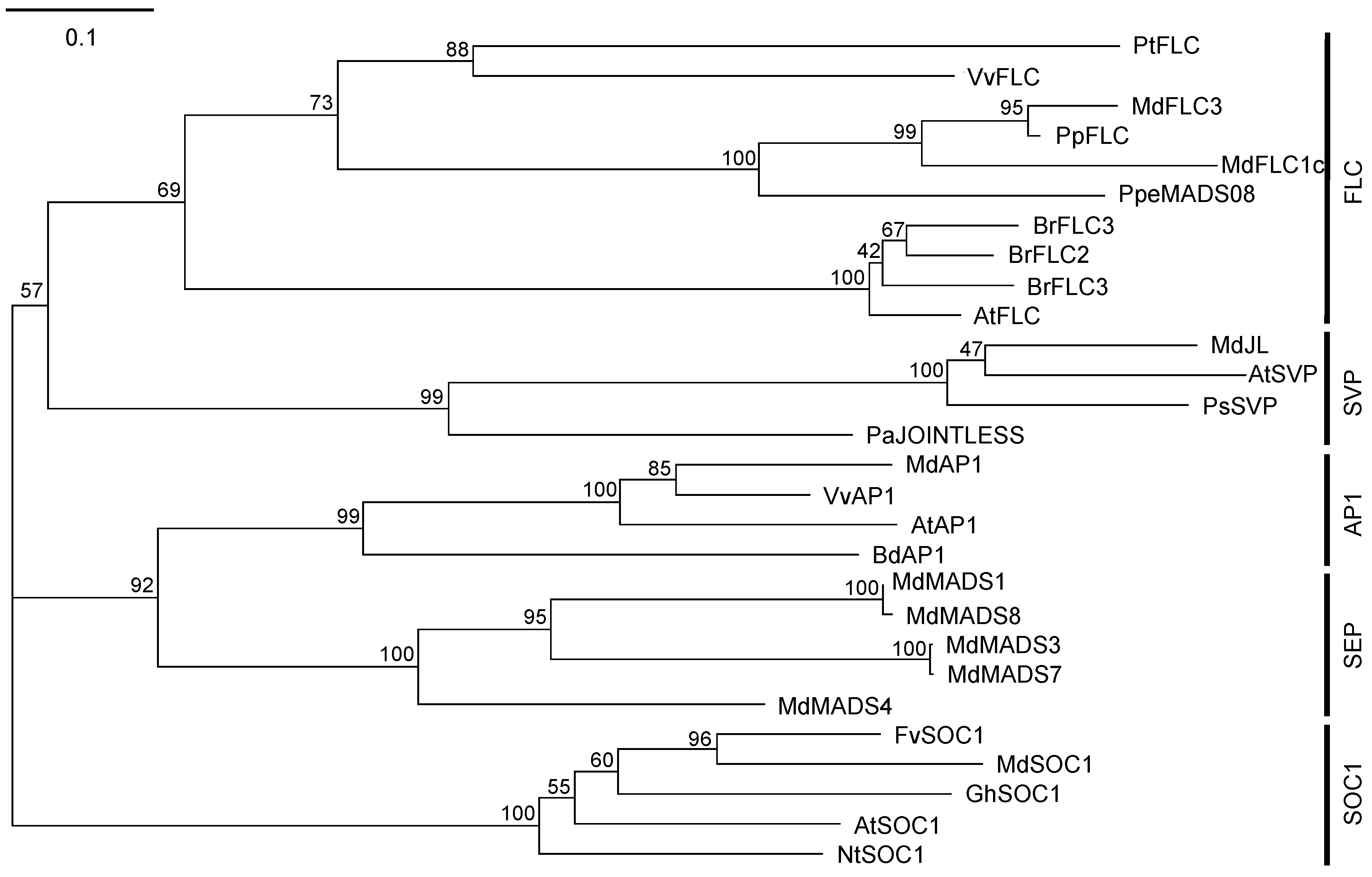
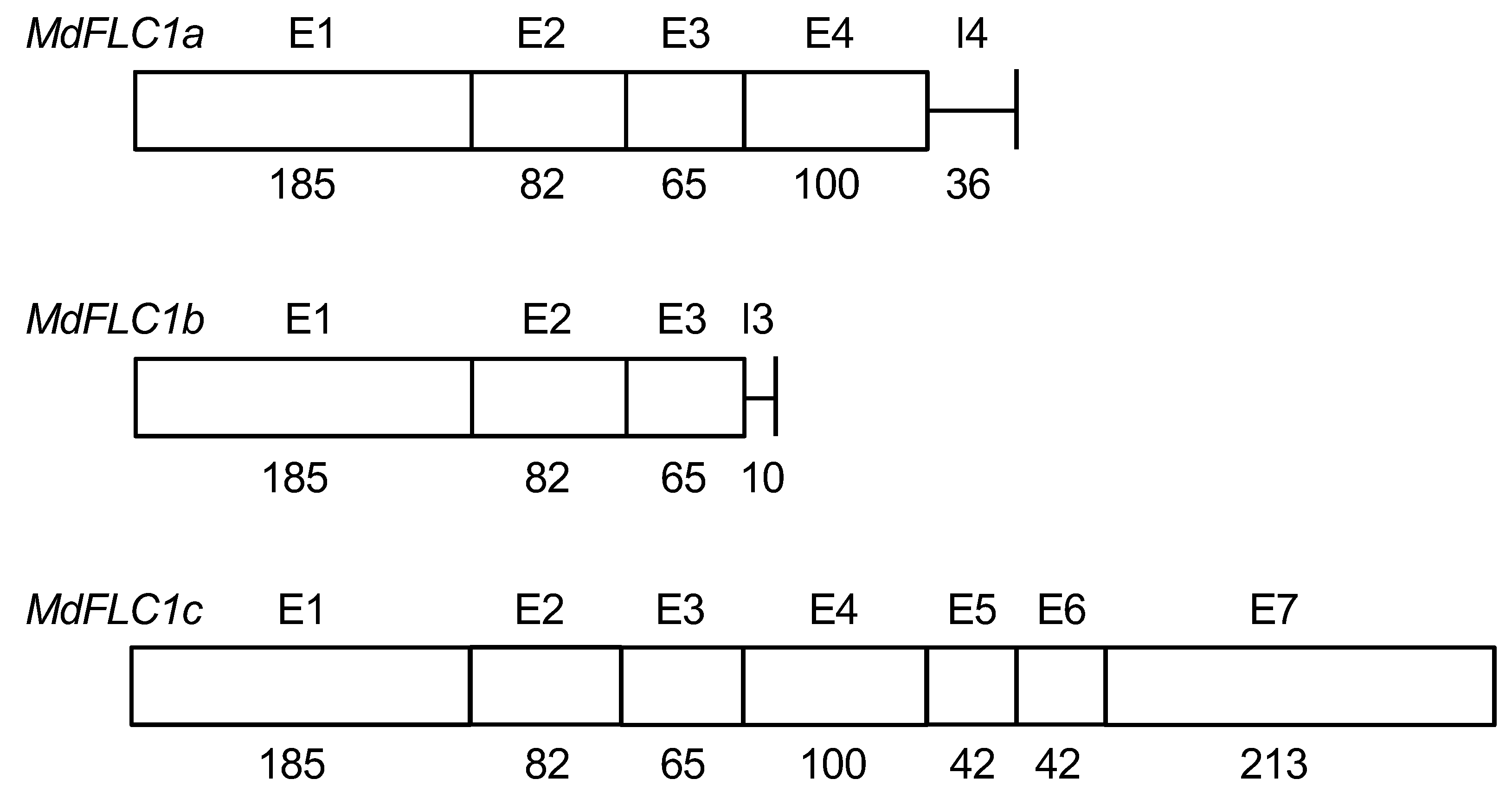
2.1. cDNA Isolation and Phylogenetic Tree Analysis of MdFLC
2.2. Expression Analysis of MdFLC in the Adult Trees
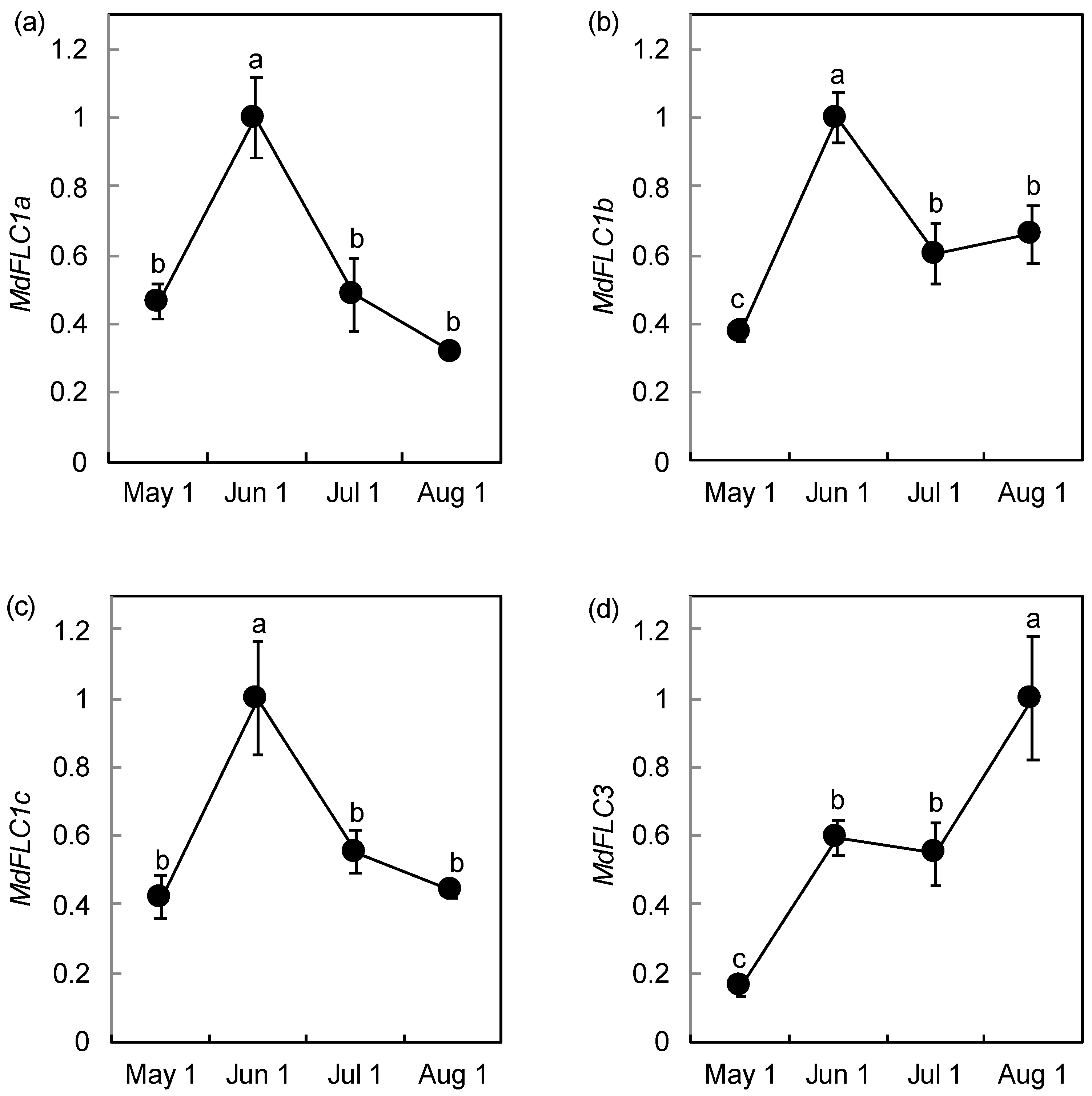
2.3. Expression Analysis of MdFLC During Phase Transition
2.4. Transformation of Arabidopsis with MdFLC3 cDNA
3. Discussion
4. Materials and Methods
4.1. Plant Materials
4.2. cDNA Isolation, Sequence Analysis, and Phylogenetic Tree Analysis of MdFLC
4.3. Expression Analysis by Real-Time PCR
4.4. Transformation of Arabidopsis with MdFLC3 cDNA
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| FLC | FLOWERING LOCUS C |
| TFL1 | TERMINAL FLOWER 1 |
| FT | FLOWERING LOCUS T |
| LFY | LEAFY |
| AP1 | APETALA1 |
| EST | expressed sequence tag |
| DDBJ | DNA Data Bank of Japan |
| WT | wild-type |
References
- Zimmerman, R.H. Juvenility and flowering in woody plants: A review. HortScience 1972, 7, 447–455. [Google Scholar]
- Visser, T. Juvenile phase and growth of apple and pear seedlings. Euphytica 1964, 13, 119–129. [Google Scholar]
- Visser, T. The effect of rootstocks on growth and flowering of apple seedlings. J. Am. Soc. Hortic. Sci. 1973, 98, 26–28. [Google Scholar]
- Hackett, W.P. Juvenility, maturation, and rejuvenation in woody plants. Hortic. Rev. 1985, 7, 109–155. [Google Scholar]
- Velasco, R.; Zharkikh, A.; Affourtit, J.; Dhingra, A.; Cestaro, A.; Kalyanaraman, A.; Fontana, P.; Bhatnagar, S.K.; Troggio, M.; Pruss, D.; et al. The genome of the domesticated apple (Malus × domestica Borkh.). Nat Genet. 2010, 42, 833–839. [Google Scholar] [CrossRef]
- Eccher, G.; Ferrero, S.; Populin, F.; Colombo, L.; Botton, A. Apple (Malus domestica L. Borkh) as an emerging model for fruit development. Plant Biosyst. 2014, 148, 157–168. [Google Scholar] [CrossRef]
- He, P.; Li, L.G.; Li, H.F.; Wang, H.B.; Yang, J.M. Development and application S-SAP molecular markers for the identification of apple (Malus Domestica Borkh.) sports. J. Hortic. Sci. Biotechnol. 2015, 90, 297–302. [Google Scholar] [CrossRef]
- Poething, R.S. Phase change and the regulation of shoot morphogenesis in plants. Science 1990, 250, 923–930. [Google Scholar] [CrossRef]
- Mimida, N.; Komori, S.; Suzuki, A.; Wada, M. Functions of the apple TFL1/FT orthologs in phase transition. Sci. Hortic. 2013, 156, 106–112. [Google Scholar] [CrossRef]
- Kotoda, N.; Wada, M.; Kusaba, S.; Kano-Murakami, Y.; Masuda, T.; Soejima, J. Overexpression of MdMADS5, an APETALA1-like gene of apple, causes early flowering in transgenic Arabidopsis. Plant Sci. 2002, 162, 679–687. [Google Scholar] [CrossRef]
- Kotoda, N.; Wada, M. MdTFL1, a TFL1-like gene of apple retards the transition from the vegetative to reproductive phase in transgenic Arabidopsis. Plant Sci. 2005, 168, 95–104. [Google Scholar] [CrossRef]
- Wada, M.; Cao, Q.; Kotoda, N.; Soejima, J.; Masuda, T. Apple has two orthologues of FLORICAULA/LEAFY involved in flowering. Plant Mol. Biol. 2002, 49, 567–577. [Google Scholar] [CrossRef] [PubMed]
- Pena, L.; Martin-Trillo, M.; Juarez, J.; Pina, J.A.; Navarro, L.; Martinez-Zapater, J.M. Constitutive expression of Arabidopsis LFAFY or APETALA1 gene in citrus reduces their generation time. Nat. Biotech. 2001, 19, 263–267. [Google Scholar] [CrossRef]
- Zhang, J.Z.; Mei, L.; Liu, R.; Khan, M.R.G.; Hu, C.G. Possible involvement of locus-specific methylation on expression regulation of LEAFY homologous gene (CiLFY) during precocious trifoliate orange phase change process. PLoS ONE 2014, 9, e88558. [Google Scholar] [CrossRef] [PubMed]
- Srikanth, A.; Schmid, M. Regulation of flowering time: All roads lead to Rome. Cell Mol. Life Sci. 2011, 68, 2013–2037. [Google Scholar] [CrossRef] [PubMed]
- Michaels, S.D.; Amasino, R.M. FLOWERING LOCUS C encodes a novel MADS domain protein that acts as a repressor of flowering. Plant Cell 1990, 11, 949–956. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Helliwell, C.A.; Wood, C.C.; Robertson, M.; James, P.W.; Dennis, E.S. The Arabidopsis FLC protein interacts directly in vivo with SOC1 and FT chromatin and is part of a high-molecular-weight protein complex. Plant J. 2006, 46, 183–192. [Google Scholar] [CrossRef]
- Hepworth, S.R.; Valverde, F.; Ravenscroft, D.; Mouradov, A.; Coupland, G. Antagonistic regulation of flowering-time gene SOC1 by CONSTANS and FLC via separate promoter motifs. EMBO J. 2002, 21, 4327–4337. [Google Scholar] [CrossRef] [Green Version]
- Searle, I.; He, Y.; Turck, F.; Vincent, C.; Fornara, F.; Krober, S.; Amasino, R.A.; Coupland, G. The transcription factor FLC confers a flowering response to vernalization by repressing meristem competence and systemic signaling in Arabidopsis. Genes Dev. 2006, 20, 898–912. [Google Scholar] [CrossRef] [Green Version]
- Kumar, G.; Arya, P.; Gupta, K.; Randhawa, V.; Achaya, V.; Singh, A.K. Comparative phylogenetic analysis and transcriptional profiling of MADS-box gene family identified DAM and FLC-like genes in apple (Malus x domestica). Sci. Rep. 2016, 6, 20695. [Google Scholar] [CrossRef] [Green Version]
- Urrestarazu, J.; Muranty, H.; Denancé, C.; Leforestier, D.; Ravon, E.; Guyader, A.; Guisnel, R.; Feugey, L.; Aubourg, S.; Celton, J.-M.; et al. Genome-wide association mapping of flowering and ripening periods in apple. Front. Plant Sci. 2017, 8, 1923. [Google Scholar] [CrossRef] [PubMed]
- Porto, D.D.; Bruneau, M.; Perini, P.; Anzanello, R.; Jean-Pierre, R.; Dos Santos, H.P.; Fialho, F.B.; Revers, L.F. Transcription profiling of the chilling requirement for bud break in apples: A putative role for FLC-like genes. J. Exp. Bot. 2015, 66, 2659–2672. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zong, X.; Zhang, Y.; Walworth, A.; Tomaszewski, E.M.; Callow, P.; Zhong, G.Y.; Song, G.Q. Constitutive expression of an apple FLC3-like gene promotes flowering in transgenic blueberry under nonchilling conditions. Int. J. Mol. Sci. 2019, 20, 2775. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wells, E.C.; Vendramin, E.; Tarodo, J.S.; Verge, I.; Bielenberg, G.D. A genome-wide analysis of MADS-box genes in peach [Prunus persica (L.) Batsch]. BMC Plant Biol. 2015, 15, 41. [Google Scholar] [CrossRef] [Green Version]
- Xu, Z.; Zhang, Q.; Sun, L.; Du, D.; Cheng, T.; Pan, H.; Yang, W.; Wang, J. Genome-wide identification, characterization and expression analysis of the MADS-box gene family in Prunus mume. Mol. Genet. Genom. 2014, 289, 903–920. [Google Scholar] [CrossRef]
- Zhang, J.Z.; Li, Z.M.; Mei, L.; Yao, J.L.; Hu, C.G. PtFLC homolog from trifoliate orange (Poncirus trifoliate) is regulated by alternative splicing and experiences seasonal fluctuation in expression level. Planta 2009, 229, 847–859. [Google Scholar] [CrossRef]
- Hatsuda, Y.; Nishio, S.; Komori, S.; Nishiyama, M.; Kanahama, K.; Kanayama, Y. Relationship between MdMADS11 gene expression and juvenility in apple. J. Soc. Hortic. Sci. 2011, 80, 396–403. [Google Scholar] [CrossRef] [Green Version]
- Zeevaart, J.A.D. Leaf-produced floral signals. Curr. Opin. Plant Biol. 2008, 11, 541–547. [Google Scholar] [CrossRef]
- Freiman, A.; Golobovitch, S.; Yablovitz, Z.; Belausov, E.; Dahan, Y.; Peer, R.; Avraham, L.; Freiman, Z.; Evenor, D.; Reuveni, M.; et al. Expression of flowering locus T2 transgene from Pyrus communis L. delays dormancy and leaf senescence in Malus x domestica Borkh, and causes early flowering in tobacco. Plant Sci. 2015, 241, 164–176. [Google Scholar] [CrossRef]
- Díaz-Riquelme, J.; Martínez-Zapater, J.M.; Carmona, M.J. Transcriptional analysis of tendril and inflorescence development in grapevine. PLoS ONE 2014, 9, e92339. [Google Scholar] [CrossRef] [Green Version]
- De Bodt, S.; Raes, J.; Florquin, K.; Rombauts, S.; Rouzé, P.; Theissen, G.; Van de Peer, Y. Genomewide structural annotation and evolutionary analysis of the type I MADS-box genes in plants. J. Mol. Evol. 2003, 56, 573–586. [Google Scholar] [CrossRef]
- Theissen, G.; Saedler, H. Floral quartets. Nature 2001, 409, 469–471. [Google Scholar] [CrossRef]
- Honma, T.; Goto, K. Complexes of MADS-box proteins are sufficient to convert leaves into floral organs. Nature 2001, 409, 525–529. [Google Scholar] [CrossRef] [PubMed]
- Kaufmann, K.; Melzer, R.; Theissen, G. MIKC-type MADS-domain proteins: Structural modularity, protein interactions and network evolution in land plants. Gene 2005, 347, 183–198. [Google Scholar] [CrossRef] [PubMed]
- Shang, X.; Cao, Y.; Ma, L. Alternative splicing in plant genes: A means of regulating the environmental fitness of plants. Int. J. Mol. Sci. 2017, 18, 432. [Google Scholar] [CrossRef] [PubMed]
- Carlsbecker, A.; Tandre, K.; Johanson, U.; Englund, M.; Engstrom, P. The MADS-box gene DAL1 is a potential mediator of the juvenile-to-adult transition in Norway spruce (Picea abies). Plant J. 2004, 40, 546–557. [Google Scholar] [CrossRef]
- Moriya, S.; Terakami, S.; Okada, K.; Shimizu, T.; Adachi, Y.; Katayose, Y.; Fujisawa, H.; Wu, J.; Kanamori, H.; Yamamoto, T.; et al. Identification of candidate genes responsible for the susceptibility of apple (Malus × domestica Borkh.) to Alternaria blotch. BMC Plant Biol. 2019, 19, 132. [Google Scholar] [CrossRef]
- Zhou, J.; Li, D.; Wang, G.; Wang, F.; Kunjal, M.; Joldersma, D.; Liu, Z. Application and future perspective of CRISPR/Cas9 genome editing in fruit crops. J. Integr. Plant Biol. 2020, 62, 269–286. [Google Scholar] [CrossRef] [Green Version]
- Ikeda, H.; Shibuya, T.; Imanishi, S.; Aso, H.; Nishiyama, M.; Kanayama, Y. Dynamic Metabolic regulation by a chromosome segment from a wild relative during fruit development in a tomato introgression line, IL8-3. Plant Cell Physiol. 2016, 57, 1257–1270. [Google Scholar] [CrossRef] [Green Version]
- Kanayama, Y.; Mizutani, R.; Yaguchi, S.; Hojo, A.; Ikeda, H.; Nishiyama, M.; Kanahama, K. Characterization of an uncharacterized aldo-keto reductase gene from peach and its role in abiotic stress tolerance. Phytochemistry 2014, 104, 30–36. [Google Scholar] [CrossRef]



| Gene | Accession Number | Putative CArG Box | Strand | Position of 1st C from ATG b |
|---|---|---|---|---|
| MdFT1 | AB458506 | AACTCCATTAATTGCAGG | Top | +289 |
| TACTCCTTATTTTGTCAA | Top | +846 | ||
| MdFT2 | AB458504 | CTAACCATTAATTGTGTT | Top | +1001 |
| AGATCCTAAAAAAGTATA | Bottom | +994 | ||
| GTATCCAAATAAGTTGC | Bottom | −159 | ||
| GTCTCCTAATTTGTTGT | Bottom | −745 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kagaya, H.; Ito, N.; Shibuya, T.; Komori, S.; Kato, K.; Kanayama, Y. Characterization of FLOWERING LOCUS C Homologs in Apple as a Model for Fruit Trees. Int. J. Mol. Sci. 2020, 21, 4562. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms21124562
Kagaya H, Ito N, Shibuya T, Komori S, Kato K, Kanayama Y. Characterization of FLOWERING LOCUS C Homologs in Apple as a Model for Fruit Trees. International Journal of Molecular Sciences. 2020; 21(12):4562. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms21124562
Chicago/Turabian StyleKagaya, Hidenao, Naoko Ito, Tomoki Shibuya, Sadao Komori, Kazuhisa Kato, and Yoshinori Kanayama. 2020. "Characterization of FLOWERING LOCUS C Homologs in Apple as a Model for Fruit Trees" International Journal of Molecular Sciences 21, no. 12: 4562. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms21124562





